Abstract
Generating responses to tumor antigens poses a challenge for immunotherapy. This phase II trial (NCT02129075) tested fms-like tyrosine kinase 3 (Flt3) ligand pre-treatment enhancement of responses to dendritic cell (DC)-targeting vaccines. We evaluated a regimen of Flt3L (CDX-301) to increase DCs and other antigen-presenting cells, poly-ICLC (TLR3 agonist that activates DCs) and a vaccine comprising anti-DEC-205-NY-ESO-1, a fusion antibody targeting CD205, linked to NY-ESO-1. High-risk melanoma patients were randomized to vaccine, with and without CDX-301. The end point was immune response to NY-ESO-1. Flt3L increased peripheral monocytes and conventional DCs (cDCs), including cross-presenting cDC1 and cDC2 and plasmacytoid DCs. Significant increases in humoral and T-cell responses and activation of DCs, natural killer cells and T cells were elicited. Transcriptional analyses revealed gene signatures associated with CDX-301 induction of an early, durable immune response. This study reveals in vivo effects of Flt3L on innate immune cells in the setting of vaccination, leading to an immunogenic vaccine regimen.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Source data are available for this study. Data supporting the findings of this study are available from the corresponding authors upon request. Data are immediately available via the CITN Labkey data-sharing platform by request for login credentials to cimldata@fredhutch.org for database access at https://ciml.labkey.com/home/project-begin.view. Source data are provided with this paper.
Code availability
Codes, raw data and analyses for gene expression analyses have been deposited at: https://github.com/patrickjdanaher/CITN07-nCounter-data-code-results.
References
Si, Z., Hersey, P. & Coates, A. S. Clinical responses and lymphoid infiltrates in metastatic melanoma following treatment with intralesional GM-CSF. Melanoma Res. 6, 247–255 (1996).
Wolchok, J. D. et al. Nivolumab plus ipilimumab in advanced melanoma. N. Engl. J. Med. 369, 122–133 (2013).
Hodi, F. S. et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 363, 711–723 (2010).
Randazzo, M., Terness, P., Opelz, G. & Kleist, C. Active-specific immunotherapy of human cancers with the heat shock protein Gp96-revisited. Int. J. Cancer 130, 2219–2231 (2012).
Wood, C. et al. An adjuvant autologous therapeutic vaccine (HSPPC-96; vitespen) versus observation alone for patients at high risk of recurrence after nephrectomy for renal cell carcinoma: a multicentre, open-label, randomised phase III trial. Lancet 372, 145–154 (2008).
Wong, H. S. & Germain, R. N. Robust control of the adaptive immune system. Semin. Immunol. 36, 17–27 (2018).
Gerner, M. Y., Casey, K. A., Kastenmuller, W. & Germain, R. N. Dendritic cell and antigen dispersal landscapes regulate T cell immunity. J. Exp. Med. 214, 3105–3122 (2017).
Maraskovsky, E. et al. Dramatic increase in the numbers of functionally mature dendritic cells in Flt3 ligand-treated mice: multiple dendritic cell subpopulations identified. J. Exp. Med. 184, 1953–1962 (1996).
Anandasabapathy, N. et al. Efficacy and safety of CDX-301, recombinant human Flt3L, at expanding dendritic cells and hematopoietic stem cells in healthy human volunteers. Bone Marrow Transplant. 50, 924–930 (2015).
Macri, C., Dumont, C., Johnston, A. P. & Mintern, J. D. Targeting dendritic cells: a promising strategy to improve vaccine effectiveness. Clin. Translat. Immunol. 5, e66 (2016).
Bonifaz, L. C. et al. In vivo targeting of antigens to maturing dendritic cells via the DEC-205 receptor improves T cell vaccination. J. Exp. Med. 199, 815–824 (2004).
Anandasabapathy, N. et al. Classical Flt3L-dependent dendritic cells control immunity to protein vaccine. J. Exp. Med. 211, 1875–1891 (2014).
Dhodapkar, M. V. et al. Induction of antigen-specific immunity with a vaccine targeting NY-ESO-1 to the dendritic cell receptor DEC-205. Sci. Transl. Med. 6, 232ra251 (2014).
Tsuji, T. et al. Antibody-targeted NY-ESO-1 to mannose receptor or DEC-205 in vitro elicits dual human CD8+ and CD4+ T cell responses with broad antigen specificity. J. Immunol. 186, 1218–1227 (2011).
Villani, A. C. et al. Single-cell RNA-seq reveals new types of human blood dendritic cells, monocytes, and progenitors. Science 356, eaah4573 (2017).
Breton, G. et al. Circulating precursors of human CD1c+ and CD141+ dendritic cells. J. Exp. Med. 212, 401–413 (2015).
See, P. et al. Mapping the human DC lineage through the integration of high-dimensional techniques. Science 356, eaag3009 (2017).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. Series B 57, 289–300 (1995).
Danaher, P. et al. Gene expression markers of tumor infiltrating leukocytes. J. Immunother. Cancer 5, 18 (2017).
Alsina, L. et al. A narrow repertoire of transcriptional modules responsive to pyogenic bacteria is impaired in patients carrying loss-of-function mutations in MYD88 or IRAK4. Nat. Immunol. 15, 1134–1142 (2014).
Altman, M. C. et al. A novel repertoire of blood transcriptome modules based on co-expression patterns across sixteen disease and physiological states. Preprint at bioRxiv https://doi.org/10.1101/525709 (2019).
Chaussabel, D. & Baldwin, N. Democratizing systems immunology with modular transcriptional repertoire analyses. Nat. Rev. Immunol. 14, 271–280 (2014).
Chaussabel, D. et al. A modular analysis framework for blood genomics studies: application to systemic lupus erythematosus. Immunity 29, 150–164 (2008).
Chiche, L. et al. Modular transcriptional repertoire analyses of adults with systemic lupus erythematosus reveal distinct type I and type II interferon signatures. Arthritis Rheum. 66, 1583–1595 (2014).
Obermoser, G. et al. Systems scale interactive exploration reveals quantitative and qualitative differences in response to influenza and pneumococcal vaccines. Immunity 38, 831–844 (2013).
Oka, Y. et al. Cancer immunotherapy targeting Wilms’ Tumor Gene WT1 product. J. Immunol. 164, 1873 (2000).
Sampson, J. H., Archer, G. E., Mitchell, D. A., Heimberger, A. B. & Bigner, D. D. Tumor-specific immunotherapy targeting the EGFRvIII mutation in patients with malignant glioma. Semin. Immunol. 20, 267–275 (2008).
Rosenberg, S. A., Yang, J. C. & Restifo, N. P. Cancer immunotherapy: moving beyond current vaccines. Nat. Med. 10, 909–915 (2004).
Scheibenbogen, C. et al. Rational peptide-based tumour vaccine development and T cell monitoring. Semin. Cancer Biol. 13, 423–429 (2003).
Pearson, F. E. et al. Activation of human CD141(+) and CD1c(+) dendritic cells in vivo with combined TLR3 and TLR7/8 ligation. Immunol. Cell Biol. 96, 390–400 (2018).
Marroquin, C. E. et al. Mobilization of dendritic cell precursors in patients with cancer by flt3 ligand allows the generation of higher yields of cultured dendritic cells. J. Immunother. 25, 278–288 (2002).
Hammerich, L. et al. Systemic clinical tumor regressions and potentiation of PD1 blockade with in situ vaccination. Nat. Med. 25, 814–824 (2019).
Ohri, N. et al. Abstract CT005: FLT3 ligand (CDX-301) and stereotactic radiotherapy for advanced non-small cell lung cancer. Cancer Res. 78, CT005 (2018).
Freedman, R. S. et al. Pilot study of Flt3 ligand comparing intraperitoneal with subcutaneous routes on hematologic and immunologic responses in patients with peritoneal carcinomatosis and mesotheliomas. Clin. Cancer Res. 9, 5228–5237 (2003).
Evans, T. G., Hasan, M., Galibert, L. & Caron, D. The use of Flt3 ligand as an adjuvant for hepatitis B vaccination of healthy adults. Vaccine 21, 322–329 (2002).
Morse, M. A. et al. Immunotherapy of surgical malignancies. Curr. Prob. Surg. 41, 15–132 (2004).
Klein, O. et al. Flt3 ligand expands CD4+ FoxP3+ regulatory T cells in human subjects. Eur. J. Immunol. 43, 533–539 (2013).
Disis, M. L. et al. Flt3 ligand as a vaccine adjuvant in association with HER-2/neu peptide-based vaccines in patients with HER-2/neu-overexpressing cancers. Blood 99, 2845–2850 (2002).
Shackleton, M. et al. The impact of imiquimod, a Toll-like receptor-7 ligand (TLR7L), on the immunogenicity of melanoma peptide vaccination with adjuvant Flt3 ligand. Cancer Immun. 4, 9 (2004).
Morse, M. A. et al. Preoperative mobilization of circulating dendritic cells by Flt3 ligand administration to patients with metastatic colon cancer. J. Clin. Oncol. 18, 3883–3893 (2000).
Rini, B. I., Paintal, A., Vogelzang, N. J., Gajewski, T. F. & Stadler, W. M. Flt-3 ligand and sequential FL/interleukin-2 in patients with metastatic renal carcinoma: clinical and biologic activity. J. Immunother. 25, 269–277 (2002).
Higano, C. S. et al. Safety and biological activity of repeated doses of recombinant human Flt3 ligand in patients with bone scan-negative hormone-refractory prostate cancer. Clin. Cancer Res. 10, 1219–1225 (2004).
Robbins, P. F. et al. Tumor regression in patients with metastatic synovial cell sarcoma and melanoma using genetically engineered lymphocytes reactive with NY-ESO-1. J. Clin. Oncol. 29, 917–924 (2011).
Nicholaou, T. et al. Directions in the immune targeting of cancer: lessons learned from the cancer-testis Ag NY-ESO-1. Immunol. Cell Biol. 84, 303–317 (2006).
Yuan, J. et al. CTLA-4 blockade enhances polyfunctional NY-ESO-1 specific T cell responses in metastatic melanoma patients with clinical benefit. PNAS 105, 20410–20415 (2008).
Yuan, J. et al. Integrated NY-ESO-1 antibody and CD8+ T-cell responses correlate with clinical benefit in advanced melanoma patients treated with ipilimumab. PNAS 108, 16723–16728 (2011).
Ebert, L. M. et al. A cancer vaccine induces expansion of NY-ESO-1-specific regulatory T cells in patients with advanced melanoma. PLoS ONE 7, e48424 (2012).
D’Angelo, S. P. et al. Antitumor activity associated with prolonged persistence of adoptively transferred NY-ESO-1 (c259)T cells in Synovial Sarcoma. Cancer Discov. 8, 944–957 (2018).
Holmes, M. L., Carotta, S., Corcoran, L. M. & Nutt, S. L. Repression of Flt3 by Pax5 is crucial for B-cell lineage commitment. Genes Devel. 20, 933–938 (2006).
Guimond, M. et al. In vivo role of Flt3 ligand and dendritic cells in NK cell homeostasis. J. Immunol. 184, 2769–2775 (2010).
Lynch, D. H. et al. Flt3 ligand induces tumor regression and antitumor immune responses in vivo. Nat. Med. 3, 625–631 (1997).
Fong, L. et al. Altered peptide ligand vaccination with Flt3 ligand expanded dendritic cells for tumor immunotherapy. PNAS 98, 8809–8814 (2001).
McNeel, D. G. et al. Pilot study of an HLA-A2 peptide vaccine using flt3 ligand as a systemic vaccine adjuvant. J. Clin. Immunol. 23, 62–72 (2003).
Chen, W. et al. FLT3 ligand administration after hematopoietic cell transplantation increases circulating dendritic cell precursors that can be activated by CpG oligodeoxynucleotides to enhance T-cell and natural killer cell function. Biology of blood and marrow transplantation 11, 23–34 (2005).
Adams, S., O’Neill, D. W. & Bhardwaj, N. Recent advances in dendritic cell biology. J. Clin. Immunol. 25, 177–188 (2005).
Vandesompele, J. et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 3, Research0034 (2002).
Moodie, Z. et al. Response definition criteria for ELISPOT assays revisited. Cancer Immunol. Immunother. 59, 1489–1501 (2010).
Agresti, A. & Caffo, B. Simple and effective confidence intervals for proportions and differences of proportions result from adding two successes and two failures. Am. Stat. 54, 280–288 (2000).
Acknowledgements
We are deeply indebted to the patients and their families and to staff at all clinical sites. This work was supported by the following grants from the National Cancer Institute: U01CA154967 and UM1CA154967 (to M.A.C. and S.P.F.); Cancer Center Support Grant P30 CA015704 (to M.A.C.); R01CA201189 (to N.B.) and R01CA180913 (to N.B.).
Author information
Authors and Affiliations
Contributions
The author contributions were as follows: protocol/trial design (N.B., M.J.Y., T.A.D., L.L., T.K., A.M.S., E.S., C.M., M.L.D. and M.A.C.); patient accrual (N.B., P.A.F., A.C.P., M.S.E., B.R.G., B.A.H., B.D.C., M.R.A. and J.J.L.); data generation/analysis (N.B., D.B., J.M.B., A.B.B., S.B., A.S.C., L.D., P.D., T.H., B.W.H., T.K., N.R., D.R., L.L., B.A.S., L.A.V., E.W., C.M., S.W., M.L.D., M.A.C. and S.P.F.); and manuscript writing (N.B., D.B., P.D., L.D., N.R., M.S.E., B.R.G., M.A.C. and S.P.F.).
Corresponding authors
Ethics declarations
Competing interests
A.S.C., T.H., T.K., L.A.V., T.A.D. and M.J.Y. are/were employed by and are stockholders in Celldex Therapeutics; J.M.B., P.D. and S.W. are employed by NanoString Technologies; A.M.S. is employed by Oncovir; N.B. and J.J.L. receive research support from Celldex (and Oncovir N.B.). L.D., L.L., N.R., B.W.H., M.A.C. and S.P.F. are employed by the Fred Hutchinson Cancer Research Center, which received partial funding from Celldex for this trial.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 T cell responses to NY-ESO-1 versus durability, NY-ESO-1 status and disease recurrence.
a, T cell responses remain durable in cohort 1 subjects for several months. IFNγ, TNFα, and IL-2 cytokine production by CD8+ T-cells (lower panel) and CD4+ T cells (upper panel) were determined by ICS. (N = 2 cohort 1, black) and N = 2 cohort 2, white). b, Baseline T cell responses vs NY-ESO-1 status. 43/60 subjects had tumor biopsies tested for NY-ESO-1 expression and 8 of these 43 subjects had tumors which expressed NY-ESO-1. The only three subjects which showed high baseline T-cell responses to NY-ESO-1 were from the group of eight subjects which had a positive NY-ESO-1 tumor status. (Ungrouped analyses: NY-ESO-1pos: N = 8 subjects; NY-ESO-1neg: N = 35 subjects; Not Stained: N = 17 subjects). (By Cohort analyses: NY-ESO-1pos: N = 3 Cohort 1, N = 5 Cohort 2; NY-ESO-1neg: N = 17 Cohort 1, N = 18 Cohort 2; Not Stained: N = 10 Cohort 1, N = 17 Cohort 2. Two-sided t-test). c, Overall T cell responses vs disease recurrence. Cohort 1 showed greater elevation in anti-NY-ESO-1 T-cell responses than cohort 2. This higher antigen-specific response, however, did not translate to lower levels of recurrence in cohort 1 (12/30 cohort 1 subjects experienced recurrence, vs 8/30 in cohort 2). Statistics for non-recurrence (NR) vs recurrence (R) within each cohort were calculated by a 2 tailed t-test. For cohort 1, n = 18 NR subjects vs n = 12R subjects. For cohort 2, n = 21 NR subjects vs n = 9R subjects. b,c, Bars indicate mean±SD.
Extended Data Fig. 2 Whole blood immunophenotyping by flow cytometry.
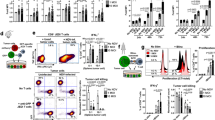
Results reveal consistent expansion of specific cell populations in cohort 1. Graphs show individual fold changes from baseline in absolute cell numbers (per mL whole blood) for 16 subjects in cohort 1 for the following cell types: monocytes, conventional DC (cDC), NK cells, B cells, plasmacytoid DC (pDC), and CD56 Bright NK cells. See Fig. 2 for mean changes by cohort.
Extended Data Fig. 3 DC subset gating and fold increase of DC subsets during Flt3L mobilization.
a, Flow cytometry gating strategy; To determine different DC subsets, gating was performed as follows: Lineage- Blue+ HLA-DR+ CD45RA- CD123- CD11c+ (CD11c+), gating on CD11c+ →CD141+ Clec9a+(DC1), gating on CD11c+ →CD141-Clec9a- CD1c+ (DC2), gating on CD11c+ →CD141-Clec9a- CD1c− CD16+(DC4), gating on Lineage- Blue+ HLA-DR+→CD123+CD45RA+ (DC6), gating on Lineage- Blue+ HLA-DR+ CD123+ CD45RA+ CD33+ AXL- (pre-DC). b, Mean fold increases of the indicated DC subsets from each cohort (analyses of n = 7 independent subjects per cohort)(Two-sided t-test).
Extended Data Fig. 5 Modular transcriptional fingerprints for group comparison (Cohort 1).
Responsive modules at each time point in comparison to baseline are mapped on a grid for visualization purposes. The color of the spots indicates the direction of changes in transcript abundancies: red for up-regulation and blue for down-regulation. The degree of intensity of the spots denotes the level of activation, which is given by the proportion of transcript coherently regulated. A legend is provided with functional interpretations indicated at each position of the grid by a color code (N = 12 cohort and N = 11 cohort 2).
Extended Data Fig. 6 Mapping perturbations of the modular repertoire across individual samples.
The responsive modules in individual subjects from cohort 1 (N = 12) after treatment as compared to baseline are mapped into grids. The expression profile of each individual subject was calculated as a FC and difference after treatment as compared to the expression of individual samples at baseline. To determine changes in individual subjects, a cut-off is set against which individual genes constitutive of a module are tested (|FC|> 1.5 and |difference|>100).
Supplementary information
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 1
Statistical source data.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 6
Statistical source data.
Rights and permissions
About this article
Cite this article
Bhardwaj, N., Friedlander, P.A., Pavlick, A.C. et al. Flt3 ligand augments immune responses to anti-DEC-205-NY-ESO-1 vaccine through expansion of dendritic cell subsets. Nat Cancer 1, 1204–1217 (2020). https://doi.org/10.1038/s43018-020-00143-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s43018-020-00143-y
This article is cited by
-
FLT3L-induced virtual memory CD8 T cells engage the immune system against tumors
Journal of Biomedical Science (2024)
-
Dendritic cell-targeted therapy expands CD8 T cell responses to bona-fide neoantigens in lung tumors
Nature Communications (2024)
-
Therapeutic targeting of tumour myeloid cells
Nature Reviews Cancer (2023)
-
Multi-antigen spherical nucleic acid cancer vaccines
Nature Biomedical Engineering (2023)
-
Close the cancer–immunity cycle by integrating lipid nanoparticle–mRNA formulations and dendritic cell therapy
Nature Nanotechnology (2023)