Abstract
Transient receptor potential ankyrin 1 (TRPA1) is an ion channel involved in thermosensation and nociception. TRPA1 is activated by exogenous irritants and also by oxidants formed in inflammatory reactions. However, our understanding of its role in inflammation is limited. Here, we tested the hypothesis that TRPA1 is involved in acute inflammatory edema. The TRPA1 agonist allyl isothiocyanate (AITC) induced inflammatory edema when injected intraplantarly to mice, mimicking the classical response to carrageenan. Interestingly, the TRPA1 antagonist HC-030031 and the cyclo-oxygenase (COX) inhibitor ibuprofen inhibited not only AITC but also carrageenan-induced edema. TRPA1-deficient mice displayed attenuated responses to carrageenan and AITC. Furthermore, AITC enhanced COX-2 expression in HEK293 cells transfected with human TRPA1, a response that was reversed by HC-030031. This study demonstrates a hitherto unknown role of TRPA1 in carrageenan-induced inflammatory edema. The results also strongly suggest that TRPA1 contributes, in a COX-dependent manner, to the development of acute inflammation.
Similar content being viewed by others
Introduction
The identification of transient receptor potential ankyrin 1 (TRPA1) as a chemosensor of potentially harmful electrophilic and non-electrophilic chemicals1,2,3,4 has opened up new avenues in our understanding of nociception and inflammatory pain5. The role of TRPA1 in noxious chemosensation has attracted considerable attention with regard to the development of TRPA1 antagonists in the treatment of pain and sensory hyperreactivity, e.g. in the therapy of the urinary bladder and airway diseases5,6,7.
TRPA1 is a membrane-associated cation channel which is involved in several physiological functions such as neurotransmission, cell proliferation and gene expression via Ca2+ influx and elevation of the cytosolic free Ca2+ concentration ([Ca2+]i)8,9. TRPA1 belongs to the transient receptor potential (TRP) ion channel superfamily which in mammals embraces six subfamilies and 28 distinct proteins with different functions in a variety of cells and tissues. TRPA1 was first discovered in 1999 by Jaquemar and colleagues10 and is the only member of its subfamily (TRP ankyrin) found in humans. Structurally, TRPA1 is composed of six transmembrane spanning segments with a pore domain between 5th and 6th segments. The TRPA1 intracellular N-terminus displays 14 ankyrin repeats11 within which lies the site of activation by the covalent modification of specific cysteines11,12,13.
Many of the oxidants formed in inflammatory reactions such as nitro-oleic acid14, 4-hydroxynonenal or hydrogen peroxide15 are endogenous agonists of TRPA1. Furthermore, a variety of exogenous agonists, for example allyl isothiocyanate (AITC)1, one of the pungent compounds in mustard oil, have been identified. TRPA1 antagonists have also been developed and, e.g. HC-0300316 has become a widely used experimental tool.
TRPA1 is primarily considered to be expressed in a sub-population of sensory neurons3,11,17, but recent findings suggest that it is also present in a number of other cells, including keratinocytes, endothelial cells, synoviocytes, odontoblasts and enterochromaffin cells18,19,20,21,22. Its physiological role remained obscure until the discovery that TRPA1 is present in mouse afferent nerves and could be activated by noxious cold, indicating a role in thermal nociception11. TRPA1 has been associated with other physiological functions including chemosensation, hearing and mechanical cognisance1,5. In addition, TRPA1 has been shown to mediate inflammatory23 and formalin-induced pain24, irritating effects of pungent compounds25,26 and neurogenic inflammation27,28. Also, mice treated with TRPA1 antagonists and TRPA1 knock out (KO) mice were found to develop a less severe ovalbumin-induced asthma reaction than untreated wild type (WT) mice29. Topical treatment with mustard oil has been shown to induce local edema, an effect also blunted in TRPA1 deficient mice25. However, it is still far from clear if TRPA1 has a role as a modulator of the inflammatory process.
In the present study, we investigated the possible role of TRPA1 in carrageenan-induced inflammatory paw edema which is a widely used model for investigating the acute inflammatory response and novel anti-inflammatory drugs. The results show that a substantial part of the mouse paw edema triggered by carrageenan is dependent on TRPA1. Furthermore, both carrageenan and AITC-induced edemas are to a large extent inhibited by ibuprofen. These findings highlight TRPA1 as a potential drug target for novel anti-inflammatory agents that could be a valuable alternative to cyclo-oxygenase (COX) inhibitors in the treatment of certain inflammatory conditions.
Results
Intraplantar (i.pl.) injection of carrageenan induced a substantial paw edema when measured at 3 h and 6 h following injection (Fig. 1A). The contralateral paw injected i.pl. with saline exhibited no measurable edema. Likewise, the TRPA1 agonist AITC evoked a severe edema when injected in the mouse paw (Fig. 1B).
TRPA1 agonist allyl isothiocyanate (AITC) and carrageenen (Car) induced an inflammatory paw edema, which could be prevented by pre-treatment with HC-030031 (HC) or ibuprofen (Ibu.).
HC-030031 (300 mg/kg) or ibuprofen (100 mg/kg) was injected intraperitoneally 2 h prior to intraplantar injection of carrageenan or AITC into the hind paw. The edema was measured 3 h and 6 h after intraplantar injection and compared to the basal level. The contralateral control paw injected with saline developed no measurable edema. Mean + SEM, n = 6, ***p<0.001.
To further study the role of TRPA1 in the inflammatory edema induced by carrageenan and AITC, we treated the mice with the selective TRPA1 antagonist HC-03003116. The mice received 300 mg/kg of HC-030031 intraperitoneally (i.p.) 2 h before carrageenan or AITC was injected into the paw. HC-030031 treatment prevented carrageenan-induced inflammatory edema by 48% and 40% when measured at 3 h and 6 h following the carrageenan injection, respectively (Fig. 1A). As expected, HC-030031 reduced the AITC-induced edema response by 67% and 69% at 3 h and 6 h, respectively (Fig. 1B).
Next we used TRPA1-deficient mice to confirm and extend the results obtained with pharmacological blockade of TRPA1. We found that such mice developed on average 62% and 50% less edema as compared to WT mice when measured 3 h and 6 h following the i.pl. carrageenan injection (Fig. 2A). AITC induced a negligible edema in TRPA1 deficient mice (Fig. 2B) providing additional evidence that the AITC-induced response is indeed dependent on TRPA1.
In TRPA1 knock out (KO) mice, the carrageenan-induced paw edema formation was blunted when compared to the corresponding wild type (WT) mice (A).
TRPA1-deficient mice showed almost no response to the TRPA1 agonist AITC (allyl isothiocyanate) in contrast to the WT mice (B). Carrageenan or AITC were injected intraplantarly. The edema was measured after 3 h and 6 h and compared to the basal level. The contralateral control paw injected with saline developed no measurable edema. Mean + SEM, n = 5, **p<0.01, ***p<0.001.
The possible involvement of COX-derived prostanoids in TRPA1-mediated inflammatory edema was investigated by treating the animals with ibuprofen. Ibuprofen (100 mg/kg i.p.) given 2 h before carrageenan reduced the edema response by 51% and 49%, respectively when measured at 3 h and 6 h following carrageenan injection (Fig. 1A). Likewise, the inflammatory edema evoked by AITC was also clearly inhibited by ibuprofen pre-treatment. The mean edema was about 60% less in ibuprofen treated than in vehicle treated mice at both 3 h and 6 h time points (Fig. 1B). These results indicate that prostanoids play an important role in mediating TRPA1-induced edema.
To further investigate the link between TRPA1 and COX, we transfected HEK 293 cells with human TRPA1. The COX-2 mRNA expression was up-regulated in TRPA1 transfected HEK 293 cells but not in non-transfected cells when they were exposed to AITC. HC-030031 given 30 min before AITC reduced the extent of the up-regulation of COX-2 mRNA triggered by TRPA1 stimulation (Fig. 3), supporting the concept that COX-derived prostanoids are involved in TRPA1-induced responses.
HEK 293 cells transfected with human TRPA1 showed an upregulation of cyclo-oxygenase-2 (COX-2) mRNA in response to the TRPA1 agonist allyl isothiocyanate (AITC).
The expression was suppressed when the TRPA1 antagonist HC-030031 (10 μM) was given before AITC. Both TRPA1 transfected and non-transfected (wild type, WT) cells were incubated with HC-030031 or vehicle for 30 min and thereafter AITC was added. After 6 h the incubations were terminated and COX-2 mRNA was assayed by real-time RT-PCR. COX-2 mRNA was normalized against GAPDH mRNA. Mean + SEM, n = 4, *p<0.05, **p<0.01, n.s. = non-significant.
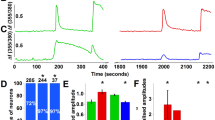
In addition, the direct activation of TRPA1 by carrageenan was investigated in HEK 293 cells expressing human TRPA1. In those cells, exposure to carrageenan (250 µg/ml) did not evoke a change in the basal calcium level, as measured by ratiometric calcium imaging, whereas subsequent addition of AITC (100 µM) always evoked robust calcium responses (Fig. 4). Higher concentrations of carrageenan could not be tested due to viscosity problems.
In calcium imaging experiments, HEK 293 cells transfected with human TRPA1 were exposed to either carrageenan or its vehicle for 60 s and subsequently to AITC (allyl isothiocyanate) for 36 s.
Whereas carrageenan or vehicle was without effect, AITC always evoked robust calcium responses as assessed by ratiometric Fura 2 imaging. Traces show the average calcium responses (A) and the bar graph (B) shows the maximum calcium responses in cells exposed to the various treatments. Mean + SEM, n = 4 (each experiment was performed in duplicate or triplicate).
Discussion
The present study confirms the effects of exogenous TRPA1 agonists on edema formation and, more interestingly, reveals a hitherto unknown role of TRPA1 in carrageenan-induced inflammatory paw edema which is a widely used model for evaluating acute inflammation and anti-inflammatory drugs. The results strongly suggest that TRPA1 has a significant role in mediating the acute inflammatory response.
Previously, TRPA1 has been shown to mediate nociceptive processes in vivo, such as mechanical and cold hyperalgesia30,31,32 and also inflammatory pain23,24. A mutation in TRPA1 resulting in hyperfunction of the ion channel was recently associated with familial episodic pain syndrome33 highlighting the significant role of TRPA1 also in human pain. Although endogenous TRPA1 agonists are produced in inflammatory reactions14, very little is known about the possible role of TRPA1 in mediating inflammatory responses in addition to pain. Experiments conducted in knock out mice revealed that TRPA1 was involved in the pathogenesis of airway hyperreactivity and inflammation during ovalbumin-induced asthma29. Exposure to a TRPA1 agonist extracted from cigarette smoke was reported to cause tracheal edema which could be reversed by local administration of a TRPA1 antagonist whereas no edema formation occurred in TRPA1 KO animals34. In the present study, we observed that activation of TRPA1 by AITC resulted in inflammatory edema and that a TRPA1-dependent mechanism was involved in the formation of the classical carrageenan-induced paw edema.
The inflammatory edema following carrageenan injection involves both neurogenic and non-neurogenic mechanisms which have been strongly associated with prostaglandin production, COX-2 up-regulation and the formation of reactive nitrogen and oxygen species as well as cytokines and other inflammatory mediators35,36,37. Many such pro-inflammatory agents can sensitize or activate TRPA1 either indirectly via G protein-coupled phospholipase C and protein kinase A pathways or by directly interacting with TRPA15. In the present study we showed that the TRPA1 antagonist HC-030031 clearly inhibited the development of carrageenan-induced edema while a direct TRPA1 agonist AITC evoked a carrageenan-like inflammatory response. To confirm our findings with the TRPA1 agonist and antagonist in acute inflammatory edema, we performed the experiments also in TRPA1 deficient animals. Indeed, the carrageenan-induced response was markedly attenuated in TRPA1 KO mice and only a minor response to AITC was detected. When compared with previous reports on the acute phase of carrageenan-induced mouse paw edema, TRPA1 deficient mice showed a quantitatively similar attenuated response as mice lacking Akt138, endothelial nitric oxide-synthase39 and the tumor necrosis factor α receptor 140,41. Taken together, our results support the conclusion that TRPA1 activation is involved in the pathogenesis of acute inflammatory edema in response to carrageenan.
Both mouse and human TRPA1 is inhibited by HC-030031, which is considered to be a selective TRPA1 antagonist as it is inactive on 48 other essential proteins involved in pain and inflammation including COX-2 and TRPV15,16,24. AITC is present in many naturally occurring plant-derived sources and has been shown to have antimicrobial42,43, cytostatic and cancer protective44 properties linked to multitudinous cellular targets. Whether TRPA1 mediates these effects is, however, unclear. As shown in the present study, AITC evoked edema in wild type but not in TRPA1 deficient animals and in WT mice the AITC-response was inhibited by the TRPA1 antagonist HC-030031. These data together suggest that the AITC-induced edema response found in the present study was indeed mediated by TRPA1.
Given that TRPA1 is a highly promiscuous chemosensor5,45, one cannot rule out the possibility that TRPA1 may also recognize carrageenan. Due to its high viscosity, we could not test the direct effect of carrageenan in concentrations above 250 µg/ml on TRPA1. This concentration is 40-60 times lower than stock solutions normally used for intraplantar injections37 and 8 times lower than the concentration known to induce inflammation46. However, the injected carrageenan is quickly diluted within the tissue and hence the local concentrations may well correspond to the in vitro test concentration that we found inactive at TRPA1 expressed in HEK 293 cells. Thus, we believe that the inflammation triggered by carrageenan is most likely not initiated by a direct interaction with TRPA1, but rather involves TRPA1 at some subsequent step in the inflammatory cascade.
The ability of ibuprofen to reduce both carrageenan and AITC-induced edema suggests that it inhibits COX downstream of TRPA1. Cellular calcium influx through TRPA1 would trigger the calcium-dependent release of prostaglandins and other pro-inflammatory agents that may further sensitize TRPA1 and increase its cell surface expression as occurs in sensory neurons47,48. It is also possible that TRPA1 activation increases prostaglandin production through enhanced COX-2 expression. This is supported by our in vitro finding that activation of TRPA1 by AITC can up-regulate the COX-2 transcript in HEK 293 cells. Accordingly, a significant role of [Ca2+]i in regulating COX-2 expression in macrophages was recently reported49. Together these events would generate and maintain a TRPA1 and COX-dependent inflammation. Our finding that the TRPA1 antagonist HC-030031 is as effective as the COX inhibitor ibuprofen to inhibit the development of carrageenan and AITC-induced edema is promising and may help to develop safer anti-inflammatory drugs than existing COX inhibitors.
An interesting question remains on the mechanisms behind TRPA1-mediated inflammatory edema which may involve neurogenic and/or non-neurogenic components. TRPA1 activation in afferent nerve endings may lead to a focal release of bioactive compounds such as calcitonin gene-related peptide (CGRP) and substance P which play a major role in neurogenic inflammation and exert vascular effects which may contribute to the formation of inflammatory edema5. However, spinal TRPA1 may also regulate the peripheral responses through retrograde afferent signaling30. In addition, activation of TRPA1 on vascular sensory neurons and/or endothelium50 can lead to vasodilatation and increased vascular permeability and edema formation51. Since endothelium is also a known source of vasodilating and edema-evoking prostaglandins and other prostanoids, this could provide an explanation for the link between TRPA1 and COX, as observed in the present study.
In humans, TRPA1 is present on nociceptive nerve endings and in non-neuronal cells such as keratinocytes and fibroblasts, from which prostaglandins and other mediators may be released after TRPA1 activation52,53. Therefore, the mouse carrageenan-induced paw edema may be a useful model to identify novel compounds and drugs targeting TRPA1 in humans.
Our study demonstrates a significant role for TRPA1 in the development of acute inflammation and suggests that TRPA1 antagonists may have anti-inflammatory properties in addition to their previously recognized analgesic effects in inflammatory pain.
Methods
Wild type and TRPA1-deficient C57BL/6 mice were used in the experiments. TRPA1-deficient mice and the corresponding WT mice were originally obtained from Dr David Julius (UCSF) and back-crossed in the laboratory of EDH and PMZ (Lund University, Sweden). TRPA1 genotype was confirmed by PCR. The mice were housed under standard conditions (12:12 h light-dark cycle, 22±1°C, 50–60% humidity) with food and water provided ad libitum.
The mice were divided into groups of six and were treated with 150 µl of saline (vehicle), ibuprofen (100 mg/kg, Sigma Chemical Co., St. Louis, MO, USA) or HC-030031 (300 mg/kg, Sigma Chemical Co.) i.p. 2 h before administering carrageenan or AITC. Before inducing the paw edema, the mice were anesthesized by i.p. injection of 0.5 mg/kg medetomidine (Domitor®, Orion Oyj, Espoo, Finland) and 75 mg/kg ketamine (Ketalar®, Pfizer Oy Animal Health, Helsinki, Finland). To induce the edema, mice received ipsilaterally i.pl. injection (30 µl) of either λ-carrageenan (15 mg/ml, Sigma Chemical Co.) or AITC (6.66 mM, Sigma Chemical Co.) into the hind paw. The control paw was injected with 30 μl of saline and developed no measurable edema. The study was approved by the National Animal Experiment Board.
Edema was measured before and at 3 h and 6 h after carrageenan or AITC injection by using a plethysmometer (Ugo Basile, Comerio, Italy). Edema is expressed as the difference between the carrageenan or AITC treated paw at the time indicated and the basal level.
HEK 293 human embryonic kidney cells (American Type Culture Collection, Manassas, VA, USA) were cultured in Eagle's Minimum Essential Medium (EMEM) supplemented with 10% heat-inactivated fetal bovine serum, sodium bicarbonate (1.5%), sodium pyruvate (1 mM), non-essential amino acids (1 mM each) (all from Lonza , Verviers SPRL, Verviers, Belgium), penicillin (100 U/ml), streptomycin (100 µg/ml) and amphotericin B (the last three compounds from Invitrogen, Paisley, UK) at 37°C in 5% CO2. The cells were transfected using 0.42 μg/cm2 human TRPA1 plasmid DNA (pCMV6-XL4 by Origene Rockville, MD, USA) with Lipofectamine 2000 (Invitrogen) according to the manufacturer's directions. After 24 h of transfection, HC-030031 (10 μM) or solvent (control) was added to the cells in fresh culture medium 30 min prior to the activation of TRPA1 by AITC (10 μM). The cells were then incubated for 6 h before being harvested for RNA extraction. Similar experiments were carried out by using non-transfected (wild type, WT) HEK 293 cells.
Total RNA extraction was carried out with the use of GenEluteTM Mammalian Total RNA Miniprep Kit (Sigma). Reverse-transcription of RNA to cDNA and quantitative RT-PCR reactions were performed as previously described54. TRPA1 expression was measured by using TaqMan Gene Expression Assay (Applied Biosystems, Foster City, CA, USA). COX-2 and GAPDH primers and probes were identical to those previously described55. COX-2 mRNA levels were normalized against GAPDH.
Fluorometric calcium imaging was used to study the effect of carrageenan on human TRPA1 expressed in HEK 293 cells. The cells were plated in 96-well black-walled plates (Costar, Cambridge, MA, USA) and loaded with Fura 2-AM (1 μM, Invitrogen), probenecid (2 mM, Sigma Chemical Co.) and pluronic acid (20%, Invitrogen) for 1 h at 37°C. The cells were then washed with physiological buffer solution (PBS), containing 140 mM NaCl, 5 mM KCl, 10 mM glucose, 10 mM HEPES, 2 mM CaCl2 and 1 mM MgCl2 and allowed to equilibrate for a period of 30 min in the dark before the start of the experiments. The intracellular calcium concentration was determined at 25°C in a Flexstation 3 (Molecular Devices, Sunnyvale, CA, USA). Basal emission (510 nm) ratios with excitation wavelengths of 340 nm and 380 nm were measured and changes in dye emission ratio (Δ ratio) determined at various times after compound addition.
Results are expressed as mean ± standard error of mean (SEM). Statistical analysis was carried out by using Student's t-test or one-way ANOVA with Bonferroni's multiple comparisons test and results were considered significant at *p<0.05, **p<0.01 and ***p<0.001.
References
Jordt, S. et al. Mustard oils and cannabinoids excite sensory nerve fibres through the TRP channel ANKTM1. Nature 427, 260–265 (2004).
Bandell, M. et al. Noxious cold ion channel TRPA1 is activated by pungent compounds and bradykinin. Neuron 41, 849–857 (2004).
Bautista, D. M. et al. Pungent products from garlic activate the sensory ion channel TRPA1. Proc. Natl. Acad. Sci. U. S. A. 102, 12248–12252 (2005).
Macpherson, L. J. et al. The pungency of garlic: activation of TRPA1 and TRPV1 in response to allicin. Curr. Biol. 15, 929–934 (2005).
Baraldi, P. G., Preti, D., Materazzi, S. & Geppetti, P. Transient Receptor Potential Ankyrin 1 (TRPA1) Channel as Emerging Target for Novel Analgesics and Anti-Inflammatory Agents. J. Med. Chem. 53, 5085–5107 (2010).
Bessac, B. F. & Jordt, S. E. Breathtaking TRP channels: TRPA1 and TRPV1 in airway chemosensation and reflex control. Physiology (Bethesda) 23, 360–370 (2008).
Andersson, K. E., Gratzke, C. & Hedlund, P. The role of the transient receptor potential (TRP) superfamily of cation-selective channels in the management of the overactive bladder. BJU Int. 106, 1114–1127 (2010).
Pedersen, S. F., Owsianik, G. & Nilius, B. TRP channels: an overview. Cell Calcium 38, 233–252 (2005).
Berridge, M. J., Lipp, P. & Bootman, M. D. The versatility and universality of calcium signalling. Nat. Rev. Mol. Cell Biol. 1, 11–21 (2000).
Jaquemar, D., Schenker, T. & Trueb, B. An Ankyrin-like Protein with Transmembrane Domains Is Specifically Lost after Oncogenic Transformation of Human Fibroblasts. Journal of Biological Chemistry 274, 7325–7333 (1999).
Story, G. M. et al. ANKTM1, a TRP-like Channel Expressed in Nociceptive Neurons, Is Activated by Cold Temperatures. Cell 112, 819–829 (2003).
Hinman, A., Chuang, H., Bautista, D. M. & Julius, D. TRP channel activation by reversible covalent modification. Proceedings of the National Academy of Sciences 103, 19564–19568 (2006).
Sadofsky, L. R. et al. TRPA1 is activated by direct addition of cysteine residues to the N-hydroxysuccinyl esters of acrylic and cinnamic acids. Pharmacol. Res. 63, 30–36 (2011).
Taylor-Clark, T. E., Ghatta, S., Bettner, W. & Undem, B. J. Nitrooleic acid, an endogenous product of nitrative stress, activates nociceptive sensory nerves via the direct activation of TRPA1. Mol. Pharmacol. 75, 820–829 (2009).
Andersson, D. A., Gentry, C., Moss, S. & Bevan, S. Transient receptor potential A1 is a sensory receptor for multiple products of oxidative stress. J. Neurosci. 28, 2485–2494 (2008).
Eid, S. R. et al. HC-030031, a TRPA1 selective antagonist, attenuates inflammatory- and neuropathy-induced mechanical hypersensitivity. Mol. Pain 4, 48 (2008).
Nagata, K., Duggan, A., Kumar, G. & Garcia-Anoveros, J. Nociceptor and hair cell transducer properties of TRPA1, a channel for pain and hearing. J. Neurosci. 25, 4052–4061 (2005).
Nozawa, K. et al. TRPA1 regulates gastrointestinal motility through serotonin release from enterochromaffin cells. Proc. Natl. Acad. Sci. U. S. A. 106, 3408–3413 (2009).
Kwan, K. Y., Glazer, J. M., Corey, D. P., Rice, F. L. & Stucky, C. L. TRPA1 modulates mechanotransduction in cutaneous sensory neurons. J. Neurosci. 29, 4808–4819 (2009).
Streng, T. et al. Distribution and function of the hydrogen sulfide-sensitive TRPA1 ion channel in rat urinary bladder. Eur. Urol. 53, 391–399 (2008).
Kochukov, M. Y., McNearney, T. A., Fu, Y. & Westlund, K. N. Thermosensitive TRP ion channels mediate cytosolic calcium response in human synoviocytes. Am. J. Physiol. Cell. Physiol. 291, C424–32 (2006).
El Karim, I. A. et al. Human odontoblasts express functional thermo-sensitive TRP channels: implications for dentin sensitivity. Pain 152, 2211–2223 (2011).
Chen, J. et al. Selective blockade of TRPA1 channel attenuates pathological pain without altering noxious cold sensation or body temperature regulation. Pain 152, 1165–1172 (2011).
McNamara, C. R. et al. TRPA1 mediates formalin-induced pain. Proc. Natl. Acad. Sci. U. S. A. 104, 13525–13530 (2007).
Bautista, D. M. et al. TRPA1 mediates the inflammatory actions of environmental irritants and proalgesic agents. Cell 124, 1269–1282 (2006).
Bessac, B. F. et al. Transient receptor potential ankyrin 1 antagonists block the noxious effects of toxic industrial isocyanates and tear gases. FASEB J. 23, 1102–1114 (2009).
Wei, H., Koivisto, A. & Pertovaara, A. Spinal TRPA1 ion channels contribute to cutaneous neurogenic inflammation in the rat. Neurosci. Lett. 479, 253–256 (2010).
Nassini, R. et al. Acetaminophen, via its reactive metabolite N-acetyl-p-benzo-quinoneimine and transient receptor potential ankyrin-1 stimulation, causes neurogenic inflammation in the airways and other tissues in rodents. FASEB J. (2010).
Caceres, A. I. et al. A sensory neuronal ion channel essential for airway inflammation and hyperreactivity in asthma. Proceedings of the National Academy of Sciences 106, 9099–9104 (2009).
da Costa, D. S. et al. The involvement of the transient receptor potential A1 (TRPA1) in the maintenance of mechanical and cold hyperalgesia in persistent inflammation. Pain 148, 431–437 (2010).
Fernandes, E. S. et al. A distinct role for transient receptor potential ankyrin 1, in addition to transient receptor potential vanilloid 1, in tumor necrosis factor α-induced inflammatory hyperalgesia and Freund's complete adjuvant-induced monarthritis. Arthritis & Rheumatism 63, 819–829 (2011).
Petrus, M. et al. A role of TRPA1 in mechanical hyperalgesia is revealed by pharmacological inhibition. Mol. Pain 3, 40 (2007).
Kremeyer, B. et al. A Gain-of-Function Mutation in TRPA1 Causes Familial Episodic Pain Syndrome. Neuron 66, 671–680 (2010).
Andrè, E. et al. Cigarette smoke-induced neurogenic inflammation is mediated by alpha,beta-unsaturated aldehydes and the TRPA1 receptor in rodents. J. Clin. Invest. 118, 2574–2582 (2008).
Posadas, I. et al. Carrageenan-induced mouse paw oedema is biphasic, age-weight dependent and displays differential nitric oxide cyclooxygenase-2 expression. Br. J. Pharmacol. 142, 331–338 (2004).
Fantone, J. C. & Ward, P. A. Role of oxygen-derived free radicals and metabolites in leukocyte-dependent inflammatory reactions. Am. J. Pathol. 107, 395–418 (1982).
Morris, C. J. Carrageenan-induced paw edema in the rat and mouse. Methods Mol. Biol. 225, 115–121 (2003).
Di Lorenzo, A., Fernandez-Hernando, C., Cirino, G. & Sessa, W. C. Akt1 is critical for acute inflammation and histamine-mediated vascular leakage. Proc. Natl. Acad. Sci. U. S. A. 106, 14552–14557 (2009).
Bucci, M. et al. Endothelial nitric oxide synthase activation is critical for vascular leakage during acute inflammation in vivo. Proc. Natl. Acad. Sci. U. S. A. 102, 904–908 (2005).
Rocha, A. C., Fernandes, E. S., Quintao, N. L., Campos, M. M. & Calixto, J. B. Relevance of tumour necrosis factor-alpha for the inflammatory and nociceptive responses evoked by carrageenan in the mouse paw. Br. J. Pharmacol. 148, 688–695 (2006).
Mazzon, E. et al. Effect of tumour necrosis factor-alpha receptor 1 genetic deletion on carrageenan-induced acute inflammation: a comparison with etanercept. Clin. Exp. Immunol. 153, 136–149 (2008).
Schirmer, B. C. & Langsrud, S. Evaluation of natural antimicrobials on typical meat spoilage bacteria in vitro and in vacuum-packed pork meat. J. Food Sci. 75, M98–M102 (2010).
Obaidat, M. M. & Frank, J. F. Inactivation of Salmonella and Escherichia coli O157:H7 on sliced and whole tomatoes by allyl isothiocyanate, carvacrol and cinnamaldehyde in vapor phase. J. Food Prot. 72, 315–324 (2009).
Zhang, Y. Allyl isothiocyanate as a cancer chemopreventive phytochemical. Molecular Nutrition & Food Research 54, 127–135 (2010).
Holzer, P. Transient receptor potential (TRP) channels as drug targets for diseases of the digestive system. Pharmacol. Ther. 131, 142–170 (2011).
Henriques, M. G. et al. Mouse paw edema. A new model for inflammation? Braz. J. Med. Biol. Res. 20, 243–249 (1987).
Materazzi, S. et al. Cox-dependent fatty acid metabolites cause pain through activation of the irritant receptor TRPA1. Proc. Natl. Acad. Sci. U. S. A. 105, 12045–12050 (2008).
Schmidt, M., Dubin, A. E., Petrus, M. J., Earley, T. J. & Patapoutian, A. Nociceptive signals induce trafficking of TRPA1 to the plasma membrane. Neuron 64, 498–509 (2009).
Jakobsson, P. Pain: How macrophages mediate inflammatory pain via ATP signaling. Nat Rev Rheumatol 6, 679–681 (2010).
Earley, S., Gonzales, A. L. & Crnich, R. Endothelium-dependent cerebral artery dilation mediated by TRPA1 and Ca2+-Activated K+ channels. Circ. Res. 104, 987–994 (2009).
Graepel, R. et al. 4-Oxo-2-nonenal (4-ONE): Evidence of Transient Receptor Potential Ankyrin 1-Dependent and -Independent Nociceptive and Vasoactive Responses In Vivo. Journal of Pharmacology and Experimental Therapeutics 337, 117–124 (2011).
Atoyan, R., Shander, D. & Botchkareva, N. V. Non-neuronal expression of transient receptor potential type A1 (TRPA1) in human skin. J. Invest. Dermatol. 129, 2312–2315 (2009).
Jain, A. et al. TRP-channel-specific cutaneous eicosanoid release patterns. Pain 152, 2765–2772 (2011).
Nieminen, R., Lahti, A., Jalonen, U., Kankaanranta, H. & Moilanen, E. JNK inhibitor SP600125 reduces COX-2 expression by attenuating mRNA in activated murine J774 macrophages. 6, 987–996 (2006).
Nieminen, R., Korhonen, R., Moilanen, T., Clark, A. R. & Moilanen, E. Aurothiomalate inhibits cyclooxygenase 2, matrix metalloproteinase 3 and interleukin-6 expression in chondrocytes by increasing MAPK phosphatase 1 expression and decreasing p38 phosphorylation: MAPK phosphatase 1 as a novel target for antirheumatic drugs. Arthritis Rheum. 62, 1650–1659 (2010).
Acknowledgements
We wish to thank Ms Petra Miikkulainen for technical assistance and Mrs Heli Määttä for secretarial help. The study was supported by grants from the Medical Research Fund of Tampere University Hospital, Finland; Tampere Tuberculosis Foundation, Finland; the Medical Faculty of Lund (ALF) and the Swedish Research Council (2007–3095, 2010–3347). The authors thank Peter McIntyre, University of Melbourne, for providing the human TRPA1-inducible cell line.
Author information
Authors and Affiliations
Contributions
LJM participated in the animal and laboratory experiments, calculated the results and statistics and drafted the manuscript. ML, MK, RK, TL and RMN participated in the animal and the laboratory experiments. EDH and PMZ provided the TRPA1 knock out and corresponding wild type animals and carried out the calcium imaging experiments. EM conceived and supervised the study and helped to draft the manuscript. All authors were involved in the planning of the study and in writing the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareALike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Moilanen, L., Laavola, M., Kukkonen, M. et al. TRPA1 Contributes to the Acute Inflammatory Response and Mediates Carrageenan-Induced Paw Edema in the Mouse. Sci Rep 2, 380 (2012). https://doi.org/10.1038/srep00380
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep00380
This article is cited by
-
Automated home-cage for the evaluation of innate non-reflexive pain behaviors in a mouse model of inflammatory pain
Scientific Reports (2021)
-
Transient Receptor Potential Ankyrin 1 (TRPA1) Mediates Lipopolysaccharide (LPS)-Induced Inflammatory Responses in Primary Human Osteoarthritic Fibroblast-Like Synoviocytes
Inflammation (2018)
-
Transient receptor potential ankyrin 1 (TRPA1) is functionally expressed in primary human osteoarthritic chondrocytes
Arthritis Research & Therapy (2016)
-
Transient receptor potential ankyrin 1 (TRPA1) receptor is involved in chronic arthritis: in vivo study using TRPA1-deficient mice
Arthritis Research & Therapy (2016)
-
Polysulfide Evokes Acute Pain through the Activation of Nociceptive TRPA1 in Mouse Sensory Neurons
Molecular Pain (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.