Key Points
-
Provides an update on the most common drugs prescribed in primary care by the GMP and the considerations the GDP should take before prescribing.
-
Highlights practical steps the clinician may take to help reduce potentially harmful drug interactions.
-
A useful reminder for the consideration of alternatives to conventional drug therapy.
Abstract
The aim of this article is to explore the diverse and complex nature of pharmacological drug-drug interactions in the general dental practice setting. Using published NHS statistics, this article will highlight medications for common medical conditions that could interact with frequently prescribed drugs by the general dental practitioner.
Similar content being viewed by others
Introduction
A drug interaction may be defined as a situation when a substance, which in this case is another prescribed drug, affects the activity of a drug already taken by a patient. The potential outcome of a drug interaction can vary and could be synergistic (causing an amplification of the intended therapeutic effect of the drug), antagonistic (depressing the therapeutic effect of the drug) or additive (two drugs combining to potentiate either or both actions). All of these outcomes may have potentially damaging or fatal consequences.
Contemporary medical care has advanced significantly in recent years, increasing patients' life expectancy. This is partly due to an increase in therapeutic drug prescribing to help with the management of many common conditions. Published NHS statistics highlighted that during 2012-2013 there were 961 million prescriptions dispensed in the UK, compared with 886 million in 2009.1,13 This increase was primarily due to an increase in the life expectancy of the UK population and the resulting polypharmacy, whereby many patients rely on a combination of drugs to maintain various essential bodily functions and minimise the effects of acquired medical conditions. Further estimates indicate that 75% of patients over the age of 55 are taking medications on a repeat prescription basis.2 Given this gradual increase in dispensed medications, it is almost certain that the general dental practitioner (GDP) will encounter patients taking medications that could potentially interact with those delivered or prescribed in dental practice. As a consequence thought should be given by the GDP before prescribing, to determine whether the potential exists for harmful drug interactions that may have an adverse effect on the patient.
It is the intention of this article to highlight common medical conditions and the drugs frequently associated with them. Furthermore, specific reference will be made to medications that can be prescribed by GDPs, and which could have potentially adverse or damaging consequences.
Drug interaction types
Drug interactions may be categorised broadly into pharmacodynamic or pharmacokinetic interactions:3
Pharmacodynamic interactions
Pharmacodynamic interactions are those that modify the pharmacological effect of a drug without altering its concentration in the tissue fluid.3 This means that the effect of one drug is changed by the presence of another drug at the same molecular site, leading to a summation, or opposition, of the effects of the drugs. These interactions may be summarised into antagonistic, additive or enhanced effects.
Antagonistic effects
This is an interaction between two drugs that have opposing pharmacological effects, leading to a reduced effectiveness of one or both of the drugs administered. A typical example would be for drugs that tend to increase blood pressure, such as non-steroidal anti-inflammatory drugs (NSAIDs); these may inhibit the antihypertensive effect of drugs such as angiotensin-converting enzyme (ACE) inhibitors or diuretics.
Additive and enhanced synergistic effects
When two or more drugs are given with the same pharmacodynamic profiles, the additive effects of the two together may result in an excessive response of the target tissue and thus toxicity. An 'additive synergy' is when the final effect is equal to the sum of the effects of both drugs. When the final effect is greater than additive, it is known as 'enhanced synergy' or potentiation. An example of a synergistic effect would be the simultaneous provision of a benzodiazepine and opioid medication. Both drugs are pharmacodynamically similar and if given together can lead to an increase in the sedative effect on the central nervous system, which may cause respiratory depression.
It is worth mentioning that not all additive effects are harmful. Some drugs are given in combination with others to encourage this additive effect. One such interaction is the anti-microbial co-amoxiclav. Penicillinase-producing bacteria are resistant to penicillin and therefore the combination of clavulanic acid (an inhibitor of penicillinase) with amoxicillin achieves the desired effect.
Pharmacokinetic interactions
Pharmacokinetic interactions are those that alter the concentration of a drug that reaches its site of action. Therefore, one drug alters the concentration of another drug in the system (either an increase or decrease). This modification could occur at any phase of absorption, distribution, metabolism or excretion of the drug.
Absorption
Drugs interacting with the gut lumen, or those that influence gastric motility or emptying can alter the rate of absorption of a second drug. For example, certain antibiotics, such as penicillin-based antimicrobials, can alter the normal gut flora; some of which are vital for the production of vitamin K, leading to a reduction in vitamin K synthesis. Vitamin K is essential for the production of several clotting factors and any deficiency will lead to an increase in the patient's international normalised ratio (INR) and bleeding tendency.4
Other absorption pharmacokinetic interactions could be associated with an alteration of the pH within the stomach. Specific drugs, such as ketoconazole (an antifungal agent), can only be absorbed in acidic conditions. Therefore any drugs that modify the gastric pH, such as antacids (eg calcium carbonate), proton-pump inhibitors (eg omeprazole) or H2-receptor blockers (eg ranitidine), subsequently reduce the absorption of ketoconazole.5
Distribution
Drugs circulating within the bloodstream are distributed in two forms; either bound to circulating plasma proteins (pharmacologically inert or inactive), or the drug is not protein bound and thus free to interact with the target receptor binding sites. The number of protein binding sites within the body is finite and when another drug competes for the same site it will result in an increase in the unbound and active drug.
An example involves warfarin and its interaction with aspirin and other NSAIDs. All these are predominantly plasma protein-bound in circulation and subsequently compete for the same plasma protein-binding site. This leads to more anticoagulant being displaced from the binding site, which in turn leads to an overall increase in the free, and therefore pharmacologically active, warfarin.6 As warfarin is 98–99% plasma protein bound, this normally leaves 1% free to exert its pharmacological effect. Therefore, if an NSAID or aspirin is given and reduces the plasma protein-bound warfarin from 98% to 97%, although that is only an increase in 1% unbound warfarin, it is effectively doubling the concentration of the active drug in the blood, therefore raising the INR.6
Metabolism
This is where there is a decrease in drug efficacy (the desired effect of the drug) due to enzymatic induction by a secondary drug in circulation. One primary site for drug metabolism is the liver, which renders the drug either inactive or alters the structure of the drug to enable it to be excreted via the kidneys. Conversely, the liver or other metabolically active organs such as the brain may increase the efficacy or activate a drug by converting a pro-drug to a more, or fully, active form. This is seen in the case of levodopa, a drug used in the treatment of Parkinson's disease. Levodopa is a pro-drug, and is only converted into active dopamine in the central nervous system after crossing the blood-brain barrier (which is impenetrable to dopamine).7
Various drug-metabolising isoenzymes are involved with drug biotransformation, the most common being cytochrome CYP450. These enzymes can either be inhibited or induced by certain drugs. Examples include rifampicin, which induces CYP450, and leads to increased metabolism of the oral contraceptive pill leading to its failure.8
Excretion
Drugs or metabolites are predominantly excreted via the kidneys. This is also a potential site for drug interactions when two drugs share the same excretion site, reducing the rate of excretion of one or both of the drugs. This means that the active form of the drug is kept in the circulation for longer, effectively prolonging its therapeutic effect with potentially harmful consequences.
Aspirin and other NSAIDs inhibit secretion of methotrexate into the urine. This could potentially lead to harmful methotrexate toxicity and care should be taken when advising patients to take analgesics such as NSAIDs when they are on these drugs.9
Therapeutic index
Although relatively common, not all interactions are clinically significant. Only if the therapeutic range of either drug involved in the interaction is narrow (Fig. 1), meaning that only a small reduction or increase in its effect could lead to a loss of efficacy or potential toxicity, does it become clinically significant.12 The converse is true for drugs that have a wide therapeutic index (Fig. 2). Any change in concentration of a drug with a wide therapeutic index is unlikely to cause adverse effects due to the wider margin of safety. Therefore, the therapeutic index of a drug provides an indication as to how safe that particular drug is. Those with lower indices (such as digoxin) could have potentially more harmful interactions than those with higher indices (eg amitriptyline). Normally, patients who take drugs with lower or narrow therapeutic indices are monitored on a regular basis.
It is beyond the scope of this article to provide details of the biochemical basis of drug interactions and the pharmacological literature3 should be sought for any further understanding. Following a review of the literature it would appear that there are no clear and concise articles exploring the common medical conditions and the drugs usually associated with them. Therefore, this article shall explore if there are any drugs commonly prescribed by GDPs that may interact with those commonly taken by patients.
Common medical conditions and associated drugs
With an ageing population and an increase in polypharmacy, it is likely the GDP will encounter a variety of medical conditions, with patients taking a plethora of drugs. While it is extremely difficult to recognise every condition and its associated medications, and to consider all the interactions to be avoided, an increased knowledge of such problems will undoubtedly be of benefit to practitioners and allied healthcare workers.
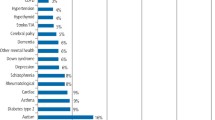
The NHS Information Centre has compiled and published data that identifies and records diseases diagnosed and treated in primary care.11 Table 1 represents 90% of the UK's disease prevalence. However, it is important to remember that more than one of these conditions may be found in the same individual; for example, coronary heart disease is frequently associated with hypertension.12
In addition, data from the Health and Social Care Information Centre (HSCIC) show the levels of drug prescription in relation to one another.13 It is likely that the most common drugs prescribed and dispensed (Table 2) are closely aligned to those diseases diagnosed in primary care settings. Nevertheless, not all diagnosed diseases require pharmacological intervention. For example, type 2 diabetes may only require exercise, diet control and regular review before any medication.14
Table 2 represents the most common drugs that are prescribed in primary care by the general medical practitioner (GMP). The majority of these medications, and therefore patients, are on repeat prescriptions, meaning that they may take these drugs indefinitely. It is therefore important to identify any potentially harmful drug interactions that could occur between these drugs and the ones commonly prescribed in the GDP setting.
Analysis of further data provided by the NHS Information Centre will allow us to compare and contrast the drugs patients are prescribed for medical reasons and those in the 'dental environment', which could precipitate potentially harmful consequences.
Table 3 shows prescription behaviour among GDPs in primary care,15 and represents nearly 98% of the overall prescribing by dentists in primary care in 2011–2012. However, there are medications commonly given in primary care that are not stated here, including the use of all local anaesthetic agents: a recent study showed that lidocaine with adrenaline is the most frequently selected and used form of local anaesthesia in primary care.16
Hypertension and associated anti-hypertensive medications
Statistical information provided by the NHS Information Centre shows that essential hypertension has a prevalence of 13.7% within the UK.5 Antihypertensive medication is one of the most commonly prescribed drugs in primary care, accounting for almost a quarter of all drugs prescribed.15 The most frequently prescribed drugs for the treatment of hypertension are thiazides and loop diuretics, calcium channel blockers (CCBs), ACE inhibitors, β-adrenoreceptor-blocking drugs, α-adrenoreceptor-blocking drugs and angiotensin II receptor antagonists. Furthermore, on many occasions a single antihypertensive drug is not adequate and supplementary antihypertensives are prescribed until the hypertension is controlled. Usually, when two antihypertensive drugs are indicated, an ACE inhibitor or an angiotensin II receptor antagonist can be combined with either a thiazide or a CCB.17 All these drugs have different pharmacological methods of action, and thus different potential harmful interactions.
Clearly, the most common drugs administered by a GDP that could affect these antihypertensive agents are local anaesthetics – typically lidocaine with adrenaline. This is one of many adrenaline-containing local anaesthetics used in primary care that may be implicated in patients who are taking antihypertensives. The most common interaction is between adrenaline and beta-adrenoreceptor blocking drugs (eg atenolol, bisoprolol fumarate). The adrenaline binds to alpha-receptors in the peripheral vasculature causing vasoconstriction, along with an increased heart rate (tachycardia) due to binding with β1-adrenergic receptors on the heart wall.18 Therefore, the overall reaction could be a potentially dangerous increase in heart rate, with associated peripheral vasoconstriction, leading to an increased blood pressure with potentially harmful consequences. To reduce this risk GDPs are advised to use infiltration injections where possible, ensure aspiration is undertaken when administering nerve blocks and keep dosages of lidocaine (with adrenaline) to an absolute minimum. Alternatively, adrenaline-free anaesthetics are available and recommended. However, this is not absolute and it has been reported that adrenaline-containing local anaesthetics are not contraindicated in the hypertensive patient unless the systolic pressure is over 200 mmHg and/or the diastolic is over 115 mmHg.19
NSAIDs are also commonly prescribed or recommended by GDPs, for example post-operatively following extractions or for relief of pulpitic pain. Previous reports and meta-analyses have shown that NSAIDs have the potential to exacerbate pre-existing hypertension and increase mean arterial pressure by as much as 5–6 mmHg in susceptible patients.20,21 NSAIDs act by inhibition of cyclooxygenase (COX)-2 enzymes, resulting in a reduction in synthesis of prostaglandins, some of which are essential in maintaining renal blood flow. Locally synthesised prostaglandins (PGI2, PGE2 and PGD2) cause vasodilation, thus reducing vascular resistance and enhancing renal perfusion.22 Therefore a reduction of prostaglandin synthesis, by inhibition of COX-2 enzymes, leads to less vasodilation and to an increased vascular resistance within the kidney, with an associated reduced renal perfusion.22
The reduction in perfusion of the kidneys causes an increase in urinary sodium retention of 30–50%.23 This antinatriuretic (sodium retention) effect, along with the vasoconstrictor effects of reduced prostaglandin synthesis, ultimately leads to an increase in fluid retention, thereby exacerbating pre-existing hypertension.23 In summary, the hypotensive effects of CCBs, ACE inhibitors, β-adrenoreceptor-blocking drugs, angiotensin II receptor antagonists and loop diuretics are all antagonised by the use of NSAIDs.23
Care must also be taken in prescribing and advising the use of NSAIDs if the patient is taking loop or thiazide diuretics, which comprise 4.0% of overall drug usage (Table 2). NSAIDs interact with diuretics in three different ways: they can cause nephrotoxicity,24 antagonize the diuretic effect and increase the risk of hyperkalaemia.23 The latter could initiate potentially harmful cardiac arrhythmias or even congestive heart failure (CHF). Patients with CHF already have reduced renal perfusion due to a reduced cardiac output and are commonly placed on diuretics. Therefore, when these patients take an NSAID (which leads to a reduced renal perfusion), the overall fluid retention could predispose the patient to dangerous manifestations of CHF.25
One particular study highlighted the potential risk of NSAIDs in CHF, due to these adverse renal effects. During the study period, the results showed that 23.3 per 1,000 hospitalisations for CHF were due to patients using diuretics with concomitant use of NSAIDs.25 The study also highlighted additional co-morbidities, the most significant of which being the patient's age (the older the patient, the higher the risk of adverse effects).
Nevertheless, interactions between diuretics and NSAIDs (for example ibuprofen) usually occur in those patients taking these medications for chronic conditions, meaning that they would have been taking the NSAID medication for an extended period of time. Normally four to five days of treatment with both drugs are needed for any potentially harmful manifestations.26 Therefore, controlled use of ibuprofen for acute dental pain may be used, provided treatment does not extend for longer than four to five days.26
All antihypertensive medications are potentiated by general anaesthesia agents,19 which could lead to potentially dangerous hypotension. A commonly used intravenous sedative in dental practice is midazolam; propofol (administered by a trained anaesthetist) is also used, though less commonly. Reports have demonstrated significant decreases in systolic blood pressure in patients taking a combination of CCBs and angiotensin II receptor blockers when undergoing general anaesthesia.27 However, intravenous sedation appears not to have the same effect and can in fact reduce the hypotensive risks in anxious patients. This is probably due to the increased release of endogenous catecholamine, leading to a provocation of pre-existing hypertension. Nevertheless, care must be taken with respect to both intravenous sedation and general anaesthesia in older patients and hypertensive patients taking CCBs and angiotensin II receptor blockers.
Calcium channel blockers are also a relatively common medication for the treatment of hypertension, accounting for 1.7% of all medications prescribed (Table 2). These drugs have significant potential interactions. One of the more harmful interactions can be with macrolide antimicrobials (eg erythromycin, clindamycin), which comprise 4.0% of all GDP prescribed medication (Table 3). Macrolide antibiotics are inhibitors of the liver isoenzyme P450 3A4, which is responsible for metaboliaing CCBs.28 The result is an increase in the serum concentration of CCBs and a prolonged and dangerous hypotensive effect. Studies have also shown that the isoenzyme is strongly inhibited by clarithromycin and erythromycin, but not by azithromycin.29 To further emphasise the hypotensive risks macrolides can pose, a recent study showed that of 7,100 patients admitted to hospital because of hazardous hypotension while taking CCBs, 176 had been prescribed a macrolide before admission.30 Although a relatively small proportion, GDPs should exercise care before prescribing any macrolide antibiotic in a patients taking CCBs, especially if the patient's age is over 65 years.30
Hypercholesterolemia and lipid-regulating drugs
As shown in Table 2, the most common individual drug prescribed in primary care is simvastatin, a lipid-lowering drug used in the treatment of hypercholesterolaemia.31 Simvastatin accounts for 6.1% of all prescribed drugs and it is likely the GDP will frequently encounter patients who are on this or other statin medications.
Statins are also metabolised by the liver cytochrome P450 enzyme CYP3A4,32 and therefore any drug that inhibits this enzyme risks increasing the serum concentration of statins and subsequently inducing potential adverse effects. The most commonly documented risk is rhabdomyolysis (a form of myopathy),32 which is a condition where skeletal muscle tissue breaks down rapidly, causing elevated levels of creatine kinase (an enzyme normally found in muscle tissue, and which is pathognomic for myopathy). While cohort studies have shown that the documented incidence of statin-related myopathy when taking statins alone is low (0.1–0.2%),33 some medications prescribed by GDPs can interact with statins to increase the potential risk of rhabdomyolysis. A 12-year study examining the risk of rhabdomyolysis showed that among 3,339 reports of statin-associated myopathy, 58% of the cases were associated with concomitant use of a variety of medications including macrolides and azole antifungals.34
Data from Table 3 show that macrolide antibiotics and azole antifungals are relatively common drugs prescribed by the GDP, accounting for approximately 5.0% of medications prescribed. Interestingly, both erythromycin and azole antifungal medications carry warnings regarding potentially harmful interactions with statins. Moreover, there is evidence demonstrating a ten-fold increase in the risk of myopathy when statins are taken with erythromycin.35 There is also a theoretical risk of drug interaction with statins leading to myopathy with concomitant use of miconazole. Although there are no documented cases of simvastatin-miconazole interaction leading to myopathy, the BNF has documented it as a drug interaction leading to potentially harmful myopathy and is best avoided.17
The most commonly reported azole and statin-related interactions are between itraconazole and lovastatin, fluvastatin or atorvastatin.36 There have also been case reports in the literature showing rhabdomyolysis likely due to interaction between fluconazole and simvastatin.37 Although dental prescribing data shows fluconazole prescribing by GDPs is only 0.04%,15 fluconazole is in the dental prescribing armamentarium and should be avoided in patients taking statins.
Cardiovascular disease and haematological medication
Data in Table 1 indicate that strokes or transient ischaemic attacks (TIAs) and coronary heart disease (CHD) have a combined prevalence of 5.1% of the medicated population. The medications commonly prescribed for these include aspirin, clopidogrel and warfarin, collectively comprising 5.6% of all drugs prescribed (Table 2). These can be given independently of one another or in combination depending upon the condition. It is highly likely that the GDP will treat patients taking these drugs and therefore knowledge regarding commonly cited interactions is important. All these drugs have different pharmacological methods of action, therapeutic indices and also different potentially harmful interactions.
Aspirin has antipyretic, anti-inflammatory and anticoagulant actions, the latter via its effect on platelets.38 The antipyretic and anti-inflammatory action means that aspirin can be categorised under non-steroidal anti-inflammatory medications. It acts like other NSAIDs in inhibiting COX enzymes, affecting COX-1 more than COX-2.39 However, unlike other NSAIDs, it does this irreversibly.39 Aspirin also has an antiplatelet function by inhibiting the production of thromboxane, which would otherwise enhance platelet aggregation in order to create the platelet plug, following damage to blood vessels.38 Again, this effect is irreversible.
The BNF recommends a low dose of 75 mg daily to reduce the risk of thromboembolic events as a primary prevention in all patients with established cardiovascular disease.17 However, a recent Cochrane review found that aspirin should not be recommended for primary prevention of cardiovascular disease in patients with hypertension, as the degree of benefit was not found to significantly outweigh the harm due to bleeding.40
One of the main interactions the GDP should be wary of is that NSAIDs (especially diclofenac and ibuprofen) can antagonise the antiplatelet effect of aspirin.41 One potential mechanism of harm with NSAIDs is their pharmacodynamic interaction with aspirin, which prevents irreversible platelet inhibition.41 Aspirin and most other NSAIDs share the same binding site:42 aspirin binds irreversibly to COX-1 thereby reducing the production of thromboxane A2 (a potent platelet-aggregating agent). NSAIDs bind to the same COX-1 site, however, they do so reversibly. Subsequently, both NSAIDs and aspirin are competitive agonists for the same binding site. If the NSAID remains reversibly bound to the COX-1 site until most of the aspirin has been metabolised, the synthesis of thromboxane A2 may resume after subsequent removal of the NSAID. Therefore, NSAIDs may impair the thromboprophylactic action of aspirin.42
One particular study investigated 7,107 patients following their discharge from hospital after admission for problems relating to established cardiovascular disease, all of whom were prescribed low-dose aspirin.43 The study found that, compared with those who used aspirin alone, patients taking aspirin in combination with ibuprofen had an increased risk of cardiovascular mortality.43 Therefore, the study concluded that their findings lend support to the hypothesis that ibuprofen interacts with the cardioprotective effects of aspirin. Furthermore, a recent meta-analysis of randomised controlled clinical trials found that COX-2-specific NSAIDs were indeed associated with an increased risk for vascular events, particularly myocardial infarction.44
Clopidogrel is another antiplatelet drug with relatively frequent use in primary care (Table 2). It is usually given for the prevention of thrombosis after placement of a coronary artery stent, or as an alternative for those patients intolerant to aspirin.45 Clopidogrel interacts significantly with NSAIDs to cause an increased risk of bleeding. One particular study highlighted 4,028 cases of gastrointestinal bleeding following the prescribing of an NSAID in combination with clopidogrel, and noted that it was associated with a greater risk of gastrointestinal bleeding.46
Conversely, the BNF states that the antiplatelet effect of clopidogrel is potentially antagonised by macrolides and azole antifungals, thus reducing its efficacy.17 This is most likely due to the inhibition of the liver cytochrome P450 enzymes involved in converting clopidogrel (a pro-drug) to its active form.
Warfarin and interactions with the dental armamentarium
Warfarin comprises 1.6% of all prescribed medications, and is the most common oral anticoagulant prescribed. Warfarin is used primarily in the treatment and prevention of thromboembolic disease and for a variety of conditions with a specific target INR for each situation. There are two types of oral anticoagulants: coumarins (which include warfarin) and indanediones. Due to their lower incidence of side effects, coumarins are the more common choice of the two.
Warfarin's mode of action is the inhibition of vitamin K-dependent proteins that are involved in the clotting cascade and therefore blood coagulation. The factors targeted are II, VII, IX and X; all of which are synthesised in the liver and require vitamin K as a co-factor for activation.
Although a popular oral anticoagulant, warfarin has important drug interactions that could occur in the dental practice setting, the most significant being with metronidazole. Metronidazole comprises 21.9% of all drugs prescribed by GDPs (Table 3), and thus there is a high potential for an interaction. There have been several reports of significant bleeding in patients taking warfarin with concomitant use of metronidazole.47,48 Warfarin consists of two stereoisomers, S(-) and R(+). It is hypothesised that metronidazole inhibits the metabolism of one of the stereoisomers, leaving the more potent stereoisomer in circulation, and therefore leading to a prolonged and dangerously enhanced anticoagulant effect.49 Interestingly, if metronidazole is unavoidable then current research advises warfarin reduction should be between one-third and one-half of the established patient dose.50 Clearly, liaison with the patient's GMP is essential before any treatment and under no circumstances should the GDP modify the patient's warfarin medication dosage.
Warfarin also has the potential to interact with macrolide antibiotics. Since macrolide antibiotics (more specifically, erythromycin) are inhibitors of the liver enzyme cytochrome P450, which otherwise metabolises warfarin, the overall result is a reduction in warfarin metabolism and thus an enhancement of its effects.51 Evidence suggests that increases in INR are detected in patients who were previously stabilised on warfarin, when simultaneously given a macrolide. Erythromycin produced a marked increase in the hypoprothrombinaemic response to warfarin.51 Therefore, concurrent use should be avoided if possible, with special attention in the elderly, where reduced drug clearance could further prolong any potentially dangerous drug interactions.
Following clinical studies, penicillins have been shown not to interact with warfarin,52 although isolated cases have been reported with broad-spectrum antibiotics. Furthermore, the BNF states that common experience in anticoagulant clinics is that INR can be altered by a course of broad-spectrum antibiotics.17 The most common drug prescribed by the GDP is amoxicillin, which comprises 46.8% of all drugs prescribed (Table 3). Therefore, close attention to detail is important with such a frequently prescribed drug and any potentially harmful interactions that may ensue.
The mechanism by which broad-spectrum penicillins may alter warfarin action is probably via alteration of gut flora. Vitamin K is not only ingested via a normal diet but is also produced by the gut flora and subsequently absorbed.53,54 As mentioned previously, vitamin K is an essential compound in the activation of blood coagulation factors and since warfarin is structurally similar to vitamin K, it binds competitively to these coagulation factors, ultimately having a detrimental effect on the clotting cascade. Nevertheless, there is controversy regarding the importance of this process in the coagulation process.55
Although there is no evidence to suggest that broad-spectrum penicillins do indeed increase INR, it would be sensible to judge each case upon its own merit. If a patient appears under or malnourished, or appears to have an increased tendency to bleeding while on warfarin,55 care must be taken in prescribing amoxicillin as there is potential to alter the serum vitamin K levels and subsequently affect the clotting cascade to increase the efficacy of warfarin.
A further area for drug interactions with warfarin is with concomitant use of azole antifungals. As mentioned previously, miconazole (Daktarin gel) is the most commonly prescribed antifungal by the GDP (Table 3). A survey of GDPs demonstrated that up to one third of practitioners are unaware of contraindications associated with the prescription of azole antifungal agents;56 thus it is important to highlight this potentially harmful interaction. Azole antifungals inhibit CYP450, a vital enzyme in the metabolism of warfarin. Consequently, warfarin remains in its active form for longer and there is decreased clearance of the drug.57 Furthermore, a controlled study examining the interaction between warfarin and miconazole (125 mg per day taken orally over a period of three days) showed a significantly decreased clearance of warfarin. It was stated that the anticoagulation effect was enhanced 5-fold,58 although this was following absorption of miconazole from the gut, rather than the intraoral epithelium. Nonetheless, there are now several reports of oral miconazole gel interacting with warfarin in the systemic circulation, confirming that the gel can be absorbed with potentially harmful interactions.56,58,59,60 The BNF states that miconazole gel is indeed absorbed and that care must be taken regarding potentially harmful interactions that may ensue.17
If there is a risk of this potential drug interaction then polyene antifungals such as nystatin should be considered as suitable alternatives. If miconazole is required for a patient undergoing warfarin treatment, liaison with the patient's GMP and careful monitoring of the INR are essential.56
Selective 5-hydroxytryptamine reuptake inhibitors (SSRIs) and tricyclic antidepressants (TCAs)
Depression accounts for 11.7% of all diagnosed diseases in primary care. According to current data it is therefore the third most commonly diagnosed illness in the UK. Most patients who have a diagnosis of depression do not require medication;61 however, for those that do, SSRIs and TCAs are the most commonly prescribed.62 While SSRIs and TCAs comprise 4.0% of all prescribed medication in the UK, SSRIs are becoming increasingly common as the first choice of treatment in depression, replacing the older and well-established TCAs.63 This is most likely due to fewer side effects and a better profile of safety with a wider therapeutic index compared to TCAs.63 Despite this, amitriptyline is the most widely used tricyclic antidepressant.64
TCAs exert their pharmacological action at synaptic junctions between neurons by blocking the reuptake of noradrenaline at the central synapses. This causes an elevation of the synaptic concentrations of noradrenaline and 5-hydroxytryptamine at cholinergic neurons, and therefore an enhancement of transmission at the neuronal junction.65 The most commonly used drug in dentistry is lidocaine containing adrenaline.16 The BNF highlights an increased risk of hypertension and arrhythmias when TCAs are given with noradrenaline.17 Since TCAs increase synaptic noradrenaline, further administration of adrenaline-containing local anaesthetics could have a significant effect on the cardiovascular system, leading to cardiac arrhythmias and hypertension. A small clinical study, examining the effects of TCAs with concomitant use of adrenaline, found that local anaesthetics used in dentistry could initiate hazardous potentiation of their cardiovascular side effects (arrhythmias and hypertension) in patients receiving TCAs.66 Careful administration of local anaesthesia is advised, with proper aspiration techniques. Furthermore, it is also recommended that no more than three cartridges of adrenaline-containing local anaesthetic should be given in one clinical setting for patients on TCAs.67 Simple procedures like these can reduce the incidence of potentially harmful hypertension or cardiac arrhythmias.
Citalopram, an SSRI, is slowly becoming more popular than TCAs, due to its reduced number of side effects.63 Nevertheless, the most likely side effects to occur with SSRIs in the GDP setting are as a result of their interaction with NSAIDs. It is a well-documented risk that concurrent use of NSAIDs with SSRIs can increase the risk of bleeding.17 When these two classes of drugs are given together there is an increased risk of upper gastro-intestinal bleeding and their concurrent use should be avoided.68 5-hydroxytryptamine is an important component of the clotting cascade and is actively taken up and stored by platelets. When platelets bind, in order to clot, they release the 5-hydroxytryptamine where it functions as a vasoconstrictor, helping regulate haemostasis in the early stages of blood clotting. SSRIs block the reuptake of 5-hydroxytryptamine in platelets,69 therefore preventing this important vasoconstrictor mechanism. Furthermore, NSAIDs exert adverse effects on the gastrointestinal mucosa, putting patients at risk of gastropathy (inflammation and destruction of the gastric lining) with a high prevalence of ulcer formation.70 Due to the risk of gastric damage with use of NSAIDs and the patient's impaired vasoconstrictive ability due to SSRIs, the risk of upper gastrointestinal haemorrhage (UGIH) is serious. A recent meta-analysis, examining the effects of concurrent usage of SSRIs and NSAIDs found that this combination substantially increases the risk of UGIH and should be avoided where possible.71
Conclusion
This article has highlighted some important drug interactions of which the dental practitioner should be aware. These have been targeted towards the most common medical conditions and drugs that patients take for their conditions. Table 4 provides a useful summary of the key drug–drug interactions the GDP is likely to encounter and advice on how to avoid them. Clearly, knowledge of patients' medical and drug histories is essential in order to avoid potentially harmful drug interactions. Consequently, we recommend that not only are patients' medical and drug histories frequently reviewed, but also that active enquiry at every visit is encouraged. This is also important not only to avoid harm to patients but also to avoid unwanted medico–legal proceedings, given an accidental prescription or usage of a drug with a potentially harmful interaction.
Other useful techniques for avoiding harmful concomitant drug interactions include use of alternative drugs where certain medications are contra-indicated.
If there is ever any doubt with regard to a patient's drug history, and whether there could be a potentially harmful drug interaction, then liaising with either the patient's GMP or a specialist in secondary care would provide useful guidance.
The BNF actively requests healthcare professionals to report any suspicious or suspected drug–drug interactions. Pharmacology is a dynamic, developing field and polypharmacy in an ageing population means it is more likely that drug-drug interactions are going to be reported in the literature. It is therefore essential that the dental practitioner update themselves with any new developments in the pharmacological aspects of dentistry.
There are sources of information that provide useful guidance on drug prescribing, and recommendation for dosages. These include the BNF and other useful texts such as the 'Drug Prescribing For Dentistry', a manual produced by the Scottish Dental Clinical Effectiveness Programme (SDCEP).
Given this information, and with continued education and vigilance by the GDP, it is hoped that potentially harmful drug interactions may be avoided and the provision of safe healthcare in an ever increasingly complex population can be maintained.
References
Health and Social Care Information Centre. Prescription cost analysis - England, 2012. HSCIC, 2013.
Seymour R A . Dentistry and the medically compromised patient. Surgeon 2003; 4: 207–214.
Rang H P, Dale M M, Ritter J R, Flower R J, Henderson G . Rang and Dale's pharmacology. 6th ed. UK: Churchill Livingstone, 2012.
Zhang Q, Simoneau G, Verstuyft C et al. Amoxicillin/clavulanic acid-warfarin drug interaction: a randomized controlled trial. Br J Clin Pharmacol 2011; 71: 232–236.
Van der Meer J W, Keuning J J, Scheijgrond H W, Heykants J, Van Cutsem J, Brugmans J . The influence of gastric acidity on the bio-availability of ketoconazole. J Antimicrob Chemother 1980; 6: 552–554.
Seymour R A . Drug interactions in dentistry. Dent Update 2009; 36: 458–470.
Rautio J, Kumpulainen H, Heimbach T et al. Prodrugs: design and clinical applications. Nat Rev Drug Discov 2008; 7: 255–270.
Dickinson B D, Altman R D, Nielsen N H et al. Drug interactions between oral contraceptives and antibiotics. Obstet Gynecol 2001; 98: 853–860.
Hoekstra M, van Ede A E, Haagsma C J et al. Factors associated with toxicity, final dose, and efficacy of methotrexate in patients with rheumatoid arthritis. Ann Rheum Dis 2003; 62: 423–426.
Ritter J, Lewis L, Mant T, Ferro A . A textbook of clinical pharmacology and therapeutics. 5th ed. London: Hodder Arnold, 2008.
Health and Social Care Information Centre. Disease Prevalence Quality and Outcomes Framework (QOF) for April 2011–March 2012, England. QMAS Database.
Weber M A . Coronary heart disease and hypertension. Am J Hypertens 1994; 7: 146S–153S.
Health and Social Care Information Centre. Prescription cost analysis - England, 2007. HSCIC, 2008.
Boulé N G, Haddad E, Kenny G P, Wells G A, Sigal R J . Effects of exercise on glycaemic control and body mass in type 2 diabetes mellitus: a meta-analysis of controlled clinical trials. JAMA 2001; 286: 1218–1227.
The NHS Information Centre, Dental and Eye Care Team, Prescribing Support Unit. Prescribing by Dentists, 2010: England. London: NHS, 2011.
Corbett I P, Ramacciato J C, Groppo F C, Meechan J G . A survey of local anaesthetic use among general dental practitioners in the UK attending postgraduate courses on pain control. Br Dent J 2005; 199: 784–787.
British Medical Association and the Royal Pharmaceutical Society of Great Britain. British national formulary. 64th ed. London: BMJ Publishing Group. 2012.
Goulet J P, Pérusse R, Turcotte J Y . Contraindications to vasoconstrictors in dentistry: Part III. Pharmacologic interactions. Oral Surg Oral Med Oral Pathol 1992; 74: 692–697.
Crispian S . Medical problems in dentistry. 6th ed. London: Churchill Livingstone; 2010.
Pope J E, Anderson J J, Felson D T . A meta-analysis of the effects of nonsteroidal anti-inflammatory drugs on blood pressure. Arch Intern Med 1993; 153: 477–484.
Johnson A G, Nguyen T V, Day R O . Do nonsteroidal anti-inflammatory drugs affect blood pressure? A meta-analysis. Ann Intern Med 1994; 121: 289–300.
Ejaz P, Bhojani K, Joshi V R . NSAIDs and kidney. J Assoc Physicians India 2004; 52: 632–640.
White W B . Cardiovascular risk, hypertension, and NSAIDs. Curr Rheumatol Rep 2007; 9: 36–43.
Swainson C P, Griffiths P . Acute and chronic effects of sulindac on renal function in chronic renal disease. Clin Pharmacol Ther 1985; 37: 298–300.
Heerdink E R, Leufkens H G, Herings R M, Ottervanger J P, Stricker B H, Bakker A . NSAIDs associated with increased risk of congestive heart failure in elderly patients taking diuretics. Arch Intern Med 1998; 158: 1108–1112.
Haas D A . Adverse drug interactions in dental practice: interactions associated with analgesics, Part III in a series. J Am Dent Assoc 1999; 130: 397–407.
Ueno D, Sato J, Nejima J et al. Effects of implant surgery on blood pressure and heart rate during sedation with propofol and midazolam. Int J Oral Maxillofac Implants 2012; 27: 1520–1526.
Henneman A, Thornby K A . Risk of hypotension with concomitant use of calcium-channel blockers and macrolide antibiotics. Am J Health Syst Pharm 2012; 69: 1038–1043.
Periti P, Mazzei T, Mini E, Novelli A . Pharmacokinetic drug interactions of macrolides. Clin Pharmacokinet 1992; 23: 106–131.
Wright A J, Gomes T, Mamdani M M, Horn J R, Juurlink D N . The risk of hypotension following co-prescription of macrolide antibiotics and calcium-channel blockers. CMAJ 2011; 183: 303–307.
Durrington P . Dyslipidaemia. Lancet 2003; 362: 717–731.
Law M, Rudnicka A R . Statin safety: a systematic review. Am J Cardiol 2006; 97: 52C–60C.
Bellosta S, Paoletti R, Corsini A . Safety of statins: focus on clinical pharmacokinetics and drug interactions. Circulation 2004; 109: III53–III57.
Thompson P D, Clarkson P, Karas R H . Statin-associated myopathy. JAMA 2003; 289: 1681–1690.
Spach D H, Bauwens J E, Clark C D, Burke W G . Rhabdomyolysis associated with lovastatin and erythromycin use. West J Med 1991; 154: 213–215.
Lees R S, Lees A M . Rhabdomyolysis from the coadministration of lovastatin and the antifungal agent itraconazole. N Engl J Med 1995; 333: 664–665.
Shaukat A, Benekli M, Vladutiu G D, Slack J L, Wetzler M, Baer M R . Simvastatin–fluconazole causing rhabdomyolysis. Ann Pharmacother 2003; 37: 1032–1035.
Lewis H D, Davis J W, Archibald D G et al. Protective effects of aspirin against acute myocardial infarction and death in men with unstable angina. Results of a Veterans Administration Cooperative Study. N Engl J Med 1983; 309: 396–403.
Brunton L, Chabner B, Knollmann B . Goodman and Gilman's the pharmacological basis of therapeutics. 12th ed. McGraw Hill Medical, 2011.
Lip G Y, Felmeden D C, Dwivedi G . Antiplatelet agents and anticoagulants for hypertension. Cochrane Database Syst Rev 2011; 12: CD003186.
Gladding P A, Webster M W, Farrell H B, Zeng I S, Park R, Ruijne . The antiplatelet effect of six non-steroidal anti-inflammatory drugs and their pharmacodynamic interaction with aspirin in healthy volunteers. Am J Cardiol 2008; 101: 1060–1063.
Schuijt M P, Huntjens-Fleuren HW, de Metz M, Vollaard E J . The interaction of ibuprofen and diclofenac with aspirin in healthy volunteers. Br J Pharmacol 2009; 157: 931–934.
MacDonald T M, Wei L . Effect of ibuprofen on cardioprotective effect of aspirin. Lancet 2003; 361: 573–574.
Kearney P M, Baigent C, Godwin J, Halls H, Emberson J R, Patrono C . Do selective cyclo-oxygenase-2 inhibitors and traditional non-steroidal anti-inflammatory drugs increase the risk of atherothrombosis? Meta-analysis of randomised trials. BMJ 2006; 332: 1302–1308.
Randall M D, Neil KE . Disease management. 2nd ed. London: Pharmaceutical Press, 2004.
Delaney J A, Opatrny L, Brophy J M, Suissa S . Drug–drug interactions between antithrombotic medications and the risk of gastrointestinal bleeding. CMAJ 2007; 177: 347–351.
Kazmier F J . A significant interaction between metronidazole and warfarin. Mayo Clin Proc 1976; 51: 782–784.
Dean R P, Talbert R L . Bleeding associated with concurrent warfarin and metronidazole therapy. Drug Intell Clin Pharm 1980; 14: 864–866.
Rice P J, Perry R J, Afzal Z, Stockley I H . Antibacterial prescribing and warfarin: a review: Br Dent J 2003; 194: 411–415.
Stockley I H . Drug interactions. 5th ed. London: Pharmaceutical Press, 1999.
Sato R I, Grey D R, Brown S E . Warfarin interaction with erythromycin. Arch Intern Med 1984; 144: 2413–2414.
Pharmacy Anticoagulant Clinic Study Group. A multicentre survey of antibiotics on the INR of anticoagulated patients. Pharm J 1996; 257: R30.
Lipsky J J . Nutritional sources of vitamin K. Mayo Clinic Proc 1994; 69: 462–466.
Conly J M, Stein K . Quantitative and qualitative measurements of K vitamins in human intestinal contents. Am J Gastroenterol 1992; 87: 311–316.
Wilson J D . Vitamin deficiency and excess. In Wilson J, Braunwald E (eds) Harrisons principles of internal medicine. 12th ed. pp 441. New York: McGraw-Hill, 1991.
Pemberton M N, Oliver R J, Theaker E D . Miconazole oral gel and drug interactions. Br Dent J 2004; 196: 529–531.
O'Reilly R A, Goulart D A, Kunze K L et al. Mechanisms of the stereoselective interaction between miconazole and racaemic warfarin in human subjects. Clin Pharmacol Ther 1992; 51: 656–667.
Pemberton M N, Sloan P, Ariyaratnam S, Thakker N S, Thornhill M H . Derangement of warfarin anticoagulation by miconazole oral gel. Br Dent J 1998; 184: 68–69.
Pillans P, Woods D J . Interaction between miconazole oral gel (Daktarin) and warfarin. NZ Med J 1996; 109: 346.
Evans J, Orme D S, Sedgwick M L, Youngs G R . Treating oral candidiasis: potentially fatal. Br Dent J 1997; 182: 452.
Regier D A, Narrow W E, Rae D S, Manderscheid R W, Locke B Z, Goodwin F K . The de Facto US Mental and Addictive Disorders Service System. Epidemiologic catchment area prospective 1-year prevalence rates of disorders and services. Arch Gen Psychiatry 1993; 50: 85–94.
Olfson M, Marcus S C, Druss B, Elinson L, Tanielian T, Pincus H A . National trends in the outpatient treatment of depression. JAMA 2002; 287: 203–209.
Anderson I M . Selective 5-hydroxytryptamine reuptake inhibitors versus tricyclic antidepressants: a meta-analysis of efficacy and tolerability. J Affect Disord 2000; 58: 19–36.
Barbui C, Hotopf M . Amitriptyline v. the rest: still the leading antidepressant after 40 years of randomised controlled trials. Br J Psychiatry 2001; 178: 129–144.
Tatsumi M, Groshan K, Blakely R D, Richelson E . Pharmacological profile of antidepressants and related compounds at human monoamine transporters. Eur J Pharmacol 1997; 340: 249–258.
Boakes A J, Laurence D R, Teoh P C, Barar F S, Benedikter L T, Prichatd B N . Interactions between sympathomimetic amines and antidepressant agents in man. BMJ 1973; 1: 311–315.
Hersh E V, Moore P A . Adverse drug interactions in dentistry. Periodontol 2000 2008; 46: 109–142.
de Jong JC, van den Berg P B, Tobi H, de Jong-van den Berg LT . Combined use of SSRIs and NSAIDs increases the risk of gastrointestinal adverse effects. Br J Clin Pharmacol 2003; 55: 591–595.
Cooper T A, Valcour V G, Gibbons RB, O'Brien-Falls K . Spontaneous ecchymoses due to paroxetine administration. Am J Med 1998; 104: 197–198.
Singh G . Recent considerations in nonsteroidal anti-inflammatory drug gastropathy. Am J Med 1998; 105: 31S–38S.
Loke Y K, Trivedi A N, Singh S . Meta-analysis: gastrointestinal bleeding due to interaction between selective 5-hydroxytryptamine uptake inhibitors and non-steroidal anti-inflammatory drugs. Aliment Pharmacol Ther 2008; 27: 31–40.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dawoud, B., Roberts, A. & Yates, J. Drug interactions in general dental practice – considerations for the dental practitioner. Br Dent J 216, 15–23 (2014). https://doi.org/10.1038/sj.bdj.2013.1237
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bdj.2013.1237
This article is cited by
-
Drug: Fraught interactions
British Dental Journal (2014)