Abstract
How microgravitational space environments affect aging is not well understood. We observed that, in Caenorhabditis elegans, spaceflight suppressed the formation of transgenically expressed polyglutamine aggregates, which normally accumulate with increasing age. Moreover, the inactivation of each of seven genes that were down-regulated in space extended lifespan on the ground. These genes encode proteins that are likely related to neuronal or endocrine signaling: acetylcholine receptor, acetylcholine transporter, choline acetyltransferase, rhodopsin-like receptor, glutamate-gated chloride channel, shaker family of potassium channel and insulin-like peptide. Most of them mediated lifespan control through the key longevity-regulating transcription factors DAF-16 or SKN-1 or through dietary-restriction signaling, singly or in combination. These results suggest that aging in C. elegans is slowed through neuronal and endocrine response to space environmental cues.
Similar content being viewed by others
Introduction
Lifespan and aging rate in metazoans are influenced by environmental factors, including temperature1, oxygen2, pheromone3 and food intake4. Accordingly, perturbation of sensory perception or signaling of mechanical, chemical, or osmotic stimuli changes the lifespan of Caenorhabditis elegans5. Microgravity has been shown to induce several physiological or pathological changes including disturbance of the sense of equilibrium and loss of muscle and bone mass, mainly from the observations in spaceflight6. However, how microgravity affects organismal aging and lifespan has not been well understood. This issue is not only intriguing from a basic scientific perspective but also important from a space medical aspect, because the duration of space missions will be increasing such as those for human exploration of other planets or colonization of the moon. To obtain some basic insights into aging in space, we examined an aging marker in space-flown C. elegans and explored the involvement of the genes whose expression was changed during spaceflight, in the control of lifespan.
Results
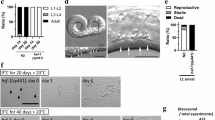
During the International C. elegans Experiment First (ICE 1st) project7, we investigated the effect of spaceflight on the formation of aggregates of a 35-glutamine repeat (Q35) in C. elegans transgenically expressing the (CAG)35-yellow fluorescent protein (YFP) gene in muscle, which normally increases with advancing age8 (Fig. 1). Q35 aggregate formation expressed as the number of aggregates per worm was found to be lower in worms flown in space from the L1 larva stage and L4/young adult stage than in matched ground control worms (Fig. 2A, B). This difference may be because of the possible changes in growth rate induced by spaceflight. However, growth of worms has been reported to be unaffected by spaceflight7,9. Moreover, the number of aggregates per body length was lower in space-flown worms than in ground control worms (Fig. 2D, E). This indicated that the spaceflight-induced suppression of Q35 aggregates was not ascribed to the spaceflight-induced changes in growth rate. Numbers of Q35 aggregates per total YFP fluorescence intensity in each worm, an indicator of Q35 expression, was also lowered by spaceflight (Fig. 2F, G). This showed that the spaceflight-induced suppression of Q35 aggregates was not due to spaceflight-induced changes in Q35 expression. These results suggest that biomarkers of aging are expressed more slowly in space-flown C. elegans in than ground control worms.
Age-dependent increases in Q35 aggregate formation on ground: simulation of procedures of spaceflight experiment.
(A) Eggs obtained by treatment of gravid-adult Q35–YFP transgenic worms with hypochlorite were allowed to hatch and maintained at L1 larva stage in CeMM for 7 days at 12°C. Then, temperature was shifted to 20°C. Worms grew to adult stage and started to lay eggs at 7 days after the temperature shift, on the other hand, the progenies produced by these worms did not grow to adult stage by 11 days after the temperature shift. Therefore, at 9 days after the temperature shift, adults seen in culture could be considered as the worms originally inoculated. (B) L4/young adult worms were incubated in CeMM with 40 μM FUdR for 7 days at 12°C and temperature was shifted to 20°C. Average numbers of Q35 aggregates/worm of more than 40 worms were plotted against time after the temperature shift to 20°C with ± S.E. Absent error bars indicate that errors fell within symbols. These figures show one of two trials, each of which gave similar results.
Spaceflight reduced Q35 aggregate formation.
Q35-YFP transgenic worms were inoculated into CeMM with 40 μM FUdR at L4/young adult stage (A, D, F) or without FUdR at egg stage (B, E, G) and incubated on ground for 5 days at 12°C. Then they were space-flown or on ground for 2 days at 12°C and for next 9 days at 20°C. In B, E and G, only adult worms in the samples containing L1, L2 and adult stage worms were measured as adult worms were considered as the worms originally inoculated (See Legend for Fig. 1). Numbers of Q35 aggregates were counted from the photographs of worms obtained by a fluorescence microscope (as in (C)) using ImageJ software (Analyze Particles). (A, B) Numbers of Q35 aggregates per worm are shown. * vs ** and # vs ##: p<0.001 (Student's t test). (C) Photographs of YFP fluorescence of the worms flown in space from L1 stage and matched ground control are shown. Bars show 100 μm. (D, E) Numbers of Q35 aggregate formation per body length are shown. The lengths of individual worms were measured using the ImageJ software. * vs ** and # vs ##: p<0.001 (Student's t test). (F, G) Ratio of Q35 aggregate formation of space-flown worms to those of ground control worms, per total body fluorescence intensity are shown. Whole-worm YFP fluorescence was quantified using the measurement and analysis software VH-HIA5 (Keyence). * vs **: p<0.05 and # vs ##: p<0.001 (Student's t test). Numbers of worms examined were as follows, A, D and F: space-flown (n = 15) and ground control worms (n = 47); B, E and G: space-flown (n = 138) and ground control worms (n = 367). Data are expressed as the mean ± S.E.
Further, these findings led us to propose a working hypothesis that the space environment changes the expression of genes involved in the control of aging. To identify the possible longevity-control genes, we first used the data from a DNA microarray experiment conducted to examine changes in gene expression in response to spaceflight10 (The data set was deposited in the Gene Expression Omnibus (GEO) database (accession number: GSE36358) and WormBase (www.wormbase.org.)), which showed that the expression of 48 genes increased by more than two-fold and that of 199 genes decreased to less than half in the spaceflight conditions relative to the ground control11. Among these genes, we noticed that eleven genes likely related to neuronal or endocrine signaling were down-regulated in space-flown worms. We confirmed these observations and quantitatively evaluated the extent of decreased mRNA expression using real-time RT-PCR (Fig. 3). Second, we examined the effects of the inactivation of these eleven genes by loss- or reduction-of-function mutations and/or feeding RNA interference (RNAi) on the lifespan under ground laboratory conditions. We found that the inactivation of each of seven genes among these eleven genes extended lifespan on NGM agar covered with killed or live bacteria as food (Fig. 4A, Supplementary Tables S1 and S2 online). These included gar-3, acetylcholine receptor; unc-17, acetylcholine transporter; cha-1, choline acetyltransferase; F57A8.4, rhodopsin-like G-protein coupled receptor (GPCR); glc-4, glutamate-gated chloride channel; shk-1, shaker family of potassium channel; and ins-35, insulin-like peptide. We also ascertained whether these lifespan extensions could be seen in the liquid culture medium (CeMM) that had been used to culture worms in the spaceflight experiment. The mutations in all of these genes, except unc-17(e245) whose lifespan could not be assessed because it could not grow in CeMM for presently unknown reasons, were also found to extend lifespan (Fig. 5).
The genes likely related to neuronal and endocrine signaling were down-regulated by spaceflight.
Relative gene expression levels in the mixed population of space-flown or ground control N2 worms are shown. gar-3: acetylcholine receptor, unc-17: acetylcholine transporter, cha-1: choline acetyltransferase, F57A8.4: rhodopsin-like GPCR, glc-4: glutamate-gated chloride channel, shk-1: shaker family of potassium channel and ins-35: insulin-like peptide, lgc-54: ligand-gated ion channel, glr-1: glutamate receptor, sym-5: synthetic lethal with mec, sra-12: serpentine receptor. Real-time RT-PCR was performed in triplicate for one sample each from the spaceflight experiment and ground control. The mRNA levels of each gene were adjusted to that of gpd-2 (glyceraldehyde-3-phosphate dehydrogenase) mRNA, which was used as the internal standard. Bar graphs are ratios of the mRNA levels in spaceflight to those on ground (%). Data are expressed as the mean ± S.E. (n = 3). All of them were significantly different (p<0.05) compared with ground control (Student's t-test).
Lifespan of the mutants in the genes down-regulated by spaceflight.
(A) The survival curves of N2 and the mutants in the genes that were down-regulated by spaceflight, on NGM with UV-killed OP50 are shown. The percentage of live worms is plotted against adult age. Lifespans of the mutants in the seven genes were longer than those of the wild type. Day 0 corresponds to the L4 molt. Data are one of two experiments, each of which gave similar results, detailed parameters of which are indicated in Supplementary Table S1. The survival curves of N2 and the mutants treated with daf-16 RNAi (B), skn-1 RNAi (C) and eat-2 RNAi (D) from the L1 stage until death. Data are one of two experiments detailed parameters of which are indicated in Supplementary Table S2 with the data of worms treated with mock-vector RNAi bacteria as control experiments.
The survival curves of N2 and the mutants in the genes that were down-regulated by spaceflight, in liquid CeMM.
The percentage of live worms is plotted against adult age. Day 0 corresponds to the L4 molt. Mean adult lifespan ± S.E. (day), number of assayed worms and statistical significance with N2 wild type are: N2: 49.5 ± 0.7, n = 364; gar-3(gk337): 62.3 ± 1.1, n = 205, p<0.001; ins-35(ok3297): 53.5 ± 1.3, n = 159, p<0.01; glc-4(ok212): 59.0 ± 0.9, n = 286, p<0.001; shk-1(ok1581): 55.2 ± 0.8, n = 204, p<0.001; F57A8.4 (tm4341): 68.1 ± 1.2, n = 179, p<0.001 and cha-1(p1152): 70.5 ± 1.1, n = 155, p<0.001.
To further explore the mechanism of lifespan extension by the inactivation of each of these genes, we investigated whether lifespan extensions are mediated through the DAF-16/FOXO transcription factor, which is a key factor in lifespan extension by reduction of insulin/IGF-1 like signaling (IIS)12, or through SKN-1, an Nrf-like xenobiotic-response factor, which is the other key factor in lifespan extension both by reduction of IIS13 and by dietary-restriction signaling14. Lifespan extensions caused by mutations either in ins-35, glc-4, unc-17, or shk-1 were totally abolished by daf-16 RNAi inactivation, whereas mutations either in gar-3, F57A8.4, or cha-1 still lived longer than wild-type worms under daf-16 RNAi (Fig. 4B, Supplementary Table S2 online). These results suggest that INS-35, GLC-4, UNC-17 and SHK-1 control lifespan thorough IIS/DAF-16 signaling. Further, the skn-1 RNAi completely abolished the lifespan extension induced by mutations in ins-35, glc-4, and shk-1, whereas mutants in unc-17, gar-3, F57A8.4 and cha-1 still lived longer than wild-type worms under skn-1 RNAi (Fig. 4C, Supplementary Table S2 online). These results suggest that INS-35, GLC-4 and SHK-1 control lifespan through SKN-1. To determine whether these lifespan extensions are mediated through dietary-restriction signaling, we examined the lifespan under the inactivation of eat-2, which induces feeding impairment-based dietary restriction15. The eat-2 RNAi further enhanced the extension of lifespan by mutations in ins-35, glc-4, unc-17, or gar-3, whereas the eat-2 RNAi shortened the lifespan of mutants of shk-1, cha-1, or F57A8.4 (Fig. 4D and Supplementary Table S2 online). These results suggest that SHK-1, CHA-1 and the F57A8.4 protein share a common lifespan control mechanism with dietary-restriction signaling.
In order to explore the involvement of these seven genes in the suppression of Q35 aggregate formation during spaceflight, we examined the effect of RNAi inactivation of some of these genes on Q35 aggregation. RNAi inactivation of gar-3, cha-1, and shk-1 reduced Q35 aggregation (Fig. 6), suggesting that GAR-3, CHA-1 and SHK-1 control Q35 aggregate formation.
The effect of RNAi of the genes on Q35 aggregate formation.
The feeding RNAi of the designated genes against Q35-YFP transgenic worms was conducted as described in Methods for RNAi. L3 larval worms were placed on RNAi-expressing or mock-vector control bacteria. Worms were incubated for 2 days at 20°C until they became gravid adults. The gravid adults were transferred to fresh RNAi-expressing bacteria lawns and allowed to lay eggs for 2 hours and removed and eggs were allowed to grow for subsequent assays. Aggregates of Q35-YFP were counted at 7 days after the treatment was started. Numbers of Q35 aggregates per worm are shown. Data are expressed as the mean ± S.E. * vs **: p<0.001 (Student's t test).
We investigated whether these seven genes relate to the formation of dauer larvae, a long-lived growth arrest state under harsh environmental conditions. Mutation and RNAi of ins-35 and mutation of shk-1 were found to enhance pheromone-induced dauer formation, whereas mutations in glc-4, unc-17, or F57A8.4 suppressed it (Fig. 7A, B). These results suggest that INS-35 and SHK-1 may be related to dauer-associated life maintenance. Alternatively, some of these five genes may be involved in sensory perception or signaling related to dauer induction.
Pheromone-induced dauer formation in the mutants.
Dauer formation of N2 and mutant worms (A) and RNAi fed rrf-3 worms (B) was scored after 3 days incubation with 1% crude pheromone extract at 25°C from the egg stage. The relative percentages of dauer arrest in the mutants compared with N2 (A) and in the RNAi fed rrf-3 worms compared with vector RNAi fed rrf-3 worms (B) are shown. Data are expressed as mean ± S.E. * vs ** and # vs ##: p<0.001 (Student's t test). Percentages of dauer larvae (mean ± S.E. n = number of worms examined): N2 (19.9 ± 1.6%, n = 367), gar-3(gk337) (16.9 ± 2.6%, n = 418), ins-35(ok3297) (71.5 ± 3.9%, n = 216), glc-4(ok212) (8.7 ± 2.0%, n = 102), unc-17(e245) (1.3 ± 1.1%, n = 171), shk-1(ok1581) (36.0 ± 3.1%, n = 306), F57A8.4(tm4341) (2.8 ± 1.4%, n = 170), vector RNAi (16.2 ± 2.3%, n = 215) and ins-35 RNAi (66.1 ± 3.1%, n = 176). These data are from one of two or three trials, each of which gave similar results. cha-1(pr1152) worms did not grow beyond the L2 stage under this condition.
Discussion
We identified seven genes, which were down-regulated in space and whose inactivation extended lifespan under ground conditions. These genes encode proteins that are conserved across animal phylogeny and that are likely related to neuronal or endocrine signaling. How do these genes play a role in longevity control? One possibility is that they are involved in longevity control through sensory perception or signaling of space environmental cues. Mutations that cause defects in sensory perception and signaling systems, related to mechanical, chemical, thermal and osmotic stimuli and pheromones, have been reported to extend the lifespan of C. elegans5. SHK-116 and GLC-417 are expressed in sensory neurons and UNC-17 in interneurons in sensory signaling18. F57A8.4 encodes a homolog of rhodopsin that is known to sense light19, suggesting that it may be involved in sensing stimuli in space environments. Pheromone-induced dauer formation was inhibited by the inactivation of unc-17, glc-4, or F57A8.4 and enhanced by that of ins-35 or shk-1 (Fig. 7A, B), suggesting that some of these genes are involved in lifespan control through sensory perception or signaling processes overlapping with dauer-switching processes. Alternatively, it may be possible that the inactivation of ins-35 or shk-1 extends lifespan, as some abnormally efficient dauer formation mutants such as daf-2 display adult-lifespan extension and it has been hypothesized that these mutants ectopically express a dauer-related efficient life maintenance capability in the adult stage20. Meanwhile, ins-35 is likely involved in endocrine signaling in the control of dauer formation and lifespan, as some of the 40 insulin-like peptides participate in regulating dauer formation and lifespan in C. elegans through a cell-nonautonomous mechanism21. The inactivation of ins-35 as well as glc-4 and shk-1 extended lifespan through the DAF-16 transcription factor (Fig. 4B). Reduction in IIS is known to extend lifespan through DAF-1612 similar to some sensory signaling mutants5. Together with the observation that the space environments and IIS reduction shared some common features in terms of their effects on gene expression22 and that a hypergravity environment induces DAF-16 localization to nuclei we suggest that DAF-16 also mediates responses to altered gravity levels (the hypergravity experiment used Escherichia coli as food instead of CeMM23). On the other hand, the lifespan extension by the inactivation of glc-4, ins-35, shk-1, or cha-1 required SKN-1 (Fig. 4C). SKN-1 in sensory neurons was reported to mediate lifespan determination during dietary-restriction signaling14, although SKN-1 in intestine also plays a role in lifespan determination through IIS13.
A second possibility is that the genes in the present study are involved in longevity control through motor neuron-muscle signaling. SHK-1 functions in muscle24 and is expressed in neurons16. CHA-1 and UNC-17, which are expressed in motor neurons25 and GAR-3, which is expressed in motor neurons and muscle26, play roles in acetylcholine transmission in motor neuron-muscle signaling. cha-1 and unc-17 are encoded by a single polycistronic cluster. They share the first noncoding exon; therefore, a common promoter termed as “cholinergic promoter” regulates the expression of both genes27. Increased acetylcholine signaling enhanced polyglutamine aggregation in post-synaptic muscle cells in C. elegans, suggesting that excess neuronal stimuli disrupt a balance in protein homeostasis in the target muscle28. Conversely, reduction in neuromuscular signaling may enhance protein structure stabilization, which has been reported to be associated with the enhancement of longevity29. Consistent with these studies, several mutations and RNAi treatments recently predicted to disrupt depolarization of the muscle membrane have been shown to alter protein homeostasis within muscle30. In fact, the inactivation of gar-3, cha-1, and shk-1 suppressed Q35 aggregation (Fig. 6), suggesting that GAR-3, CHA-1 and SHK-1 play a role in protein structure stability in muscle during motor neuron-muscle signaling. In case of cha-1, it has been shown that loss-of-function mutants evoke muscle protein degradation, which is suppressed by inhibitors of the ubiquitin-proteasome activity31. PolyQ aggregation was also reported to be regulated by genes related to the ubiquitin-proteasome system32. These neuromuscular signaling changes may affect the muscle protein metabolism observed during spaceflight10. Alternatively, the possibility remains that SHK-1, which is expressed in sensory neurons16, controls polyQ aggregation through a mechanism such as thermosensory neuronal circuit nonautonoumously modulates protein misfolding in muscle and intestine cells via Ca2+-dependent dense core vesicle neurosecretion33. The Ca2+-dependent dense core vesicle neurosecretion mechanism modulates the HSP-70 levels in muscle to affect polyQ aggregation33 and HSP-70 levels are elevated in response to spaceflight22. In either case, the suppression of Q35 aggregation observed in space might be explained in part by reduced expression of gar-3, cha-1, and/or shk-1. Given that polyglutamine aggregation is considered to be regulated by protein synthesis, degradation, or transport32, it seems likely that reduction of gar-3, cha-1, and/or shk-1 affect proteostasis in muscle; this also may partially account for the decreased expression of muscle proteins during spaceflight10.
Lifespan extension by eat-2 inactivation inducing the dietary-restriction state15 was totally abolished by the inactivation of either cha-1, shk-1 or F57A8.4 (Fig. 4D), suggesting that CHA-1, SHK-1, or F57A8.4 protein function in processes related to sensory perception of food or signaling in response to food. Alternatively, some of these may be involved in motor neuron-muscle signaling related to food intake. In support of this idea, CHA-1 was reported to participate in determining pharyngeal pumping rate to affect food intake34.
The combined results suggest that environmental factors associated with spaceflight, including microgravity, modulate neuronal or endocrine signaling to induce some of “longevity-promoting” processes, including dietary-restriction signaling, stabilization of protein structure, or dauer-related efficient life maintenance during harsh conditions (Fig. 8). However, it should be noted that whether suppression of Q35 aggregation and the expression of the seven studied genes is due to microgravity or other environmental factors such as radiation is still uncertain, because the necessary experiments under the artificial gravitational field of the earth (1 G) in space environments have not been performed. Although further investigation regarding the effects of a decrease in the expression of the present genes comparable to the level in spaceflight and a simultaneous decrease in the expression of all these genes on lifespan is required, the present findings suggest that space-flown worms age more slowly compared with ground control worms and further predict that spaceflight extends worm lifespan. Male Drosophila lived shorter on the ground after spaceflight compared to controls that maintained on the ground through their lifetimes35. Further investigation is needed to compare aging and lifespan under microgravity and 1 G in space environments.
Model for the action of longevity-control genes whose expressions are suppressed by spaceflight.
In response to space-environmental stimuli including microgravity, sensory perception, neuronal and endocrine signaling and motorneuron-muscle signaling work through changes in these gene activities. These changes, some of which activate DAF-16 and/or SKN-1 transcription factors, induce “longevity-promoting” processes including dietary-restriction signaling, stabilization of protein structure or dauer-related efficient life maintenance during harsh conditions.
Methods
Nematode strains and culture conditions
Unless otherwise stated, the C. elegans strains were maintained at 20°C on nematode growth medium (NGM) agar with E. coli OP50 as a food source, as previously described36. The N2 Bristol strain was used as the wild-type C. elegans. The mutant strains used in this study were JD31: glc-4(ok212) II, NL2099: rrf-3(pk1426) II, RB1392: shk-1(ok1581) II, PR1152: cha-1(p1152) IV, TY1652: cha-1(y226) IV, CB933: unc-17(e245) IV, F57A8.4(tm4341) V, VC670: gar-3(gk337) V, TM290 (x5 outcrossed): ins-35(ok3297) V and Q35-YFP strain.
Spaceflight experiment
The spaceflight experiment was conducted as described previously7. Briefly, eggs isolated after treatment of gravid adult worms with hypochlorite were incubated in a chemically defined liquid medium, C. elegans Maintenance Medium (CeMM)37 and allowed to hatch and grow. Approximately 70 worms at the L4 larva/young adult stage were placed into a gas-permeable culture bag7 containing 2.5 mL CeMM with 40 µM 5-fluoro-2′-deoxyuridine (FUdU, Sigma Aldrich, St. Louis, MO, USA) which was used to prevent self-fertilization. Approximately 100 hatched L1 worms were also placed in a culture bag containing 2.5 mL CeMM without FUdR. Both of them were incubated for 5 days at 12°C on the ground. After the launch, they were flown for 2 days at 12°C and on board the International Space Station for 9 days at 20°C. When samples were returned to earth, they were flash frozen (freezing time <1 min) in liquid nitrogen within 2 h of landing7. Control worms underwent the same procedures at the same time on the ground.
PolyQ aggregate measurement
To measure polyQ aggregates, a C. elegans transgenic line expressing a chimeric fusion protein of a 35-glutamine repeat (Q35) and yellow fluorescent protein (YFP) in the body wall muscle, as described previously8, was used. The Q35 aggregates of worms were counted with a fluorescence microscope system (VB-G25/S20/L10/7010; Keyence CO., Osaka, Japan). Aggregates that were more than 1 µm long were defined as discrete structures.
Real-time RT-PCR
To confirm the gene expression differences between ground control worms and space-flown worms, real-time PCR was performed. Total RNA of a mixed population of space-flown and ground control N2 worms was reverse-transcribed to cDNA using ExScript RT reagent kit (TaKaRa Bio Inc., Shiga, Japan) and iCycler thermal cycler (Bio-Rad Laboratories Inc., Hercules, CA, USA). The reaction mixture for measuring the cDNA quantity was prepared using Premix EX Taq (TaKaRa Bio) according to the manufacturer's instructions. Reaction and fluorescence monitoring were done using a Thermal Cycler Dice Real Time System (Takara Bio). The following primers were used:
gpd-2 forward, 5′-accggagtcttcaccaccatc-3′; gpd-2 reverse, 5′-acgactacgaggttacaagca-3′;
gar-3 forward, 5′-tgcacaccgatgccgacta-3′; gar-3 reverse, 5′-gcgacttcgaaaccttgctga-3′;
ins-35 forward, 5′-actgccaacgcgcactaaag-3′; ins-35 reverse, 5′-tggctctgggcagcagataag-3′;
glc-4 forward, 5′-ccgttggtgcagtgttctgt-3′; glc-4 reverse, 5′-catgactcgtccggatcctc-3′;
unc-17 forward, 5′-ccacggctatatttgctcttgga-3′; unc-17 reverse, 5′-ctaggcacccgaacgagatga-3′;
shk-1 forward, 5′-tgcgcaatgattggtgtctt-3′; shk-1 reverse, 5′-atgtcgtcgtctgcatcgtc-3′;
F57A8.4 forward, 5′-tcgcatcagacgctgtcagta-3′; F57A8.4 reverse, 5′-tccgtcacttccacaggtgag-3′;
cha-1 forward, 5′-cgtggtgtgtcttgacatgga-3′; cha-1 reverse, 5′-cgattgagcccgaactttga-3′;
lgc-54 forward, 5′-ccaatgcaaatgcagcagag-3′; lgc-54 reverse, 5′-tgtgcgagagcgtgcaaag-3′;
glr-1 forward, 5′-aggtgcagttgcacaaccag-3′; glr-1 reverse, 5′-gtttgcaggtcgatccacatta-3′;
sym-5 forward, 5′-aagccggagccagtgctagt-3′; sym-5 reverse, 5′-cctgtcgttgtatggacgtttc-3′;
sra-12 forward, 5′-gacgtcacccagcgatatttg-3′; sra-12 reverse, 5′-gaaacacaggcatatggtggag-3′.
Real-time RT-PCR was performed in triplicate for one sample each from the spaceflight experiment and ground control. The mRNA levels of each gene were adjusted to that of gpd-2 (glyceraldehyde-3-phosphate dehydrogenase), which was used as the internal standard. Flight/ground ratios of these were statistically analyzed by Student's t-test using PRISM software (GraphPad Software Inc., San Diego, CA, USA). Significance was accepted at p<0.05.
RNAi
RNAi experiments were performed according to the RNAi feeding method described previously38. E. coli HT115 strains expressing double-stranded RNA that inactivated the target genes were obtained from Open Biosystems (Huntsville, AL, USA) and Source BioScience (Nottingham, UK).
Determination of lifespan
Eggs isolated after treatment of gravid adult worms with hypochlorite were incubated in S basal at 20°C for 1 day. Lifespan was determined in three ways. First, the hatched L1 larvae were placed on NGM agar covered with UV-killed OP50. UV killing was used to avoid any toxic effects of live E. coli on C. elegans lifespan39. Second, the hatched L1 worms were placed on NGM agar covered with RNAi-expressing or vector control bacteria. In these two assay systems, worms were transferred to fresh plates every other day. Third, the hatched L1 worms were inoculated into CeMM. In every case, synchronous L4/young adult stage worms estimated as 0-day adults in the lifespan assay were treated with 40 µM FUdU to prevent self-fertilization. The number of surviving worms was monitored until death as judged by nonresponse to a mechanical stimulus. To focus on aging, we excluded worms that had become desiccated on the side of the plate after crawling off in the former two assay systems and that displayed extruded internal organs and those that died because of progeny hatching inside the uterus (matricidal death). The results of the survival assays were analyzed using the Kaplan-Meier method and significance was measured with the log-rank test using the statistical analysis package StatMate III (ATMS, Tokyo, Japan).
Dauer formation
Dauer forming ability was tested in the presence of crude pheromone extract3. Crude pheromone extract was added to a final concentration of 1% to liquid NGM that had been autoclaved and cooled to 50°C. The media were immediately dispensed into petri plates. Gravid adult hermaphrodites were allowed to lay eggs on the plates covered with UV-killed OP50 for 6 h. After removal of the adult hermaphrodites, the plates were incubated at 25°C for 3 days and scored for dauer arrest. The dauer stage was confirmed by visual inspection of the animals as the presence of dark pigment granules and constriction of the body and pharynx and the loss of pharyngeal pumping.
References
Klass, M. R. Aging in the nematode Caenorhabditis elegans: major biological and environmental factors influencing life span. Mech. Ageing Dev. 6, 413–429 (1977).
Honda, S., Ishii, N., Suzuki, K. & Matsuo, M. Oxygen-dependent perturbation of life span and aging rate in the nematode. J. Gerontol. 48, B57–61 (1993).
Kawano, K. et al. Lifespan extending activity of substances secreted by the nematode Caenorhabditis elegans that include the dauer-inducing pheromone. Biosci. Biotechnol. Biochem. 69, 2479–2481 (2005).
Masoro, E. J. Overview of caloric restriction and ageing. Mech. Ageing Dev. 126, 913–922 (2005).
Apfeld, J. & Kenyon, C. Regulation of lifespan by sensory perception in Caenorhabditis elegans. Nature 402, 804–809 (1999).
Vernikos, J. Human physiology in space. Bioessays 18, 1029–1037 (1996).
Szewczyk, N. J. et al. Description of International Caenorhabditis elegans Experiment first flight (ICE-FIRST). Adv. Space Res. 42, 1072–1079 (2008).
Morley, J. F., Brignull, H. R., Weyers, J. J. & Morimoto, R. I. The threshold for polyglutamine-expansion protein aggregation and cellular toxicity is dynamic and influenced by aging in Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 99, 10417–10422 (2002).
Oczypok, E. A. et al. Remote automated multi-generational growth and observation of an animal in low Earth orbit. J. R. Soc. Interface 9, 596–599 (2012).
Higashibata, A. et al. Decreased expression of myogenic transcription factors and myosin heavy chains in Caenorhabditis elegans muscles developed during spaceflight. J. Exp. Biol. 209, 3209–3218 (2006).
Higashitani, A. et al. Checkpoint and physiological apoptosis in germ cells proceeds normally in spaceflown Caenorhabditis elegans. Apoptosis 10, 949–954 (2005).
Kenyon, C. J. The genetics of ageing. Nature 464, 504–512 (2010).
Tullet, J. M. A. et al. Direct inhibition of the longevity promoting factor SKN-1 by Insulin-like signaling in C. elegans. Cell 132, 1025–1038 (2008).
Bishop, N. A. & Guarente, L. Two neurons mediate diet-restriction-induced longevity in C. elegans. Nature 447, 545–549 (2007).
Lakowski, B.. & Hekimi, S. The genetics of caloric restriction in Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 95, 13091–13096 (1998).
Fawcett, G. L. et al. Mutant analysis of the Shal (Kv4) voltage-gated fast transient K+ channel in Caenorhabditis elegans. J. Biol. Chem. 281, 30725–30735 (2006).
Hunt-Newbury, R. et al. High-throughput in vivo analysis of gene expression in Caenorhabditis elegans. PLoS Biol. 5, e237 (2007).
Altun-Gultekin, Z. et al. A regulatory cascade of three homeobox genes, ceh-10, ttx-3 and ceh-23, controls cell fate specification of a defined interneuron class in C. elegans. Development 128, 1951–1969 (2001).
Palczewski, K. G protein-coupled receptor rhodopsin. Annu. Rev. Biochem. 75, 743–767 (2006).
Kenyon, C., Chang, J., Gensch, E., Rudner, A. & Tabtiang, R. A C. elegans mutant that lives twice as long as wild type. Nature 366, 461–464 (1993).
Tatar, M., Bartke, A. & Antebi, A. The endocrine regulation of aging by insulin-like signals. Science 299, 1346–1351 (2003).
Selch, F. et al. Genomic response of the nematode Caenorhabditis elegans to spaceflight. Adv. Space Res. 41, 807–815 (2008).
Kim, N. et al. Gravity Force Transduced by the MEC-4/MEC-10 DEG/ENaC Channel Modulates DAF-16/FoxO Activity in Caenorhabditis elegans. Genetics 177, 835–845 (2007).
Liu, P. et al. Genetic dissection of ion currents underlying all-or-none action potentials in C. elegans body-wall muscle cells. J. Physiol. 589, 101–117 (2011).
Duerr, J. S., Han, H. P., Fields, S. D. & Rand, J. B. Identification of major classes of cholinergic neurons in the nematode Caenorhabditis elegans. J. Comp. Neurol. 506, 398–408 (2008).
Liu, Y., LeBoeuf, B. & Garcia, L. R. Gαq-coupled muscarinic acetylcholine receptors enhance nicotinic acetylcholine receptor signaling in Caenorhabditis elegans mating behavior. J. Neurosci. 27, 1411–1421 (2007).
Alfonso, A., Grundahl, K., McManus, J. R., Asbury, J. M. & Rand, J. B. Alternative splicing leads to two cholinergic proteins in Caenorhabditis elegans. J Mol Biol. 241, 627–630 (1994)
Garcia, S. M., Casanueva, M. O., Silva, M. C., Amaral, M. D. & Morimoto, R. I. Neuronal signaling modulates protein homeostasis in Caenorhabditis elegans post-synaptic muscle cells. Genes Dev. 21, 3006–3016 (2007).
Taylor, R. C. & Dillin, A. Aging as an event of proteostasis collapse. Cold Spring Harb. Perspect. Biol. 3, a004440 (2011).
Shephard, F., Adenle, A. A., Jacobson, L. A. & Szewczyk, N. J. Identification and functional clustering of genes regulating muscle protein degradation from amongst the known C. elegans muscle mutants. PLoS ONE. 6, e24686 (2011)
Szewczyk, N. J., Hartman, J. J., Barmada, S. J. & Jacobson, L. A. Genetic defects in acetylcholine signalling promote protein degradation in muscle cells of Caenorhabditis elegans. J Cell Sci. 113, 2003–2010 (2000).
Nollen E. A, A. et al. Genome-wide RNA interference screen identifies previously undescribed regulators of polyglutamine aggregation. Proc. Natl. Acad. Sci. USA. 101, 6403–6408 (2004).
Prahlad, V. & Morimoto, R. I. Neuronal circuitry regulates the response of Caenorhabditis elegans to misfolded proteins. Proc. Natl. Acad. Sci. USA. 108, 14204–14209 (2011).
Rand, J. B. & Russell, R. L. Choline acetyltransferase-deficient mutants of the nematode Caenorhabditis elegans. Genetics 106, 227–248 (1984).
Bengur, A. et al. Microgravity effects on Drosophila melanogaster behavior and aging. Implications of the IML-2 experiment. J. Biotech. 47, 191–201 (1996).
Brenner, S. the genetics of Caenorbabditis elegans. Genetics 77, 71–94 (1974).
Szewczyk, N. J., Kozak, E. & Conley, C. A. Chemically defined medium and Caenorhabditis elegans. BMC Biotechnol. 3, 19 (2003).
Timmons, L., Court, D. L. & Fire, A. Ingestion of bacterially expressed dsRNAs can produce specific and potent genetic interference in Caenorhabditis elegans. Gene 263, 103–112 (2001).
Garigan, D. et al. Genetic analysis of tissue aging in Caenorhabditis elegans: a role for heat-shock factor and bacterial proliferation. Genetics 161, 1101–1112 (2002).
Acknowledgements
We are grateful to the entire crew of the ICE 1st for operation in orbit. The ICE 1st was well organized with the support of the European Space Agency, the Space Research Organization of the Netherlands, the Japan Aerospace Exploration Agency, National Aeronautics and Space Administration (NASA), the Canadian Space Agency and the French Space Agency. We thank Drs. M. Viso, G. Gasset, B. Eche and K. Fukui for their kind support in the space experiment. We acknowledge NASA Ames Research Center for preparation of CeMM. We also thank Dr. R. Morimoto for kindly supplying the Q35-YFP strain. We are grateful to N. Endo for his support with respect to fluorescence microscope observation. Some strains were provided by the Caenorhabditis Genetics Center funded by the National Institutes of Health National Center for Research Resources (NCRR) and the International C. elegans Gene Knockout Consortium and National Bioresource Project for the nematode. YH was supported by a grant-in-aid from the Ministry of Education, Science and Culture of Japan and by the program “Grand-based Research Announcement for Space Utilization” promoted by the Japan Space Forum. NJS was supported by NIAMS AR054342.
Author information
Authors and Affiliations
Contributions
YH and SH conceived and designed the experiments. YH, SH, A. Higashibata, YM, YY, TK, A. Higashitani and NI carried out the experiments and analysed the data. KK and TS prepared spaceflight experiments. NJS contributed materials. MT and NJS discussed results. YH and SH wrote the main manuscript text. NJS edited and all authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplementary Tables 1 & 2
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareALike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Honda, Y., Higashibata, A., Matsunaga, Y. et al. Genes down-regulated in spaceflight are involved in the control of longevity in Caenorhabditis elegans. Sci Rep 2, 487 (2012). https://doi.org/10.1038/srep00487
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep00487
This article is cited by
-
Integrated Analysis of MRNA and MiRNA Expression Profiles in dys-1 Mutants of C. Elegans After Spaceflight and Simulated Microgravity
Microgravity Science and Technology (2023)
-
Unconscious mind activates central cardiovascular network and promotes adaptation to microgravity possibly anti-aging during 1-year-long spaceflight
Scientific Reports (2022)
-
Microfluidics-integrated spaceflight hardware for measuring muscle strength of Caenorhabditis elegans on the International Space Station
npj Microgravity (2022)
-
Astronauts well-being and possibly anti-aging improved during long-duration spaceflight
Scientific Reports (2021)
-
Anti-aging effects of long-term space missions, estimated by heart rate variability
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.