Abstract
Soil phosphorus (P) fractions are critical for understanding soil P dynamics and availability. This paper provides a global dataset of soil P fractions separated by the Hedley method. The dataset also includes key environmental factors associated with soil P dynamics and availability, including climate factors, vegetation, soil and parent material types, soil age, and soil physiochemical properties such as particle size, bulk density, pH in water, organic carbon, total nitrogen, and extractable iron and aluminium concentrations. This dataset includes measures of Hedley P fractions of 802 soil samples and was gathered through a literature survey of 99 published studies. Plant availability of each soil P fraction was noted. We anticipate that the global dataset will provide valuable information for studying soil P dynamics and availability, and it will be fused into earth system models to better predict how terrestrial ecosystems will respond to global environmental changes.
Design Type(s) | systematic review study design • database creation objective • data integration objective |
Measurement Type(s) | phosphorus content |
Technology Type(s) | data item extraction from journal article |
Factor Type(s) | |
Sample Characteristic(s) | Brazil • Switzerland • French Republic • United States of America • Ethiopia • Canada • Colombia • Chile • Sri Lanka • Argentina • Indonesia • Germany • Kingdom of Spain • Costa Rica • Tanzania • Puerto Rico • Mexico • Malaysia • Cameroon • Peru • Nigeria • New Zealand • Australia • Panama • Japan • China • Sweden • Senegal • Republic of South Africa • Finland • Israel • Nepal • Jamaica • Pakistan • Bolivia • soil |
Machine-accessible metadata file describing the reported data (ISA-Tab format)
Similar content being viewed by others
Background & Summary
Phosphorus (P) is a key limiting nutrient of plant growth and soil microbial activity. Atmospheric P input is extremely low (<0.1 kg P ha yr−1) in most global land areas1. Therefore, soil is typically the major source of P to plants and soil microbes in terrestrial ecosystems2,3. Soil P supply or availability plays a vital role in determining the structures, functions, and processes of terrestrial ecosystems2,4–7. For example, insufficient soil P supply accounts for P limitation on plant production in terrestrial ecosystems worldwide8,9. The growth of different plant or soil microbe species varied in their dependencies on soil P supply6. Soil P supply is, therefore, an important driver of the composition and diversity of plant and soil microbial communities6,10. Since low soil P supply can limit soil microbial activity11, soil P supply also regulates litter decomposition and soil organic C dynamics7,12,13. Improved understanding of soil P dynamics and availability is critical for better understanding of nutrient regulation on key ecosystem properties in terrestrial ecosystems.
Total P stock of soil is always large compared to that of vegetation P stock14,15. However, soil P supply is rarely adequate in meeting the P demands of plants in terrestrial ecosystems9,16. This is largely due to multiple forms of P existing in the soils, which differ in their availability for plant uptake across time scales17,18. Soil P availability is controlled by sorption/desorption, precipitation/dissolution, immobilization/mineralization, weathering, and solid-phase P transformations such as solid-phase diffusion or penetration, recrystallization, and migration in aggregates2,19–22. A detailed investigation of P dynamics and bioavailability requires the separation and identification of different forms of P in soils23. The method of Hedley, et al.17 and its modification by Tiessen and Moir24 (Fig. 1) are the most commonly used procedures for the sequential fractionation of P in soils. These procedures enable the characterization of different inorganic P (Pi) and organic P (Po) fractions based on their solubility. Investigating changes in these soil P fractions along environmental (e.g. climate) gradients can provide important insights into environmental controls over long-term soil P dynamics and availability25–30.
The flow chart follows the procedures of Hedley, et al.17 and Tiessen and Moir24. Microbial biomass P estimates in Hedley, et al.17 had not been in common use and therefore was not included in the flow chart. “Sonicate and extract with 0.1 M NaOH” was available only in the procedure of Hedley, et al.17; “Extract with hot concentrated HCl” was available only in the procedure of Tiessen and Moir24. Soil P pools were assigned according to previous studies25,30–33,38.
Several studies have summarized data of Hedley P fractions in worldwide natural soils25,31,32. In 1995, Cross and Schlesinger25 compiled Hedley P fractions of 88 natural, unfertilized or uncultivated soil samples worldwide from 16 published studies. Based on this dataset, the authors explored changes in soil P dynamics across a soil weathered gradient. In 2011, Yang and Post32 updated this global dataset to 178 soil samples from 28 published studies, to further explore soil P dynamics along a pedogenesis gradient. Based on data of the 178 soil samples, Yang et al.33 provided the first spatially explicit estimates of different forms of P in 0–50 cm mineral soils at the global scale in 2013. These estimates (usually only labile P, sometimes in combination with a soil P dynamics model) have been used as an index of soil P supply in a few earth system models to diagnose P limitation on plant growth at the global scale34–37. However, a large uncertainty in P limitation has been found in these studies, largely due to a deficiency in the number of soil P fraction measurements34–37. In 2015, Hou et al.31 updated this global dataset to 626 soil samples from 85 published studies, to test hypothetical models of soil P transformations. In 2017, Hou et al.30 further updated this global dataset to 802 soil samples from 98 published studies and one unpublished study (i.e., ‘Hou et al., unpublished’), to examine climate effects on soil P cycle and availability. The ‘Hou et al., unpublished’ was published recently29. Therefore, our global database now included 802 soil samples from 99 published studies.
Here, we provide a detailed description of the global database for use in further studies. Besides data of Hedley P fractions, the dataset also contained various environmental factors that potentially affect soil P dynamics and availability, such as mean annual temperature (MAT), mean annual precipitation (MAP), soil pH, organic C and extractable iron (Fe) and aluminium (Al) concentrations. This global dataset is expected to permit detailed analysis of the controls of environmental factors, e.g. climate and soil pH, on soil P dynamics and availability30. It can also be incorporated into earth system models7,34–37, to constrain the prediction of climate effect on primary productivity and C storage in global terrestrial ecosystems. Our dataset is about four times as large as that of Yang and Post32 in terms of both soil number and publication number. In particular, our dataset included much more data from Asia (269), Africa (40), and Oceania (20) which were underrepresented previously (3, 4, and 0, respectively).
Methods
Soil P fractionation procedure
Hedley P fractionation procedure and its modifications are designed to indicate soil P pools17,24,25. The terminology of Hedley P fractions have been different in different studies25. Some consensus, however, has been reached during the last three decades25. In general, the resin Pi fraction represents the soil solution or soluble Pi pool, which can be immediately accessed by plants31,38. If depleted, the soluble Pi will be replenished by solid-phase Pi pools via desorption, dissolution, or solid-phase P transformation and by solid-phase Po pools via mineralization20,22. The HCO3 Pi fraction is considered a labile Pi pool that can be released by ligand exchange with the bicarbonate ion; this Pi pool is available to plants and persists for only short periods, e.g., a growing season25,32. The HCO3 Po fraction represents a labile Po pool that can be utilized by plants after being mineralized22. The OH P (Pi and Po) fractions index moderately labile P (Pi and Po) pools that are bound with amorphous and some crystalline Al and Fe22, with low availability to plants22,25. The dilute HCl Pi fraction indexes a primary mineral P pool that is bound with calcium (Ca) and that can be utilized by plants after it is released by weathering25,31. Other P fractions such as residual P (Fig. 1) usually indicate the occluded P pool that is least available to plants due to their particularly low solubility23,24,39.
A summarized description of the procedures of Hedley, et al.17 and Tiessen and Moir24 was shown in Fig. 1 and also as follows:
1. Resin extract: weigh 0.5 g air-dried soil into a 50 ml centrifuge tube, add 2 resin strips (in HCO3− form) + 30 ml deionized water, and shake 16 h. Remove resin strips from the tube. Then place resin strips in a clean 50 ml centrifuge tube, add 20 ml 0.5 M HCl, set aside for 1 h, and collect the 0.5 M HCl extract for P measurement. Centrifuge the tube with soil suspension, discard the supernatant, and keep the soil for further extractions.
(In the Hedley procedure, duplicate 0.5 g air-dried soils are prepared; one of the soil samples is added with 1 ml CHCl3 for the determination of soil microbial biomass P. However, this procedure has rarely been used by later studies. In our database, only 3 of the 41 measurements of soil microbial biomass P were determined according to the Hedley procedure.)
2. HCO3− extract: add 30 ml 0.5 M NaHCO3 at pH 8.5 to the soil, shake 16 h, centrifuge, filter (<0.45 mm) and collect the supernatant for P measurement, and keep the soil for further extractions.
3. OH− extract: add 30 ml 0.1 M NaOH to the soil, shake 16 h, centrifuge; filter (<0.45 mm) and collect the supernatant for P measurement, and keep the soil for further extractions.
(In the Hedley procedure, a second extraction with 30 ml 0.1 M NaOH and sonication is used after step 3. This procedure has been, however, usually absent in later studies, e.g. Paré and Bernier40.)
4. HCl extract: add 30 ml 0.1 M HCl to the soil, shake 16 h, centrifuge; filter (<0.45 mm) and collect the supernatant for P measurement, and keep the soil for the following digestion.
(An important modification of the Hedley procedure by Tiessen and Moir24 is removing the second OH− extraction in the Hedley procedure but adding an extraction of 10 ml hot (at 80 °C) concentrated HCl after step 4.)
5. Residual fraction: Digest the soil with 5 ml concentrated H2SO4 and H2O2, filter (<0.45 mm) and collect the solution for P measurement.
Inorganic P in all the above extracts are determined using the molybdate blue method41. Organic P of the HCO3− extract, OH− extract, and second OH− extract or hot conc. HCl extract are calculated as the difference between total P determined after persulphate digestion42 and inorganic P. Phosphorus in the resin and HCl extracts are usually considered to be totally in inorganic form22.
Literature search
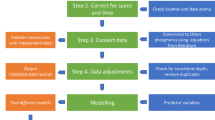
In general, we compiled a database of soil P fractions by surveying the peer-reviewed, published research that used the sequential fractionation techniques developed by Hedley, et al.17 and modified by Tiessen and Moir24. Our survey was restricted to studies of unfertilized, uncultivated, and (semi-) natural soils. We defined (semi-) natural soils as those in sites with primary vegetation or with a stand age greater than 10 years for forests, which is consistent with a study of Hedley P fractions in tropical soils26. Our survey included papers published as recently as April 2017 except one study published in 2018 (ref. 29). We collected data of Hedley P fractions in soils at all reported depths in various habits/landscapes (e.g. forest lands, grasslands, and savanna lands). There is no additional criteria for studies to be included. In general, we collected data in four steps, as summarized in Fig. 2 and described in detail as follows:
Yang and Post32 comprehensively surveyed peer-reviewed published research that reported values of Hedley P fractions (following the procedure of Hedley, et al.17 or Tiessen and Moir24) in worldwide unfertilized, uncultivated, and natural soils before 2010 (data of 178 soil samples from 28 studies). Gama-Rodrigues, et al.26 collected data of Hedley P fractions in tropical soils using the similar method (data of 81 soil samples from 23 studies). To simplify our survey of early studies (published before 2010), we resurveyed papers listed in these studies. It is noted that these two studies collected data of only surface soils (mostly at a mineral depth of 0–15 cm). Since deep (>15 cm) soils are important components of terrestrial ecosystems that can intensively interact with surface soils43, we collected data of Hedley P fractions in soils at all reported depths. (1) The resurvey of published papers referred by Yang and Post32 resulted in a dataset of 294 soil samples from 26 published papers. (2) The resurvey of published papers referred by Gama-Rodrigues, et al.26 resulted in a dataset of 72 soil samples from 16 published papers.
And then, (3) we comprehensively surveyed all peer-reviewed papers that cited Hedley, et al.17 or Tiessen and Moir24 and published during the period of 2010 to April, 2017 on Google Scholar. An exception was a study published in 2018 (ref. 29). We did the survey by reading the title, abstract, and/or the full text of each of the papers. During this literature survey, data from 408 additional soil samples in 50 additional studies were collected. Finally, (4) we comprehensively surveyed peer-reviewed papers published in Chinese before April, 2017, using keywords of “soil” and “phosphorus fraction” (in Chinese) on the website of Chinese National Knowledge Infrastructure (CNKI, website: http://www.cnki.net/). Since the database of CNKI is not well linked to English journals, we surveyed papers in Chinese using keywords rather than tracing the citations of Hedley, et al.17 or Tiessen and Moir 24. During this literature surveying, data from 28 soil samples in 7 studies were collected.
In total, we collected data of Hedley P fractions in 802 natural soil samples from 99 published studies. In our database, all data were collected at the plot scale. For data with sample replicates in the same plots, the average values per plot were calculated and used. Typically, there is no analytical duplicate for Hedley P fractionation. Some descriptions and analyses of this database or its sub-databases were given in previous studies30,31.
Data Records
The database file is in xlsx format and the reference list in pdf format. Both files were archived in PANGAEA (Data Citation 1). Blank denotes missing data. The database included both raw data from the published studies and the data derived from global maps or recalculated by the authors (Fig. 2).
Raw data compiled from the published studies are listed as follows in the format of ‘variable name (location in the database; unit): variable description’:
Code (column 1 of the database (C1 in abbreviation, the same below)): label of the soil sample.
Reference (C2): the referred studies
Country (C3): the country where the study site located
Site (C4): name of the site where the study performed
Latitude (C5; −43.25 to 69.35): in decimal degrees
Longitude (C6; −117.86 to 171.58): in decimal degrees
MAT (C7; °C): mean annual temperature
MAP (C8; mm yr−1): Mean annual precipitation
Elevation (C9; m, a.s.l.)
Slope (C10; o): site slope with unit of degree or percentage
Vegetation type (C11): as described in the referred study, mostly of forest and grass.
Stand age (C12; yr): stand age of forest ecosystems. It is either a specific stand age (e.g. 20) or a description such as ‘Native’ or ’Primary’ forest.
Parent materials (C13): as described in the referred study.
Soil type (C14): mostly classified according to the soil classification system of the country where the study performed.
Soil classification system (C15): soil classification systems used to define soil types in the referred studies.
Soil age (C16; yr)
Soil note (C17): label of the soil, as described in the referred studies
Soil horizon (C18): either a range of soil depth (e.g. 0–15 cm) or a description of soil horizon (e.g. A horizon)
Water Pi (C19; mg kg−1): some studies modified Hedley procedure by replacing resin extraction with water or KCl extraction, e.g. Vu, et al.44.
Resin Pi (C20; mg kg−1)
HCO3 Pi (C21; mg kg−1)
HCO3 Pi2 (C22; mg kg−1): some studies modified Hedley procedure by extracting P from soils firstly with 0.5 M NaHCO3 at pH 8.5 (i.e. without a resin extract), e.g., Lilienfein, et al.45, or reported only the sum of, but not the individual values of, the HCO3 Pi fraction and the resin (or water) Pi fraction, e.g., Satti, et al.46.
HCO3 Po (C23; mg kg−1)
OH Pi (C24; mg kg−1)
OH Po (C25; mg kg−1)
HCl Pi (C26; mg kg−1)
Sonic Pi (C27; mg kg−1)
Sonic Po (C28; mg kg−1)
CHCl Pi (C29; mg kg−1)
CHCl Po (C30; mg kg−1)
Residual P (C31; mg kg−1)
Total Po (C32; mg kg−1): total organic P measured separately
Sum of P fractions (C33; mg kg−1): sum of all Hedley P fractions, generally equal to soil total P
Soil total P (C34; mg kg−1): measured separately using a digestion method47
Comment (C35): Notes about soil P fractions
pH (C36): soil pH in water
TOC (C37; %): soil total organic carbon
TN (C38; %): soil total nitrogen
DCB_Al (C39; mg kg−1): dithionite-citrate-bicarbonate extractable soil Al48
DCB_Fe (C40; mg kg−1): dithionite-citrate-bicarbonate extractable soil Fe48
Oxa_Al (C41; mg kg−1): oxalate extractable soil Al48
Oxa_Fe (C42; mg kg−1): oxalate extractable soil Fe48
DBD (C43; g cm−3): soil bulk density
Texture (C44): soil texture as described in the referred studies (e.g. coarse loamy)
Sand (C45; %): soil sand content (diameter between 0.05 mm and 2.00 mm)
Silt (C46; %): soil silt content (diameter between 0.002 mm and 0.05 mm)
Clay (C47; %): soil clay content (diameter<0.002 mm)
Method comment (C48): method for the determination of soil particle size, mostly with the pipette or hydrometer method49.
MBP (C49; mg kg−1): soil microbial biomass P; a total of 41 values, 38 of which had been separately determined by a fumigation-extraction method50 rather than as a fraction of the Hedley procedure17
Reorganized data by the authors as follows:
Latitude2 (C50) and Longitude2 (C51): in decimal degrees. In cases where the referred studies did not report the latitude or longitude of the measurement, the approximate latitude or longitude were derived by geocoding site name in Google Earth 7.0
MAT2 (C52); MAP2 (C53), Elevation2 (C56): In cases where the referenced studies did not report MAT, MAP, or elevation, the values were derived from WorldClim51 using site geographic location (i.e., latitude and longitude)
Aridity index (C54): aridity index that was derived from CGIAR-CSI52 using site geographic location
Soil type2 (C56): soil type classified according to the USDA soil classification system53. For soil types that were initially not classified according to the USDA soil classification system, they were reclassified according to the USDA soil classification system by referring the descriptions in published studies through searching the soil type described by the referred study (e.g. ‘Ferric Acrisol’) and ‘USDA’ in Google Scholar.
Parent material2 (C57): parent materials grouped mainly according to Porder and Ramachandran54, except glacial till and volcanic ash which were treated as two separate groups
Vegetation type2 (C58): vegetation type grouped into seven groups, i.e. forest, shrub, savanna, grass, meadow, pasture, tundra
Slope2 (C59; o): soil slope with unit expressed in degree. Site slope expressed in percentage was transformed to data in degree
Depth_soil (C60; m): soil depth ranges were recoded into average value (e.g., ‘0–15 cm depth’ was recoded as ‘0.075’)
Depth_note (C61): soils of organic layer or mineral layer classified according to soil genesis
Code (C62): 0 indicates organic layer; 1 indicates averaged soil depth between 0 and 10 cm; 2 indicates averaged soil depth between 10 cm and 20 cm; 3 indicates averaged soil depth >20 cm; 4 indicates mineral soil at unknown soil depth; 5 indicates unknown soil horizon
Total P2 (C63): soil total P; mostly of the sum of P fractions; if sum of P fractions was not given or can't be calculated from available data, separately measured soil total P was used
Labile Pi or available P (C64; mg kg−1): sum of HCO3 Pi (C20) and resin Pi (C19)/water Pi (C18), or HCO3 Pi2 (C21)
Organic P (C65; mg kg−1): calculated as the sum of HCO3 Po (C22) and OH Po (C24)
Primary P (C66; mg kg−1): primary mineral P, which was HCl Pi (C25)
Secondary P (C67; mg kg−1): secondary mineral P, which was OH Pi (C23)
Occluded P (C68; mg kg−1): the sum of residual P (C30), sonic Pi (C26), and sonic Po (C27) obtained by the Hedley procedure; the sum of residual P (C30), CHCl Pi (C28), and CHCl Po (C29) obtained by the Tiessen and Moir procedure; or the difference between total P (C62) and the sum of resin Pi (C19), HCO3 Pi (C20) and HCO3 Po (C22), OH Pi (C23) and OH Po (C24), and HCl Pi (C25) obtained by the studies in which neither a second OH− extract nor a hot conc. HCl extract was included.
Data overview
Sites in our database were located on all continents except Antarctica (Fig. 3). The database spanned over 112o in latitude (43.3oS–69.4oN; Table 1 and Fig. 3). MAT ranged from -7.1°C to 29.0°C. MAP ranged from 31 to 6000 mm yr−1. Elevation ranged from 11 m to 4235 m. Average soil depth ranged from 1 cm to 450 cm. Soil pH in water ranged from 3.2 to 9.5. Soil P fractions generally varied over three orders (Table 1).
Among the 802 soil samples, values for the sonic Pi, sonic Po, CHCl Pi, and CHCl Po fractions were missing for about 85% (84.2–85.3%) of the samples (Table 1). This was partly because studies that used the procedure of Hedley, et al.17 did not have values of the CHCl Pi, and CHCl Po fractions, and studies that used the procedure of Tiessen and Moir24 did not have values of the sonic Pi and sonic Po fractions. Moreover, some studies, e.g. Vu, et al.44, modified the procedure of Hedley, et al.17 by omitting the extract of second 0.1 M NaOH and sonication. Values for the resin Pi and HCO3 Pi fractions were missing for 32.3 and 26.1%, respectively, of the samples. Data were missing for these two fractions partly because the resin Pi fraction was not separated from the HCO3 Pi fraction (e.g., Lilienfein, et al.45) (Table 1). Data were missing for the resin Pi fraction also because resin was replaced by deionized water or KCl solution to extract the most soluble P pool in some studies (e.g., Vu, et al.44) (Table 1). For the other P fractions, data were missing mainly because the specific P fraction value (Pi or Po) was not indicated (e.g., HCO3 Po in Garcia-Montiel, et al.55). For other parameters in the database, data were missing either because the values were not indicated or because the measurement method did not fulfill our survey requirements. Missing data would not hinder the use of our dataset by most researchers, as shown in our previous studies30,31. Missing data may be either deleted or filled using multiple imputation methods before statistical analyses. The dataset may be also analyzed with statistical methods that can deal with missing data such as boosting regression tree.
Technical Validation
A test of relationships between data of climate and altitude reported in the referred studies and those derived from WorldClim (mean annual temperature: r=0.95, P<0.001, n=407; mean annual precipitation: r=0.85, P<0.001, n=459; elevation: r=0.88, P<0.001, n=328) indicates that the derived data from WorldClim were generally reliable for our study sites.
Usage Notes
The availability of P in soil to plant is strongly time-dependent18,22,56. Definition of the availability of a soil P fraction to plant is also time-dependent22,25. Here, we provide some advices for the definition of plant available and unavailable P in soils derived by Hedley fractionation, as summarized in some previous studies22,25,31. The resin Pi and HCO3 Pi fractions function similarly in soils25,32, with turnover times likely of a few days18,57; therefore the two P fractions can be always defined as plant available P25,32. There is probably a continuum of solubility among the resin Pi, HCO3 Pi, and OH Pi fractions31,58. However, the OH Pi fraction turnovers more slowly than the resin Pi and HCO3 Pi fractions, which have a likely turnover time of months18. Therefore, the OH Pi fraction may be available to plant in months or over longer terms18,56. Similar to the Pi fractions, there is also a continuum of solubility between the HCO3 Po and OH Po fractions, with the former having a somewhat faster turnover than the latter one22,31. The HCO3 Po fraction may be considered as soil available P in weeks or longer terms25; while the OH Po fraction could be also available to plants in seasons or longer terms56,59. The HCl Pi fraction is typically slow-changing60 and can be available to plants in decades or longer terms31,61. The sonic Pi, sonic Po, conc. HCl Pi, conc. HCl Po, and residual P fractions all turnover slowly in soils25,59, but their roles (either as a source or as a sink of soil available P) in controlling soil P availability should be considered in decades or longer terms31,61. Finally, it’s noted that the same soil P fraction is not of equal availability to plants in all soils56, but is influenced by soil conditions (e.g. weathered extent)56, plant species62, and environmental conditions (e.g. temperature and precipitation)30.
Hedley P fractions are usually grouped according to the similarity of their functions and chemical natures, to simplify statistical analysis and/or facilitate data interpretation. Here, we have several suggestions inline with this. (1) Sum of the resin Pi fraction and the HCO3 Pi fraction may be used as an index of labile inorganic P or available P, as frequently used in some previous studies18,25,30. This is because resin used for the Hedley P fractionation is typically in HCO3− form, which extract P from soil in a similar manner (i.e. through ion exchange) as 0.5 M NaHCO3 (pH 8.5)25. Functional similarity between the resin Pi fraction and the HCO3 Pi fraction was also suggested by the close relationship between them found in previous studies31. (2) Sum of the HCO3 Po fraction and the OH Po fraction, and also the second OH Po fraction or the CHCl Po fraction if available, may be used as an index of soil organic P. (3) To reconcile the difference in defining the residual P fraction among publications25, a measure of occluded P, recalcitrant P, or residual P may be calculated in one of the three following ways31: the sum of residual P, sonic Pi, and sonic Po fractions obtained by the Hedley procedure; the sum of residual P, conc. HCl Pi, and conc. HCl Po fractions obtained by the Tiessen and Moir procedure; or the difference between total P and the sum of resin Pi, HCO3 Pi and Po, OH Pi and Po, and HCl Pi fractions.
Soils in our database varied largely in their depths, of which half had an average soil depth ≤ 10 cm (Table 1). Biogeochemistry-climate models typically rely on the properties of soils with the same depth (e.g. 0-50 cm)34–37. Soil P fractions in our database need to be unified before its usage by biogeochemistry-climate models. One possible way to do this is recalculating the soil P fraction values using the empirical relationships between soil depth and soil P fractions.
A full list of references used to build our database is given in the References section25,29,40,44,45,46,55,59,63–153.
Additional information
How to cite this article: Hou, E. et al. A global dataset of plant available and unavailable phosphorus in natural soils derived by Hedley method. Sci. Data 5:180166 doi: 10.1038/sdata.2018.166 (2018).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
References
Mahowald, N. et al. Global distribution of atmospheric phosphorus sources, concentrations and deposition rates, and anthropogenic impacts. Global Biogeochem. Cy 22, GB4026 (2008).
Vitousek, P. M., Porder, S., Houlton, B. Z. & Chadwick, O. A. Terrestrial phosphorus limitation: mechanisms, implications, and nitrogen-phosphorus interactions. Ecol. Appl. 20, 5–15 ( (2010).
Chadwick, O. A., Derry, L. A., Vitousek, P. M., Huebert, B. J. & Hedin, L. O. Changing sources of nutrients during four million years of ecosystem development. Nature 397, 491–497 (1999).
Penuelas, J. et al. Human-induced nitrogen-phosphorus imbalances alter natural and managed ecosystems across the globe. Nat. Commun 4, 2934 (2013).
Reed, S. C., Yang, X. & Thornton, P. E. Incorporating phosphorus cycling into global modeling efforts: a worthwhile, tractable endeavor. New Phytol. 208, 324–329 (2015).
Turner, B. L. Resource partitioning for soil phosphorus: a hypothesis. J. Ecol. 96, 698–702 (2008).
Wang, Y. P., Law, R. M. & Pak, B. A global model of carbon, nitrogen and phosphorus cycles for the terrestrial biosphere. Biogeosciences 7, 2261–2282 (2010).
Augusto, L., Achat, D. L., Jonard, M., Vidal, D. & Ringeval, B. Soil parent material - a major driver of plant nutrient limitations in terrestrial ecosystems. Global Change Biol. 23, 3808–3824 (2017).
Elser, J. J. et al. Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol. Lett. 10, 1135–1142 (2007).
Zemunik, G., Turner, B. L., Lambers, H. & Laliberté, E. Diversity of plant nutrient-acquisition strategies increases during long-term ecosystem development. Nat. Plants 1, 15050 (2015).
Camenzind, T., Hättenschwiler, S., Treseder, K. K., Lehmann, A. & Rillig, M. C. Nutrient limitation of soil microbial processes in tropical forests. Ecol. Monogr. 88, 4–21 (2017).
Hou, E., Chen, C., McGroddy, M. E. & Wen, D. Nutrient limitation on ecosystem productivity and processes of mature and old-growth subtropical forests in China. PLoS ONE 7, e52071 (2012).
Mori, T. et al. Effects of phosphorus addition on nitrogen cycle and fluxes of N2O and CH4 in tropical tree plantation soils in Thailand. Agr. Nat. Res 51, 91–95 (2017).
Smil, V. Phosphorus in the environment: natural flows and human interferences. Annu. Rev. Energ. Env 25, 53–88 (2000).
Imai, N., Kitayama, K. & Titin, J. Distribution of phosphorus in an above-to-below-ground profile in a Bornean tropical rain forest. J. Trop. Ecol. 26, 627–636 (2010).
Aerts, R. & Chapin, F. S. The mineral nutrition of wild plants revisited: a re-evaluation of processes and patterns. Adv. Ecol. Res 30, 1–67 (1999).
Hedley, M., Stewart, J. & Chauhan, B. Changes in inorganic and organic soil phosphorus fractions induced by cultivation practices and by laboratory incubations. Soil Sci. Soc. Am. J 46, 970–976 (1982).
Chen, C. R. et al. Characterization of phosphorus availability in selected New Zealand grassland soils. Nutr. Cycl. Agroecosys 65, 89–100 (2003).
Barrow, N. A mechanistic model for describing the sorption and desorption of phosphate by soil. Eur. J. Soil Sci. 66, 9–18 (2015).
Frossard, E., Condron, L. M., Oberson, A., Sinaj, S. & Fardeau, J. C. Processes governing phosphorus availability in temperate soils. J. Environ. Qual. 29, 15–23 (2000).
Turner, B. L. Storage-induced changes in phosphorus solubility of air-dried soils. Soil Sci. Soc. Am. J 69, 630–633 (2005).
Tiessen, H., Moir, J. in Soil Sampling and Methods of Analysis, 2nd edn (eds Carter, M. R. & Gregorich, E. G. ) 293–306 (CRC Press, 2007).
Condron, L. M. & Newman, S. Revisiting the fundamentals of phosphorus fractionation of sediments and soils. J. Soils Sediments. 11, 830–840 (2011).
Tiessen, H., Moir, J. in Soil Sampling and Methods of Analysis (ed. Carter, M.R ) 75–86 (Lewis Publishers, 1993).
Cross, A. F. & Schlesinger, W. H. A literature review and evaluation of the Hedley fractionation - Applications to the biogeochemical cycle of soil phosphorus in natural ecosystems. Geoderma 64, 197–214 (1995).
Gama-Rodrigues, A. C. et al. An exploratory analysis of phosphorus transformations in tropical soils using structural equation modeling. Biogeochemistry 118, 453–469 (2014).
Harrison, A. F. Soil Organic Phosphorus: A Review of World Literature (Commonwealth Agricultural Bureaux International, 1987).
Negassa, W. & Leinweber, P. How does the Hedley sequential phosphorus fractionation reflect impacts of land use and management on soil phosphorus: A review. J. Plant Nutr. Soil Sc 172, 305–325 (2009).
Hou, E. et al. Soil pH predominantly controls the forms of organic phosphorus in topsoils under natural broadleaved forests along a 2500 km latitudinal gradient. Geoderma 315, 65–74 (2018).
Hou, E. et al. Effects of climate on soil phosphorus cycle and availability in natural terrestrial ecosystems. Global Change Biol https://doi.org/10.1111/gcb.14093 (2018).
Hou, E. et al. A structural equation model analysis of phosphorus transformations in global unfertilized and uncultivated soils. Global Biogeochem. Cy 30, 1300–1309 (2016).
Yang, X. & Post, W. M. Phosphorus transformations as a function of pedogenesis: A synthesis of soil phosphorus data using Hedley fractionation method. Biogeosciences 8, 2907–2916 (2011).
Yang, X., Post, W. M., Thornton, P. E. & Jain, A. The distribution of soil phosphorus for global biogeochemical modeling. Biogeosciences 10, 2525–2537 (2013).
Sun, Y. et al. Diagnosing phosphorus limitations in natural terrestrial ecosystems in carbon cycle models. Earth's Future 5, 730–749 (2017).
Ringeval, B. et al. Phosphorus in agricultural soils: drivers of its distribution at the global scale. Global Change Biol. 23, 3418–3432 (2017).
Kvakić, M. et al. Quantifying the limitation to world cereal production due to soil phosphorus status. Global Biogeochem. Cy 30, 143–157 (2018).
Goll, D.S. et al. A representation of the phosphorus cycle for ORCHIDEE (revision 4520). Geosci. Model Dev 10, 3745–3770 (2017).
Tiessen, H., Stewart, J. W. B. & Cole, C. V. Pathways of phosphorus transformations in soils of differing pedogenesis. Soil Sci. Soc. Am. J 48, 853–858 (1984).
Velásquez, G. et al. Chemical nature of residual phosphorus in Andisols. Geoderma 271, 27–31 (2016).
Paré, D. & Bernier, B. Origin of the phosphorus deficiency observed in declining sugar maple stands in the Quebec Appalachians. Can. J. Forest Res. 19, 24–34 (1989).
Murphy, J. & Riley, J. P. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta 27, 31–36 (1962).
Ormaza-González, F. I. & Statham, P. J. A comparison of methods for the determination of dissolved and particulate phosphorus in natural waters. Water Res 30, 2739–2747 (1996).
Jobbágy, E. & Jackson, R. The distribution of soil nutrients with depth: Global patterns and the imprint of plants. Biogeochemistry 53, 51–77 (2001).
Vu, D., Tang, C. & Armstrong, R. Transformations and availability of phosphorus in three contrasting soil types from native and farming systems: A study using fractionation and isotopic labeling techniques. J. Soils Sediments 10, 18–29 (2010).
Lilienfein, J. et al. Chemical fractionation of phosphorus, sulphur, and molybdenum in Brazilian savannah Oxisols under different land use. Geoderma 96, 31–46 (2000).
Satti, P., Mazzarino, M. J., Roselli, L. & Crego, P. Factors affecting soil P dynamics in temperate volcanic soils of southern Argentina. Geoderma 139, 229–240 (2007).
O’halloran, I. & Cade-Menun, B. in Soil Sampling and Methods of Analysis (ed. Carter, M.R ) 213–229 (Lewis Publishers, 1993).
Ross, G. & Wang, C. in Soil Sampling and Methods of Analysis, Vol. (ed. Carter, M.R ) 239–246 (Lewis Publishers, 1993).
Kroetsch, D. & Wang, C. in Soil Sampling and Methods of Analysis, 2nd edn (eds Carter, M. R. & Gregorich, E. G. ) 713–725 (CRC Press, 2007).
Brookes, P., Powlson, D. & Jenkinson, D. Measurement of microbial biomass phosphorus in soil. Soil Biol. Biochem. 14, 319–329 (1982).
Hijmans, R. J., Cameron, S. E., Parra, J. L., Jones, P. G. & Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 25, 1965–1978 (2005).
Trabucco, A. & Zomer, R. . G Global Geospatial Potential EvapoTranspiration & Aridity Index CGIAR Consortium for Spatial Information (CGIAR-CSI, 2009).
Soil Survey Staff. Soil Taxonomy: A Basic System of Soil Classification for Making and Interpreting Soil Surveys. 2nd edn (Natural Resources Conservation Service. U.S. Department of Agriculture Handbook 436, 1999).
Porder, S. & Ramachandran, S. The phosphorus concentration of common rocks—a potential driver of ecosystem P status. Plant Soil 367, 41–55 (2013).
Garcia-Montiel, D. C. et al. Soil phosphorus transformations following forest clearing for pasture in the Brazilian Amazon. Soil Sci. Soc. Am. J 64, 1792–1804 (2000).
Guo, F., Yost, R., Hue, N., Evensen, C. & Silva, J. Changes in phosphorus fractions in soils under intensive plant growth. Soil Sci. Soc. Am. J 64, 1681–1689 (2000).
Helfenstein, J., Jegminat, J., McLaren, T. I. & Frossard, E. Soil solution phosphorus turnover: derivation, interpretation, and insights from a global compilation of isotope exchange kinetic studies. Biogeosciences 15, 105–114 (2018).
Guo, F. & Yost, R. S. Partitioning soil phosphorus into three discrete pools of differing availability. Soil Sci. 163, 822–833 (1998).
Richter, D. D., Allen, H. L., Li, J. W., Markewitz, D. & Raikes, J. Bioavailability of slowly cycling soil phosphorus: major restructuring of soil P fractions over four decades in an aggrading forest. Oecologia 150, 259–271 (2006).
Goll, D. S., Moosdorf, N., Hartmann, J. & Brovkin, V. Climate-driven changes in chemical weathering and associated phosphorus release since 1850: Implications for the land carbon balance. Geophys. Res. Lett. 41, 3553–3558 (2014).
Wang, Y. P., Houlton, B. Z. & Field, C. B. A model of biogeochemical cycles of carbon, nitrogen, and phosphorus including symbiotic nitrogen fixation and phosphatase production. Global Biogeochem. Cy 21, GB1018 (2007).
Turner, B. L. Resource partitioning for soil phosphorus: a hypothesis. J. Ecol. 96, 698–702 (2008).
Agbenin, J. & Goladi, J. Dynamics of phosphorus fractions in a savanna Alfisol under continuous cultivation. Soil Use Manage. 14, 59–64 (1998).
Aguiar, A. D. C. F., Cândido, C. S., Carvalho, C. S., Monroe, P. H. M. & De Moura, E. G. Organic matter fraction and pools of phosphorus as indicators of the impact of land use in the Amazonian periphery. Ecol. Indic. 30, 158–164 (2013).
Alamgir, M., Mcneill, A., Tang, C. & Marschner, P. Changes in soil P pools during legume residue decomposition. Soil Biol. Biochem. 49, 70–77 (2012).
Alt, F., Oelmann, Y., Herold, N., Schrumpf, M. & Wilcke, W. Phosphorus partitioning in grassland and forest soils of Germany as related to land‐use type, management intensity, and land use–related pH. J. Plant Nutr. Soil Sci. 174, 195–209 (2011).
Araújo, M. S., Schaefer, C. E. & Sampaio, E. V. Soil phosphorus fractions from toposequences of semi-arid Latosols and Luvisols in northeastern Brazil. Geoderma 119, 309–321 (2004).
Beck, M. & Elsenbeer, H. Biogeochemical cycles of soil phosphorus in southern Alpine spodosols. Geoderma 91, 249–260 (1999).
Beck, M. A. & Sanchez, P. A. Soil phosphorus fraction dynamics during 18 years of cultivation on a Typic Paleudult. Soil Sci. Soc. Am. J 58, 1424–1431 (1994).
Brandtberg, P. O., Davis, M., Clinton, P. & Allen, R. Forms of soil phosphorus affected by stand development of mountain beech (Nothofagus) forests in New Zealand. Geoderma 157, 228–234 (2010).
Cassagne, N., Remaury, M., Gauquelin, T. & Fabre, A. Forms and profile distribution of soil phosphorus in alpine Inceptisols and Spodosols (Pyrenees, France). Geoderma 95, 161–172 (2000).
Cherubin, M. R. et al. Phosphorus pools responses to land-use change for sugarcane expansion in weathered Brazilian soils. Geoderma 265, 27–38 (2016).
Chimdi, A., Esala, M. & Ylivainio, K. Sequential fractionation patterns of soil ohosphorus collected from different land use systems of Dire Inchine District, West Shawa Zone, Ethiopia. Am. Eurasian J. Sci. Res 9, 51–57 (2014).
Cross, A. F. & Schlesinger, W. H. Biological and geochemical controls on phosphorus fractions in semiarid soils. Biogeochemistry 52, 155–172 (2001).
Da Silva, V. M. et al. Influence of the tropical millipede, Glyphiulus granulatus (Gervais, 1847), on aggregation, enzymatic activity, and phosphorus fractions in the soil. Geoderma 289, 135–141 (2017).
De Oliveira, C. M. B., Erich, M. S., Gatiboni, L. C. & Ohno, T. Phosphorus fractions and organic matter chemistry under different land use on Humic Cambisols in Southern Brazil. Geoderma Reg 5, 140–149 (2015).
Dieter, D., Elsenbeer, H. & Turner, B. L. Phosphorus fractionation in lowland tropical rainforest soils in central Panama. Catena 82, 118–125 (2010).
Dossa, E., Diedhiou, S., Compton, J., Assigbetse, K. & Dick, R. Spatial patterns of P fractions and chemical properties in soils of two native shrub communities in Senegal. Plant Soil 327, 185–198 (2010).
Duffera, M. & Robarge, W. P. Characterization of organic and inorganic phosphorus in the highland plateau soils of Ethiopia. Commun. Soil Sci. Plant Anal. 27, 2799–2814 (1996).
Esberg, C., Du Toit, B., Olsson, R., Ilstedt, U. & Giesler, R. Microbial responses to P addition in six South African forest soils. Plant Soil 329, 209–225 (2010).
Feng, J. et al. Phosphorus transformations along a large‐scale climosequence in arid and semiarid grasslands of northern China. Global Biogeochem. Cy 30, 1264–1275 (2016).
Frizano, J., Johnson, A. H., Vann, D. R. & Scatena, F. N. Soil phosphorus fractionation during forest development on landslide scars in the Luquillo Mountains, Puerto Rico. Biotropica 34, 17–26 (2002).
Frizano, J. et al. Labile phosphorus in soils of forest fallows and primary forest in the Bragantina region, Brazil. Biotropica 35, 2–11 (2003).
Giardina, C., Sanford, R. & Døckersmith, I. Changes in soil phosphorus and nitrogen during slash-and-burn clearing of a dry tropical forest. Soil Sci. Soc. Am. J 64, 399–405 (2000).
Giesler, R., Esberg, C., Lagerström, A. & Graae, B. J. Phosphorus availability and microbial respiration across different tundra vegetation types. Biogeochemistry 108, 429–445 (2012).
Gross, A. & Angert, A. What processes control the oxygen isotopes of soil bio-available phosphate? Geochim. Cosmochim. Ac 159, 100–111 (2015).
Guo, H., Zhou, J., Luo, X., Wang, W. & Wu, X. Phosphorus fractions of Latosols developed from different parent materials in rubber plantation of Hainan Province. Chin. J. Trop. Crops 33, 1724–1730 (2012).
Hashimoto, Y. & Watanabe, Y. Combined applications of chemical fractionation, solution 31P-NMR and P K-edge XANES to determine phosphorus speciation in soils formed on serpentine landscapes. Geoderma 230, 143–150 (2014).
Hinojosa, M. B. et al. Effects of drought on soil phosphorus availability and fluxes in a burned Mediterranean shrubland. Geoderma 191, 61–69 (2012).
Izquierdo, J. E., Houlton, B. Z. & Van Huysen, T. L. Evidence for progressive phosphorus limitation over long-term ecosystem development: examination of a biogeochemical paradigm. Plant Soil 367, 135–147 (2013).
Jien, S. H. et al. Forms and distribution of phosphorus in a placic podzolic toposequence in a subtropical subalpine forest, Taiwan. Catena 140, 145–154 (2016).
Julich, D., Julich, S. & Feger, K. H. Phosphorus fractions in preferential flow pathways and soil matrix in hillslope soils in the Thuringian Forest (Central Germany). J. Plant Nutr. Soil Sci. 180, 407–417 (2017).
Lin, K., Guo, J., Yang, Z., Ji, S. & Yang, Y. Soil phosphorus forms and availability in natural regeneration by man-aided Castanopsis carlesii forests. J. Cent. S. U. Forest. Tech 34, 6–11 (2014).
Khan, K. S. & Joergensen, R. G. Relationships between P fractions and the microbial biomass in soils under different land use management. Geoderma 173, 274–281 (2012).
Kitayama, K., Majalap-Lee, N. & Aiba, S. Soil phosphorus fractionation and phosphorus-use efficiencies of tropical rainforests along altitudinal gradients of Mount Kinabalu, Borneo. Oecologia 123, 342–349 (2000).
Kunito, T. et al. Soil properties affecting phosphorus forms and phosphatase activities in Japanese forest soils: soil microorganisms may be limited by phosphorus. Soil Sci. 177, 39–46 (2012).
Lajtha, K & Schlesinger, W. H. The biogeochemistry of phosphorus cycling and phosphorus availability along a desert soil chronosequence. Ecology 69, 24–39 (1988).
Lee, D., Han, X. & Jordan, C. Soil phosphorus fractions, aluminum, and water retention as affected by microbial activity in an Ultisol. Plant Soil 121, 125–136 (1990).
Lehmann, J. et al. Inorganic and organic soil phosphorus and sulfur pools in an Amazonian multistrata agroforestry system. Agroforest. Syst. 53, 113–124 (2001).
Levy, E. T & Schlesinger, W. H. A comparison of fractionation methods for forms of phosphorus in soils. Biogeochemistry 47, 25–38 (1999).
Liptzin, D., Sanford, R. L. Jr. & Seastedt, T. R Spatial patterns of total and available N and P at alpine treeline. Plant Soil 365, 127–140 (2013).
Lloyd, J. et al. Edaphic, structural and physiological contrasts across Amazon Basin forest–savanna ecotones suggest a role for potassium as a key modulator of tropical woody vegetation structure and function. Biogeosciences 12, 6529–6571 (2015).
Lu, X. et al. Characteristics of phosphorus contents in the rhizosphere soil of different shrubs on sandy grassland in Maqu. Pratacultural Sci 29, 167–173 (2012).
Mage, S. M. & Porder, S. Parent material and topography determine soil phosphorus status in the Luquillo Mountains of Puerto Rico. Ecosystems 16, 284–294 (2013).
Maranguit, D., Guillaume, T. & Kuzyakov, Y. Land-use change affects phosphorus fractions in highly weathered tropical soils. Catena 149, 385–393 (2017).
Mcclintock, M. A. et al. Spatial variability of African dust in soils in a montane tropical landscape in Puerto Rico. Chem. Geol. 412, 69–81 (2015).
Mckenzie, R., Stewart, J., Dormaar, J. & Schaalje, G. Long-term crop rotation and fertilizer effects on phosphorus transformations: I. In a Chernozemic soil. Can. J. Soil Sci. 72, 569–579 (1992).
Mckenzie, R., Stewart, J., Dormaar, J. & Schaalje, G. Long-term crop rotation and fertilizer effects on phosphorus transformations: II. In a Luvisolic soil. Can. J. Soil Sci. 72, 581–589 (1992).
Mirabello, M. et al. Soil phosphorus responses to chronic nutrient fertilisation and seasonal drought in a humid lowland forest, Panama. Soil Res 51, 215–221 (2013).
Muller, M., Oelmann, Y., Schickhoff, U., Bohner, J. & Scholten, T. Himalayan treeline soil and foliar C:N:P stoichiometry indicate nutrient shortage with elevation. Geoderma 291, 21–32 (2017).
Newbery, D. M., Alexander, I. & Rother, J. Phosphorus dynamics in a lowland African rainforest: the influence of ectomycorrhizal trees. Ecol. Monogr. 67, 367–409 (1997).
O’halloran, I., Stewart, J. W. & De Jong, E. Changes in P forms and availability as influenced by management practices. Plant Soil 100, 113–126 (1987).
Oberson, A., Friesen, D. K., Rao, I. M., Bühler, S. & Frossard, E. Phosphorus transformations in an oxisol under contrasting land-use systems: the role of the soil microbial biomass. Plant Soil 237, 197–210 (2001).
Olander, L. P. et al. Surface soil changes following selective logging in an eastern Amazon forest. Earth Interact. 9, 1–19 (2005).
Perroni, Y., García-Oliva, F., Tapia-Torres, Y. & Souza, V. Relationship between soil P fractions and microbial biomass in an oligotrophic grassland-desert scrub system. Ecol. Res 29, 463–472 (2014).
Qin, S., Liu, J., Wang, G. & Zhou, W. Phosphorus fractions under different land uses in Sanjiang plain. Environ. Sci. 28, 2777–2782 (2007).
Quesada, C. et al. Variations in chemical and physical properties of Amazon forest soils in relation to their genesis. Biogeosciences 7, 1515–1541 (2010).
Redel, Y., Escudey, M., Alvear, M., Conrad, J. & Borie, F. Effects of land use change on P bioavailability determined by chemical fractionation and 31P-NMR spectroscopy in a Nothofagus forest and adjacent grassland. J. Soil Sci. Plant Nutr. 15, 1061–1070 (2015).
Redel, Y., Rubio, R., Godoy, R. & Borie, F. Phosphorus fractions and phosphatase activity in an Andisol under different forest ecosystems. Geoderma 145, 216–221 (2008).
Resende, J. C. F., Markewitz, D., Klink, C. A., Da Cunha Bustamante, M. M. & Davidson, E. A. Phosphorus cycling in a small watershed in the Brazilian Cerrado: impacts of frequent burning. Biogeochemistry 105, 105–118 (2011).
Roberts, T., Stewart, J. & Bettany, J. The influence of topography on the distribution of organic and inorganic soil phosphorus across a narrow environmental gradient. Can. J. Soil Sci. 65, 651–665 (1985).
Rückamp, D., Amelung, W., Theisz, N., Bandeira, A. G. & Martius, C. Phosphorus forms in Brazilian termite nests and soils: relevance of feeding guild and ecosystems. Geoderma 155, 269–279 (2010).
Sattell, R. & Morris, R. Phosphorus fractions and availability in Sri Lankan Alfisols. Soil Sci. Soc. Am. J 56, 1510–1515 (1992).
Schlesinger, W. H. et al. The biogeochemistry of phosphorus after the first century of soil development on Rakata Island, Krakatau, Indonesia. Biogeochemistry 40, 37–55 (1998).
Schlichting, A., Leinweber, P., Meissner, R. & Altermann, M. Sequentially extracted phosphorus fractions in peat‐derived soils. J. Plant Nutr. Soil Sci. 165, 290–298 (2002).
Schoenau, J., Stewart, J. & Bettany, J. Forms and cycling of phosphorus in prairie and boreal forest soils. Biogeochemistry 8, 223–237 (1989).
Selmants, P. C. & Hart, S. C. Phosphorus and soil development: does the Walker and Syers model apply to semiarid ecosystems? Ecology 91, 474–484 (2010).
Shafqat, M. N., Shahid, S., Shah, S. H. & Waseem, A. Soil phosphorus fractionation as a tool for monitoring dust phosphorus signature underneath a Blue Pine (Pinus wallichiana) canopy in a Temperate Forest. Forest Syst 25, e70 (2016).
Sharpley, A. N. et al. A Detailed Phosphorus Characterization of Seventy-eight Soils, US Department of Agriculture. Agricultural Research Service, ARS-31, U.S. Government Printing Office, Washington, D.C (1985).
Soinne, H., Räty, M. & Hartikainen, H. Effect of air‐drying on phosphorus fractions in clay soil. J. Plant Nutr. Soil Sci. 173, 332–336 (2010).
Solomon, D. & Lehman, N. Loss of phosphorus from soil in semi-arid northern Tanzania as a result of cropping: evidence from sequential extraction and 31P-NMR spectroscopy. Eur. J. Soil Sci. 51, 699–708 (2000).
Solomon, D., Lehmann, J., Mamo, T., Fritzsche, F. & Zech, W. Phosphorus forms and dynamics as influenced by land use changes in the sub-humid Ethiopian highlands. Geoderma 105, 21–48 (2002).
Sun, H., Wu, Y., Yu, D. & Zhou, J. Altitudinal gradient of microbial biomass phosphorus and its relationship with microbial biomass carbon, nitrogen, and rhizosphere soil phosphorus on the eastern slope of Gongga Mountain, SW China. PLoS ONE 8, e72952 (2013).
Szott, L. & Melendez, G. Phosphorus availability under annual cropping, alley cropping, and multistrata agroforestry systems. Agroforest. Syst. 53, 125–132 (2001).
Tchienkoua, M. & Zech, W. Chemical and spectral characterization of soil phosphorus under three land uses from an Andic Palehumult in West Cameroon. Agr. Ecosyst. Environ. 100, 193–200 (2003).
Thomas, S. M. et al. Phosphorus fractions in montane forest soils of the Cordillera de Piuchué, Chile: biogeochemical implications. Plant Soil 211, 139–148 (1999).
Tiessen, H., Salcedo, I. & Sampaio, E. Nutrient and soil organic matter dynamics under shifting cultivation in semi-arid northeastern Brazil. Agr. Ecosyst. Environ. 38, 139–151 (1992).
Trasar-Cepeda, M. C., Carballas, T., Gil-Sotres, F. & De Blas, E. Liming and the phosphatase activity and mineralization of phosphorus in an andic soil. Soil Biol. Biochem. 23, 209–215 (1991).
Trasar-Cepeda, M. C., Gil-Sotres, F. & Guitian-Ojea, F. Relation between phosphorus fractions and development of soils from Galicia (NW Spain). Geoderma 47, 139–150 (1990).
Turner, B. L. & Laliberté, E. Soil development and nutrient availability along a 2 million-year coastal dune chronosequence under species-rich Mediterranean shrubland in southwestern Australia. Ecosystems 18, 287–309 (2015).
Vincent, A. G., Sundqvist, M. K., Wardle, D. A. & Giesler, R. Bioavailable soil phosphorus decreases with increasing elevation in a subarctic tundra landscape. PLoS ONE 9, e92942 (2014).
Vu, D., Armstrong, R., Newton, P. & Tang, C. Long-term changes in phosphorus fractions in growers’ paddocks in the northern Victorian grain belt. Nutr. Cycl. Agroecosys 89, 351–362 (2011).
Wang, J. et al. Conversion of rainforest into agroforestry and monoculture plantation in China: Consequences for soil phosphorus forms and microbial community. Sci. Total Environ. 595, 769–778 (2017).
Wardle, D. A., Bellingham, P. J., Kardol, P., Giesler, R. & Tanner, E. V. J. Coordination of aboveground and belowground responses to local-scale soil fertility differences between two contrasting Jamaican rain forest types. Oikos 124, 285–297 (2015).
Wu, R., Bao, L. & Tiessen, H. Study on the dynamic of soil phosphorus in the transitional areas of grasslands and crop fields. Plant Nutr. Fert. Sci 9, 131–138 (2003).
Wu, Y. et al. Soil phosphorus bioavailability assessed by XANES and Hedley sequential fractionation technique in a glacier foreland chronosequence in Gongga Mountain, Southwestern China. Sci. China Earth Sci 57, 1860–1868 (2014).
Xu, G et al. Effects of air-drying and freezing on phosphorus fractions in soils with different organic matter contents. Plant Soil Environ. 57, 228–234 (2011).
Xue, Q. Y. et al. Impact of manure application on forms and quantities of phosphorus in a Chinese Cambisol under different land use. J. Soil. Sediment 13, 837–845 (2013).
Yang, H. et al. Effects of land use types on phosphorus forms and their contents in soil aggregates in watershed of Hongfeng lake. J. Agro-Environ. Sci 33, 2214–2220 (2013).
Yang, K., Zhu, J., Gu, J., Yu, L. & Wang, Z. Changes in soil phosphorus fractions after 9 years of continuous nitrogen addition in a Larix gmelinii plantation. Ann. For. Sci. 72, 435–442 (2014).
Zhang, J. & Chen, A. Phosphorus fractions and availability of tropical soil in rubber plantation. Soil Environ. Sci. 8, 284–286 (1999).
Zhang, L. et al. Impacts of vegetation type on soil phosphorus availability and fractions near the alpine timberline of the Tibetan Plateau. Pol. J. Ecol. 59, 307–316 (2011).
Zhou, J. et al. Variations in soil phosphorus biogeochemistry across six vegetation types along an altitudinal gradient in SW China. Catena 142, 102–111 (2016).
Data Citations
Hou, E PANGAEA https://doi.org/10.1594/PANGAEA.883611 (2017)
Acknowledgements
We thank all the authors whose work were included in our database and apologize to those whose work were not included in our database. We thank Dr. Stefanie Schumacher for her help with the deposit of our data in PANGAEA. This work was supported by the National Natural Science Foundation of China (31130010, 31570483, 41401326).
Author information
Authors and Affiliations
Contributions
X.T., E.H., and D.W. conceived this paper. E.H. compiled the dataset. E.H. and X.T. wrote the manuscript, with significant inputs from M.H. and D.W.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
ISA-Tab metadata
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/ The Creative Commons Public Domain Dedication waiver http://creativecommons.org/publicdomain/zero/1.0/ applies to the metadata files made available in this article.
About this article
Cite this article
Hou, E., Tan, X., Heenan, M. et al. A global dataset of plant available and unavailable phosphorus in natural soils derived by Hedley method. Sci Data 5, 180166 (2018). https://doi.org/10.1038/sdata.2018.166
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/sdata.2018.166
This article is cited by
-
Phosphorus applications adjusted to optimal crop yields can help sustain global phosphorus reserves
Nature Food (2024)
-
A Global Database of Soil Plant Available Phosphorus
Scientific Data (2023)
-
Unraveling the consequences of nitrogen addition on soil phosphorus fractions in alpine grasslands: insights from field experiments and global analysis
Journal of Soils and Sediments (2023)
-
Impacts of Climate Warming on Soil Phosphorus Forms and Transformation in a Tibetan Alpine Meadow
Journal of Soil Science and Plant Nutrition (2022)
-
Biochemical inhibition of acid phosphatase activity in two mountain spruce forest soils
Biology and Fertility of Soils (2021)