Abstract
Satellite observations show that the 2010 Antarctic ozone hole is characterized by anomalously small amounts of photochemical ozone destruction (40-60% less than the 2005-2009 average). Observations from the MLS instrument show that this is mainly related to reduced photochemical ozone destruction between 20-25 km altitude. Lower down between 15-20 km the atmospheric chemical composition and photochemical ozone destruction is unaffected. The modified chemical composition and chemistry between 20-25 km altitude in 2010 is related to the occurrence of a mid-winter minor Antarctic Sudden Stratospheric Warming (SSW). The measurements indicate that the changes in chemical composition are related to downward motion of air masses rather than horizontal mixing and affect stratospheric chemistry for several months. Since 1979, years with similar anomalously small amounts of ozone destruction are all characterized by either minor or major SSWs, illustrating that their presence has been a necessary pre-condition for reduced Antarctic stratospheric ozone destruction.
Similar content being viewed by others
Introduction
The annual occurrence of the Antarctic ozone hole is a well understood and documented phenomenon1. Its existence is related to the gradual increase in atmospheric concentrations of chlorine as a result of the anthropogenic production of chlorofluorcarbons (CFC). Effective international efforts to reduce emissions have led to a gradual decline of these substances by about 0.5–1% per year2. However, despite these gradual changes, year-to-year variations in ozone depletion over the last 30 years show a number of years with anomalously small amounts of ozone depletion.
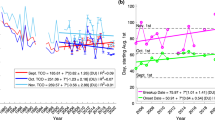
Figure 1A and 1B present the Antarctic ozone hole area and ozone mass deficit (OMD), respectively, based on total ozone columns for the period 1979–2010 for three indices of ozone destruction. The development of the ozone hole in the 1980's and the stabilization during the 1990's are easily recognized. However, several years show strongly reduced ozone destruction: 1986, 1988, 2002, 2004 and 2010. Given that ozone destruction is related to the amount of ozone-depleting substances (ODS) present in the stratosphere and that the amount of ozone-depleting substances steadily increased from the late 1970's onwards and has been slowly decreasing since about 1995, the gradual changes in ODS cannot explain these large year to year reductions in depletion3.
Antarctic ozone mass deficit (A) and ozone hole area (B) for the years 1979–2010 for three different “ozone destruction” definitions: D1 is the average for the period 21 to 30 September, D2 is the period 7 September to 13 October and D3 denotes the “worst” 30-day period, i.e. the largest deficit for any 30-day period.
The ozone mass deficit is given in 109 kg ozone with regard to the 220 Dobson Unit total ozone column value (Dobson Unit = DU = 2.69 1016 molecules/cm2) for total ozone columns south of 45°S. The vortex area is the total area where total ozone columns south of 45°S are smaller than 220 DU. For 1979–2008 MSR data is used, for 2009 and 2010 SCIAMACHY total ozone column assimilation data is used.
Although the OMD is a commonly used metric for determining Antarctic ozone depletion1,4 and other methods for estimating ozone loss also reveal similar years with anomalous ozone loss5, it nevertheless is conceivable that the reduction in OMD in 2004 and 2010 could be caused by a smaller vortex area. The OMD based vortex area is indeed smaller in 2004 and 2010 by about 20–25%, as can be seen in figure 1B. However, the reduction in size is insufficient to explain the 50% reduction in OMD for both years. Furthermore, there is a caveat in the use of OMD to estimate the vortex area. At the edges of the vortex ozone depleted layers in the lower stratosphere can be partly compensated for by ozone rich air in the upper troposphere, leading to total ozone column values larger than 220 DU even though one would clearly identify the area as being inside the vortex6,7. However, the MLS measurements used in this study confirm that total ozone column values in 2004 and 2010 within the vortex are 25% larger than in 2005–2009. Furthermore, it should be noted the MLS measurements only cover part of the vortex and cannot be used to estimate the total vortex OMD. Finally, dynamical estimates of the vortex area show that in 2004 the area is of average size and in 2010 it is even well above average8. Hence, it can be safely concluded that at least half of the OMD reduction in 2004 and 2010 is unexplained by vortex dynamics.
Another possible explanation for this decrease in OMD would be changes in stratospheric aerosols related to volcanic eruptions, which are known to enhance heterogeneous ozone depletion9,10,11. Since 1980, enhanced stratospheric aerosol related to volcanic eruptions was present from 1982–1985 and 1991–1994. However, outside those periods stratospheric aerosols were at background levels. Given the strong year-top-year fluctuation of years with and without strongly reduced stratospheric ozone, changes in stratospheric aerosols cannot explain these variations. Hence, other explanations must be sought.
The depth of the ozone hole depends on meteorological conditions and the tropospheric wave driving of the stratosphere12, affecting mixing across the Antarctic vortex edge, either directly mixing in ozone rich air or affecting the chemical composition of the vortex air. Variations in meteorological conditions are more likely13 to be the cause of interannual variability in Antarctic stratospheric ozone at the top edge of the hole, possibly linked to temperature and Polar Stratospheric Clouds (PSC) formation.
The year 2002 was characterized by an early Antarctic vortex break up due to a strong Sudden Stratospheric Warming (SSW) causing the vortex to split in September and to quickly dissipate14. The years 1986 and 1988 have been described15,16 as years with a “warm” vortex and the occurrence of significant SSWs17, in which fewer PSCs were formed, denitrification was reduced and thus catalytic ozone destruction was less efficient13. Previous studies18,19 have noted that 2004 appears atypical in a sense that there were no signs of dehydration above approximately 20 km, but due to lack of EOS MLS20 (Earth Observing System Microwave Limb Sounder) measurements from the beginning of the winter in 2004 no firm conclusion was drawn on the causes of this lack of dehydration.
Temperatures and wave activity as well as Antarctic stratospheric chemistry are thus important for understanding the existence of years with significantly reduced Antarctic stratospheric ozone depletion. Nevertheless, it remains unclear how the dynamical and chemical effects change ozone depletion. EOS MLS provides observations of multiple trace gases to study specifically the role of SSWs in year-to-year variations of ozone depletion18,19. The reduced ozone depletion in 2010 – based on SCIAMACHY21 (SCanning Imaging Absorption spectroMeter for Atmospheric CartograpHY) total ozone column observations - is 40–60% below the 2005–2009 average.
The current length of the MLS record, covering five “normal” Antarctic ozone hole years (2005–2009) provides a climatology to which the anomalous 2010 Antarctic ozone depletion and the underlying dynamical and chemical mechanisms can be compared. Combined with the recently developed 30 year total ozone column reanalysis (see methods) the anomalous conditions of the 2010 ozone hole can be related to the observed interannual variability in Antarctic ozone depletion over the last 30 years.
Results
During Austral winter, a strong dynamic polar vortex forms over Antarctica that inhibits the mixing of warm mid-latitude air and enhances radiative cooling in absence of solar radiation during part of the winter. If temperatures drop below approximately 196 K, PSCs are formed which consist of pure water (H2O), nitric-acid (HNO3) hydrate solution (Nitric Acid Trihydrate or NAT and Nitric Acid Di-hydrate or NAD) or liquid H2O/HNO3/H2SO4 solutions, known as supercooled ternary solution or STS22. Pure water PSCs are referred to as type 2, the others as type 1. Type 2 PSCs occur at slightly lower temperatures than type 1 PSCs. The role of PSC's in Antarctic ozone depletion is twofold. On the one hand they act as surface on which halogen species like chlorine can be activated which is necessary for catalytic ozone depletion. On the other hand, PSC formation removes odd nitrogen (odd nitrogen = NOy = abundance of N atoms that are not bound up in either nitrogen (N2) or nitrous oxide (N2O); NOy includes HNO3 and nitrogen dioxide (NO2)). PSC formation also removes water vapor from the stratosphere through sedimentation of the cloud particles. This “denitrification” further enhances ozone depletion as deactivation of chlorine via formation of reservoir HCl becomes less effective. Depending on the temperature, denitrification can occur with or without dehydration.
Once the Antarctic polar vortex is established and PSC formation, denitrification and/or dehydration have occurred, catalytic ozone destruction can start due to photochemical activation of the halogens once the sun returns during early spring. As long as temperatures remain sufficiently low, PSC formation will continue and prevent the buildup of halogen reservoirs. However, once the Antarctic vortex warms due to increasing sunlight, the PSCs evaporate and halogen species are deactivated within weeks by the re-formation of reservoir species.
Figure 2 shows nine-day running mean MLS measurements of ClO, HCl, HNO3, O3, H2O and temperature as function of time for all years from 2004–2010 at 68 hPa altitude (∼ 19 km) for MLS measurements south of 80°S, the typical altitude of the ozone hole. The nine-day period for the running mean was chosen for visualization purposes in order to reduce noise and better distinguish between different years. Indicated in the temperature plot are also the NAT (upper dotted line) and pure ice formation temperatures (lower two dotted lines). ClO is in particular an important proxy for catalytic halogen chemistry.
Nine-day running mean MLS measurements of ClO, HCl, HNO 3 , O 3 , H 2 O and temperatures at approximately 68 hPa for the years 2004–2010 between 9 or 10 April (DOY 100) to 15 or 16 December (DOY 350) depending on whether the year is a leap year or not.
Only MLS measurements south of 80°S are used. Ozone is given in Dobson Units (DU), all other chemical species are given in parts per billion (ppbv) and temperature is given in Kelvin. The horizontal dotted lines in the temperature graph indicate the PSC formation temperatures for: NAT39 (upper black line), saturation over ice39 (middle blue line) and saturation over liquid water40 (bottom red line).
Once temperatures drop below the NAT formation temperature around the Day Of Year (DOY) 150 (∼ 1 June), denitrification starts as evidenced by the decrease in HNO3. Full denitrification is accomplished within about 20 days. At the same time the chlorine reservoir HCl is “emptied”23 due to chemical reactions of HCl on PSC's. Dehydration starts about 20 days later as the pure ice formation temperatures are reached 20 days later than NAT formation temperatures. Once the sun returns halfway August (∼ DOY 225) ozone is being destroyed by halogens as evidenced by the increase in ClO around DOY 225. Ozone destruction maximizes between DOY 250 and 270 (mid-September) when ClO is abundant. Around DOY 270, temperatures get above the NAT formation temperature, PSCs evaporate and the active halogens are rapidly deactivated back into reservoir species like HCl. The slow increase in HNO3 and H2O starting around DOY 270 also shows that mixing is taking place. Finally, after DOY 300 (∼ late October – early November) ozone slowly increases again, mainly by mixing of mid-latitude air. This behavior is very similar for all years, showing that catalytic ozone depletion at 68 hPa in both 2004 and 2010 is not unusual. The same behavior is observed lower down at 100 hPa and higher up at 46 hPa (approximately 16 and 21 km; not shown here).
However, at 32 and 22 hPa (23.5 and 26 km; figures 3 and 4) the variations for 2004 and 2010 differ from the other years on a few but crucial points. Temperatures still drop below NAT and ice PSC formation temperatures, denitrification and dehydration still occur and chlorine is activated. At sunrise ozone depletion still occurs but the larger ozone amounts at this altitude, typical17 for the Antarctic vortex for these altitudes, prevent complete destruction.
As figure 2 but at an altitude of approximately 32 hPa
As figure 2 but at an altitude of approximately 22 hPa.
At 22 hPa, shortly after the full dehydration around DOY 200 the amount of H2O already starts to increase again, simultaneously with HNO3. Furthermore, less ClO is formed and HCl starts to increase earlier while ozone destruction is absent, unlike the years 2005–2009 when there is still some ozone destruction at 22 hPa. Note that for both 2004 and 2010 between DOY 200–250 a small but distinct increase in temperature occurs, reflecting the occurrence of a SSW.
At 32 hPa H2O also increases shortly after the full dehydration, but this is not accompanied by an increase in HNO3. Although there is less ClO, the reduction is smaller than at 22 hPa and there is no early increase in HCl. However, after DOY 250 HNO3 increases a little bit faster in 2004 and 2010 compared to 2005–2009. Finally, ozone is being destroyed but depletion is smaller compared to 2005–2009.
The left image of figure 5 shows the average MLS N2O profile for DOY 190. Clearly N2O decreases in concentration between 100 and 10 hPa. The right image of figure 5 shows the 20-day difference in N2O at 32 hPa. Because of the general slow diabatic decent of air within the vortex this differences is negative for all years throughout the Antarctic winter. However, in 2010, around DOY 200, this difference increases by about a factor of 3–4. N2O concentrations outside of the Antarctic vortex are larger than within the vortex, hence this acceleration in decrease in N2O cannot be explained by horizontal transport and mixing and thus must be caused by increased diabatic descend due to the SSW. Based on the N2O profile we estimate that at 32 hPa the air has descended by about 10 hPa or approximately 3 km starting around DOY 190 concurrent with the reversal in H2O at 22 hPa (Figure 4).
MLS nine-day running mean N2O vertical profile (left plot DOY 190) and 20-day lag difference in N2O concentrations at 32 hPa.
This difference is calculated as the differences in N2O concentration for ± 10 days around the central date. From 14 July 2007 (DOY 194) – 8 august 2007 (DOY 219) N2O data is lacking due to the MLS 640 GHz radiometer (R4) anomaly. The large difference for 2007 (green) around DOY 210 is related to the startup after this anomaly.
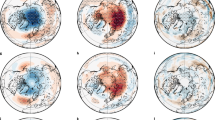
Figure 6 shows temperature and humidity anomalies for 2004 and 2010 relative to the mean for the period 2005–2009 as a function of altitude. During both 2004 and 2010 there are clear SSWs, with a strong SSW developing in 2010 around DOY 190, whereas in 2004 there is a whole series of weaker SSWs. The SSWs quickly descend and dissipate in a termination zone between 10 and 50 hPa. At the same time a clear H2O anomaly develops during both years in this termination zone. The H2O anomalies follow the general slow descending motion within the Antarctic winter vortex. Note that in both 200419 and 2010 the positive H2O anomalies are accompanied by positive HNO3 anomalies. The re-hydration and re-nitrification clearly are related to the occurrence of the SSW and corresponding downward propagation of the temperature anomaly.
MLS temperature (K) and water vapor (ppbv) anomaly profiles between 1–100 hPa for the same period and latitude as figures 2 to 4 for 2010 (upper two panels) and 2004 (lower panel, H 2 O only) and the same nine-day running means.
The anomaly is calculated compared to the 2005–2009 MLS averages. Note that in 2004 no MLS observations are available before 1 August (∼ DOY 215).
Discussion
We have shown, using MLS measurements, that the strongly reduced Antarctic ozone hole destruction in 2010 and 2004 is directly related to SSWs occurring during the polar winter. The SSW causes humidity-rich air in the middle stratosphere (10–50 hPa) to descend but not below 50 hPa or 18 km. This descent strongly modifies the chemical composition of the air between 10 and 50 hPa. As the SSW does not reach altitudes below 50 hPa, the ozone destruction below 50 hPa is unaffected by what happens above.
However, based on the MLS observations it remains largely unclear how the SSW affects the chemistry which causes the observed changes in chemical composition. At 22 hPa the chemical pathway appears straightforward. More HNO3 and H2O (via HOx) promote the formation of NO2, which in turn causes formation of ClONO2. This reduces ClO and as ozone destruction goes by the square of ClO as it involves the dimmer ClOOCl, net ozone destruction at 22 hPa is prevented (Figure 4). Also the early increase in the chlorine reservoir HCl could be the result of the increase in HOx resulting from H2O.
Although there are no early increases in HNO3 and HCl at 32 hPa, once ClO starts to form around DOY 250 there is also an increase in HNO3 that in 2004 and 2010 is larger than during 2005–2009. And once HCl is starting to form after DOY 250, ClO starts to decrease again. It thus appears that during the buildup of ClO there still is some form of nitrogen oxide present – as indicated by increase in HNO3 after DOY 250 – and that this reduces the effectiveness of the ClO formation, which in turn affects ozone destruction. One possible pathway could be that there is some nitrogen oxide present in the form of N2O5, which at some point is converted to NO2 by reaction with the more abundant H2O. However, this remains speculative and for a proper understanding of the chemistry between 10 and 50 hPa detailed photochemistry and/or 3D chemistry-transport model calculations are required, which is beyond the scope of this paper. However, the behavior of Antarctic stratospheric ozone in 2004 and 2010 provide an interesting test case for the current understanding of stratospheric chemistry as captured in models.
The MLS observations clearly show that ozone destruction between 10 and 50 hPa is strongly reduced in 2004 and 2010 and that the SSW had a significant impact on the chemical composition. Although 2010 differs from 2004 in terms of the number and severity of SSWs, the similarities of the water vapor and HNO3 anomalies and their link with the SSW confirm the importance of the SSW for middle-stratospheric Antarctic ozone chemistry and destruction. Interestingly, it has been suggested that enhanced (lower) stratospheric H2O due to climate change might actually increase polar ozone destruction by enabling chlorine activation at higher temperatures24 and lead to more PSC formation25. The observations presented in this paper strongly suggest that enhanced H2O in the middle to upper Antarctic stratosphere reduces ozone destruction.
We also note that in 1988 an SSW occurred that was very similar to the SSW of 2010 in terms of timing and amplitude. The year 1988 was characterized17 by very little ozone destruction that occurred primarily in the lower stratosphere between 15 and 19 km altitude. This observation fits with the 2004 and 2010 sequence of events, i.e. the SSW affecting middle stratospheric ozone but not lower stratospheric ozone. The lesser ozone destruction in 1988 compared to 2010 may be related to the lower total amount26 of halogen species in 1988, which was significantly smaller than in 2010.
Our analysis suggests that the occurrence of an early minor SSW during polar winter has a strong impact on the ozone destruction at polar sunrise. Both timing and magnitude of the SSW are important, i.e. the SSW should occur in winter and temperatures should rise sufficiently to prevent denitrification of the middle stratosphere. The major SSW in 2002 which occurred later in the year led to a vortex split and the dynamical transport and mixing effects of this event dominated the evolution of the 2002 ozone depletion season.
Finally, the IPCC/AR4 climate models including 20th century ozone destruction consistently predict26 an increase in the surface pressure difference between middle and high southern latitudes. The same models show27 that 21st century ozone hole recovery reduces these surface pressure differences. Models without ozone destruction and recovery do not show these changes. Therefore, based on the assertion that climate models including ozone destruction and recovery produce the rights signs in their surface pressure difference response we conclude that while ozone depletion and greenhouse gas increases worked together in the 20th century, the anticipated ozone recovery in the 21st century will weaken the trend in the surface pressure difference induced by the increases in greenhouse gases.
There are indications28 from stratosphere-resolving general circulation model simulations that the weakening in surface pressure differences could increase the frequency of SSW occurrences at southern latitudes, although no consensus has been reached yet28. Nevertheless, according to our analysis such an increase in SSW occurrences would be accompanied with more frequent years of strongly reduced ozone loss like in 2010.
In this way the 2010 ozone hole season could be seen as a fingerprint for possible positive dynamical feedbacks accompanying the ozone hole recovery in the next decades in response to the decline in ozone depleting substances.
Methods
Total ozone columns used for 2009 and 2010 are derived from satellite observations of total ozone columns from SCIAMACHY that are assimilated28 in the chemistry transport model TM3 to provide an analysis of global ozone and UV Index. The SCIAMACHY total ozone columns are retrieved using the TOSOMI29 algorithm. These measurements are corrected30 for instrument drift. Daily total ozone columns for 1979–2008 are obtained from the Multi Sensor Reanalysis project30 (MSR), which produced a 30-year total ozone column assimilation dataset based on a total of eleven satellite instruments measuring total ozone columns – including SCIAMACHY - that were operating during various periods within these 30 years. Data from both the MSR and SCIAMACHY assimilated total ozone columns are used in figure 1.
For the ozone (O3), temperature (T), water vapor (H2O) and other stratospheric trace gases including chlorine monoxide (ClO), hydrogen chloride (HCl) and nitric acid (HNO3) we use EOS MLS v2.2 measurements. The MLS instrument was launched on 15 July 2004 on board of the EOS Aura platform. Table 1 provides an overview of the precision and vertical resolution as well as the useful altitude range of the MLS measurements, including references to validation results. The ClO product in the lower stratosphere (100-32 hPa) suffers from a negative bias which is corrected according to published recommendations31. Finally, quality control with regard to “precision values”, “status”, “quality” and “convergence” has been implemented for the various species as described in the validation reports referred to in table 1. For all MLS observations we focus on the altitude range of 1–100 hPa, for which as a rule of thumb the estimated precision is 10% or better (for temperature the precision is 1K). All are of sufficient quality to be used within this altitude range.
References
World Meteorological Organization, The scientific assessment of ozone depletion: 2006. Global Ozone Research and Monitoring Project – Report No. 50, WMO, Geneva (2006).
Clerbaux, C. & Cunnold, D. M. “Long-lived compounds”, in: WMO, The Scientific Assessment of Ozone Depletion: 2006. Chapter 1, Global Ozone Research and Monitoring Project – Report No. 50, World Meteorological Organization, Geneva (2006).
Huck, P. E., McDonald, A. J., Bodeker, G. E. & Struthers, H. Interannual variability in Antarctic ozone depletion controlled by planetary waves and polar temperature. Geophys. Res. Lett, 32, L13819, doi:10.1029/2005GL022943 (2005).
World Meteorological Organization, Scientific assessment of ozone depletion: 2010. Global Ozone Research and Monitoring Project – Report No. 52, WMO, Geneva (2010).
Bodeker, G. E., Shiona, H. & Eskes, H. Indicators of Antarctic ozone depletion.. Atmos. Chem. Phys., 5, 2603–2615, 10.5194/acp-5-2603-2005 (2005).
van Peet, J. C. A. et al. Height resolved ozone hole structure as observed by the Global Ozone Monitoring Experiment-2. Geophys. Res. Lett., 36, L11816, doi:10.1029/2009GL038603 (2009).
Struthers, H. et al. The simulation of the Antarctic ozone hole by chemistry-climate models. Atmos. Chem. Phys., 9, 6363–6376, 10.5194/acp-9-6363-2009 (2009).
Hofmann, D. J. & Solomon, S. Ozone Destruction through heterogeneous chemistry following the eruption of El Chichón. J. Geophys. Res., 94, 5029–5041 (1989).
Portmann, R. et al. Role of aerosol variations in anthropogenic ozone depletion in polar regions. J. Geophys. Res., 101, 22991–23006 (1996).
Solomon, S., Portmann, R. W., Sasaki, T., Hofmann, D. J. & Thompson, D. W. J. Four decades of ozonesonde measurements over Antarctica. J. Geophys. Res., 110, D21311, 10.1029/2005JD005917 (2005).
Newman, P. A., Nash, E. R., Kawa, S. R., Montzka, S. A. & Schauffler, S. M. When will the Antarctic ozone hole recover.. Geophys. Res. Lett., 33, L12814, 10.1029/2005GL025232 (2006).
Hoppel, K. et al. Reduced ozone loss at the upper edge of the Antarctic ozone hole during 2001–2004. Geophys. Res. Lett., 32, L20816, 10.1029/2005GL023968 (2005).
Shepherd, T. G., Plumb, R. A. & Wofsy, S. C. Preface to the Special Issue on the Antarctic stratospheric sudden warming and split ozone hole of 2002. J. Atmos. Sci., 62, 565–566 (2005).
Kanzawa, H. & Kawaguchi, S. Large stratospheric sudden warming in the Antarctic late winter and shallow ozone hole in 1988. Geophys. Res. Lett., 17, pages 77–80 (1990).
Schoeberl, M. R., Stolarksi, R. S. & Krueger, A. J. the 1988 Antarctic ozone depletion: comparison with previous year depletions. Geophys. Res. Lett., 16, 377–380, 89GL00399 (1989).
Hofmann, D., Oltmans, S., Harris, J., Johnson, B. & Lathrop, J. Ten years of ozonesonde measurements at the south pole: implications for recovery of springtime Antarctic ozone. J. Geophys. Res., 102, D7, 8931–8943, 10.1029/96JD03749 (1997).
Jiménez, C. et al. EOS MLS observations of dehydration in the 2004–2005 polar winters. Geophys. Res. Lett., 33, L16806, 10.1029/2006GL025926 (2005).
Santee, M. L. et al. Polar processing and development of the 2004 Antarctic ozone hole: First results from MLS on Aura. Geophys. Res. Lett., 32, L12817, 10.1029/2005GL022582 (2005).
Waters, J. W. et al. The Earth Observing System Microwave Limb Sounder (EOS MLS) on the Aura Satellite. IEEE Trans. Geosci. Remote Sens., 44, 1075–1092 (2006).
Bovensmann, H. et al. SCIAMACHY – Mission Objectives and Measurement Modes. J. Atmos. Sci., 56, 127–150, 10.1175/1520-0469 (1999).
Carslaw, K. S. et al. Stratospheric aerosol growth and HNO3 gas phase depletion from coupled HNO3 and water uptake by liquid particles. Geophys. Res. Lett., 21, 10.1029/94GL02799 (1994).
Shindell, D. T. & de Zafra, R. L. Limits on heterogeneous processing in the Antarctic spring vortex from a comparison of measured and modeled chlorine. J. Geophys. Res., 102, 1441–1449, 10.1029/96JD00519 (1997).
Kirk-Davidoff, D. B., Hintsa, E. J., Anderson, J. G. & Keith, D. W. The effect of climate change on ozone depletion through changes in stratospheric water vapour. Nature, 402, 10.1038/4652 (1999).
Heck, T. J., Grooß, U. & Riese, M. Sensitivity of Arctic ozone loss to stratospheric H2O. Geophys. Res. Lett., 35, L01803, 10.1029/2007GL031334 (2008).
Yang, E. S. et al. first stage of the Antarctic ozone recovery. J. Geophys. Res., 113, D20308, 10.1029/2007JD009675 (2008).
Son, S.-W., Tandon, N. F., Polvani, L. M. & Waugh, D. W. Ozone hole and Southern Hemisphere climate change. Geophys. Res. Lett., 36, L15705, 10.1029/2009GL038671 (2010).
See “Vortex integrity and mixing”, section 3.2.4.2 of the Scientific Assessment of Ozone Depletion 2010, Global Ozone Research and Monitoring Project—Report No. 52, WMO, Geneva, Switzerland (2010).
Eskes, H. J., van Velthoven, P. F. J., Valks, P. J. M. & Kelder, H. M. Assimilation of GOME total ozone satellite observations in a three-dimensional tracer transport model. Q. J. R. Meteorol. Soc., 129, 1663 (2003).
Eskes, H. J. et al. Retrieval and validation of ozone columns derived from measurements of SCIAMACHY on Envisat. Atmos. Chem. Phys. Discuss., 5, 4429–4475, 10.5194/acpd-5-4429-2005 (2005).
van der A, R. J., Allaart, M. A. F. & Eskes, H. J. Multi sensor reanalysis of total ozone. Atmos. Chem. Phys., 10, 11277–11294, 10.5194/acp-10-11277-2010 (2010).
See table 2 of Santee, M. L. et al. Validation of Aura Microwave Limb Sounder HNO3 Measurements. J. Geophys. Res., 112, D24S40, 10.1029/2007JD008721 (2007).
Santee, M. L. et al. Validation of the Aura Microwave Limb Sounder ClO Measurements. J. Geophys. Res., 113, D15S22, 10.1029/2007JD008762 (2008).
Lambert, A. et al. Validation of the Aura Microwave Limb Sounder middle atmosphere water vapor and nitrous oxide measurements. J. Geophys. Res., 112, D24S36, 10.1029/2007JD008724 (2007).
Froidevaux, L. et al. Validation of Aura Microwave Limb Sounder HCl measurements. J. Geophys. Res., 113, D15S25, 10.1029/2007JD009025 (2008).
Santee, M. L. et al. Validation of Aura Microwave Limb Sounder HNO3 Measurements . J. Geophys. Res., 112, D24S40, 10.1029/2007JD008721 (2007).
Froidevaux, L. et al. Validation of Aura Microwave Limb Sounder stratospheric ozone measurements . J. Geophys. Res., 113, D15S20, 10.1029/2007JD008771 (2008).
Schwartz, M. J. et al. Validation of the Aura Microwave Limb Sounder Temperature and Geopotential Height Measurements. J. Geophys. Res., 113, D15S11, 10.1029/2007JD008783 (2008).
Hanson, D. & Mauersberger, K. Laboratory studies of the nitric acid trihydrate: Implications for the south polar stratosphere. Geophys. Res. Lett., 15, 855–858, 10.1029/GL015i008p00855 (1988).
Hyland, D. & Wexler, A. Formulations for the Thermodynamic Properties of the saturated Phases of H2O from 173.15K to 473.15K. ASHRAE Trans, 89 (2A), 500–519 (1983).
Acknowledgements
The authors thank Marc Allaart (KNMI) for his contributions to the calculation of PSC formation temperatures, Holger Vomel at CIRES, University of Colorado (USA) for providing a webpage with a list of available water vapor saturation pressure formula (http://cires.colorado.edu/∼voemel/vp.html) and Ronald van der A, Peter van Velthoven and Peter Siegmund (KNM) for their comments and suggestions. This work was partly financed by the Netherlands Space Office as part of the SCIAVISIE project.
Author information
Authors and Affiliations
Contributions
A.T.J. de Laat and M van Weele both wrote the manuscript with A.T.J. de Laat as the leading author.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareALike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
de Laat, A., van Weele, M. The 2010 Antarctic ozone hole: Observed reduction in ozone destruction by minor sudden stratospheric warmings. Sci Rep 1, 38 (2011). https://doi.org/10.1038/srep00038
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep00038
This article is cited by
-
The sudden stratospheric warming and chemical ozone loss in the Antarctic winter 2019: comparison with the winters of 1988 and 2002
Theoretical and Applied Climatology (2022)
-
Emergence of ozone recovery evidenced by reduction in the occurrence of Antarctic ozone loss saturation
npj Climate and Atmospheric Science (2018)
-
The signs of Antarctic ozone hole recovery
Scientific Reports (2017)
-
Continuous ozone depletion over Antarctica after 2000 and its relationship with the polar vortex
Acta Meteorologica Sinica (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.