Abstract
Correlated evolution of traits can act synergistically to facilitate organism function. But, what happens when constraints exist on the evolvability of some traits, but not others? The orb web was a key innovation in the origin of >12,000 species of spiders. Orb evolution hinged upon the origin of novel spinning behaviors and innovations in silk material properties. In particular, a new major ampullate spidroin protein (MaSp2) increased silk extensibility and toughness, playing a critical role in how orb webs stop flying insects. Here, we show convergence between pseudo-orb-weaving Fecenia and true orb spiders. As in the origin of true orbs, Fecenia dragline silk improved significantly compared to relatives. But, Fecenia silk lacks the high compliance and extensibility found in true orb spiders, likely due in part to the absence of MaSp2. Our results suggest how constraints limit convergent evolution and provide insight into the evolution of nature's toughest fibers.
Similar content being viewed by others
Introduction
Much of Darwin's strongest support for the theory of natural selection came from observations of convergent evolution1,2. When organismal function is determined by interactions among different suites of traits, such as morphology and behavior influencing feeding in stickleback fish or locomotion in Anolis lizards, then deterministic evolution should extend to those interactions3,4. However, natural selection only acts upon existing genetic variation, so that it is important to ask “what happens when genetic constraints may resist that facilitation”?
The orb web was a key innovation in the origin of more than 12,000 species of spiders in Orbiculariae5,6,7 – the orb-weaving spiders and descendant taxa such as cobweb spiders. The success of the orb web hinged upon the evolution of two discrete suites of phenotypic traits – novel web spinning behaviors and innovations in the material properties of silks8. Aerial orb webs are built by first constructing discrete frames of dragline silk threads and then depositing adhesive silks. The individual behaviors involved in spinning orb webs are well documented9 and suggest no a priori constraint preventing other taxa from evolving similar web architectures. Yet, substantial phylogenetic evidence argues that orb webs evolved only once8,9,10,11. While famous for their gluey adhesive silks, orb webs depend upon a network of dragline silk for support and for the primary work of dissipating the kinetic energy of flying insect prey12,13. All araneomorph spiders produce dragline silk using major ampullate spidroin 1 (MaSp1) proteins14 that contain varying amounts of crystalline domains. These domains are determined in part by the prevalence of poly-alanine and glycine-alanine repeats, which interlock the proteins, thereby strengthening and stiffening the silk15,16. However, spiders in the orbicularian subclade, the “true” orb spiders, produce dragline silk that is notably tougher, more compliant and more extensible than other clades17. These changes are due in part to the origin of a novel protein, MaSp218. Proline in the MaSp2 disrupts crystalline intermolecular structures, thereby increasing the compliance and extensibility of the silk19. Major ampullate silk spun by orbicularian spiders is initially stiff, but then extends greatly under prey impacts, allowing orb webs to arrest prey in midair without the insects ricocheting out of the webs12,13. The resulting fibers are some of the toughest known biological materials20,21, with great biomimetic potential22, but which are also exceedingly difficult to replicate in the laboratory23.
A little known spider genus, Fecenia, constructs a web, termed a “pseudo-orb”, that is spectacularly similar to the true orb webs spun by Orbiculariae (Fig. 1). However, Fecenia's web is considered an example of convergent evolution to true orb webs because of the morphological similarity of Fecenia to retrolateral tibial apophysis (RTA) clade spiders24,25 – a group that includes wolf spiders, crab spiders and jumping spiders as well as many other taxa that often do not construct prey capture webs. If coevolution of complex suites of traits, such as web spinning behaviors and silk biomaterial properties, occurred during the origin of orb webs then the “convergence” hypothesis predicts that Fecenia should produce silk with material properties more like that of true orb spiders than its phylogenetic relatives. Fecenia silk should be particularly extensible, compliant and tough. However, all investigations to date of dragline silk in RTA clade show that RTA clade spiders lack the MaSp2 that is critical for shaping the functional properties of orb spider dragline silk26,27. The incorporation of MaSp2 into dragline silk is associated with a dramatic increase in the plasticity of dragline silk properties, particularly its overall extensibility and stiffness after yield28. The end result is that spiders lacking MaSp2 produce dragline silk that occupies only one third of the total performance space defined by interspecific variation in silk properties among Orbiculariae28. Thus, Fecenia dragline silk lacks much of the potential variation in mechanical performance that natural selection could act upon to evolve the high extensibility and hence also toughness, that underlies orb web function. Here, we first use molecular data to test the convergent evolution hypothesis (see also29). We then characterize the material properties of Fecenia dragline silk and place them in a comparative framework with orbicularian, RTA clade and outgroup species of spiders.
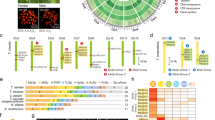
Comparison of pseudo- and true orb web architectures.Fecenia constructs pseudo orb webs that are remarkably similar in architecture to the true orb webs spun by orbicularian spiders such as Nephila.
Both types of webs consist of radiating support threads spun from major ampullate (MA) silk and overlaid spiral-like arrays of adhesive capture silk. The supporting silk is the focus here because of its dominant role in dissipating prey energy12,13.
Results
Our analysis clearly shows the pseudo-orb web's independent origin from true orbs. The phylogenetic analysis recovered a monophyletic Orbiculariae sister to a monophyletic RTA clade (Fig. 2), together forming the morphologically recognized “canoe tapetum” clade30. Fecenia nests deep within the RTA clade (Fig. 2; see also29).
Phylogenetic placement of Fecenia.
Pseudo-orb webs are nested deeply within RTA clade and clearly derived independently of the origin of true orb webs in Orbiculariae. The RTA clade, containing Fecenia, diverged from the ancestor of orb-weaving spiders ~225 mya, prior to the known origin of the MaSp2 protein. Posterior probabilities are indicated for most taxa, except where omitted from intra-familial relationships for clarity.
We found that Fecenia's major ampullate silk is both tougher and stronger than major ampullate silk produced by its RTA clade relatives (toughness and true stress respectively in Fig. 3 and Table S1; P = 0.0001 in post-hoc comparisons for both parameters). This supports the hypothesis that the evolution of aerial web-spinning behaviors is accompanied by strong selection on silk biomaterial properties in spiders. However, Fecenia's silk resembles that of its phylogenetic relatives in RTA clade in being both stiff and non-extensible (Young's modulus and true strain respectively in Fig. 3 and Table S1; P = 0.15 and P = 0.051 respectively in post-hoc comparisons).
Major ampullate silk material properties across the spider phylogeny17,49.
The performance of Fecenia silk is compared to basal taxa of spiders (Haplogynae and Austrochiloidea in black), its own clade (RTA clade in gray) and true orb spiders (Orbiculariae in red). Boxes represent ±SEM and tails represent ±SD for individual species. The bands represent the 95% confidence for mean performance of silk from species in the RTA clade (gray) and Orbiculariae (red). Fecenia shares the relatively high stiffness and low extensibility characteristic of non-orb-weaving spiders, but outperforms taxa outside Orbiculariae in strength and toughness.
Table 1 compares the amino acid composition of major ampullate silk from Fecenia webs to published compositions for a variety of Orbiculariae, including Latrodectus which spins an evolutionarily derived three-dimensional cobweb. Fecenia major ampullate silk contains (mean ± SD) 3.3 ± 0.3% proline. Fecenia's major ampullate silk also contrasts with orbicularian taxa in its relatively lower glycine content and higher content of aspargine/aspartic acid, serine, tyrosine and valine. These differences are consistent with cDNA data that show MaSp composition of orbicularian taxa to be more homogenous, dominated by ~5 common amino acids, compared to other taxa26,31.
Discussion
Fecenia's placement deep within RTA clade shows that the orb-like architectures of their webs evolved convergently with true orb webs. The origin of the orb web placed new demands on the function of silk threads. In contrast to plesiomorphic web types, orb webs target primarily flying insects that transfer tremendous amounts of kinetic energy to the webs, which must be dissipated quickly by the silk. Orb webs accomplish this feat through the very tough major ampullate silk in the radial threads of the webs. Energy is dissipated by the high damping capacity of the MA silk32. Moreover, the substantial softening of MA silk after yield, coupled with its high extensibility and strain hardening, help to localize damage during impact and ensure robust performance despite possible defects12. Thus, a suite of biomechanical traits interact with web spinning behaviors in optimizing orb web function.
If coevolution of complex suites of traits, such as web spinning behaviors and the material properties of spider silks, occurred during the origin of orb webs then Fecenia should produce silk with material properties more like that of true orb spiders than its phylogenetic relatives. We show that Fecenia's MA silk is both tougher and stronger than MA silk produced by its RTA clade relatives (Fig. 3), as predicted by the “coevolution” hypothesis. However, Fecenia's silk resembles that of its phylogenetic relatives in being both stiff and non-extensible. Compared to Orbiculariae, Fecenia MA silk likely achieves similarly high toughness through mechanisms that enhance inter-molecular bonding and hence improve stiffness and strength.
The extensibility and non-linear behavior of Orbiculariae MA silk was facilitated by the origin of MaSp2 because proline residues fold the protein backbone into molecular “nanosprings” that disrupt intermolecular bonding in the amorphous regions of the silk19,33,34. This increases the mobility of the proteins when strained and is critical for decelerating prey while also optimizing the dissipation of impact energy within discrete regions of the orbs12,13. Much of the interspecific variation among Orbiculariae in MA silk properties, such as extensibility and energy damping, correlates with differences among species in the proline composition of the silk33,35 – determined by the amino acid sequence of MaSp2 and its expression level. The low proline content in threads from the webs of Fecenia is consistent with its MA silk containing mostly MaSp1. Lack of MaSp2 is further supported because cDNA from all RTA clade taxa examined to date lack proline-rich MaSp226,27, with one possible exception - Euprosthenops expressed a MaSp2-like sequence with some isolated proline residues36. Many structures induced by amino acid sequence, such as helical glycine repeats and crystalline poly-alanine regions, clearly play critical roles in determining MA silk's remarkable properties16,20,37. Yet, proline plays a dominant role in determining the elasticity of many fibrous proteins34. Moreover, the evolution of new amino acid sequences in silk proteins is quite rare among spiders, compared to changes in expression levels of existing proteins38,39 or the effects of concerted evolution on amino acid sequences26,31,40,41. We therefore argue that Fecenia and perhaps all RTA clade taxa, face substantial phylogenetic constraint on the evolvability of their MA silk properties.
Orbiculariae is a remarkably speciose and diverse clade, while Fecenia is species-poor and restricted to habitats in South East Asia29. Why are true orb spiders so successful compared to pseudo orb-weaving Fecenia? We speculate that constraints on web function imposed by lack of MaSp2 are important, although many other factors likely help to explain Fecenia's low species richness. For instance, Fecenia uses cribellate adhesive silk in its capture threads8, a primitive and behaviorally costly type of silk utilized by few true orb spiders. However, while cribellate Orbiculariae are also species-poor5,8, the materials properties of their MA silk are clearly more like other orbicularians than like Fecenia (e.g. Hyptiotes and Uloborus in Fig. 3). Thus, the type of adhesive silk used in a web does not itself inherently constrain the evolution of MA silk properties. Behavioral differences may also contribute because the radiating threads in Fecenia webs are not as regularly spaced as in true orbs42. Fecenia also does not produce a complete capture spiral, instead spinning discrete bands of capture silk in segments that zigzag back and forth across the web surface29. Finally, the two core fibers of the capture silk are broadly separated in Fecenia29. However, orb webs are generally robust to minor perturbations in the shape of the capture spiral12 and often include substantial zig-zagging, rather than spiraling, of the capture silk43,44,45. Fecenia is also younger than Orbiculariae (Fig. 2). Yet, changes in the expression levels of MA silk genes allow even relatively young clades within Orbiculariae to show large variation in silk performance (e.g. within Argiope46 and between Argiope and its sister clade Cyrtophora33,35) so that age also appears insufficient to explain constraint on the success of pseudo orb-weaving spiders. Instead, many orb spiders depend upon rare, but very large insects for the bulk of their calories47,48. Dissipating the high kinetic energy of these insects is a formidable challenge for orb webs49. Our investigation shows that Fecenia MA silk lacks many aspects of mechanical performance that are essential for energy dissipation by true orb webs, therefore suggesting that Fecenia may have reduced ability to capture large or fast-moving prey. Furthermore, the absence of MaSp2 proteins likely limited the evolution of Fecenia's MA silk properties during the origin of the pseudo orb. Thus, our study adds to growing evidence that the evolution of innovations in silk production, such as the origin of novel silk proteins like MaSp2, plays a critical role in determining patterns of spider diversification5,6,50.
Methods
We collected Fecenia cf protensa from MacRichie, Singapore, Fecenia ochracea from New Britain, Papua New Guinea and one unidentified species from the morphologically related, confamilial genus Psechrus, collected from Pulau Ubin, Singapore and preserved them in 100% ethanol. Vouchers are deposited in the invertebrate collection of the University of Vermont. We then used standard DNA extraction, amplification and sequencing methods to sequence partial fragments for two mitochondrial (16 S, COI) and four nuclear (18 S, 28 S, H3, wingless) loci, resulting in ~4600 bp of data. We added these sequences to the preferred alignment of the orbicularian phylogenetic analysis of Blackledge et al.8 by hand to minimize change to the existing data sets, using Mesquite51. We used jModeltest v0.1.152,53 to select the best model of nucleotide substitution for each gene and position for the protein coding genes COI and H3 and then used Bayesian inference to analyze the matrices. This resulted in a total of 10 data partitions. The resulting models were: (GTR + Γ + I for COI1st, COI2nd, 16 S, 18 S, 28 S, H33rd; GTR + Γ for H31st and Wingless; HYK + Γ + I for COI3rd and JC for H32nd). We ran the MC3 (Metropolis coupled Markov chain Monte Carlo) chain in MrBayes V3.1.254 for 10,000,000 generations while sampling every 1000 generations. The first 50% of the trees were discarded as burnin, after stationarity was reached.
We also collected silk from the pseudo-radii of Fecenia webs and characterized its properties using a Nano Bionix testing system. We then constructed the broadest data set of material properties for naturally produced spider dragline silks to compare Fecenia silk to both its relatives and true orb spiders. Fecenia specimens were collected in Jiangfeng, Hainan, China and brought into the laboratory where they were housed in 20 cm diameter, 20 cm high cages with removable sides. Small leaves were placed in the cages for the construction of retreats. Spiders were then allowed to construct webs for ~2 days before silk was collected for testing. We collected silk from the pseudo-radii of Fecenia webs using standard protocols that are well-established for orb spiders49. In total, 65 samples of silk from 14 webs spun by a total of nine different spiders were obtained from regions of webs lacking capture spiral onto cardboard mounts and secured with cyanoacrylate adhesive. We then tested the 10–12 mm long samples using a Nano Bionix UTM (Agilent Technologies) at a strain rate of 0.10/s. We compared the data to two existing data sets on the material properties of MA silk from spiders. Swanson et al.55 provides the phylogenetically broadest dataset on the properties of MA silk. Most of the silk in this study was collected from naturally spun silk structures, such as draglines and webs. However, silk from the orb spider taxa were collected not from webs, but rather through forcible silking of restrained spiders, which alters the alignment of silk proteins such that the resulting fibers are unusually stiff compared to naturally spun silk in webs. Therefore, we substituted data from Sensenig et al.49 because it provides the most diverse dataset on the properties of major ampullate silk from actual radii in naturally spun orb webs. Silk in both of these studies was collected using techniques similar to our own and was tested using a Nano Bionix under similar conditions.
For each species, we used individual spiders or webs as the primary sampling unit. We then constructed 95% confidence intervals for the mean performance of silk for two major clades – the RTA clade, which is the phylogenetic “home” of Fecenia and the orbicularian clade, with whom Fecenia has converged behaviorally. We also used Tukey's HSD tests to make post-hoc comparisons of the performance of Fecenia silk to non-Orbiculariae.
Finally, we analyzed the amino acid composition of the MA silk produced by Fecenia to test for proline content. Because proline is largely confined to the MaSp2 locus in Orbiculariae26, the percentage of proline in MA silk is a strong indicator of the presence of MaSp2 and its proportion relative to MaSp135. The vapor phase hydrolization and amino acid analysis were performed using an established protocol described by Smith (2003)56. Briefly, we collected four 5–10 cm samples of frame and radial threads from Fecenia webs. The samples were transferred to the bottoms of 6×50 mm glass tubes that were vacuum-sealed into a glass vial containing 300 ul of 6 N HCl. We then hydrolyzed the samples in this vial-in-vial setup under the vapor of 6 N HCl at 112°C for 20 hr. Then, the inner sample tubes were removed, dried under vacuum and free amines derivatized with an aminoquinolyl-NHS compound (AQC) using a Waters AccQ Tag kit (Milford, MA). The derivatized amino-acids were separated by the 3.9×150 mm C18 column from the AccQ Tag kit using an ÄKTAPurifier HPLC system. Chromatographs were analyzed and peak areas integrated using Unicorn 5.20 software and compared to identically derivatized amino-acids standards run under the same conditions.
References
Darwin, C. On the origin of species by means of natural selection, or the preservation of favoured races in the struggle for life. (John murray, 1859).
Schluter, D. The ecology of adaptive radiation. (Oxford University Press, 2000).
Losos, J. B., Jackman, T. R., Larson, A., de Queiroz, K. & Rodriguez-Schettino, L. Contingency and determinism in replicated adaptive radiations of island lizards. Science 279, 2115–2118 (1998).
Schluter, D. & McPhail, J. D. Ecological character displacement and speciation in sticklebacks. Am. Nat. 140, 85–108 (1992).
Bond, J. E. & Opell, B. D. Testing adaptive radiation and key innovation hypotheses in spiders. Evolution 52, 403–414 (1998).
Blackledge, T. A., Kuntner, M. & Agnarsson, I. The form and function of spider orb webs: evolution from silk to ecosystems. Advances in Insect Physiology 41, 175–262 (2011).
Platnick, N. I. The world spider catalog, version 13. American Museum of Natural History, online at http://research.amnh.org/iz/spiders/catalog (2012).
Blackledge, T. A. et al. Reconstructing web evolution and spider diversification in the molecular era. Proc. Natl. Acad. Sci. U. S. A. 106, 5229–5234 (2009).
Coddington, J. in Spiders: webs, behavior and evolution (ed W. A. Shear) 319–363 (Stanford University Press, 1986).
Griswold, C. E., Coddington, J. A., Hormiga, G. & Scharff, N. Phylogeny of the orb-web building spiders (Araneae, Orbiculariae : Deinopoidea, Araneoidea). Zool. J. Linn. Soc. 123, 1–99 (1998).
Dimitrov, D. et al. Tangled in a sparse spider web: single origin of orb weavers and their spinning work unravelled by denser taxonomic sampling. Proc. Roy. Soc. B. 279, 1341–1350 (2012).
Cranford, S. W., Tarakanova, A., Pugno, N. M. & Buehler, M. J. Nonlinear material behaviour of spider silk yields robust webs. Nature 482, 72–76 (2012).
Sensenig, A., Lorentz, K. A., Kelly, S. P. & Blackledge, T. A. Spider orb webs rely on radial threads to absorb prey energy. J. Roy. Soc. Interface 9, 1880–1891 (2012).
Gatesy, J., Amato, G., Norell, M., DeSalle, R. & Hayashi, C. Combined support for wholesale taxic atavism in gavialine crocodylians. Syst. Biol. 52, 403–422 (2003).
Xu, M. & Lewis, R. V. Structure of a protein superfiber - spider dragline silk. Proc. Natl. Acad. Sci. U. S. A. 87, 7120–7124 (1990).
Termonia, Y. Molecular modeling of spider silk elasticity. Macromolecules 27, 7378–7381 (1994).
Swanson, B. O., Blackledge, T. A., Summers, A. P. & Hayashi, C. Y. Spider dragline silk: Correlated and mosaic evolution in high performance biological materials. Evolution 60, 2539–2551 (2006).
Hinman, M. B. & Lewis, R. V. Isolation of a clone encoding a second dragline silk fibroin - Nephila clavipes dragline silk is a two protein fiber. J. Biol. Chem. 267, 19320–19324 (1992).
Hayashi, C. Y. & Lewis, R. V. Evidence from flagelliform silk cDNA for the structural basis of elasticity and modular nature of spider silks. J. Mol. Biol. 275, 773–784 (1998).
Gosline, J. M., Guerette, P. A., Ortlepp, C. S. & Savage, K. N. The mechanical design of spider silks: From fibroin sequence to mechanical function. J. Exp. Biol. 202, 3295–3303 (1999).
Agnarsson, I., Kuntner, M. & Blackledge, T. A. Bioprospecting finds the toughest biological material: extraordinary silk from a giant riverine orb spider. PLoS One 5, e11234 (2010).
Altman, G. H. et al. Silk-based biomaterials. Biomaterials 24, 401–416 (2003).
Eisoldt, L., Smith, A. & Scheibel, T. Decoding the secrets of spider silk. Materials Today 14, 80–86 (2011).
Bayer, S. Revision of the pseudo-orbweavers of the genus Fecenia Simon, 1887 (Araneae, Psechridae), with emphasis on their pre-epigyne. Zookeys 153, 1–56 (2011).
Levi, H. W. The spider genera Psechrus and Fecenia (Araneae, Psechridae). Pacific Insects 24, 114–138 (1982).
Gatesy, J., Hayashi, C., Motriuk, D., Woods, J. & Lewis, R. Extreme diversity, conservation and convergence of spider silk fibroin sequences. Science 291, 2603–2605 (2001).
Pérez-Rigueiro, J. et al. Supercontraction of dragline silk spun by lynx spiders (Oxyopidae). Int. J. Biol. Macromol. 46, 555–557 (2010).
Blackledge, T. A. et al. Sequential origin in the high performance properties of orb spider dragline silk. Scientific Reports. 782, 10.1038/srep00782 (2012).
Agnarsson, I., Gregorič, M., Blackledge, T. A. & Kuntner, M. The phylogenetic placement of Psechridae within Entelegynae and the convergent origin of orb-like spider webs. J. Zoological Sys. Evol. Res. in press (2012).
Griswold, C. E., Coddington, J. A., Platnick, N. I. & Forster, R. R. Towards a phylogeny of entelegyne spiders (Araneae, Araneomorphae, Entelegynae). J. Arachnol. 27, 53–63 (1999).
Garb, J. E., Ayoub, N. A. & Hayashi, C. Y. Untangling spider silk evolution with spidroin terminal domains. BMC Evolutionary Biology 10, 243 (2010).
Kelly, S. P., Sensenig, A., Lorentz, K. A. & Blackledge, T. A. Damping capacity is evolutionarily conserved in the radial silk of orb-weaving spiders. Zoology 114, 233–238 (2011).
Liu, Y., Shao, Z. Z. & Vollrath, F. Elasticity of spider silks. Biomacromolecules 9, 1782–1786 (2008).
Rauscher, S., Baud, S., Miao, M., Keeley, F. W. & Pomes, R. Proline and glycine control protein self-organization into elastomeric or amyloid fibrils. Structure 14, 1667–1676 (2006).
Liu, Y., Sponner, A., Porter, D. & Vollrath, F. Proline and processing of spider silks. Biomacromolecules 9, 116–121 (2008).
Rising, A. et al. Major ampullate spidroins from Euprosthenops australis: multiplicity at protein, mRNA and gene levels. Insect Mol. Biol. 16, 551–561 (2007).
Hayashi, C. Y., Shipley, N. H. & Lewis, R. V. Hypotheses that correlate the sequence, structure and mechanical properties of spider silk proteins. Int. J. Biol. Macromol. 24, 271–275 (1999).
Craig, C. L. et al. Evidence for diet effects on the composition of silk proteins produced by spiders. Mol. Biol. Evol. 17, 1904–1913 (2000).
Tso, I. M., Wu, H. C. & Hwang, I. R. Giant wood spider Nephila pilipes alters silk protein in response to prey variation. J. Exp. Biol. 208, 1053–1061 (2005).
Hayashi, C. Y. & Lewis, R. V. Molecular architecture and evolution of a modular spider silk protein gene. Science 287, 1477–1479 (2000).
Starrett, J., Garb, J. E., Kuelbs, A., Azubuike, U. O. & Hayashi, C. Y. Early events in the evolution of spider silk genes. PLoS One 7, e38084 (2012).
Robinson, M. H. & Lubin, Y. D. Specialists and generalists - ecology and behavior of some web-building spiders from Papua New Guinea. 2. Psechrus argentatus and Fecenia sp. (Araneae, Psechridae). Pacific Insects 21, 133–164 (1979).
Kuntner, M., Kralj-Fišer, S. & Gregorič, M. Ladder webs in orb-web spiders: ontogenetic and evolutionary patterns in Nephilidae. Biol. J. Linnean Soc. 99, 849–866 (2010).
Coslovsky, M. & Zschokke, S. Asymmetry in orb-webs: An adaptation to web building costs? J. Insect Behav. 22, 29–38 (2009).
Herberstein, M. E. & Heiling, A. M. Asymmetry in spider orb webs: a result of physical constraints? Anim. Behav. 58, 1241–1246 (1999).
Elices, M. et al. Mechanical behavior of silk during the evolution of orb-web spinning spiders. Biomacromolecules 10, 1904–1910 (2009).
Blackledge, T. A. Prey capture in orb weaving spiders: Are we using the best metric? J. Arachnol. 39, 205–210 (2011).
Venner, S. & Casas, J. Spider webs designed for rare but life-saving catches. Proc. Roy. Soc. B. 272, 1587–1592 (2005).
Sensenig, A., Agnarsson, I. & Blackledge, T. A. Behavioural and biomaterial coevolution in spider orb webs. J. Evol. Biol. 23, 1839–1856 (2010).
Craig, C. L. Spider webs and silk: tracing evolution from molecules to genes to phenotypes. (Oxford University Press, 2003).
Mesquite: a modular system for evolutionary analysis.. Version 2.75 http://mesquiteproject.org v2.5 (2010).
Posada, D. jModelTest: Phylogenetic model averaging. Mol. Biol. Evol. 25, 1253–1256 (2008).
Guindon, S. & Gascuel, O. A simple, fast and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst. Biol. 52, 696–704 (2003).
Huelsenbeck, J. P. & Ronquist, R. MRBAYES: Bayesian inference of phylogenetic trees. Bioinfomatics 17, 754–755 (2001).
Swanson, B. O., Blackledge, T. A., Beltrán, J. & Hayashi, C. Y. Variation in the material properties of spider dragline silk across species. Appl. Phys. A - Mater. 82, 213–218 (2006).
Smith, B. J. in Methods in Molecular Biology Vol. 211 (Human Press, Totowa, New Jersey, 2003).
Acknowledgements
We thank S. Zschokke, M. Herberstein and an anonymous reviewer for insightful comments on earlier drafts of the manuscript. D. Piorkowski provided assistance with the collection and mechanical testing of silk. S. Bilinovich provided support for the HPLC. M. Gregorič and D. Li helped with field collection and/or DNA sequencing. S. Richards and the Conservation International RAP program sponsored and organized fieldwork in Papua. Work was supported by NSF grants no. IOS-0745379 and DEB-1050187–1050253 and by the Slovenian Research Agency grant no. J1 – 2063. Sequences were deposited in GenBank with accession numbers (KC011009-11021).
Author information
Authors and Affiliations
Contributions
T.A.B., M.K. and I.A. designed the experiment. M.K. and I.A. collected the spiders. T.A.B. collected and analyzed the silk data. M.M. and T.C.L collected and analyzed the amino acid composition data. T.A.B. sequenced DNA and I.A. conducted the phylogenetic analysis. T.A.B., M.K. and I.A. wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplementary Dataset 1
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareALike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Blackledge, T., Kuntner, M., Marhabaie, M. et al. Biomaterial evolution parallels behavioral innovation in the origin of orb-like spider webs. Sci Rep 2, 833 (2012). https://doi.org/10.1038/srep00833
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep00833
This article is cited by
-
The secondary frame in spider orb webs: the detail that makes the difference
Scientific Reports (2016)
-
Predictive modelling-based design and experiments for synthesis and spinning of bioinspired silk fibres
Nature Communications (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.