Abstract
Graphene and related materials have come to the forefront of research in electrochemical sensors during recent years due to the promising properties of these nanomaterials. Further applications of these nanomaterials have been hampered by insufficient sensitivity offered by these nanohybrids for the type of molecules requiring lower detection ranges. Here, we report a signal amplification strategy based on magneto-electrochemical immunoassay which combines the advantages of carbon nanotube and reduced graphene oxide together with electrochemical bursting of magnetic nanoparticles into a large number of metal ions. Sensitive detection was achieved by precisely designing the nanohybrid and correlating the available metal ions with analyte concentration. We confirmed the ultrahigh sensitivity of this method for a new generation herbicide diuron and its analogues up to sub-picomolar concentration in standard water samples. The novel immune-detection platform showed the excellent potential applicability in rapid and sensitive screening of environmental pollutants or toxins in samples.
Similar content being viewed by others
Introduction
Graphene-based nanocomposite films have recently been used as enhanced sensing platform for the development of electrochemical sensors and biosensors because of their unique facile surface modification characteristics and high charge mobility1,2,3. Zhang et al., have recently reported a hybrid film consisting of graphene oxide (GO) nanosheets together with the prussian blue films for electrochemical sensing applications4. In a different approach, an in-situ chemical synthesis approach has been developed to prepare graphene-gold nanoparticles based nanocomposite, demonstrating its good potential as a highly sensitive electrochemical sensing platform5. A GO sheet consists of two randomly distributed regions namely, aromatic regions with unoxidised benzene rings and regions with aliphatic six-membered rings making it to behave like an amphiphilic molecule6. The oxygen containing groups render GO sheets hydrophilic and highly dispersible in water, whereas the aromatic regions offer active sites to make it possible to interact with other aromatic molecules through π-π supramolecular interactions. This chemical nature makes GO a unique dispersant to suspend CNTs in water and to develop a new strategy for making graphene/CNT hybrids7,8. Similarities in structure and physical properties between CNTs and graphene, their hybridization would presumably have useful synergistic effects in biosensing applications9,10,11.
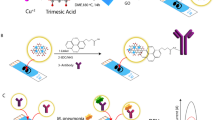
Nanometer-sized magnetic particles of iron are potential candidates in catalysis, magnetic separation and biomedical applications12. However, pure iron nanoparticles are chemically unstable and easily oxidize, which limits their utility in biosensing and other applications. These particles are therefore coated with another inert layer such as metal-oxide (iron oxide), inorganic material (SiO2) and noble metals (gold and silver), thereby making core–shell nano-structures showing favorable magnetic properties of metal iron while preventing them from oxidation13. Gold has been one of the potential coating materials owing to its chemical inertness, biocompatibility, non-toxic and diverse cluster geometries14. Very recently, inorganic or semiconductor nanoparticles tagged with receptor molecules has generated good interest for electrochemical detection of analyte15,16. Anodic stripping voltammetry (ASV) has proved to be a very sensitive method for trace determination of metal ions liberated from nanoparticles. Recently, Liu developed multi-QDs functionalized silica nanoparticles based electrochemical amplification platform which dramatically enhanced the intensity of the signal and led to ultrasensitive detection17. Our previous study reported the use of gold nanoparticles mediated ASV technique based upon oxidative gold nanoparticles dissolution in an acidic solution. The consequent release of large amount of gold (Au) metal ions after dissolution lead to the development of sensitive stripping voltammetry based immunoassay18. However, it suffers from the use of strongly corrosive and hazardous agents such as HBr/Br2 for the oxidation of gold nanoparticles, which minimizes it's usage in common lab practices. Although great achievement has been obtained in this field, the finding of more sensitive, environment friendly convenient assay still attracts increasing interest for improving the sensitivity of bioassays. The present work demonstrates the development of metal ion derivatized electrochemical immunoassay based on gold-iron (Au/Fe) nanobioprobes for the detection of commonly used phenylurea herbicide diuron on reduced graphene oxide-carbon nanotubes (rGO/CNT) modified biosensing platform using specific anti-diuron antibodies (Figure 1). The use of core magnetic nanoparticles offers rapid immunocomplex formation on magneto-microtitre plates and their further electrochemical bursting into a large number of Fe2+ ions presented ultra-high sensitivity for diuron detection on SPE. The novel immune-detection platform demonstrated excellent potential applicability in rapid and sensitive screening of environmental pollutants or toxins in samples.
Results
High resolution transmission electron microscopy (HR-TEM) was done to characterize the surface morphology and elemental mapping of synthesized nanobioprobes (Figure 2). Figure 2a shows the morphology of the synthesized Au/Fe nanoparticles with an average dia of ~30 nm. In the micrographs, the nanoparticles after coating with gold appear much darker than the Fe3O4 nanoparticles because gold is more electron dense than iron oxide. The line mapping of the selected nanoparticles indicates the inclusion of Fe core and Au shell as single Au/Fe nanostructure. The relative ratios of Au:Fe in the selected nanoparticle was found to be nearly 11:1 as depicted in figure 2b and has also been concluded by atomic absorption spectrometry (see supplementary information S1). Further, the elemental compositions of the selected area scan as determined by the energy dispersive X-ray spectroscopy (EDX) (Fig. 2c) demonstrates Au LR, Au Lâ, Fe KR and Fe Kâ lines at 9.8 keV, 11.6 keV, 6.4 keV and 7.0 keV respectively. Similarly, whole area mapping analysis shows Fe NPs with pink dots (Fig. 2e) and Au NPs in red dots (Fig. 2f) and their merging (2d) exemplifies the formation of mosaic type Au/Fe nanoparticles.
(a) TEM micrograph of Au/Fe nanoparticles.(b) The line map curve showing the ratio of Au:Fe in a single selected nanoparticle (c) EDX spectra of the whole scan area (d) The whole area mapping analysis of nanoparticles in dark field showing the distribution of Fe and Au in the synthesized nanoparticles. In (e) and (f), pink and red dots represent Fe and Au respectively.
The Au/Fe nanoparticles were conjugated with specific anti-diuron antibodies and the synthesized nanobioprobes were characterized by TEM analysis after negative staining using phosphotungstic acid (Fig. 3 a&b). The micrographs revealed that the Au/Fe nanoparticles appear to be surrounded by transparent material with a dark background clearly indicating antibodies tagging around them. Dynamic light scattering (DLS) data demonstrated the binding of antibody on Au/Fe by showing an increase (~10 nm) in size of nanobioprobes with respect to native Au/Fe nanostructures (inset of Fig. 3 a&b). Atomic force microscopy (AFM) further confirms the Au/Fe nanostructures as single colloids that display homogeneous lateral and vertical dimensions, with no evidence of aggregates even after antibody immobilization (see supplementary information, S2). Superconducting quantum interference device (SQUID) analysis demonstrated the magnetic properties of synthesized Au/Fe nanoparticles and their subsequent functionalization with specific anti-diuron antibodies. The saturation magnetization values for the uncoated and gold-coated Au/Fe nanoparticles were determined at 300 K and found to be 49.5 and 46 emu g−1 respectively (Fig. 3c). Increasing coating layers increase the amount of non magnetic substance (gold) on iron oxide between the nanoparticles leading to decrease of saturated magnetization19. Further decrease in magnetization was observed on immobilizing the antibody on the nanoparticles as the inter particle separation increases thereby reducing the magnetic dipole-dipole interactions indicating the coverage of immobilized antibody on the nanoparticles surface.
The, as prepared, Ab-Au/Fe nanobioprobes were employed for their potential application in bio-assay development. For this, rGO/CNT nanohybrid system was developed where synergistic properties of rGO and CNTs were used for sensitive detection of the analyte of interest. GO was synthesized by the oxidation of exfoliated graphite using modified Hummer's method from graphite powders as reported in the literature7. The use of high aspect ratio multiwalled CNTs promoted the electrochemical conversion of GO to reduced GO. For the confirmation of efficient electrochemical reduction of GO, three successive reductive scans were carried out in PBS, leading to formation of the nanocomposite on the SPE surface (see supplementary information, S3). From the voltammograms, it was confirmed that the GO in rGO/CNT nanocomposite was reduced during 1st scan itself. The 2nd and 3rd scan did not lead to a significant change in the reductive peak. The corresponding cyclic voltammogram (CV) scans recorded for the redox of small ion (Fe2+) for rGO/CNT showed maximum current signal for anodic and cathodic peak currents for the first reductive scan and consecutive reductive scans did not lead to any enhancement of peak current (Fig. 4a). However, GO and CNTs dropcasted individually on separate electrodes and further reduced electrochemically could not yield comparable enhancements in terms of anodic/cathodic current response as shown in Fig. 4a. The scan rate response of the prepared nanocomposite modified SPE surface was also carried out at different scan rates (25–200 mV/s at an interval of 25 mV/s) as depicted in figure 4b. The anodic potential shifts more towards the positive potential and the cathodic peak potential shifts in the reverse direction as we move towards a higher scan rate. The anodic and cathodic peak currents increase linearly with increase in scan rates indicating the process is under diffusion control20. Besides this, the redox peak currents show linear behavior with the square root of the scan rate, ν1/2 (inset in Figure 4 b), revealing it as a diffusion controlled electron-transfer process. The diffusion coeffcient value (D) of the redox species estimated from the slope of Ip versus ν1/2 plot using the Randel Sevcik equation:

Where Ip is the peak current, n is electron stoichiometry, A is electrode area (7.065 mm2), D is diffusion coeffcient, C is the concentration of the redox species (2.5 mM ferrocyanide in mol/cm3 and ν is scan rate. The D value for bare electrode was calculated to be 9.728 x 10−5 cm2/s. With this, the electro-active area of nanocomposite modified electrodes was estimated to be ~18.8 mm2 which resulted a significant increase in signal response in the developed electrochemical assay.
Raman spectroscopy was used to investigate the structural aspects of rGO/CNT modification on SPE. The experimental data was fitted using Microcal Origin 6.1 in order to elucidate the peak position and full width of half-maxima (FWHM) of D, G and 2D bands (Fig. 5a). The different peak positions of rGO/CNT depict a shift at higher wave number compared to CNT, which is shown in Fig. 5b. The changes in the crystallinity analyzed by comparing the calculated FWHM values of all the bands are observed in the spectrum. The FWHM of D band for CNT and GO was found to be ~114 and 149 cm−1 respectively, whereas for rGO/CNT it was enhanced up to 154 cm−1. This enhancement indicates the increase in disorderness due to the intercalation GO and CNT as compared to their parent molecules (GO and CNT). Raman spectra interpretations are supported by HR-TEM micrograph of the rGO/CNT nanocomposite (inset of Fig. 4a) displaying a view of CNT bundles attached to GO layer.
The formation of rGO/CNT nanocomposite on the working electrode of SPE increased the surface area enabling it to capture a large amount of metal ions liberated from Au/Fe nanoparticles after electrochemical conversion in the developed magneto-electrochemical immunoassay (MEI). The assay was carried out by coating the ELISA plates with hapten-protein (DCPU–BSA) conjugate. Optimum dilution of synthesized Ab-Au/Fe nanobioprobes pre-mixed with different concentrations of standard samples were used and incubated on the plates. The quick formation of nanobioprobe mediated immunocomplex on the plates was assisted by the application of magnetic field using bar magnet. The plates were washed and acid dissolved for the desorption of nanoparticles by using a mild acid followed by partial neutralization to retain pH 5.2. The intact magnetic core remain unaffected as mild weak acids are not capable of forming complexes with Fe in order to break the Fe-O bonds in the nanoparticles21. However, a reductive electrochemical scan is capable of liberating the Fe2+ ions from the magnetic nanoparticles22. The Fe2O3 nanoparticles were converted into metal ions (Fe2+) by applying a potential sweep between 0 to −1.6 V vs. Ag/AgCl. A characteristic broad reductive peak appeared at −0.75 V as shown in inset of figure S4 (see supplementary information, S4). Thus liberated reduced metal ions (Fe2+) on rGO/CNT modified SPE were monitored by differential pulse voltammetry (DPV). The competitive inhibition curve for diuron and other herbicides prepared at different concentrations in double distilled water ranging from 0.01 pg/mL to 1 µg/mL is shown in figure 6. The figure shows a linear decrease in current signal response with increasing concentrations of diuron (i to viii). At the highest concentration of diuron (1 µg/mL), the signal response was almost negligible demonstrating that there were no available metal ions on modified electrode surface. The percentage cross reactivity to related diuron analogues (linuron and fenuron) and other non related herbicides (atrazine and 2, 4-dichlorophenoxyacetic acid) was calculated on the basis of standard calibration curves see supplementary information Fig. S5). In the figure 6b, the standard inhibition curve for diuron is depicted. Data analysis was performed by normalizing the absorbance values using the following formula:

Where I, I0 and Iex are the relative current intensities of the sample, hapten at zero concentration and hapten at excess concentration, respectively. The limit of detection for diuron was found to be approximately 0.01 ng/mL in standard samples. The sensitivity of the assay was higher than the existing chromatography techniques (GC or HPLC) where the detection limits were 0.25 and 2.0 µg/L respectively for diuron samples23. The developed immunoassay showed excellent sensitivity and specificity demonstrating detection limit up to 0.1 pg/mL (sub-ppt) for diuron samples with high degree of reproducibility (n = 3) and good signal precision (~2%).
(a) Response curves of rGO/CNT modified SPE The signal response was measured by a differential pulse voltammetry technique at amplitude 50 mV, pulse width 0.2 s, pulse period 0.5 s.(b) Competitive inhibition response curve for diuron at different concentrations. Each point in the graph was the mean of three successive measurements (n = 3).
Discussion
The usage of Au/Fe nanoparticles offered an advantage over Fe3O4 nanoparticles by presenting an easy bioconjugation with antibodies and provides an extra stability by encapsulating the magnetic cores that prevented them from rusting. Elemental point and line mapping analysis identified and quantified the constituents of elements present in the spectrum according to their K, L, or M energy lines. The whole area mapping analysis exemplifies the formation of mosaic type Au/Fe nanoparticles. To further understand the interaction between the Au/Fe nanoparticles with specific antibodies, SQUID analysis demonstrates the alteration of magnetic properties due to various surface modifications24. The gold-coated iron nanoparticles were superparamagnetic at room temperature and the saturation magnetization values could be as high up to 60 emu/g25. The decrease in magnetization was mainly attributed due to the increase in inter particle separation after gold coating and antibodies immobilization respectively, thereby reducing magnetic dipole-dipole interactions. This substantiates the coverage of immobilized antibody on the Au/Fe nanoparticles surface.
The structural characterization of rGO/CNT modification on SPE, as characterized by Raman spectroscopy shows the D band at 1342–1353 cm−1 (due to disorder), the G band at 1575–1583 cm−1 (because of graphitic nature) and the 2D band at 2688–2716 cm−1 (second order of D band). The 2D peak indicator of strong dispersive character was detected in the Raman spectra of all carbon based nanomaterials, which is essentially the second harmonic of the D band, comes out at ~2ωD wavenumber position, when sp2-bonded carbon atoms are present26. The changes in crystallinity and orderness by measuring peak positions along with the FWHM of D, G and 2D band of the rGO/CNT nanocomposite with respect to the parent molecule supports their formation on SPE. rGO/CNT nanocomposite was formed due to the convenient integration of 1D CNT with the 2D graphene to form hierarchically structured carbon nanohybrid with enhanced electrochemical properties. The formed nanocomposite on SPE was having a larger surface area which potentially helped in enhancing the redox conversion, thus resulting in an increase in the redox current. Further, the restacking of thus formed rGO in the nanocomposite was prevented because of the intercalation of CNT which are essentially rolled graphene sheets. This is in agreement with previous studies that CNT can serve as conducting wires to promote charge transfer in electrochemical reactions27.
Metal ions derivatized electrochemical immunoassay based on electrochemical bursting of Au/Fe nanoparticles to release large number of Fe ions that are assayed on rGO/CNT modified biosensing platform. The enormous signal enhancement associated with the use of nanohybrid rGO/CNT, with its unique facile surface modification characteristics and high charge mobility, which can improve the electronic transmission rate as well as the surface area to capture a large number of Fe2+ ions on sensor surface. Liberation of the large number of (Fe2+) ions were detected by their oxidation response on rGO/CNT nanostructured electrodes, which possess the enhanced electrochemical response due to the oxygen containing groups leading to rapid electron transfer28. These oxygen containing groups present in GO sheets make it more hydrophilic and highly dispersible in water, whereas the aromatic regions of GO offer active sites to make it possible to interact with other aromatic molecules through π-π supramolecular interactions. The contact angle measurements further revealed the hydrophilic/hydrophobic character of the modified SPE surface due to the decrease in value of the contact angle after surface modification of the SPE with rGO/CNT. A large number of hydrophilic (-COOH) groups present in rGO and CNT makes the surface more hydrophilic resulting in reduced contact angle value (see supplementary information S6).
The developed sensing platform combines the advantages of GO and CNT nanohybrid offering enhanced electrochemical properties giving a synergistic effect in electroanalytical performance of the resulting electrode material along with a large number of metal ions (Fe2+) available on electrode which are detected by differential pulse voltammetry technique. This combined strategy improves the immunoassay sensitivity and thus provides a novel promising platform for environmental or clinical applications where sensitivity is a major issue. The in-house produced anti-diuron antibody was checked for its cross reactivity with diuron analogues fenuron and linuron as well as other non-related herbicides (atrazine and 2, 4-D). As observed in the electrochemical response curves the non-related herbicides show insignificant reactivity towards anti-diuron antibody because of their structural dissimilarity with parent compound diuron. A similar trend was also observed in standard ELISA for these herbicides (see supplementary information S7). The developed immunosensor showed good precision, high stability and reproducibility. High throughput screening of the large number of samples is possible because of requirement of low volume of sample in microtiter plate.
Methods
Synthesis of Au/Fe nanoparticles
The Au/Fe nanoparticles were synthesized by first preparing Fe3O4 seeds using modified co-precipitation method27 which were further oxidized to encapsulate with gold shells. Briefly, 1.28 M FeCl3 and 0.64 M FeSO4.7H2O in 1:2 ratios were dissolved in deoxygenated water and stirred vigorously in nitrogen environment. Subsequently, a solution of 1.5 M NaOH was added dropwise into the mixture followed by stirring for 40 min. A black colored precipitate of magnetite was formed and collected on a permanent magnet and washed with deionized water. The magnetite solution was diluted 200 times to have a final volume of 200 mL. Sodium citrate (155 mM) was added to the solution under constant boiling and stirring for 15 min to oxidize the magnetic particles before Au shell coating. 10 mL of gold chloride (10 mM) was injected immediately into the oxidized magnetic solution on a stirring sonicator to encapsulate the iron nanoparticles completely with gold shells. The Au/Fe nanoparticles were collected by magnetic separation followed by washings with deionised water and finally reconstituted in 0.5 mL water.
Labelling of Au/Fe nanoparticles with antibodies
The specific anti-diuron antibodies were generated by immunizing New Zealand white rabbits (4-6 months old) with well characterized hapten-protein conjugate29. Antibody solution (100 µg/mL) was prepared in phosphate buffer and was added into 1 mL Au/Fe solution under mild stirring condition. The pH of the colloidal gold solution was maintained at 7.4 by adding 0.1 M K2CO3 before adding antibody. The mixture was incubated overnight at 4°C and centrifuged at 12,000 rpm for 30 min to remove traces of unconjugated antibody. The pellet obtained was further washed twice with 10 mM Tris (pH 8.0) containing 3% BSA. The pellet was resuspended in 1 mL of phosphate buffer (pH 7.4) and stored at 4°C.
Nanobioprobe characterization
The synthesized nanoparticles and the magneto-nanobioprobes were characterized by STEM, equipped with Oxford EDS and Gatan GIF system for the atomic resolution Z-contrast imaging at sub-nanoscale resolution in point mapping and line scanning analysis. The synthesized nanoparticles and their labelling with antibody were characterized by analytical Scanning Transmission Atomic Resolution (A STAR) Electron Microscope (JEOL JEM-2100F). Further, size profiling of Au/Fe NPs and antibody tagged NPs was carried out by particle size distribution using Malvern Zetasizer dynamic light scattering (DLS) system by illuminating the colloidal gold solution with He–Ne Laser (633 nm) in a sample cell. SQUID analysis (Quantum Design, MPMS) was carried out as the characterization tools to reveal the magnetic properties after Au coating over Fe (core–shell structure) and their functionalization with specific anti-diuron antibodies.
Synthesis and characterization of rGO/CNT nanohybrid
Reduced graphene oxide/CNT nanohybrid system was developed where synergistic properties of GO and CNTs were used for sensitive detection of the analyte of interest. Graphite oxide was synthesized by the oxidation of exfoliated graphite using modified Hummer's method from graphite powders using NaNO3, H2SO4 and KMnO4 in an ice bath as reported in the literature7. GO thin films were obtained by filtration through anodized aluminium oxide (AAO) membrane with a nominal pore size of 0.02 μm. The thin GO film can be peeled off from the AAO filter after drying. For preparing rGO/CNT nanocomposite on screen printed electrodes (CH instruments USA; TE 100), a suspension (1:1) of multiwalled CNTs and GO was first prepared by dissolving in water followed by sonication (1h, 96% power). 5 µL of the suspension was drop-casted on the working area of SPEs followed by incubation in vacuum oven for 1 h at 60°C. A potential reductive scan from 0 to −1.5 V with the scan rate 0.1 V/s was applied for the electrochemical conversion of rGO/CNT nanocomposites on SPE. The thus formed nanohybrid was characterized by TEM and Raman spectroscopy. Contact angle measurements were carried out using the Sessile drop method (DSA 100, DSA/V 1.9, Kruss Gmbh Hamburg) (see supplementary information, S6). For characterization, sample was prepared electrochemically on ITO films by applying the potential between 0 to −1.5 V. The samples were scraped off from the surface. Raman scans were performed using Renishaw Invia setup attached with 785-HP-NIR laser (1.58 eV). The optical microscope imaging was used to locate randomly the selected area on SPE. The spectra of first order Raman scattering (D and G peaks) were observed around 1350 cm−1 and 1600 cm−1 respectively.
Magneto-electrochemical immunoassay
Magneto-electrochemical immunoassay (MEI) was carried out by first coating the microtiter ELISA plates with 100 µL of hapten-protein conjugate (10 µg/mL) prepared in carbonate buffer (20 mM, pH 9.6). The plates were incubated overnight at 4°C and subsequently washed with PBS (50 mM, pH 7.2). Unbound sites of the plates were blocked with 10% defatted skimmed milk in PB for 1 h at 37°C. After incubation, the plates were added with the mixture of the optimized concentration of Ab-Au/Fe nanobioprobes (1:5) and varying concentrations of diuron (0.01 pg/mL to 1 μg/mL) 50 µL each were mixed and incubated for 20 minutes at RT. A strong magnet was put beneath the plate to speed up the formation of immunocomplex followed by its separation and subsequent washing with phosphate buffer. For the dissolution of bound immobilized antibody complex, 0.1N HCl was used followed by partial neutralization with 0.1 N NaOH to retain pH ~5.2. The solution (50 µL) was transferred to rGO/CNT modified SPE surface. After reductive scan (0 to −1.5 V), differential pulse voltammetry (DPV) was carried out using an electrochemical workstation (CH Instruments, 600D). A competitive inhibition immunoassay format was developed by coating the ELISA plates with DCPU–BSA conjugate. Optimum dilution of synthesized Ab-Au/Fe nanobioprobes pre-mixed with different concentrations of standard diuron samples were used and incubated on the plates. Nanobioprobe mediated immunocomplex formed on the plates were washed and acid dissolved for the desorption of nanoparticles from the immobilized antibody by using a mild acid (1 N HCl) followed by partial neutralization with 1N NaOH.
References
Yang, W., Ratinac, K. R., Ringer, S. P., Thordarson, P., Gooding, J. J. & Braet, F. Carbon nanomaterials in biosensors: should you use nanotubes or graphene? Angew. Chem. Int. Ed. Engl. 49, 2114–38 (2010).
Shao, Y., Wang, J., Engelhard, M., Wang, C. & Lin, Y. Facile and controllable electrochemical reduction of graphene oxide and its applications. J. Mat. Chem. 20, 743–748 (2010).
Wu, X. M. et al. Electrochemical approach for detection of extracellular oxygen released from erythrocytes based on graphene film integrated with laccase and 2, 2-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid). Anal. Chem. 82, 3588–3596 (2010).
Zhang, Y., Sun, X., Zhu, L., Shen, H. & Jia, N. Electrochemical sensing based on graphene oxide/Prussian blue hybrid film modified electrode. Electrochimica Acta 56, 1239–1245 (2011).
Dong, X., Huang, W. & Chen, P. In Situ Synthesis of Reduced Graphene Oxide and Gold Nanocomposites for Nanoelectronics and Biosensing. Nanoscale Research 6, 60 (2011).
Cote, L. J., Kim, F. & Huang, J. X. Langmuir−Blodgett Assembly of Graphite Oxide Single Layers. J. Am. Chem. Soc. 131, 1043–1049 (2009).
Salzmann, C. G., Llewellyn, S. A., Tobias, G., Ward, M. A. H., Huh, Y. & Green, M. L. H. The Role of Carboxylated Carbonaceous Fragments in the Functionalization and Spectroscopy of a Single-Walled Carbon-Nanotube Material. Adv. Mater. 19, 883–887 (2007).
Kim, J., Tung, V. C. & Huang, J. Water Processable Graphene Oxide:Single Walled Carbon Nanotube Composite as Anode Modifier for Polymer Solar Cells. Adv. Energy Mater. 1, 1052–1057 (2011).
Tung, V. C. et al. Low-temperature solution processing of graphene-carbon nanotube hybrid materials for high-performance transparent conductors. Nano Lett 9, 1949–1955 (2009).
Dimitrakakis, G. K., Tylianakis, E. & Froudakis. G. E. . Pillared Graphene: A New 3-D Network Nanostructure for Enhanced Hydrogen Storage. Nano Lett 8, 3166–3170 (2008).
Qiu, L., Yang, X., Gou, X., Yang, W., Ma, Z. F., Wallace, G. G. & Li, D. Dispersing carbon nanotubes with graphene oxide in water and synergistic effects between graphene derivatives. Chem. Eur. J. 16, 10653–10658 (2010).
Chen, M., Yamamuro, S., Farrell, D. & Majetich, S.A. Gold-coated iron nanoparticles for biomedical applications. J. Appl. Phys. 93, 7551–7553 (2003).
Ban, Z., Barnaov, Y. A., Li, F., Golup, V. O. & O'Conner, C. J. The synthesis of core–shell iron@gold nanoparticles and their characterization J. Mater. Chem. 15, 4660–4662 (2005).
Cho, S. J., Kauzlarich, S. M., Olamit, J., Liu, K., Grandjean, F., Rebbouh, L., Long, G. J. Characterization and magnetic properties of core–shell structured Fe–Au nanoparticles. J. Appl. Phys. 95, 6803–6806 (2004).
Wang, J., Liu, G., Wu, H. & Lin, Y. Quantum-Dot-Based Electrochemical Immunoassay for High-Throughput Screening of the Prostate-Specific Antigen. Small 4, 82–86 (2008).
Chu, X., Fu, X., Chen, K., Shen, G. L. & Yu, R. Q. An electrochemical stripping metallo immunoassay based on silver-enhanced gold nanoparticle label. Biosens. and Bioelectron. 20, 1805–1812 (2005).
Chen, L. Y., Chen, C. L., Li, R. N., Li, Y. & Liu, S. Q. CdTe quantum dot functionalized silica nanosphere labels for ultrasensitive detection of biomarker. Chem. Commun. 2670–2672 (2009).
Nangia, Y., Bhalla, V., Kumar, B. & Suri, C. R. Electrochemical stripping voltammetry of gold ions for development of ultra-sensitive immunoassay for chlorsulfuron. Electrochem. Comm. 14, 51–54 (2012).
Gupta, A. K. & Gupta, M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 26, 3995–4021 (2005).
Srivastava, R. K., et al. Functionalized Multilayered Graphene Platform for Urea Sensor. ACS Nano 1, 168–175 (2012).
Sidhu, P. S., Gilkes, R. J., Cornell, R. M., Posner, A. M. & Quirk, J. P. Dissolution of iron oxides and oxyhydroxides in hydrochloride and perchloric acids. Clays and Clay Minerals 29, 269–276 (1981).
Mckenzie, K. J. & Marken, F. Direct electrochemistry of nanoparticulate Fe2O3 in aqueous solution and adsorbed onto tin-doped indium oxide. Pure Appl. Chem. 73, 1885–1894 (2001).
Wong, J. W. C., Fang, M., Zhao, Z. & Xing, B. Effect of surfactants on solubilisation and degradation of phenanthrene under thermophilic conditions. J. Environ. Qual. 33, 2015–2025 (2004).
Xu, C. et al. Au-Fe3O4 dumbbell nanoparticles as dual-functional probes. Angew Chem. Int. Ed 47, 173–176 (2008).
Wang, L. & Gan, X. X. Antibody-functionalized magnetic nanoparticles for electrochemical immunoassay of α-1-fetoprotein in human serum. Microchim. Acta 164, 231–237 (2009).
Srivastava, R. K., Srivastava, S., Narayanan, T. N., Mahlotra, B. D., Vajtai, R., Ajayan, P. M., Srivastava, A. Functionalized Multilayered Graphene Platform for Urea Sensor. ACS Nano 6(1), 168–175 (2012).
Gooding, J. J. et al. Protein Electrochemistry using Aligned Carbon Nanotube Arrays. J. Am. Chem. Soc. 125, 9006–9007 (2003).
Huang, C., Jiang, J., Muangphat, C., Sun, X. & Hao, Y. Trapping Iron Oxide into Hollow Gold Nanoparticles. Nanoscale Res. Lett. 6, 43 (2011).
Sharma, P. & Suri, C. R. Biotransformation and biomonitoring of phenylurea herbicide diuron. Bioresource Tech 102, 3119–3125 (2011).
Acknowledgements
This work was supported by Indo-Russia joint research grant ILTP (A- 1.29). The authors greatly acknowledge Prof. Jiaxing Huang and Laura Cote, NWU, IL for valuable suggestions and discussions on GO synthesis.
Author information
Authors and Affiliations
Contributions
CRS originated the proposed strategy and supervised the experimental works along with VD and GS. PS and VB performed all the experiments with the help of ESP. J-Wu did STEM imaging with point mapping and line scanning analysis of magneto-nanobioprobes.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplementary Infornation
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Sharma, P., Bhalla, V., Dravid, V. et al. Enhancing electrochemical detection on graphene oxide-CNT nanostructured electrodes using magneto-nanobioprobes. Sci Rep 2, 877 (2012). https://doi.org/10.1038/srep00877
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep00877
This article is cited by
-
Socio-economic impacts of solar energy technologies for sustainable green energy: a review
Environment, Development and Sustainability (2023)
-
Overcoming humidity-induced swelling of graphene oxide-based hydrogen membranes using charge-compensating nanodiamonds
Nature Energy (2021)
-
Electroconductive performance of polypyrrole/reduced graphene oxide/carbon nanotube composites synthesized via in situ oxidative polymerization
Journal of Materials Science (2019)
-
A comparative Study of Aptasensor Vs Immunosensor for Label-Free PSA Cancer Detection on GQDs-AuNRs Modified Screen-Printed Electrodes
Scientific Reports (2018)
-
Label-free Detection of Influenza Viruses using a Reduced Graphene Oxide-based Electrochemical Immunosensor Integrated with a Microfluidic Platform
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.