Abstract
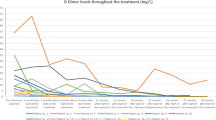
Arteriovenous malformations (AVMs) are fast-flow lesions that may be destructive and are the most difficult-to-treat vascular anomalies. Embolization followed by surgical resection is commonly used; however, complete resection is rarely possible and partial resection often leads to dramatic worsening. Accumulating data implicate abnormal angiogenic activity in the development of AVMs. Thalidomide has been reported as an inhibitor of vascular proliferation. Here, we report a prospective experimental observational study testing the effects of the angiogenesis inhibitor thalidomide on 18 patients with a severely symptomatic AVM that is refractory to conventional therapies. All patients experienced a rapid reduction in pain, cessation of bleeding, and ulcer healing. Cardiac failure resolved in all three affected patients. Reduced vascularity on arteriography was observed in two patients. One AVM appeared to be cured after 19 months of thalidomide and an 8-year follow-up. Eight AVMs were stable after a mean thalidomide cessation of 58 months, and four lesions recurred after 11.5 months. Combined treatment with embolization permitted dose reduction in five patients with clinical improvement. Grade 3 side effects were dose dependent, including asthenia (n = 2) and erythroderma (n = 2). The results suggest that thalidomide is efficacious in the management of chronic pain, bleeding and ulceration of extensive AVMs that are refractory to conventional therapy. Further clinical study is needed to confirm the safety and efficacy of thalidomide treatment in AVM.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
All data generated and analyzed during this study are included in this published article.
References
Liu, A. S., Mulliken, J. B., Zurakowski, D., Fishman, S. J. & Greene, A. K. Extracranial arteriovenous malformations: natural progression and recurrence after treatment. Plast. Reconstr. Surg. 125, 1185–1194 (2010).
Boon, L. M. & Vikkula, M. in Harper’s Textbook of Pediatric Dermatology 4th edn (eds Irvine, A. D., Hoeger, P. H. & Yan, A. C.) Ch. 118 (Wiley-Blackwell, 2019).
Young, E. J. & Fishman S. J. in Mulliken and Young’s Vascular Anomalies: Hemangiomas and Malformations 2nd edn (eds Mulliken, J. B., Burrows, P. E. & Fishman, S. J.) Ch. 15 (Oxford University Press, 2013).
Boon, L. M. & Vikkula M. in Mulliken and Young’s Vascular Anomalies: Hemangiomas and Malformations 2nd edn (eds Mulliken, J. B., Burrows, P. E. & Fishman, S. J.) Ch. 9 (Oxford University Press, 2013).
Revencu, N. et al. RASA1 mosaic mutations in patients with capillary malformation-arteriovenous malformation. J. Med. Genet. 57, 48–52 (2020).
Amyere, M. et al. Germline loss-of-function mutations in EPHB4 cause a second form of capillary malformation-arteriovenous malformation (CM-AVM2) deregulating RAS-MAPK signaling. Circulation 136, 1037–1048 (2017).
Shovlin, C. L., Hughes, J. M. B., Scott, J., Seidman, C. E. & Seidman, J. G. Characterization of endoglin and identification of novel mutations in hereditary hemorrhagic telangiectasia. Am. J. Hum. Genet. 61, 68–79 (1997).
Shovlin, C. L. Hereditary haemorrhagic telangiectasia: pathophysiology, diagnosis and treatment. Blood Rev. 24, 203–219 (2010).
Marsh, D. J. et al. Germline PTEN mutations in Cowden syndrome-like families. J. Med. Genet. 35, 881–885 (1998).
Marsh, D. J. et al. PTEN mutation spectrum and genotype-phenotype correlations in Bannayan-Riley-Ruvalcaba syndrome suggest a single entity with Cowden syndrome. Hum. Mol. Genet. 8, 1461–1472 (1999).
Kohout, M. P., Hansen, M., Pribaz, J. J. & Mulliken, J. B. Arteriovenous malformations of the head and neck: natural history and management. Plast. Reconstr. Surg. 102, 643–654 (1998).
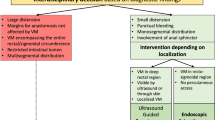
Soulez, G., Gilbert, P., Giroux, M.-F., Racicot, J.-N. & Dubois, J. Interventional management of arteriovenous malformations. Tech. Vasc. Interv. Radiol. 22, 100633 (2019).
Koshima, I. et al. Free perforator flap for the treatment of defects after resection of huge arteriovenous malformations in the head and neck regions. Ann. Plast. Surg. 51, 194–199 (2003).
Wong, M. et al. Soft tissue reconstruction and facial reanimation with bilateral latissimus dorsi flaps after extensive resection of head and neck arteriovenous malformation: a case report. Ann. Plast. Surg. 83, 73–77 (2019).
Lee, B. B. et al. Consensus Document of the International Union of Angiology (IUA)-2013. Current concept on the management of arterio-venous management. Int. Angiol. 32, 9–36 (2013).
Hammer, F. D., Boon, L. M., Mathurin, P. & Vanwijck, R. R. Ethanol sclerotherapy of venous malformations: evaluation of systemic ethanol contamination. J. Vasc. Interv. Radiol. 12, 595–600 (2001).
Burrows, P. E., Mulliken, J. B., Fishman, S. J., Klement, G. L. & Folkman, J. Pharmacological treatment of a diffuse arteriovenous malformation of the upper extremity in a child. J. Craniofac. Surg. 20, 597–602 (2009).
McBride, W. G. Thalidomide embryopathy. Teratology 16, 79–82 (1977).
D’Amato, R. J., Loughnan, M. S., Flynn, E. & Folkman, J. Thalidomide is an inhibitor of angiogenesis. Proc. Natl Acad. Sci. USA 91, 4082–4085 (1994).
Lebrin, F. et al. Thalidomide stimulates vessel maturation and reduces epistaxis in individuals with hereditary hemorrhagic telangiectasia. Nat. Med. 16, 420–428 (2010).
Buscarini, E. et al. Safety of thalidomide and bevacizumab in patients with hereditary hemorrhagic telangiectasia. Orphanet J. Rare Dis. 14, 28 (2019).
Zhu, W. et al. Thalidomide reduces hemorrhage of brain arteriovenous malformations in a mouse model. Stroke 49, 1232–1240 (2018).
Fink, E. C. & Ebert, B. L. The novel mechanism of lenalidomide activity. Blood 126, 2366–2369 (2015).
Burrows, P. E. Endovascular treatment of slow-flow vascular malformations. Tech. Vasc. Interv. Radiol. 16, 12–21 (2013).
Mulliken J. B. in Mulliken and Young’s Vascular Anomalies: Hemangiomas and Malformations 2nd edn (eds Mulliken, J. B., Burrows, P. E. & Fishman, S. J.) Ch. 23 (Oxford University Press, 2013).
Park, K. B. et al. Predictive factors for response of peripheral arteriovenous malformations to embolization therapy: analysis of clinical data and imaging findings. J. Vasc. Interv. Radiol. 23, 1478–1486 (2012).
Pugh, C. W. & Ratcliffe, P. J. Regulation of angiogenesis by hypoxia: role of the HIF system. Nat. Med. 9, 677–684 (2003).
Carmeliet, P. & Jain, R. K. Angiogenesis in cancer and other diseases. Nature 407, 249–257 (2000).
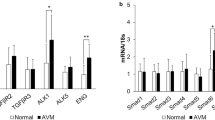
Lu, L. et al. Increased endothelial progenitor cells and vasculogenic factors in higher-staged arteriovenous malformations. Plast. Reconstr. Surg. 128, 260e–269e (2011).
Komorowski, J. et al. Effect of thalidomide affecting VEGF secretion, cell migration, adhesion and capillary tube formation of human endothelial EA.hy 926 cells. Life Sci. 78, 2558–2563 (2006).
Grinspan, D. Significant response of oral aphthosis to thalidomide treatment. J. Am. Acad. Dermatol. 12, 85–90 (1985).
Ghobrial, I. M. & Rajkumar, S. V. Management of thalidomide toxicity. J. Support. Oncol. 1, 194–205 (2003).
Couto, J. A. et al. Somatic MAP2K1 mutations are associated with extracranial arteriovenous malformation. Am. J. Hum. Genet. 100, 546–554 (2017).
Nikolaev, S. I., Fish, J. E. & Radovanovic, I. Somatic activating KRAS mutations in arteriovenous malformations of the brain. N. Engl. J. Med. 378, 1561–1562 (2018).
Van Damme, A., Seront, E., Dekeuleneer, V., Boon, L. M. & Vikkula, M. New and emerging targeted therapies for vascular malformations. Am. J. Clin. Dermatol. 21, 657–668 (2020).
Seront, E., Van Damme, A., Boon, L. M. & Vikkula, M. Rapamycin and treatment of venous malformations. Curr. Opin. Hematol. 26, 185–192 (2019).
Gabeff, R. et al. Efficacy and tolerance of sirolimus (rapamycin) for extracranial arteriovenous malformations in children and adults. Acta Derm. Venereol. 99, 1105–1109 (2019).
Lekwuttikarn, R., Lim, Y. H., Admani, S., Choate, K. A. & Teng, J. M. C. Genotype-guided medical treatment of an arteriovenous malformation in a child. JAMA Dermatol. 155, 256–257 (2019).
Edwards, E. A. Monitoring arteriovenous malformation response to genotype-targeted therapy. Pediatrics 146, e20193206 (2020).
Wu, J. J., Huang, D. B., Pang, K. R., Hsu, S. & Tyring, S. K. Thalidomide: dermatological indications, mechanisms of action and side-effects. Br. J. Dermatol. 153, 254–273 (2005).
Lokhorst, M. M. et al. Development of a condition-specific patient-reported outcome measure for measuring symptoms and appearance in vascular malformations: the OVAMA questionnaire. Br. J. Dermatol. 185, 797–803 (2021).
Gagne, J. J., Thompson, L., O’Keefe, K. & Kesselheim, A. S. Innovative research methods for studying treatments for rare diseases: methodological review. BMJ 349, g6802 (2014).
Acknowledgements
All authors but one (A.D.) are members of the VASCA Working Group of VASCERN, project no. 769036. We are grateful to all of the patients for their invaluable participation. This work is part of the thesis project of J.C., who was supported by a Fond de la Recherche Clinique (FRC) des Cliniques universitaires Saint-Luc. These studies were financially supported by Fonds de la Recherche Scientifique (FNRS) grants T.0247.19 (to M.V.) and P.C013.20 (to L.M.B.), and the Fund Generet managed by the King Baudouin Foundation (grant 2018-J1810250-211305) (to M.V.). The authors are grateful to J.B. Mulliken, P.E. Burrows and J. Dubois for their expert review of and suggestions for the manuscript; the nurse coordinator, O. Debue, for his excellent assistance in patient follow-up; and L. Niculescu and L. Huybrechts for their skilled secretarial help.
Author information
Authors and Affiliations
Contributions
L.M.B. and M.V. conceived and designed the study; L.M.B., J.C., V.D., L.M., A.-C.B. and A.J. followed the patients; F.H. and P.C. analyzed the radiological data; and A.D. L.M.B., J.C., V.D., L.M. and A.-C.B. analyzed the data. All authors contributed to writing the paper and reviewed the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Cardiovascular Research thanks Joyce M. C. Teng and the other, anonymous, reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1
Patient #1 with dramatic progressive aggravation of her AVM due to several partial surgical resection and embolization. (a) stage I AVM involving her upper lip at the age of 20. (b) stage IV AVM at the age of 50 now involving her entire central face and causing labial and palatal ulceration. (c) arteriography showing the extensiveness of her AVM. (D) 1 month after thalidomide initiation (d,e) showing cessation of mucosal bleeding of the lip, cessation of drooling and complete healing of her palate.
Supplementary information
Rights and permissions
About this article
Cite this article
Boon, L.M., Dekeuleneer, V., Coulie, J. et al. Case report study of thalidomide therapy in 18 patients with severe arteriovenous malformations. Nat Cardiovasc Res 1, 562–567 (2022). https://doi.org/10.1038/s44161-022-00080-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44161-022-00080-2
This article is cited by
-
Targeting the microenvironment in the treatment of arteriovenous malformations
Angiogenesis (2024)
-
Somatic BrafV600E mutation in the cerebral endothelium induces brain arteriovenous malformations
Angiogenesis (2024)
-
A new tale of thalidomide repurposing
Nature Cardiovascular Research (2022)