Abstract
Carbon accounting in the land sector requires a reference level from which to calculate past losses of carbon and potential for gains using a stock-based target. Carbon carrying capacity represented by the carbon stock in primary forests is an ecologically-based reference level that allows estimation of the mitigation potential derived from protecting and restoring forests to increase their carbon stocks. Here we measured and collated tree inventory data at primary forest sites including from research studies, literature and forest inventories (7982 sites, 288,262 trees, 27 countries) across boreal, temperate, and subtropical Global Ecological Zones within Europe. We calculated total biomass carbon stock per hectare (above- and below-ground, dead biomass) and found it was 1.6 times larger on average than modelled global maps for primary forests and 2.3 times for all forests. Large trees (diameter greater than 60 cm) accounted for 50% of biomass and are important carbon reservoirs. Carbon stock foregone by harvesting of 12–52% demonstrated the mitigation potential. Estimated carbon gain by protecting, restoring and ongoing growth of existing forests equated to 309 megatons carbon dioxide equivalents per year, additional to, and higher than, the current forest sink, and comparable to the Green Deal 2030 target for carbon dioxide removals.
Similar content being viewed by others
Introduction
International negotiations over climate change have reached agreements and targets - Paris Agreement1 and Glasgow Climate Pact2 - with the goal of stabilising atmospheric CO2 concentration and temperature increase to well below 2 °C above pre-industrial levels and pursue efforts to limit warming to 1.5 °C3. However, operationalising these commitments to achieve a true reduction in the carbon stock in the atmosphere requires re-thinking some fundamental concepts of carbon accounting, revising specific guidelines and rules, and collecting the necessary data. The land sector is particularly problematic where flows of carbon between the biosphere and atmosphere are naturally two-way and the reservoirs that store carbon in the biosphere differ in their ecosystem condition, in terms of stability, longevity, and resilience to disturbances. Current accounting methods do not distinguish these differences in quality and the gross flows of emissions and removals adequately in reporting of net emissions reductions towards targets. Unintended consequences of mitigation actions have resulted, such as emissions from forest harvesting being netted out against removals from the entire forest area, carbon stocks in forests having different levels of ecosystem condition not being differentiated, the historical debt in carbon stock due to human activities is a permanent loss not being counted, and the effect of the time difference between instantaneous emissions and future removals by tree growth on the resultant carbon stock change in the atmosphere not being included in life-cycle assessments4,5,6,7,8.
We address a key issue in accounting concerning the reference level from which past changes in forest carbon stocks are calculated and future changes are predicted. Using an “ecologically-based reference level” for forest ecosystems is crucial for ensuring consistent information regarding: (i) assessing the carbon stock loss that has occurred in the past due to human activities; (ii) predicting the potential gains in stocks by changing forest management; and (iii) determining the foregone mitigation benefits due to managing forests at carbon stocks below their maximum. This context using a carbon stock-based target provides an alternative accounting solution which can be implemented under the Paris Agreement.
Management of forests is critical for mitigation in terms of both avoiding emissions to, and increasing removals from, the atmosphere. Forest ecosystem are large stores of carbon (~660 PgC in living and dead biomass and soil organic carbon9 and have high potential to sequester additional carbon (with forests constituting most of the terrestrial sink10). Systems for evaluating the mitigation outcomes of forest management strategies, including all carbon stocks and flows through forests, products and energy, are thus crucial and require consistency in monitoring and accounting for changes in stocks. A consistent reference level has been an issue particularly prevalent in continents with a long history of land use, such as Europe, where the effect of human activities on ecosystem carbon stocks is difficult to define because of the transformations of primary forest into managed or degraded ecosystems11,12. Describing the reference level in terms of an ecological state across all forest areas and management systems provides consistency for interpretation of carbon stock changes in the long-term, rather than an historical, business-as-usual or near-term projected level. Reporting in greenhouse gas inventories is only required for managed lands13, although unmanaged forests can be included, and for the European Union, 98% of forest area is monitored and reported14 (S1.1).
The current carbon stock (CCS) of forest ecosystems is a function of natural history, environmental conditions, and natural disturbance regimes, but also human land use, particularly the harvesting and regeneration of trees. The reference level used currently under the UNFCCC to assess each country’s compliance against emissions reduction targets (previously under Kyoto Protocol rules or European LULUCF Regulation), projects future net annual emissions based on the CCS together with forecasted dynamics resulting from management as either the continuation of documented historical practices or future implementation of approved policies15. Using this approach to the reference level may show changes in net emissions due to human activities. However, it cannot be used to predict the potential carbon stock that forests could store if management changed to allow restoration of the maximum stocks for the site conditions given the natural potential of forest ecosystems. To answer the question of potential stock requires an ecologically-based local reference level derived from a forest ecosystem’s carbon-carrying capacity (CCC).
We show that estimating the carbon stored in a primary forest represents a given forest ecosystem type’s CCC - defined as the mass of carbon stored in an ecosystem at landscape scales given the life history traits of the tree species (e.g. longevity), prevailing environmental conditions (noting these are varying due to climate change), and the impacts of natural disturbance regimes, but excluding direct anthropogenic disturbance16. The stock at CCC is the potential stock that can be maintained by natural processes within an ecosystem in a resilient and self-sustaining manner. The residence time of the stored carbon is centennial scales, with cycling of carbon maintained by frequent low- to moderate-severity natural disturbances, as well as some infrequent larger-scale disturbances, such that the carbon stocks remain relatively stable when averaged over long spatial and temporal scales inclusive of the regenerative capacity of ecosystems12,17. However, carbon stocks and relative proportions among ecosystem compartments may be changing with changing climate.
Primary forests are defined by the FAO18 as naturally regenerated forest of native tree species, with no clearly visible indications of direct human activities, and whose composition, structure and dynamics are dominated by ecological and evolutionary processes, including natural disturbance regimes. Resulting forests include a range of tree ages and seral stages at stand and landscape scales (S1.2). In Europe, the long history of land use means that the term is more appropriately interpreted as referring to forests that are long unlogged and have reached a level of maturity including many with old-growth characteristics and a high degree of naturalness and ecological functioning, without implying that there was never human disturbance19,20,21. Compared to secondary forests managed for commodity production, these primary forests have the highest levels of ecosystem integrity22 and store the largest carbon stocks for a given forest type and environmental conditions, because of the relationship between high biomass in large trees and total biomass density23,24,25,26.
We present the theoretical basis and data to support the proposition that using the CCC in primary forest as the ecologically-based reference level can improve accounting for past and predicting future changes in forest carbon stocks. Changes can then be tracked in the condition of forests resulting from increases/decreases in their carbon stocks due to human activities and management strategies. This accounting would improve the transparency and soundness of the predicted mitigation potential of forests because the reference level is defined as an ecological state rather than a construct of human activities that can vary depending on timeframe, location and management legacy. We address this proposition through two research questions.
-
1.
What is the CCC of forest types in Europe that is represented by the extant primary forests and how well are these carbon stocks represented by global default values and models?
-
2.
What is the potential contribution to climate change mitigation by protecting primary forests to avoid carbon stock losses, and restoring secondary forests to gain carbon stocks?
The foci of our study are the primary forests of Europe which serve as a case study of how our approach could have global applications. Extensive human land use (S1.1) present challenges to identifying primary forests. Even though these identified forest areas have least disturbance and highest levels of ecosystem integrity, our estimates of CCC are uncertain and may be conservative. The concept of CCC has not been applied in Europe nor has it been used as the reference level for carbon accounting. The data on primary forests presented here now make this approach to accounting feasible. We found that the large carbon stocks in primary forests have been underestimated previously. Applying an ecologically-based reference level of the CCC revealed the foregone losses due to maintaining forests at carbon stocks below their maximum, and the mitigation potential from gains by ongoing forest growth. Hence, the protection and restoration of primary forests is a critical action for climate mitigation.
Results
Carbon stock density in primary forests across Europe
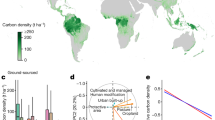
The extant forest area of Europe was studied with field sites located in areas of primary forest from which carbon stocks were calculated (Fig. 1). Above- and below-ground biomass and dead biomass for primary forest sites were aggregated by country and forest type (Fig. 2). To complete the carbon budget, soil carbon data for the site locations was extracted from the global map because there was insufficient soil data from the field measurements (Supplementary Fig. S5). Biomass carbon stock varied by an order of magnitude across countries, from the lowest in alpine birch forest in Sweden (21 MgC ha−1) to the highest in mixed spruce-fir-beech forest in Bosnia-Herzegovina (346 MgC ha−1) (Fig. 2). Variation in dead biomass was not coincident with living biomass; it is influenced by the total amount of biomass but also disturbance factors causing mortality. For example, the highest amount of 113 MgC ha−1 in Czech Republic conifer forest was measured after a severe windstorm (Fig. 2).
Primary forest sites included (i) research sites and (ii) sites reported in the literature that were selected by observation of ecological characteristics, and (iii) forest inventory sites described as primary forest or as natural forest >100 years old and under some protection status. Forest cover was defined by area of CORINE forest types94 and forest canopy cover ≥30% (S1.7) (See Methods: Identification of primary forest sites).
Coloured bars are the mean value and lines are standard deviations for the total biomass. Countries ordered approximately by latitude. Numbers for each column refer to the numbered rows in Supplementary Table S2. Forest types classified by the European CORINE Land Cover94 and then by dominant species according to national systems. Data from national forest inventories (NFIs) were used for Sweden (1, 2, 3), Germany (8), and Romania (25) (no dead biomass data) and so do not have the same level of strict definition of primary forest (see S1.2).
Classified by Global Ecological Zones (GEZs)27 (Supplementary Fig. S1), biomass carbon stocks were similar in temperate and subtropical (Mediterranean) zones, and lower in the boreal. Mountain systems had higher biomass than continental forests. Increasing biomass occurred from conifer to broadleaf and mixed forest types within the temperate and subtropical zones, but the reverse in the boreal (Fig. 3).
Within GEZs, forest types are classified by the European CORINE Land Cover94 (conifer, broadleaf, mixed forest). Coloured bars are mean value from the primary forest site data, lines are standard deviations, and the numbers in each column are the number of sites per class.
Distribution of sites within the environmental space for forest cover within each GEZ illustrates their representativeness in terms of productive potential (Fig. 4). The sites are generally at higher elevation (except inventory sites in temperate zones and all sites in temperate continental and oceanic), sites have lower temperatures in the boreal and subtropical mountain zones, and research sites are drier and hotter in temperate oceanic forest.
Environmental space defined by elevation, mean annual temperature and water availability index (S1.5) for each GEZ. The environmental space is shown by 2000 random points within the current forest cover that occurs within each GEZ. Primary forest sites distinguished as research sites, literature sites and forest inventory sites.
Comparison with global modelled and default biomass
The spatial distribution of global modelled aboveground biomass (Supplementary Fig. S2) and soil carbon (Supplementary Fig. S3) show high carbon stock densities in some of the forest areas mapped as remaining primary forest28 (Supplementary Fig. S4), such as Carpathian Mountains in eastern Europe, Dinaric Mountains in the Balkans, and Pyrenees in north-eastern Spain. High soil carbon contents are related more to cooler temperatures and high elevation, particularly where peat or transitions occur.
Frequency distributions of biomass carbon stock density at the sites were consistently higher than the modelled data from the global biomass maps across all forest types, with the greatest difference for mixed species, then broadleaf, and least for conifers (Fig. 5).
Distribution of carbon densities compared for the site data and global modelled data from GlobBiomass97 and GeoCarbon98 for the primary forest site locations, for a all forest types, b conifer species, c broadleaf species, d mixed species. The dataset for all forest types included sites of unidentified forest types and thus was larger than the combined broadleaf plus conifer plus mixed datasets. Results for paired two sample t-test one-tail for the site level data are presented for each forest category to show the statistical differences. All forests: Site data and GlobBiomass t = 59.47, P < 0.0001, n = 5942. All forests: Site data and GeoCarbon t = 53.16, P < 0.0001, n = 3778. Conifer: Site data and GlobBiomass t = 20.70, P < 0.0001, n = 720. Conifer: Site data and GeoCarbon t = 19.80, P < 0.0001, n = 597. Broadleaf: Site data and GlobBiomass t = 29.55, P < 0.0001, n = 1176. Broadleaf: Site data and GeoCarbon t = 38.21, P < 0.0001, n = 774. Mixed forest: Site data and GlobBiomass t = 24.24, P < 0.0001, n = 182. Mixed forest: Site data and GeoCarbon t = 33.61, P < 0.0001, n = 178.
The cumulative biomass from the global models was lower than the site data by proportions of 0.57 for GeoCarbon and 0.59 for GlobBiomass for all forest types, 0.64 and 0.65 for conifer species, 0.53 and 0.61 for broadleaf species, and 0.28 and 0.46 for mixed species, respectively (Supplementary Table S5). The cumulative biomass from the different data sources showed the greatest difference for research and literature sites, and least for inventory sites (Supplementary Fig. S6). The difference was least in Sweden and greatest in the carbon-dense forests, such as Montenegro, Bosnia-Herzegovina, Romania, Albania and Bulgaria (Supplementary Fig. S7).
Biomass carbon stocks at primary forest sites classified by GEZ compared with global modelled biomass (GlobBiomass) within the mapped primary forest area of the GEZ28 were similar for boreal forests, 2-times higher for temperate, and 2.5–3-times higher for subtropical forests (Fig. 6, Table 1). Comparison for each site location showed the field data were 3-times higher on average than the global modelled biomass.
Coloured bars are mean value from the primary forest site data within each GEZ and lines are standard errors. Primary forest sites distinguished as research sites, literature sites and forest inventory sites. Default values include the IPCC within GEZs29 (primary forest values for Europe were used for the temperate GEZs, no data were available for boreal mountain, data from the Americas was used for boreal coniferous, data for Asia for subtropical mountain, and average data for all age categories from Asian, Americas and African for subtropical dry), the modelled GlobBiomass within GEZs97 and the mapped area of primary forest28, and the Forests Europe average for all forest types81,99.
Field measurements of biomass at sites showed higher carbon stocks than IPCC default values within GEZs29 (Fig. 6), with more than twice the stock in temperate oceanic forest, but similar or slightly lower in temperate continental forest. The database presented here consists of many more site measurements than were used in the IPCC guidelines, which have uncertainty levels of 90% and many data gaps (references cited for GEZs in Europe include eight research studies, one inventory study and three remote sensing studies, with only two studies in primary forests). The 2019 updated default values are an improvement to the 61 MgC ha−1 used in the original 2006 guidelines30 and now recognise that carbon stocks are higher and the importance of differentiating GEZs and primary and secondary forests.
Tree size distribution
Distributions of biomass carbon stock density and tree density in relation to tree diameter size class show the contributions of each size class to the total carbon stock of the forest (Fig. 7, Supplementary Fig. S8, Supplementary Table S6). The highest density of tree numbers occurred in the smaller diameter classes with a long positive skew. In contrast, carbon stock density had a normal to positive skew distribution, with a small number of large trees having high stocks. Cumulative biomass showed the proportion of the tree size range that contributed to the total biomass carbon stock. Across all primary forest sites sampled, 50% of total carbon stock in living biomass was contained in trees with DBH ≥ 60 cm (median value), but this represented only 15% of the number of trees. (Tree density depended on the minimum diameter measured, commonly 5 cm, but varied among countries from 1 to 10 cm) (Supplementary Table S2). The proportion of total biomass in the primary forest contributed by large trees varied depending on forest type (broadleaf, conifer and mixed forest), and DBH threshold, with ranges in biomass carbon stocks from 23 to 43% at the high tree DBH threshold to 68–75% at the low threshold (Supplementary Figs. S8 and S9).
The light green area represents the distribution of tree density, the dark green histogram represents the distribution of total live biomass carbon stock, the red curve represents the cumulative biomass over the range in tree sizes, the blue dashed line represents the proportion of total biomass carbon stock contributed by large trees (DBH ≥ 60 cm). Vertical bars are standard errors.
Total carbon stocks and potential stock gain
The current total forest area in Europe is 174 Mha and 1.07% is classified and mapped as primary forest28 (Supplementary Fig. S4), with 78% in the boreal region. The aboveground biomass carbon stock in the total forest area is estimated to be 9861 TgC (based on current data and assumptions), and the contribution of primary forests is 1.02%. The total carbon stock in the area mapped as primary forest28 was estimated from the collated primary forest site data and this represents the CCC (see Methods: Spatial estimation of carbon stocks and stock changes and Supplementary Information S1.8). This updated carbon stock was 1.6-times more than that currently estimated from global modelled biomass within the mapped primary forest area, with temperate and subtropical forests double or more, and boreal forests similar stocks (Table 2(1), Supplementary Table S9, Fig. 6). However, the boreal and subtropical dry (Mediterranean) biomes have low numbers of sites.
The potential gain in carbon stock in secondary forests by ongoing growth was calculated as the increasing carbon stock over time from CCS towards its CCC. The estimated CCC in these secondary forest areas was 2.3-times the current stock with an additional 12,659 MtC, indicating the potential for high carbon sequestration through restoration (Table 2(2), Supplementary Table S9).
The area of mapped primary forest28 is likely on overestimate in some countries, such as Sweden, Finland and the Iberian Peninsula, based on observations of researchers (S1.7). Such overestimates in area suggest that the high carbon stock densities from the site data assigned to areas of primary forest would result in a higher total carbon stock for primary forest. Conversely, the effect of mapping some primary forest that should have been assigned to secondary forest, is a larger area for potential gain in carbon stock. This would amplify the result of mitigation potential in Table 2(2). Additionally, if the area of primary forest is even smaller than that mapped, this amplifies the significance of their rarity, and hence, importance of their protection.
Discussion
Carbon stocks in primary forests have been underestimated
The larger carbon stocks identified at primary forest sites compared to estimates based on modelled or default values is consistent with that found in individual forest types in Europe31. Our updated estimate of CCC means the mitigation benefit of primary forests by protecting their accumulated carbon stocks and avoiding their loss has been undervalued in climate and forest management policies.
The main reason for the discrepancy in estimates of aboveground biomass is that the global models based on remotely sensed data have high uncertainties at high biomass densities (>125 MgC ha−1) where the retrieval was effectively based on a single radar observation32. Comparison of the biomass map with independent reference data concluded from the validation process that biomass was underestimated at ≥75 MgC ha−1 in the temperate forest biome33. The models use plot data to calibrate the satellite-based estimates of extant forest cover and thus estimate CCS. However, our results from the site data show that even in the locations where primary forests exist and their structural characteristics should be detected by remote sensing, those calibrations do not accurately reflect observed carbon stocks (Fig. 5). The modelled data lacks differentiation between forest types (conifer, broadleaf, mixed) suggesting that the models are not able to account adequately for the different biomass densities of these forest types. Consequently, underestimation of biomass occurs particularly for broadleaf species.
Biomass estimates using plot data also have limitations, particularly in fragmented landscapes. First, the area sampled is a very small proportion of the area of primary forest and total forest cover. Second, the representativeness of plot data for ecosystem types at the landscape scale is uncertain as such small proportions of natural forest ecosystems remain. Third, the distribution of sites is spatially biased, and some forest types are poorly represented, such as subtropical dry and mountain systems, coastal boreal and coastal temperate oceanic systems. Therefore, the full distribution of forest types and their carbon stocks are not evident. Fourth, patch heterogeneity is a characteristic of old-growth forest structures but measuring the full extent of spatial structural complexity of forests, including canopy gaps and variations in stand density related to fine-scale natural disturbance events, is challenging with plot-based sampling, as seen by the high variance around mean plot values for a forest type. However, carefully randomised and replicated plot locations have been shown to record robust estimates of biomass34,35,36.
Three additional factors are likely to amplify the underestimation of biomass from global models. First, carbon stock densities estimated from site data may be lower than the forest type within GEZ average because the patches of primary forest remaining often occur lower on the productivity gradient because they are less accessible or desirable for human intervention37 (Fig. 4). However, there may be cases of sites preferentially sampled to select large growth forms and areas of productive forests selectively preserved for private use historically38,39. Particularly in Europe with the long history of land use (S1.1), the available forest areas with least disturbance likely do not fully represent the CCC of the forest type, which means estimates of the potential carbon storage may be conservative. Second, total carbon stock includes not only aboveground living biomass but also belowground biomass and dead biomass of standing dead trees, coarse woody debris, litter and soil. These components are higher in primary forests with old trees and complex structures40. The importance of including dead biomass and belowground biomass is demonstrated, adding on average 16% and 22% to the aboveground living biomass, respectively (averages from Fig. 2). Improved data are needed for soil carbon. Third, most plot-based forest biomass data are derived from allometric equations. Sampling of trees to derive these equations often underrepresents large trees because destructive measurement of total biomass is difficult and often done in production forests that do not contain large trees. Underestimation has been demonstrated in the biomass of forest plots derived from allometric equations compared with detailed three-dimensional laser measurements across the full range of tree sizes41, and this increased with tree size42. Extrapolation of biomass calculated for trees larger than the calibration data increases uncertainty in biomass estimates, thus particularly affecting results for primary forests.
The large carbon stocks in primary forests are due to the presence of large old trees, where tree size is relative depending on forest type and environmental conditions. These large trees are rarely present in forests managed for commercial production by clear cutting, and reduced under selective cutting, as the trees only attain harvest maturity and not the large sizes of old trees, as well as their associated dead biomass43,44. Within the production cycle, the remaining old forests (for example, >100 years old with a high density of moderately large trees) may attain similar biomass density to that of long-unmanaged forests31, but this represents a small proportion of the total production forest area. Our results provide a comprehensive assessment of the contribution to carbon stocks by large tree-size classes for primary forests in all biomes across Europe24,45,46,47, (Supplementary Table S8).
While modelled and default values of carbon stocks have been refined over the last decade, our synthesis of site data for primary forests within GEZs across Europe (Table 1, Supplementary Table S9) further improves the input data for deriving biomass carbon stocks and contributes to the next update of IPCC default values.
Mitigation potential from protecting and restoring primary forests
Protecting and restoring primary forests contribute to climate mitigation by (i) retaining an accumulated stock of carbon in living and dead biomass and soil, (ii) maintaining the natural terrestrial carbon sink to buffer some of the impact of elevated atmospheric CO2 concentration, and (iii) removing CO2 from the atmosphere through restoration of secondary and degraded forests43.
(i) The priority for mitigation action is retaining carbon stored in forests and avoiding emissions. Thus, the mitigation benefit of primary forests is the magnitude and longevity of their accumulated ecosystem carbon stocks. The large amounts of carbon stored in primary forests, as revealed in this study, represent long-lived, stable and resilient stocks in ecosystems with a high level of integrity22. Primary forest ecosystems have lower risk of emissions to the atmosphere caused by either human activities or increases in extent, frequency, and severity of disturbance events48, compared with forests that have lower levels of ecosystem integrity, such as secondary forests or plantations. Area and biomass harvested annually, and use of wood resources, have been increasing since 2016, thus reducing carbon stocks, although rates vary between different data sources49,50,51,52,53. A documented increase in canopy mortality of 2.4% over the last three decades was attributed to intensified tree harvesting as the prime agent and correlated with reported wood removal statistics54. Disturbance regimes under the influence of climate change have been intensifying, for example windstorms, fires, insect outbreaks, and are likely to impact carbon storage55, although much of the effect is transference from living to dead biomass56. The biodiversity, structural complexity and ecological and evolutionary processes operating in primary forest ecosystems confer greater stability, resilience and adaptive capacity that support accumulation and maintenance of carbon stocks57,58,59,60. Hence, primary forests provide the benefit of avoided damages.
(ii) Global forests act as sinks with positive net ecosystem productivity as trees continue to grow, accumulate biomass, produce dead biomass, and store soil carbon61,62,63. In general, secondary forests have higher rates of biomass productivity because their management aims to increase wood production by maintaining trees at younger ages, often increasing plant-available resources, sometimes selecting more efficient genetics, and locating on more productive sites61,64. However, a continuing sink has been demonstrated in older forests through many studies, both at the stand scale and from tree ring analysis12,40,65,66,67,68,69,70,71, and rates of net primary productivity can be similar in primary and production secondary forests63. The complex heterogeneity of older forests maintained by natural disturbance regimes contribute to their high rates of productivity by creating structural diversity of tree sizes, ages, spatial distribution, vertical canopy structure, leaf area index and accumulation in the soil carbon pool72,73. The carbon sink in forests globally absorbs approximately 18% of annual anthropogenic CO2 emissions (2001–2019)74 and unharvested mature forests account for about half of this sink75.
(iii) Ecological restoration of secondary and degraded forests provides a pathway for removals of atmospheric carbon, through gains in carbon stocks with growth toward the CCC and reconnecting fragmented landscapes4,59,76,77. The ecologically-based reference level of CCC that is characterised by primary forests provides information about the composition, structure, ecological processes and natural disturbance regimes of these ecosystems that can help guide restoration activities and the selection of appropriate indicators for assessing progress.
The potential carbon stock gain by allowing trees to continue growing beyond currently applied harvest maturity was estimated from the analysis of tree size distribution (Supplementary Fig. S8). The carbon stock foregone by harvesting at a given tree size was simulated by restricting trees at or below the harvest diameter threshold but maintaining the same tree density. The foregone stock varied from 12 to 21% at the high tree diameter to 46-52% at the low tree diameter (Supplementary Table S7). The biomass carbon stock of forests managed on a harvesting rotation would be lower than the estimate here when the full age distribution of stands since regeneration are included at the landscape scale. Hence, this estimate of foregone carbon stock is conservative.
This potential for carbon stock gain has been demonstrated in other studies with forests naturally developing following protection from human intervention78,79. Historical change in forests over the last 250 years in Europe shows 41.7 Mha being converted to production management involving wood extraction and tree species conversion. This has resulted in a 38% average reduction in biomass carbon storage80, observed under a range of conditions63,81,82, (Supplementary Table S9). The ensuing wood products constitute a small pool of 10% or less of the carbon stored in aboveground living forest biomass83,84,85, and have a shorter lifetime than that of trees30. However, increasing the proportion of long-lived wood products and their longevity, reducing waste on-site and during processing, and substituting wood for products with large carbon footprints due to their processing such as concrete, would contribute to increasing carbon storage compared to current forest management.
We estimated the potential for carbon stock gain by regrowing secondary forests to be more than double the current stock, with an additional 12,659 MtC (46,415 MtCO2) (Table 2(2)). Similar results have been reported, including modelling of European forests using estimated maximum carbon stock in broadleaf forests of 170 tC ha−1 (compared to our site measurements of 187 tC ha−1) and in conifer forests of 130 tC ha−1 (current study 145 tC ha−1)86, and estimated 42–47% carbon stock loss due to degradation in the land sector25. Based on historical and ecological sources, carbon storage of temperate deciduous/ mixed forests was estimated at about half of the potential storage87. Further work is required to refine these estimates, for example using site matching between secondary and primary forests, improved representativeness of sites within forest types, and finer spatial scale of aggregation for the comparison. The actual gains in carbon stock in the future will, however, depend on the impacts of climate change on forest productivity and the relative rates of carbon fluxes through the vegetation and soil that determine carbon accumulation, its stability and resilience.
The potential mitigation benefits from protecting and restoring forests can be considered relative to the overall European target for net annual removals in the LULUCF sector under the Fit for 55 package in the European Green Deal of 310 MtCO2_e by 203014 and the simulated trajectory for forest land of 410 MtCO2_e removals required to reach this target88. Our estimated gain in carbon stock by regrowing secondary forests (Table 2(2)) equates to an annual rate of removals of 309 MtCO2_e; assuming the CCC would be achieved after 150 years with a linear rate of accumulation (or 464 MtCO2_e after 100 years). (Carbon accumulation is likely to have a logarithmic form, in which case annual removal rates would be higher initially and then slow but reaching the same stock). These estimated removals are additional to, and higher than, the current forest sink in the EU (2021: 289 MtCO2_e89. Thus, restoration of the existing forest area through carbon accumulation by ongoing growth could achieve the net removals target in the land sector and fill the carbon debt created by historical forest management. However, all emissions reduction activities should be pursued and the requirement to maintain wood provisioning is recognised. Our results demonstrate the considerable opportunities for increasing carbon storage in the existing forest area, for example through restoration of 30% of forest area (EU Biodiversity Strategy target of 30% protection90, protecting biodiversity habitats, optimising wood production, retaining large trees, restoring coppices, and lengthening rotation periods, as well as opportunities for increasing the forest area through reforestation and regeneration (cognisant of requirements for other land uses, natural ecosystems and ecosystem services).
Conclusions
Retaining and increasing carbon stocks in forests and avoiding emissions should be recognised as priorities for global environmental policy progressing towards more ambitious climate and biodiversity targets for the European Green Deal. Achieving these targets requires conservation management and governance to ensure formal and enforced protection that prevents degradation by logging and mining, encroachment by roads and other infrastructure developments. In Europe, only just over half of the identified primary forests are strictly protected81,91 and degradation continues in some places even within designated protected areas37. Under the EU Biodiversity Strategy for 2030, the aim is to strictly protect all remaining primary and old-growth forests, restore and manage them and their buffer zones for biodiversity conservation92. This protection is a mitigation activity that can be implemented rapidly and contribute to many co-benefits for biodiversity conservation and provision of ecosystem services, cognisant of their complementarities and interdependencies17,93. Europe has few areas of forest with minimal human disturbance and so protecting these remaining areas should be a priority for climate and forest policy. Restoration of forest ecosystems by allowing continued growth of regenerating forests, active restoration measures, and re-connecting fragmented remnants across landscapes, will provide crucial mitigation benefits that contribute to emissions reduction targets as well as existing and future co-benefits.
Methods
Identification of primary forest sites
The status of primary forests in Europe is described (S1.1) and the characteristics used for their identification detailed (S1.2, Supplementary Table S1). As much data as possible was collated from existing studies in primary forest areas in boreal, temperate and subtropical (Mediterranean) zones within Europe. Site data were collected from three sources: (i) research sites were from existing studies (the network used for mapping primary forest in Europe by Sabatini et al.28), and the primary forest status was determined from observed characteristics (Supplementary Table S2); (ii) site data from the literature where characteristics of the forest are described (S1.4, Supplementary Data 1); and (iii) inventory data were obtained from NFIs where the data custodians filtered the database to extract sites that were described as primary forest, or as natural forest under some protection status, and stands older than 100 years (Supplementary Table S2, Fig. 1). The inventory sites do not necessarily comprise all the characteristics of primary forests but provide an estimate of maximum carbon stocks. Site selection was based on observation of forest characteristics with the assumption that there was no systematic selection for site conditions. However, this synthesis of sites is currently the most comprehensive compilation of primary forest site data. The database of primary forest sites collated consists of 1818 research sites with 242,461 trees across 16 countries; 6015 inventory sites with 45,801 trees across 3 countries; and 149 literature sites across 17 countries.
The locations for these sites are not necessarily representative of all remaining primary forests or forest types and the distribution of sites across Europe shows a spatially biased sample (Fig. 1). Patches of primary forest remaining are often located on extreme sites, for example steep slopes, shallow soils and high elevation, that are likely to be at the lower end of the productivity gradient for the forest type. To test the representativeness of the sites, they were located within the environmental space for current forest cover within each GEZ defined by elevation, mean annual temperature and a water availability index (S1.5).
Sites were classified by forest type - broadleaf, conifer, mixed forest – (according to the European classification from Corine Land Cover 201894), and Global Ecological Zones (GEZ27) (Supplementary Fig. S1) for the purpose of aggregation and reporting of results and for comparability with that used by IPCC.
Carbon stock components
The basic inventory measurements at all sites included the DBH of all trees within a defined plot area, tree height (in some cases), species or forest type, living and dead trees, and dimensions of coarse woody debris. Individual tree biomass was calculated for above- and below-ground biomass of living and dead standing trees, and biomass of coarse woody debris (or lying dead wood).
Tree dimensions were converted to biomass using regional and species-specific allometric equations where available. Wood volume of stems and coarse woody debris was converted to biomass using species-specific wood density and carbon concentration, and wood decay classes where available. Methods used for each dataset are described in Supplementary Table S1. Additional data for dead wood biomass in various forest types in Europe was sourced from the literature (Supplementary Table S3). Data for soil organic carbon was sourced from global modelled data (GSOC95, S1.7).
Tree size distribution
The relationship between tree size and biomass carbon stock for individual trees was analysed where individual tree data were available for the sites. The distribution of biomass carbon stock in the primary forests was differentiated according to the stock contributed by trees larger than the harvesting diameter threshold, that is the large trees that would not occur in a forest managed on a harvesting rotation. The size of trees at harvest varies depending on many factors, including species, environmental growth conditions, silvicultural systems, industry regulations or protocols, products and their markets. Harvesting may be determined by age or size of the trees and ranges are reported (Supplementary Table S4). As a conservative estimate of the tree size at harvest, we selected a minimum and maximum for each forest category: 50–80 cm for broadleaf, 40–60 cm for conifers, and 50–70 cm for mixed forest. These tree sizes relate to single-stemmed trees or high forest and the harvesting of coppice forests at smaller sizes has not been analysed here, although a common silvicultural practice in Mediterranean forests96.
The biomass contributed by the large trees is reported as a percentage of the total biomass in the primary forest. The carbon stock that is foregone by harvesting was estimated as the difference in biomass between the primary forest and the same number of trees but limited to the diameter threshold at harvest. In the harvesting scenario, the biomass of all trees per hectare were included but the maximum size was set at the threshold DBH representing harvest maturity. Calculated biomass included all trees below the diameter threshold plus the biomass of the equivalent number of trees above the threshold assuming their size was at the threshold, that is, at harvest maturity or rotation forest age. The foregone stock was represented as the percent of the biomass carbon stock for a range of tree sizes at harvest maturity.
Spatial estimation of carbon stocks and stock change
Global spatial data were used for forest extent, global modelled biomass (GlobBiomass97 and GeoCarbon98), and soil carbon (GSOC95) (S1.7). Carbon stock densities were extracted from the global maps for each site location, and for the spatial extent within the forest areas of each GEZ, to allow comparison of the modelled and measured data. The extant forest cover in Europe was differentiated into the mapped area of primary forest28 (Supplementary Fig. S4), and the remainder that was described as secondary forest, and thus had the potential to continue growing and accumulating carbon stocks.
First, the difference in carbon stock density (MgC ha−1) was estimated for primary forests between the site data and modelled values at the site locations and within the area of mapped primary forest28 for each GEZ. Applying the ratio of this difference to the area of primary forest provided an updated estimate of the total carbon stock as the CCC of the extant primary forest (MgC). Second, the potential for carbon stocks to change in relation to natural ecological processes or human intervention was estimated from the difference between the current carbon stock (CCS) and the carbon carrying capacity (CCC). CCS of the secondary forest area within each GEZ was represented by the global modelled biomass. CCC was represented by the average carbon stock density (MgC ha−1) measured at primary forest sites within each GEZ and used to calculate the ratio of site data to global spatial data. This ratio was applied to convert the global spatial biomass in secondary forest for each GEZ to the equivalent of CCC. Results reported in Table 2 are for the sum of GEZs within each biome (boreal, temperate, subtropical). The workflow is described in S1.8 and details of the data in S2.3. The estimated potential carbon stock gain is only for the existing forest area and does not include the area that has been cleared of forest, that is, the potential natural forest vegetation.
Data availability
Data are available in the Supplementary Information, a Supplementary Data file and online at https://doi.org/10.6084/m9.figshare.25632561 with summaries for all datasets and the data for results presented. Data for individual research sites would need to be requested from country data custodians.
References
UNFCCC Paris Agreement. https://unfccc.int/sites/default/files/english_paris_agreement.pdf (2015).
UNFCCC Glasgow Climate Pact. https://unfccc.int/sites/default/files/resource/cop26_auv_2f_cover_decision.pdf (2021).
IPCC Global Warming of 1.5 °C: IPCC Special Report on Impacts of Global Warming of 1.5 °C above Pre-industrial Levels in Context of Strengthening Response to Climate Change, Sustainable Development, and Efforts to Eradicate Poverty. Cambridge University Press. https://doi.org/10.1017/9781009157940 (2022).
Mackey, B. G., et al. Untangling the confusion around land carbon science and climate change mitigation policy. Nat. Clim. Change 3, https://doi.org/10.1038/NCLIMATE1804 (2013).
Hudiburg, T. W. et al. Meeting GHG reduction targets requires accounting for all forest sector emissions. Environ. Res. Lett. 14, 095005 (2019).
Carton, W., Lund, J. F. & Dooley, K. Undoing equivalence: rethinking carbon accounting for just carbon removal. Front. Clim. 3, 664130 (2021).
Keith, H. et al. Evaluating nature-based solutions for climate mitigation and conservation requires comprehensive carbon accounting. Sci. Total Environ. 769, 144341 (2021).
Peng, L., Searchinger, T. D., Zionts, J. & Waite, R. The carbon costs of global wood harvests. Nature https://doi.org/10.1038/s41586-023-06187-1 (2023).
FAO Global Forest Resources Assessment 2020. Main Report. Rome. https://doi.org/10.4060/ca9825en (2020a).
Pan, Y. et al. A large and persistent carbon sink in the world’s forests. Science 333, 988–993 (2011).
Garbarino, M. & Weisberg, P. J. Land-use legacies and forest change. Landsc. Ecol. 35, 2641–2644 (2020).
Martin-Benito, D., Pederson, N., Férriz, M. & Gea-Izquierdo, G. Old forests and old carbon: a case study on the stand dynamics and longevity of aboveground carbon. Sci. Total Environ. 765, 142737 (2021).
Grassi, G. et al. The key role of forests in meeting climate targets requires science for credible mitigation. Nat. Clim. Change 7, 220–226 (2017).
EU Regulation of the European Parliament and of the Council amending Regulation (EU) 2018/841 as regards the scope, simplifying the compliance rules and setting out the targets of the Member States for 2030, and (EU) 2018/1999 as regards improvement in monitoring, reporting, tracking of progress and review. https://data.consilium.europa.eu/doc/document/ST-10774-2022-INIT/x/pdf (2022).
Grassi, G., Pilli, R., House, J., Federici, S. & Kurz, W. A. Science-based approach for credible accounting of mitigation in managed forests. Carbon Balance Manag. 13, 8 (2018).
Keith, H., Mackey, B. G., Berry, S., Lindenmayer, D. B. & Gibbons, P. Estimating carbon carrying capacity in natural forest ecosystems across heterogeneous landscapes: addressing sources of error. Global Change Biol. 16, 2971–2989 (2010).
Mikoláš, M., et al. Natural disturbance impacts on trade-offs and co-benefits of forest biodiversity and carbon. Proc. R. Soc. B 288, 20211631 https://doi.org/10.1098/rspb.2021.1631 (2021).
FAO. Global Forest Resource Assessment 2020. Terms and definitions (FAO, 2020b).
Buchwald, E. A. hierarchical terminology for more or less natural forests in relation to sustainable management and biodiversity conservation. In Proceedings: Third expert meeting on harmonizing forest-related definitions for use by various stakeholders (Food and Agriculture Organization of the United Nations, 2005).
Sabatini, F. M. et al. Where are Europe’s last primary forests? Divers. Distrib. 2018, 1–14 (2017).
Vandekerkhove, K., et al. Old-growth criteria and indicators for beech forests (Fageta). LIFE_PROGNOSES Work Package 1.11. https://purews.inbo.be/ws/portalfiles/portal/74600061/Criteria_oldgrowth_PROGNOSES_Finalversion.pdf (2021).
Rogers, B. M., et al. Using ecosystem integrity to maximize climate mitigation and minimize risk in international forest policy. Front. Forests Global Change https://doi.org/10.3389/ffgc.2022.929281 (2022).
Stegen, J. C. et al. Variation in aboveground forest biomass across broad climatic gradients. Global Ecol. Biogeogr. 20, 744–754 (2011).
Bastin, J.-F. et al. Pan-tropical prediction of forest structure from the largest trees. Global Ecol. Biogeogr. 27, 1366–1383 (2018).
Erb, K.-H. et al. Unexpectedly large impact of forest management and grazing on global vegetation biomass. Nature 553, 73–76 (2018).
Lutz, J. A. et al. Global importance of large-diameter trees. Global Ecol. Biogeogr. 8, 1–16 (2018).
FAO. Global Ecological Zones for FAO forest reporting: 2010 update. Forest Resource Assessment Working Paper 179, Rome (FAO, 2012).
Sabatini, F. M. et al. European primary forest database v2.0. Sci. Data 8, 220 (2021).
IPCC Refinement to the 2006 IPCC guidelines for national greenhouse gas inventories. https://www.ipcc.ch/report/2019-refinement-to-the-2006-ipcc-guidelines-for-national-greenhouse-gas-inventories/ (2019).
Eggleston, H. S., et al. (eds) IPCC Guidelines for National Greenhouse Gas Inventories, prepared by the National Greenhouse Gas Inventories Programme (IGES, Japan, 2006).
Nord-Larsen, T., Vesterdal, L., Bentsen, N. S. & Larsen, J. B. Ecosystem carbon stocks and their temporal resilience in a semi-natural beech-dominated forest. Forest Ecol. Manag. 447, 67–76 (2019).
Santoro, M. et al. The global forest above-ground biomass pool for 2010 estimated from high-resolution satellite observations. Earth Syst. Sci. Data 13, 3927–3950 (2021).
Rozendaal, D. M. A., Santoro, M., Schepaschenko, D., Avitabile, V., Herold, M. GlobBiomass Validation Report D17 v06. Prepared for European Space Agency (ESA-ESRIN). Friedrich Schiller University of Jena, Department of Earth Observation, Germany. https://www.dropbox.com/s/hplshh3ipm7hrak/GlobBiomass_D_17_Validation_Report_V06.pdf?dl=0 (2017).
Král, K. et al. Local variability of stand structural features in beech dominated natural forests on Central Europe: implications for sampling. For. Ecol. Manage 260, 2196–2203 (2010).
Král, K., Valtera, M., Janík, D., Šamonil, P. & Vrška, T. Spatial variability of general stand characteristics in central European beech-dominated natural stands – effects of scale. For. Ecol. Manag. 328, 353–364 (2014).
Martin-Benito, D., Molina-Valero, J. A., Pérez-Cruzado, C., Bigler, C. & Bugmann, H. Development and long-term dynamics of old-growth beech-fir forests in the Pyrenees: Evidence from dendroecology and dynamic vegetation modelling. For. Ecol. Manag. 524, 120541 (2022).
Mikoláš, M. et al. Primary forest distribution and representation in a Central European landscape: Results of a large-scale field-based census. For. Ecol. Manag. 449, 117466 (2019).
Holeksa, J. et al. A giant tree stand in the West Carpathians – an exception or a relic of formerly widespread mountain European forests? For. Ecol. Manag. 257, 1577–1585 (2009).
Matuszkiewicz, J. M. et al. Current and potential carbon stock in the forest communities of the Bialowieza Biosphere Reserve. Forest Ecol. Manag. 502, 119702 (2021).
Schulze, E.-D., et al. Temperate and Boreal Old-Growth Forests: How do Their Growth Dynamics and Biodiversity Differ from Young Stands and Managed Forests? In Old-Growth Forests: Function, Fate and Value, (eds, Wirth, C., Gleixner, G., Heimann, M.) 343–366, Springer Berlin Heidelberg. https://doi.org/10.1007/978-3-540-92706-8_1 (2009).
Calders, K. et al. Laser scanning reveals potential underestimation of biomass carbon in temperate forest. Ecol. Solutions Evid. 3, e12197 (2021).
Calders, K. et al. Nondestructive estimates of above-ground biomass using terrestrial laser scanning. Methods Ecol. Evol. 6, 198–208 (2015).
Keith, H., et al. The Land Gap Report. https://www.landgap.org/ (2022).
Ķēniņa, L., Elferts, D., Jaunslaviete, I., Bāders, E., Jansons, Ā. Sustaining carbon storage: lessons from hemiboreal old-growth coniferous and deciduous forest stands. Forest Science 1–9, https://doi.org/10.1093/forsci/fxac055 (2022a).
Bastin, J.-F. et al. Seeing central African forests through their largest trees. Scientific Reports 5, 13156 (2015).
Slik, J. W. F. et al. Large trees drive forest aboveground biomass variation in moist lowland forests across the tropics. Glob. Ecol. Biogeogr. 22, 1261–1271 (2013).
Ķēniņa, L. et al. Tree biomass – a fragile carbon storage in old-growth birch and aspen stands in hemiboreal Latvia. Baltic Forestry 28, 654 (2022b).
Senf, C. & Seidl, R. Mapping the forest disturbance regimes of Europe. Nat. Sustain. 4, 63–70 (2020).
Ceccherini, G. et al. Abrupt increase in harvested forest area over Europe after 2015. Nature 583, 72–77 (2020).
Ceccherini, G. et al. Reply to Wernick IK et al.; Palahi M et al. 2021. Nature 592, E18–E23 (2021).
Palahi, M. et al. Concerns about reported harvests in European forests. Nature 592, E15–E17 (2021).
Wernick, I. K. et al. Quantifying forest change in the European Union. Nature 592, E13–E14 (2021).
Camia, A. et al. The use of woody biomass for energy production in the EU and impacts on forests. JRC Science for Policy Report EUR 30548 EN (Publications Office of the European Union, 2020). https://doi.org/10.2760/831621 (2020).
Senf, C. et al. Canopy mortality has doubled in Europe’s temperate forests over the last three decades. Nat. Commun. 9, 4978 (2018).
Seidl, R., Schelhaas, M. J., Rammer, W. & Verkerk, P. J. Increasing forest disturbances in Europe and their impact on carbon storage. Nat. Clim. Change 4, 806–810 (2014).
Keith, H. et al. Accounting for biomass carbon stock change due to wildfire in temperate forest landscapes in Australia. Public Library Sci. PLoS One 9, e10712 (2014).
Thompson, I., Mackey, B., McNulty, S., Mosseler, A. Forest Resilience, Biodiversity, and Climate Change. A synthesis of the biodiversity/resilience/stability relationship in forest ecosystems. Secretariat of the Convention on Biological Diversity, Montreal. Technical Series no. 43. p. 67. https://doi.org/10.1111/ele.12054 (2009).
Alberto, F. et al. Potential for evolutionary responses to climate change – evidence from tree populations. Global Change Biol. 19, 1645–1661 (2013).
Barber, C. V., Petersen, R., Young, V., Mackey, B., Kormos, C. The Nexus Report: Nature Based Solutions to the Biodiversity and Climate Crisis. F20 Foundations, Campaign for Nature and SEE Foundation. https://foundations-20.org/publication/the-nexus-report-nature-based-solutions-to-the-biodiversity-and-climate-crisis/ (2020).
Di Filippo, A., Biondi, F., Piovesan, G. & Ziaco, E. Tree ring‐based metrics for assessing old‐growth forest naturalness. J. Appl. Ecol. 54, 737–749 (2017).
Carey, E. V., Sala, A., Keane, R. & Callaway, R. M. Are old forests underestimated as global carbon sinks. Glob. Chang. Biol. 7, 339–344 (2001).
Zhou, G. et al. Old-Growth Forests Can Accumulate Carbon in Soils. Science 314, 1417–1417 (2006).
Glatthorn, J., Feldmann, E., Pichler, V., Hauck, M. & Leuschner, C. Biomass Stock and Productivity of Primeval and Production Beech Forests: Greater Canopy Structural Diversity Promotes Productivity. Ecosystems 21, 704–722 (2018).
Campioli, M. et al. Biomass production efficiency controlled by management in temperate and boreal ecosystems. Nat. Geosci. 8, 843–846 (2015).
Knohl, A., Schulze, E.-D., Kolle, O. & Buchmann, N. Large carbon uptake by an unmanaged 250-year-old deciduous forest in Central Germany. Agric. For. Meteorol. 118, 151–167 (2003).
Gundersen, P. et al. Old-growth forest carbon sinks overestimated. Nature 591, E21–E23 (2021).
Luyssaert, S. et al. Reply to: Old-growth forest carbon sinks overestimated. Nature 591, E24–E25 (2021).
Stephenson, N. L. et al. Rate of tree carbon accumulation increases continuously with tree size. Nature 507, 90–93 (2014).
Gough, C. M., Curtis, P. S., Hardiman, B. S., Scheuermann, C. M. & Bond-Lamberty, B. Disturbance, complexity, and succession of net ecosystem production in North America’s temperate deciduous forests. Ecosphere 7, e01375 (2016).
Vandekerkhove, K. et al. Very large trees in a lowland old-growth beech (Fagus sylvatica L.) forest: density, size, growth and spatial patterns in comparison to reference sites in Europe. Forest Ecol. Manag. 417, 1–17 (2018).
Birdsey, R. A., et al. Assessing carbon stocks and accumulation potential of mature forests and larger trees in U.S. federal lands. Frontiers in Forests and Global Change. https://doi.org/10.3389/ffgc.2022.1074508 (2023).
Achat, D. L., Fortin, M., Landmann, G., Ringeval, B. & Augusto, L. Forest soil carbon is threatened by intensive biomass harvesting. Sci. Rep. 5, 15991 (2015).
Mayer, M. et al. Influence of forest management activities on soil organic carbon stocks: a knowledge synthesis. For. Ecol. Manage. 466, 118127 (2020).
Harris, N. et al. Global maps of twenty-first century forest carbon fluxes. Nat. Clim. Change 11, 234–240 (2021).
Bellassen, V. & Luyssaert, S. Managing forests in uncertain times. Nature 506, 153–155 (2014).
Houghton, R. A., Byers, B. & Nassikas, A. A. A role for tropical forests in stabilizing atmospheric CO2. Nat. Clim. Change 5, 1022–1023, (2015).
Keith, H., Lindenmayer, D., Macintosh, A. & Mackey, B. Under What Circumstances Do Wood Products from Native Forests Benefit Climate Change Mitigation? Plos One 10, e0139640 (2015).
Meyer, P., Nagel, R. & Feldmann, E. Limited sink but large storage: Biomass dynamics in naturally developing beech (Fagus sylvatica) and oak (Quercus robur, Quercus petraea) forests of north-western Germany. J. Ecol. 2021, 1–15 (2021). 00.
Idoate-Lacasia, J. et al. Long-term biomass dynamics of temperate forests in Europe after cessation of management. Forest Ecol. Manag. 554, 121697 (2024).
Naudts, K. et al. Europe’s forest management did not mitigate climate warming. Science 351, 597–600 (2016).
Barredo, J. I. Mapping and assessment of primary and old-growth forests in Europe. JRC Science for Policy Report, European Commission. EUR 30661 EN, Publications Office of the European Union, https://doi.org/10.2760/797591 (2021).
Burrascano, S., Keeton, W. S., Sabatini, F. M. & Blasi, C. Commonality and variability in the structural attributes of moist temperate old-growth forests: A global review. Forest Ecol. Manag. 291, 458–479 (2013).
Eggers, T. The impacts of manufacturing and utilisation of wood products on the European carbon budget. European Forest Institute, Internal Report 9. https://efi.int/sites/default/files/files/publication-bank/2018/ir_09.pdf (2002).
Brunet-Navarro, P., Jochheim, H. & Muys, B. The effect of increasing lifespan and recycling rate on carbon storage in wood products from theoretical model to application for the European wood sector. Mitig Adapt Strateg Glob Change 22, 1193–1205 (2017).
European Commission. The European Commission’s Knowledge Centre for Bioeconomy. Brief on the role of the forest-based bioeconomy in mitigating climate change through carbon storage and material substitution. https://knowledge4policy.ec.europa.eu/bioeconomy/topic/forest-bioeconomy-cc-mitigation_en (2022).
Ciais, P. et al. Carbon accumulation in European forests. Nat. Geosci. 1, 425–429 (2008).
Adams, J. M. & Piovesan, G. Uncertainties in the role of land vegetation in the carbon cycle. Chemosphere 49, 805–819 (2002).
Korosuo, A. et al. The role of forests in the EU climate policy: are we on the right track? Carbon Balance Manag. 18, 15 (2023).
UNFCCC 2023. European Union Common Reporting Format Table https://unfccc.int/documents/627830 (2023).
European Commission Biodiversity Strategy for 2030. https://environment.ec.europa.eu/strategy/biodiversity-strategy-2030_en (2023).
Sabatini, F. M. et al. Protection gaps and restoration opportunities for primary forests in Europe. Divers. Distrib. 26, 1646–1662 (2020).
Maes, J., et al. Mapping and assessment of ecosystems and their services: an EU ecosystem assessment. JRC Science for Policy Report. EUR 30161, Publications Office of the European Union. https://doi.org/10.2760/757183 (2020).
Thomas, C. D. et al. Reconciling biodiversity and carbon conservation. Ecol. Lett. 16, 39–47 (2013).
European Environment Agency, CORINE Land Cover https://land.copernicus.eu/pan-european/corine-land-cover (2018).
FAO Global Soil Organic Carbon GSOC map v 1.5. Technical Report, Intergovernmental Technical Panel on Soils, Rome FAO. https://www.fao.org/documents/card/en/c/ca7597en. https://www.fao.org/global-soil-partnership/pillars-action/4-information-and-data-new/global-soil-organic-carbon-gsoc-map/en/ (2020c).
Bottero, A., Meloni, F., Garbarino, M. & Motta, R. Temperate coppice forests in north-western Italy are resilient to wild ungulate browsing in the short to medium term. For Ecol Manage 523, 120484 (2022).
Santoro, M., et al. GlobBiomass: global above-ground biomass and growing stock volume datasets available on-line at http://globbiomass.org/products/global-mapping (2018).
Avitabile, V. et al. An integrated pan-tropical biomass map using multiple reference datasets. Global Change Biol. 22, 1406–1420 (2016).
Forest Europe State of Europe’s Forests 2020. https://foresteurope.org/wp-content/uploads/2016/08/SoEF_2020.pdf (2020).
Acknowledgements
We thank the many people involved with the collection and provision of the site data and recognise the significant resources, people and time required to collect this invaluable data. The research for the synthesis, analysis and writing (H.K., Z.K., S.H., B.M.) was supported by a grant from a charitable organisation which neither seeks nor permits publicity for its efforts. The funder had no involvement in the study design, results or publication of the paper. Site data from Spain was funded by the Spanish Ministry of Science, Innovation and Universities [AGL2016-76769-C2-2-R]. C.P.C. was supported by the Spanish Ministry of Science and Innovation [RYC2018-024939-I]. J.A.M.V. was supported by the Ramón Areces Foundation Grants for Postdoctoral Studies. Contribution of D.A., K.K. and P.S. as well as data collection and processing from Czech natural forests was supported by Czech Science Foundation, project no. 24-11119S. D.M.-B. was funded by projects AGL2015-73190-JIN, PID2019-110273RB-I00 and contract RYC-2017-23389 by the Spanish Ministry of Science and Innovation MCIN/AEI. V.B. and I.D. were supported by the FORCLIMIT project funded in the frame of the ERA-NET FACCE ERA-GAS and with national support from Romanian National Authority for Scientific Research and Innovation, CCCDI – UEFISCDI [grant number 82/2017]. FACCE ERA-GAS has received funding from the European Union’s Horizon 2020 research and innovation programme [grant agreement 696356. T.Z. was funded by The WWF Bulgaria through the project IKEA № 9E0710.05 and by The National Roadmap for Research Infrastructure (2020-2027), Ministry of Education and Science of Republic of Bulgaria, through agreements No DO1-405/18.12.2020 and DO1-163/28.07.2022 (LTER-BG). L.D. was funded by the project of the National Research, Development and Innovation Office NKFIH K 131837. T.N. received support from the Slovenian Research Agency (Project No. J4-1765). For additional assistance with site data, we thank Dr. Raúl Sanchez-Salguero and Dr. Andrea Hevia for evaluating the age in the dendrochronological samples in Spain, and Nesibe Köse, Mehmet Doğan, Daniel Bishop, Marco Mina, Timothy Thrippleton, Neil Pederson, Guillermo Gea-Izquierdo and Macarena Férriz for their help during fieldwork, and Cengiz Cihan and the Turkish General Directorate of Forestry (OGM) in Borçka (Artvin) for their assistance in the field in Turkey.
Author information
Authors and Affiliations
Contributions
H.K., Z.K., B.M. conceptualised the study; Z.K. coordinated data providers; H.K. and S.H. analysed the data; H.K. wrote the paper; M.S., M.M., D.A., D.B., V.B., F.B., J.J.C., L.D., A.D.F., I.D., M.G., A.J., F.H., V.I., L.K., K.K., D.M.B., J.A.M.V., R.M., T.A.N., M.P., C.P.C., G.P., C.C.R., P.S., O.V., M.Y., T.Z. contributed site data and interpretation, all authors contributed to editing and review of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Earth & Environment thanks Per Gundersen and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Alireza Bahadori and Aliénor Lavergne. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Keith, H., Kun, Z., Hugh, S. et al. Carbon carrying capacity in primary forests shows potential for mitigation achieving the European Green Deal 2030 target. Commun Earth Environ 5, 256 (2024). https://doi.org/10.1038/s43247-024-01416-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s43247-024-01416-5
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.