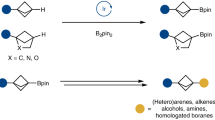
Abstract
Alkynes are fundamental molecular building blocks of great importance in synthetic chemistry and materials science. The efficient construction of alkynyl functionality from widely accessible functional groups is therefore highly valuable. Here we report the development of a modular synthesis of alkynes through reaction of carboxylic esters with lithiated gem-diborylalkanes and aryl triflimides. 1H, 13C and 11B NMR spectroscopic and X-ray crystallographic analyses identify and characterize an intermediate α-boryl lithium enolate, which upon triflation with an aryl triflimide followed by water quenching generates the alkyne product. The developed strategy allows a range of carboxylic acid esters to be converted into both internal and terminal alkynes, within a short reaction time, including the transformation of chiral α-substituted esters into chiral propargyl compounds without racemization. The method has been applied to the synthesis of 13C-labelled alkynes, using 13C-labelled gem-diborylmethane and 13C-labelled carboxylic esters, which enables access to 13C-labelled terminal alkyne fragments in drugs and natural products.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data are available in the main text or the Supplementary Information. Solid-state structure of I-B is freely available from the Cambridge Crystallographic Data Centre under the associated CCDC codes: 2183417.
References
Knochel, P. & Molander, G. A. Comprehensive Organic Synthesis 2nd edn (Elsevier, 2014).
Olah, G. A., Molnár, Á. & Prakash, G. K. S. Hydrocarbon Chemistry 3rd edn (John Wiley, 2017).
Trost, B. M. & Li, C.-J. Modern Alkyne Chemistry: Catalytic and Atom-Economic Transformations (Wiley-VCH, 2014).
Hopf, H. Science of Synthesis Vol. 43 (Thieme Chemistry, 2008).
Lehmann, J., Wright, M. H. & Sieber, S. A. Making a long journey short: alkyne functionalization of natural product scaffolds. Chem. Eur. J. 22, 4666–4678 (2016).
Steins, M., Thomas, M. & Geißler, M. in Small Molecules in Oncology (ed. Martens, U. M.) 1–17 (Springer, 2018).
Brunner, H. Narcotic drug methohexital: synthesis by enantioselective catalysis. Chirality 13, 420–424 (2001).
Madar, D. J. et al.Discovery of 2-[4-{{2-(2S,5R)-2-Cyano-5-ethynyl-1-pyrrolidinyl]-2-oxoethyl]amino]-4-methyl-1-piperidinyl]-4-pyridinecarboxylic acid (ABT-279): a very potent, selective, effective, and well-tolerated inhibitor of dipeptidyl peptidase-IV, useful for the treatment of diabetes. J. Med. Chem. 49, 6416–6420 (2006).
Thirumurugan, P., Matosiuk, D. & Jozwiak, K. Click chemistry for drug development and diverse chemical–biology applications. Chem. Rev. 113, 4905–4979 (2013).
Parker, C. G. & Pratt, M. R. Click chemistry in proteomic investigations. Cell 180, 605–632 (2020).
Bi, X., Miao, K. & Wei, L. Alkyne-tagged Raman probes for local environmental sensing by hydrogen–deuterium exchange. J. Am. Chem. Soc. 144, 8504–8514 (2022).
Lam, J., Breteler, H., Arnason, T. & Hansen, L. Chemistry and Biology of Naturally-Occurring Acetylenes and Related Compounds (Elsevier, 1988).
Chinchilla, R. & Nájera, C. The Sonogashira reaction: a booming methodology in synthetic organic chemistry. Chem. Rev. 107, 874–922 (2007).
Chinchilla, R. & Nájera, C. Recent advances in Sonogashira reactions. Chem. Soc. Rev. 40, 5084–5121 (2011).
Fürstner, A. Alkyne metathesis on the rise. Angew. Chem. Int. Ed. 52, 2794–2819 (2013).
Habrant, D., Rauhala, V. & Koskinen, A. M. P. Conversion of carbonyl compounds to alkynes: general overview and recent developments. Chem. Soc. Rev. 39, 2007–2017 (2010).
Shaw, R., Elagamy, A., Althagafi, I. & Pratap, R. Synthesis of alkynes from non-alkyne sources. Org. Biomol. Chem. 18, 3797–3817 (2020).
Lyapkalo, I. M., Vogel, M. A. K., Boltukhina, E. V. & Vavřík, J. Thieme Chemistry Journal awardees—where are they now? A general one-step synthesis of alkynes from enolisable carbonyl compounds. Synlett 2009, 558–561 (2009).
Zha, G.-F. et al. SO2F2-mediated oxidative dehydrogenation and dehydration of alcohols to alkynes. J. Am. Chem. Soc. 140, 17666–17673 (2018).
Fleming, I. & Ramarao, C. Decarboxylative elimination of enol triflates as a general synthesis of acetylenes. Chem. Commun. 1113-1114 (1999).
Katritzky, A. R., Wang, J., Karodia, N. & Li, J. A novel transformation of esters to alkynes with 1-substituted benzotriazoles. J. Org. Chem. 62, 4142–4147 (1997).
Eymery, F., Iorga, B. & Savignac, P. The usefulness of phosphorus compounds in alkyne synthesis. Synthesis 2000, 185–213 (2000).
Yang, K. & Song, Q. Tetracoordinate boron intermediates enable unconventional transformations. Acc. Chem. Res. 54, 2298–2312 (2021).
Taniguchi, T. Substituent effects of tetracoordinate boron in organic synthesis. Chem. Eur. J. 28, e202104333 (2022).
Shi, D., Xia, C. & Liu, C. Photoinduced transition-metal-free alkynylation of alkyl pinacol boronates. CCS Chem. 3, 1718–1728 (2021).
Sumida, Y., Kato, T. & Hosoya, T. Generation of arynes via ate complexes of arylboronic esters with an ortho-leaving group. Org. Lett. 15, 2806–2809 (2013).
Sun, W., Wang, L., Xia, C. & Liu, C. Dual functionalization of α-monoboryl carbanions through deoxygenative enolization with carboxylic acids. Angew. Chem. Int. Ed. 57, 5501–5505 (2018).
Sun, W. et al. Chemodivergent transformations of amides using gem-diborylalkanes as pro-nucleophiles. Nat. Commun. 11, 3113 (2020).
Lee, B. & Chirik, P. J. Ketone synthesis from benzyldiboronates and esters: leveraging α-boryl carbanions for carbon–carbon bond formation. J. Am. Chem. Soc. 142, 2429–2437 (2020).
Mukaiyama, T., Murakami, M., Oriyama, T. & Yamaguchi, M. A new aldol reaction: a method for the generation of vinyloxyboranes by the acylation of boron-stabilized carbanions. Chem. Lett. 10, 1193–1196 (1981).
Matteson, D. S. & Moody, R. J. Carbanions from deprotonation of gem-diboronic esters. J. Am. Chem. Soc. 99, 3196–3197 (1977).
Iacono, C. E., Stephens, T. C., Rajan, T. S. & Pattison, G. A coupling approach for the generation of α,α-bis(enolate) equivalents: regioselective synthesis of gem-difunctionalized ketones. J. Am. Chem. Soc. 140, 2036–2040 (2018).
Shi, D., Wang, L., Xia, C. & Liu, C. Synthesis of secondary and tertiary alkyl boronic esters by gem-carboborylation: carbonyl compounds as bis(electrophile) equivalents. Angew. Chem. Int. Ed. 57, 10318–10322 (2018).
Lee, Y., Han, S. & Cho, S. H. Catalytic chemo- and enantioselective transformations of gem-diborylalkanes and (diborylmethyl)metallic species. Acc. Chem. Res. 54, 3917–3929 (2021).
Miralles, N., Maza, R. J. & Fernández, E. Synthesis and reactivity of 1,1-diborylalkanes towards C–C bond formation and related mechanisms. Adv. Synth. Catal. 360, 1306–1327 (2018).
Wu, C. & Wang, J. Geminal bis(boron) compounds: their preparation and synthetic applications. Tetrahedron Lett. 59, 2128–2140 (2018).
Nallagonda, R., Padala, K. & Masarwa, A. gem-Diborylalkanes: recent advances in their preparation, transformation and application. Org. Biomol. Chem. 16, 1050–1064 (2018).
Nallagonda, R. et al. in Patai’s Chemistry of Functional Groups 1–72 (Wiley, 2020).
Wang, L. et al. C–O functionalization of α-oxyboronates: a deoxygenative gem-diborylation and gem-silylborylation of aldehydes and ketones. J. Am. Chem. Soc. 139, 5257–5264 (2017).
He, Z. et al. Cooperation between an alcoholic proton and boryl species in the catalytic gem-hydrodiborylation of carboxylic esters to access 1,1-diborylalkanes. Org. Chem. Front. 6, 900–907 (2019).
Li, H. et al. Formal carbon insertion of N-tosylhydrazone into B–B and B–Si bonds: gem-diborylation and gem-silylborylation of sp3 carbon. Org. Lett. 16, 448–451 (2014).
Palmer, W. N., Obligacion, J. V., Pappas, I. & Chirik, P. J. Cobalt-catalyzed benzylic borylation: enabling polyborylation and functionalization of remote, unactivated C(sp3)–H bonds. J. Am. Chem. Soc. 138, 766–769 (2016).
Li, L., Gong, T., Lu, X., Xiao, B. & Fu, Y. Nickel-catalyzed synthesis of 1,1-diborylalkanes from terminal alkenes. Nat. Commun. 8, 345 (2017).
Teo, W. J. & Ge, S. Cobalt-catalyzed diborylation of 1,1-disubstituted vinylarenes: a practical route to branched gem-bis(boryl)alkanes. Angew. Chem. Int. Ed. 57, 1654–1658 (2018).
Wang, X. et al. Zirconium-catalyzed atom-economical synthesis of 1,1-diborylalkanes from terminal and internal alkenes. Angew. Chem. Int. Ed. 59, 13608–13612 (2020).
Docherty, J. H., Nicholson, K., Dominey, A. P. & Thomas, S. P. A boron–boron double transborylation strategy for the synthesis of gem-diborylalkanes. ACS Catal. 10, 4686–4691 (2020).
Lee, H., Lee, Y. & Cho, S. H. Palladium-catalyzed chemoselective Negishi cross-coupling of bis[(pinacolato)boryl]methylzinc halides with aryl (pseudo)halides. Org. Lett. 21, 5912–5916 (2019).
Kumar, N., Eghbarieh, N., Stein, T., Shames, A. I. & Masarwa, A. Photoredox-mediated reaction of gem-diborylalkenes: reactivity toward diverse 1,1-bisborylalkanes. Chem. Eur. J. 26, 5360–5364 (2020).
Endo, K., Hirokami, M. & Shibata, T. Synthesis of 1,1-organodiboronates via Rh(I)Cl-catalyzed sequential regioselective hydroboration of 1-alkynes. Synlett 2009, 1331–1335 (2009).
Lee, J. C. H., McDonald, R. & Hall, D. G. Enantioselective preparation and chemoselective cross-coupling of 1,1-diboron compounds. Nat. Chem. 3, 894–899 (2011).
Chassaing, S., Specklin, S., Weibel, J.-M. & Pale, P. Vinyl and aryl sulfonates: preparations and applications in total synthesis. Curr. Org. Synth. 9, 806–827 (2012).
Elmore, C. S. in Annual Reports in Medicinal Chemistry Vol. 44 (ed. Macor, J. E.) 515–534 (Academic Press, 2009).
Wang, Z. J. et al. Hyperpolarized 13C MRI: state of the art and future directions. Radiology 291, 273–284 (2019).
Dale, H. J. A., Nottingham, C., Poree, C. & Lloyd-Jones, G. C. Systematic evaluation of 1,2-migratory aptitude in alkylidene carbenes. J. Am. Chem. Soc. 143, 2097–2107 (2021).
Masui, Y., Hattori, T. & Onaka, M. Reversible generation of labile secondary carbocations from alcohols in the nanospace of H-mordenite and their long-lasting preservation at ambient temperature. J. Am. Chem. Soc. 139, 8612–8620 (2017).
Lahann, J. Click Chemistry for Biotechnology and Materials Science (Wiley, 2009).
Lee, Y., Park, J. & Cho, S. H. Generation and application of (diborylmethyl)zinc(II) species: access to enantioenriched gem-diborylalkanes by an asymmetric allylic substitution. Angew. Chem. Int. Ed. 57, 12930–12934 (2018).
Konno, T. Trifluoromethylated internal alkynes: versatile building blocks for the preparation of various fluorine-containing molecules. Synlett 25, 1350–1370 (2014).
Zhou, Y., Zhang, Y. & Wang, J. Recent advances in transition-metal-catalyzed synthesis of conjugated enynes. Org. Biomol. Chem. 14, 6638–6650 (2016).
Dickson, H. D., Smith, S. C. & Hinkle, K. W. A convenient scalable one-pot conversion of esters and Weinreb amides to terminal alkynes. Tetrahedron Lett. 45, 5597–5599 (2004).
Stephens, T. C. & Pattison, G. Transition-metal-free homologative cross-coupling of aldehydes and ketones with geminal bis(boron) compounds. Org. Lett. 19, 3498–3501 (2017).
Liu, J., Lam, J. W. & Tang, B. Z. Acetylenic polymers: syntheses, structures, and functions. Chem. Rev. 109, 5799–5867 (2009).
McCormick, K. D. et al. Preparation of imidazolylmethylbenzoxazines and related compounds as α2c adrenoreceptor agonists. US patent 20080039439 (2008).
Zhu, M. & Messaoudi, S. Diastereoselective decarboxylative alkynylation of anomeric carboxylic acids using Cu/photoredox dual catalysis. ACS Catal. 11, 6334–6342 (2021).
Liu, X., Ming, W., Zhang, Y., Friedrich, A. & Marder, T. B. Copper-catalyzed triboration: straightforward. atom-economical synthesis of 1,1,1-triborylalkanes from terminal alkynes and HBpin. Angew. Chem. Int. Ed. 58, 18923–18927 (2019).
Matuszewski, M., Kiliszek, A., Rypniewski, W., Lesnikowski, Z. J. & Olejniczak, A. B. Nucleoside bearing boron clusters and their phosphoramidites—building blocks for modified oligonucleotide synthesis. New J. Chem. 39, 1202–1221 (2015).
Tietze, L. F. et al. Novel carboranyl C-glycosides for the treatment of cancer by boron neutron capture therapy. Chem. Eur. J. 9, 1296–1302 (2003).
Valliant, J. F. et al. The medicinal chemistry of carboranes. Coord. Chem. Rev. 232, 173–230 (2002).
Acknowledgements
We thank the National Natural Science Foundation of China (number 22022113 to C.L.) and the Natural Science Foundation of Jiangsu Province (number BK20190002 to C.L.) for financial support. We also thank J. Song and K. Xiao (NJUPT) for assistance with the crystal structure analysis.
Author information
Authors and Affiliations
Contributions
W.S. and L.X. conducted the experiments and collected data. Y.Q. helped to collect some data. C.L. directed the project and wrote the manuscript with input from all authors. All authors analysed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Synthesis thanks Ahmad Masarwa and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Thomas West, in collaboration with the Nature Synthesis team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary sections 1–3, Figs. 1–5 and Tables 1 and 2.
Supplementary Data 1
Crystallographic data for compound I-B; CCDC 2183417.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, W., Xu, L., Qin, Y. et al. Alkyne synthesis through coupling of gem-diborylalkanes with carboxylic acid esters. Nat. Synth 2, 413–422 (2023). https://doi.org/10.1038/s44160-023-00243-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44160-023-00243-4
This article is cited by
-
Triple coupling to make alkynes
Nature Synthesis (2023)