Abstract
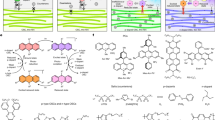
Graphene-like molecules with a zigzag periphery are interesting because of their ability to host spin-polarized electronic edge states. Although progress has been made for the preparation of armchair-edged graphene-like molecules, a general synthetic strategy to zigzag type remains elusive. Herein, using acetylenedicarboxylates as the C2 insertion unit, we report a rapid and modular strategy to extend the π-conjugation in a zigzag fashion through a rhodium-catalysed sequential C2–H and C8–H activation-annulation of naphthalene ketones. Different from the reported C–H activation-annulation of aryl ketones with alkynes to form a five-membered indenol, fulvene or six-membered pyran oxonium, this [4 + 2] and [4 + 2] annulation sequence selectively undergoes C(sp3)–H cyclization to extend the naphthalene fragment. This programmable zigzag π-extension can be compared to the stacking of toy blocks, as the naphthalene fragments are added step by step along an alkyl chain, demonstrated by the synthesis of anthanthrene and subsequently peri-naphthacenonaphthacene. These products are easily transformed to electron transport materials and thermally activated delayed fluorescent materials.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the article and its Supplementary Information. The X-ray crystallographic coordinates for structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre (CCDC) under deposition number CCDC 2153377 (26), 2170028 (34), 2153378 (37) and 2153379 (52). These data can be obtained free of charge from the CCDC via www.ccdc.cam.ac.uk/data_request/cif. Source data are provided with this paper.
References
Anthony, J. E. Functionalized acenes and heteroacenes for organic electronics. Chem. Rev. 106, 5028–5048 (2006).
Wu, J., Pisula, W. & Müllen, K. Graphenes as potential material for electronics. Chem. Rev. 107, 718–747 (2007).
Figueira-Duarte, T. M. & Müllen, K. Pyrene-based materials for organic electronics. Chem. Rev. 111, 7260–7314 (2011).
Narita, A., Wang, X.-Y., Feng, X. & Müllen, K. New advances in nanographene chemistry. Chem. Soc. Rev. 44, 6616–6643 (2015).
Segawa, Y., Ito, H. & Itami, K. Structurally uniform and atomically precise carbon nanostructures. Nat. Rev. Mater. 1, 15002 (2016).
Ito, H., Ozaki, K. & Itami, K. Annulative π-extension (APEX): rapid access to fused arenes, heteroarenes, and nanographenes. Angew. Chem. Int. Ed. 56, 11144–11164 (2017).
Koga, Y., Kaneda, T., Saito, Y., Murakami, K. & Itami, K. Synthesis of partially and fully fused polyaromatics by annulative chlorophenylene dimerization. Science 359, 435–439 (2018).
Ozaki, K., Kawasumi, K., Shibata, M., Ito, H. & Itami, K. One-shot K-region-selective annulative π-extension for nanographene synthesis and functionalization. Nat. Commun. 6, 6251 (2015).
Matsuoka, W., Ito, H., Sarlah, D. & Itami, K. Diversity-oriented synthesis of nanographenes enabled by dearomative annulative π-extension. Nat. Commun. 12, 3940 (2021).
Hein, S. J., Lehnherr, D., Arslan, H., Uribe-Romo, F. J. & Dichtel, W. R. Alkyne benzannulation reactions for the synthesis of novel aromatic architectures. Acc. Chem. Res. 50, 2776–2788 (2017).
Senese, A. D. & Chalifoux, W. A. Nanographene and graphene nanoribbon synthesis via alkyne benzannulations. Molecules 24, 118 (2019).
Chen, Q., Schollmeyer, D., Müllen, K. & Narita, A. Synthesis of circumpyrene by alkyne benzannulation of brominated dibenzo[hi,st]ovalene. J. Am. Chem. Soc. 141, 19994–19999 (2019).
Steiner, A.-K. & Amsharov, K. Y. The rolling-up of oligophenylenes to nanographenes by a HF-zipping approach. Angew. Chem. Int. Ed. 56, 14732–14736 (2017).
Kolmer, M. et al. Fluorine-programmed nanozipping to tailored nanographenes on rutile TiO2 surfaces. Science 363, 57–60 (2019).
Ruffieux, P. et al. On-surface synthesis of graphene nanoribbons with zigzag edge topology. Nature 531, 489–492 (2016).
Mishra, S. et al. Large magnetic exchange coupling in rhombus-shaped nanographenes with zigzag periphery. Nat. Chem. 13, 581–586 (2021).
Lungerich, D. et al. Dehydrative π-extension to nanographenes with zig-zag edges. Nat. Commun. 9, 4756 (2018).
Gu, Y., Wu, X., Gopalakrishna, T. Y., Phan, H. & Wu, J. Graphene-like molecules with four zigzag edges. Angew. Chem. Int. Ed. 57, 6541–6545 (2018).
Zeng, W. et al. Superoctazethrene: an open-shell graphene-like molecule possessing large diradical character but still with reasonable stability. J. Am. Chem. Soc. 140, 14054–14058 (2018).
Ajayakumar, M. R. et al. Toward full zigzag-edged nanographenes: peri-tetracene and its corresponding circumanthracene. J. Am. Chem. Soc. 140, 6240–6244 (2018).
Colby, D. A., Bergman, R. G. & Ellman, J. A. Rhodium-catalyzed C–C bond formation via heteroatom-directed C–H bond activation. Chem. Rev. 110, 624–655 (2010).
Song, G., Wang, F. & Li, X. C–C, C–O and C–N bond formation via rhodium(III)-catalyzed oxidative C–H activation. Chem. Soc. Rev. 41, 3651–3678 (2012).
Yang, Y. et al. Rhodium-catalyzed annulation of arenes with alkynes through weak chelation-assisted C–H activation. Chem. Commun. 52, 2872–2884 (2016).
Huang, Z., Lim, H. N., Mo, F., Young, M. C. & Dong, G. Transition metal-catalyzed ketone-directed or mediated C–H functionalization. Chem. Soc. Rev. 44, 7764–7786 (2015).
Patureau, F. W., Besset, T., Kuhl, N. & Glorius, F. Diverse strategies toward indenol and fulvene derivatives: Rh-catalyzed C–H activation of aryl ketones followed by coupling with internal alkynes. J. Am. Chem. Soc. 133, 2154–2156 (2011).
Muralirajan, K., Parthasarathy, K. & Cheng, C.-H. Regioselective synthesis of indenols by rhodium-catalyzed C–H activation and carbocyclization of aryl ketones and alkynes. Angew. Chem. Int. Ed. 50, 4169–4172 (2011).
Liu, X., Li, G., Song, F. & You, J. Unexpected regioselective carbon–hydrogen bond activation/cyclization of indolyl aldehydes or ketones with alkynes to benzo-fused oxindoles. Nat. Commun. 5, 5030 (2014).
Yin, J. & You, J. Concise synthesis of polysubstituted carbohelicenes by a C–H activation/radical reaction/C–H activation sequence. Angew. Chem. Int. Ed. 58, 302–306 (2019).
Yin, J. et al. Acyl radical to rhodacycle addition and cyclization relay to access butterfly flavylium fluorophores. Nat. Commun. 10, 5664 (2019).
Stuart, D. R., Alsabeh, P., Kuhn, M. & Fagnou, K. Rhodium(III)-catalyzed arene and alkene C–H bond functionalization leading to indoles and pyrroles. J. Am. Chem. Soc. 132, 18326–18339 (2010).
Mochida, S., Shimizu, M., Hirano, K., Satoh, T. & Miura, M. Synthesis of naphtho[1,8-bc]pyran drivatives and related cmpounds through hydroxy group directed C–H bond cleavage under rhodium catalysis. Chem. Asian J. 5, 847–851 (2010).
Tan, X. et al. Rhodium-catalyzed cascade oxidative annulation leading to substituted naphtho[1,8-bc]pyrans by sequential cleavage of C(sp2)–H/C(sp3)–H and C(sp2)–H/O–H bonds. J. Am. Chem. Soc. 134, 16163–16166 (2012).
Yin, J. et al. Synthesis of phenalenyl-fused pyrylium cations: divergent C–H activation/annulation reaction sequence of naphthalene aldehydes with alkynes. Angew. Chem. Int. Ed. 56, 13094–13098 (2017).
Yin, J. et al. Annulation cascade of arylnitriles with alkynes to stable delocalized PAH carbocations via intramolecular rhodium migration. Chem. Sci. 9, 5488–5493 (2018).
Neochoritis, C. G., Zarganes-Tzitzikas, T. & Stephanidou-Stephanatou, J. Dimethyl acetylenedicarboxylate: a versatile tool in organic synthesis. Synthesis 46, 0537–0585 (2014).
Gerfaud, T., Neuville, L. & Zhu, J. Palladium-catalyzed annulation of acyloximes with arynes (or alkynes): synthesis of phenanthridines and isoquinolines. Angew. Chem. Int. Ed. 48, 572–577 (2009).
Uoyama, H., Goushi, K., Shizu, K., Nomura, H. & Adachi, C. Highly efficient organic light-emitting diodes from delayed fluorescence. Nature 492, 234–238 (2012).
Huang, Z. et al. Molecular design of non-doped OLEDs based on a twisted heptagonal acceptor: A delicate balance between rigidity and rotatability. Angew. Chem. Int. Ed. 59, 9992–9996 (2020).
Frisch, M. J. et al. Gaussian 09, Revision D.01 (Gaussian, Inc., 2013).
Becke, A. D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993).
Lee, C., Yang, W. & Parr, R. G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37, 785–789 (1988).
Zhao, Y. & Truhlar, D. G. The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor. Chem. Acc. 120, 215–241 (2008).
Cossi, M., Barone, V., Cammi, R. & Tomasi, J. Ab initio study of solvated molecules: a new implementation of the polarizable continuum model. Chem. Phys. Lett. 255, 327–335 (1996).
Hruszkewycz, D. P., Miles, K. C., Thielb, O. R. & Stahl, S. S. Co/NHPI-mediated aerobic oxygenation of benzylic C–H bonds in pharmaceutically relevant molecules. Chem. Sci. 8, 1282 (2017).
Quinn, J. T. E., Zhu, J., Li, X., Wang, J. & Li, Y. Recent progress in the development of n-type organic semiconductors for organic field effect transistors. J. Mater. Chem. C 5, 8654 (2017).
Minder, N. A., Ono, S., Chen, Z., Facchetti, A. & Morpurgo, A. F. Band-like electron transport in organic transistors and implication of the molecular structure for performance optimization. Adv. Mater. 24, 503–508 (2012).
Wu, Z.-H. et al. 4,5,9,10-Pyrene diimides: a family of aromatic diimides exhibiting high electron mobility and two-photon excited emission. Angew. Chem. Int. Ed. 56, 13031–13035 (2017).
Tao, Y. et al. Thermally activated delayed fluorescence materials towards the breakthrough of organoelectronics. Adv. Mater. 26, 7931–7958 (2014).
Wong, M. Y. & Zysman-Colman, E. Purely organic thermally activated delayed fluorescence materials for organic light-emitting diodes. Adv. Mater. 29, 1605444 (2017).
Acknowledgements
We acknowledge support for this work from the National Natural Science Foundation of China (No. 22031007, J.You) and from the Office of China Postdoctoral Council, the International Postdoctoral Exchange Fellowship Program (No. 20190088, J.Yin).
Author information
Authors and Affiliations
Contributions
J. Yin, J.L., Y.W. and Y.Z. performed the experiments and analysed the data. H.C. performed the DFT calculations. J. You designed and directed the project. Y.L. directed the DFT calculations. C.Z. directed the OTFT measurements. Z.B. directed the OLED measurements. J. Yin, J. You, J.L., H.C., D.P., Y.Y. and Y.L. wrote the manuscript. All authors contributed to discussions.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Haibo Ge, Andreas Hirsch and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Alison Stoddart, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Experimental Details, Sections I–XIV, Supplementary Tables 1–10 and Figs. 1–10.
Supplementary Data 1
Cif file for compound 26, CCDC 2153377.
Supplementary Data 2
Structure factors for compound 26, CCDC 2153377.
Supplementary Data 3
Cif file for compound 34, CCDC 2170028.
Supplementary Data 4
Structure factors for compound 34, CCDC 2170028.
Supplementary Data 5
Cif file for compound 37, CCDC 2153378.
Supplementary Data 6
Structure factors for compound 37, CCDC 2153378.
Supplementary Data 7
Cif file for compound 52, CCDC 2153379.
Supplementary Data 8
Structure factors for compound 52, CCDC 2153379.
Source data
Source Data Fig. 6
Source Data for Fig. 6b and 6d.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yin, J., Li, J., Chen, H. et al. Programmable zigzag π-extension toward graphene-like molecules by the stacking of naphthalene blocks. Nat. Synth 2, 838–847 (2023). https://doi.org/10.1038/s44160-023-00306-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44160-023-00306-6