Abstract
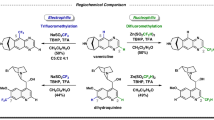
α-Quaternary amino and glycolic acids have found application in many biologically relevant compounds and pharmaceuticals. For example, α-quaternary amino acids can act as modifiers of peptide conformation, compared with natural amino acids. Although there are numerous enantioselective methods for the synthesis of α-quaternary amino and glycolic acids through α-alkylation, α-arylation routes are challenging. Here we report two protocols for the enantioselective synthesis of chiral α-aryl quaternary amino acids and glycolic acids derivatives, respectively, using palladium catalysis with two unique sulfinamide phosphine (Sadphos) ligands. The methods employ two common heterocycles, azlactones and 5H-oxazol-4-ones, as amino acid and glycolic acid precursors and show a broad substrate scope, with high yields and excellent enantioselectivity. Density functional theory calculations of the transition-state structures, studying non-covalent interactions and using natural bond orbital analysis, reveal that C–H···O interactions between the ligand and the substrate play a critical role in the efficient stereocontrol.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its Supplementary Information. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2233239 (5ab). These data are provided free of charge by the joint Cambridge Crystallographic Data Centre and Fachinformationszentrum Karlsruhe Access Structures service, www.ccdc.cam.ac.uk/structures.
References
Vogt, H. & Bräse, S. Recent approaches towards the asymmetric synthesis of α,α-disubstituted α-amino acids. Org. Biomol. Chem. 5, 406–430 (2007).
Michaux, J., Niel, G. & Campagne, J.-M. Stereocontrolled routes to β,β′-disubstituted α-amino acids. Chem. Soc. Rev. 38, 2093–2116 (2009).
Toniolo, C., Crisma, M., Formaggio, F. & Peggion, C. Control of peptide conformation by the Thorpe–Ingold effect (Cα-tetrasubstitution). Pept. Sci. 60, 396–419 (2001).
Sjöquist, B., Johnson, H. A. & Borg, S. The influence of acute ethanol on the catecholamine system in man as reflected in cerebrospinal fluid and urine. A new condensation product, 1-carboxysalsolinol. Drug Alcohol Depend. 16, 241–249 (1985).
Ma, D. Conformationally constrained analogues of l-glutamate as subtype-selective modulators of metabotropic glutamate receptors. Bioorg. Chem. 27, 20–34 (1999).
Schoepp, D. D., Jane, D. E. & Monn, J. A. Pharmacological agents acting at subtypes of metabotropic glutamate receptors. Neuropharmacology 38, 1431–1476 (1999).
Meusel, M. & Gütschow, M. Recent developments in hydantoin chemistry. A review. Org. Prep. Proced. Int. 36, 391–443 (2004).
Sonowal, H. et al. Aldose reductase inhibitor increases doxorubicin-sensitivity of colon cancer cells and decreases cardiotoxicity. Sci Rep. 7, 3182 (2017).
Pal, R., Kumar, B., Akhtar, M. J. & Chawla, P. A. Voltage gated sodium channel inhibitors as anticonvulsant drugs: a systematic review on recent developments and structure activity relationship studies. Bioorg. Chem. 115, 105230 (2021).
Grimm, J. B., Stables, J. P. & Brown, M. L. Design, synthesis, and development of novel caprolactam anticonvulsants. Biorg. Med. Chem. 11, 4133–4141 (2003).
Kakuda, R., Machida, K., Yaoita, Y., Kikuchi, M. & Kikuchi, M. Studies on the constituents of Gentiana species. II. A new triterpenoid, and (S)-(+)- and (R)-(−)-gentiolactones from Gentiana lutea. Chem. Pharm. Bull. 51, 885–887 (2003).
Davis, G. C. et al. Asymmetric synthesis and evaluation of a hydroxyphenylamide voltage-gated sodium channel blocker in human prostate cancer xenografts. Biorg. Med. Chem. 20, 2180–2188 (2012).
Yarker, Y. E., Goa, K. L. & Fitton, A. Oxybutynin. Drugs Aging 6, 243–262 (1995).
Tomohara, K., Yoshimura, T., Hyakutake, R., Yang, P. & Kawabata, T. Asymmetric α-arylation of amino acid derivatives by Clayden rearrangement of ester enolates via memory of chirality. J. Am. Chem. Soc. 135, 13294–13297 (2013).
Atkinson, R. C., Fernández-Nieto, F., Mas Roselló, J. & Clayden, J. Pseudoephedrine-directed asymmetric α-arylation of α-amino acid derivatives. Angew. Chem. Int. Ed. 54, 8961–8965 (2015).
Leonard, D. J., Ward, J. W. & Clayden, J. Asymmetric α-arylation of amino acids. Nature 562, 105–109 (2018).
Shirakawa, S., Yamamoto, K. & Maruoka, K. Phase-transfer-catalyzed asymmetric SNAr reaction of α-amino acid derivatives with arene chromium complexes. Angew. Chem. Int. Ed. 54, 838–840 (2015).
Shirakawa, S., Yamamoto, K., Tokuda, T. & Maruoka, K. Phase-transfer-catalyzed asymmetric α-arylation of α-amino acid derivatives. Asian J. Org. Chem. 3, 433–436 (2014).
Ye, C.-X., Shen, X., Chen, S. & Meggers, E. Stereocontrolled 1,3-nitrogen migration to access chiral α-amino acids. Nat. Chem. 14, 566–573 (2022).
Tian, S.-K. & Deng, L. A highly enantioselective chiral Lewis base-catalyzed asymmetric cyanation of ketones. J. Am. Chem. Soc. 123, 6195–6196 (2001).
Yabu, K. et al. Switching enantiofacial selectivities using one chiral source: catalytic enantioselective synthesis of the key intermediate for (20S)-camptothecin family by (S)-selective cyanosilylation of ketones. J. Am. Chem. Soc. 123, 9908–9909 (2001).
Deng, H., Isler, M. P., Snapper, M. L. & Hoveyda, A. H. Aluminum-catalyzed asymmetric addition of TMSCN to aromatic and aliphatic ketones promoted by an easily accessible and recyclable peptide ligand. Angew. Chem. Int. Ed. 41, 1009–1012 (2002).
Tokuda, O., Kano, T., Gao, W.-G., Ikemoto, T. & Maruoka, K. A practical synthesis of (S)-2-cyclohexyl-2-phenylglycolic acid via organocatalytic asymmetric construction of a tetrasubstituted carbon center. Org. Lett. 7, 5103–5105 (2005).
Liu, J. et al. Asymmetric direct aldol reaction of functionalized ketones catalyzed by amine organocatalysts based on bispidine. J. Am. Chem. Soc. 130, 5654–5655 (2008).
Roy, S., Sharma, A., Chattopadhyay, N. & Chattopadhyay, S. An efficient asymmetric synthesis of (S)-2-cyclohexyl-2-phenylglycolic acid, the acid segment of oxybutynin. Tetrahedron Lett. 47, 7067–7069 (2006).
Wang, F., Liu, X., Zhang, Y., Lin, L. & Feng, X. Highly enantioselective synthesis of tertiary alcohols: C2-symmetric N,N′-dioxide–Sc(III) complex promoted direct aldol reaction of α-ketoesters and diazoacetate esters. Chem. Commun., 7297−7299, (2009).
Shaw, S. A., Aleman, P. & Vedejs, E. Development of chiral nucleophilic pyridine catalysts: applications in asymmetric quaternary carbon synthesis. J. Am. Chem. Soc. 125, 13368–13369 (2003).
Fisk, J. S., Mosey, R. A. & Tepe, J. J. The diverse chemistry of oxazol-5-(4H)-ones. Chem. Soc. Rev. 36, 1432–1440 (2007).
Marra, I. F. S., de Castro, P. P., Carpanez, A. G. & Amarante, G. W. Azlactone reaction developments. Chem. Eur. J. 22, 10294–10318 (2016).
Marra, I. F. S., de Castro, P. P. & Amarante, G. W. Recent advances in azlactone transformations. Eur. J. Org. Chem. 2019, 5830–5855 (2019).
Jain, A. & Rana, N. K. Review on asymmetric catalysis employing 5H-oxazol-4-ones as α-hydroxy carboxylic acid surrogates. Adv. Synth. Catal. 363, 3879–3912 (2021).
Bellina, F. & Rossi, R. Transition metal-catalyzed direct arylation of substrates with activated sp3-hybridized C–H bonds and some of their synthetic equivalents with aryl halides and pseudohalides. Chem. Rev. 110, 1082–1146 (2010).
Johansson, C. C. C. & Colacot, T. J. Metal-catalyzed α-arylation of carbonyl and related molecules: novel trends in C–C bond formation by C–H bond functionalization. Angew. Chem. Int. Ed. 49, 676–707 (2010).
Hao, Y.-J., Hu, X.-S., Zhou, Y., Zhou, J. & Yu, J.-S. Catalytic enantioselective α-arylation of carbonyl enolates and related compounds. ACS Catal. 10, 955–993 (2020).
Satoh, T., Kawamura, Y., Miura, M. & Nomura, M. Palladium-catalyzed regioselective mono- and diarylation reactions of 2-phenylphenols and naphthols with aryl halides. Angew. Chem. Int. Ed. Engl. 36, 1740–1742 (1997).
Hamann, B. C. & Hartwig, J. F. Palladium-catalyzed direct α-arylation of ketones. Rate acceleration by sterically hindered chelating ligands and reductive elimination from a transition metal enolate complex. J. Am. Chem. Soc. 119, 12382–12383 (1997).
Palucki, M. & Buchwald, S. L. Palladium-catalyzed α-arylation of ketones. J. Am. Chem. Soc. 119, 11108–11109 (1997).
Liu, X. & Hartwig, J. F. Palladium-catalyzed α-arylation of azlactones to form quaternary amino acid derivatives. Org. Lett. 5, 1915–1918 (2003).
Chai, Z., Wang, B., Chen, J.-N. & Yang, G. Arylation of azlactones using diaryliodonium bromides and silver carbonate: facile access to α-tetrasubstituted amino acid derivatives. Adv. Synth. Catal. 356, 2714–2718 (2014).
Uraguchi, D., Ueki, Y. & Ooi, T. Chiral organic ion pair catalysts assembled through a hydrogen-bonding network. Science 326, 120–123 (2009).
Pan, Z. et al. Palladium/TY-Phos-catalyzed asymmetric intermolecular α-arylation of aldehydes with aryl bromides. Angew. Chem. Int. Ed. 60, 18542–18546 (2021).
Li, S., Chen, Q., Yang, J. & Zhang, J. Palladium-catalyzed enantioselective γ-arylation of β,γ-unsaturated butenolides. Angew. Chem. Int. Ed. 61, e202202046 (2022).
Xu, B. et al. Palladium/Xu-Phos-catalyzed enantioselective cascade Heck/remote C(sp2)–H alkylation reaction. Chem 8, 836–849 (2022).
Netz, I., Kucukdisli, M. & Opatz, T. Enantioselective synthesis of α-quaternary amino acids by alkylation of deprotonated α-aminonitriles. J. Org. Chem. 80, 6864–6869 (2015).
Huang, Z. et al. Arene CH–O hydrogen bonding: a stereocontrolling tool in palladium-catalyzed arylation and vinylation of ketones. Angew. Chem. Int. Ed. 52, 4906–4911 (2013).
Pinheiro, D. L. J., Nielsen, D. U., Amarante, G. W. & Skrydstrup, T. Pd-catalyzed carbonylative Α-arylation of azlactones: a formal four-component coupling route to α, α-disubstituted amino acids. J. Catal. 364, 366–370 (2018).
Johnson, E. R. et al. Revealing noncovalent interactions. J. Am. Chem. Soc. 132, 6498–6506 (2010).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2012).
Wiberg, K. B. Application of the Pople–Santry–Segal CNDO method to the cyclopropylcarbinyl and cyclobutyl cation and to bicyclobutane. Tetrahedron 24, 1083–1096 (1968).
Huang, Z. et al. Weak arene C−H⋅⋅⋅O hydrogen bonding in palladium-catalyzed arylation and vinylation of lactones. Angew. Chem. Int. Ed. 52, 5807–5812 (2013).
Yang, J. & Zhou, J. A general method for asymmetric arylation and vinylation of silyl ketene acetals. Org. Chem. Front. 1, 365–367 (2014).
Kalkman, E. D. & Hartwig, J. F. Direct observation of diastereomeric α-C-bound enolates during enantioselective α-arylations: synthesis, characterization, and reactivity of arylpalladium fluorooxindole complexes. J. Am. Chem. Soc. 143, 11741–11750 (2021).
Acknowledgements
We gratefully acknowledge funding support from the National Key R&D Program of China (2021YFF0701600, J.Y. and J.Z.), NSFC (21901043, J.Y.; 21921003, J.Z.; 22031004, J.Z.), STCSM (21ZR1445900, J.Y.) and Shanghai Municipal Education Commission (20212308, J.Z.).
Author information
Authors and Affiliations
Contributions
J.Y. conceived the project and analysed the data. J.Y. and J.Z wrote the manuscript. S.Q. performed most of the experiments. W.Y., Y.H. and L.P. contributed to the synthesis of ligands and substrate. J.Y. did the DFT calculations. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Giovanni Amarante and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary handling editor: Thomas West, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–6, Discussion and Tables 1–9.
Supplementary Data 1
Crystallographic Data for 5ab, CCDC 2233239.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qi, S., Ye, W., Hua, Y. et al. Ligand-enabled palladium-catalysed enantioselective synthesis of α-quaternary amino and glycolic acids derivatives. Nat. Synth 3, 357–367 (2024). https://doi.org/10.1038/s44160-023-00448-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44160-023-00448-7
This article is cited by
-
Making α-aryl quaternary stereocentres
Nature Synthesis (2024)