Abstract
The prevalence of metalloenzymes with multinuclear metal complexes in their active sites inspires chemists’ interest in the development of multinuclear catalysts. Studies in this area commonly focus on binuclear catalysts containing either metal-metal bond or electronically discrete, conformationally advantageous metal centres connected by multidentate ligands, while in many multinuclear metalloenzymes the metal centres are bridged through μ2-ligands without a metal-metal bond. We report herein a μ2-iodide-bridged binuclear palladium catalyst which accelerates the C-H nitrosation/annulation reaction and significantly enhances its yield compared with palladium acetate catalyst. The superior activity of this binuclear palladium catalyst is attributed to the trans effect-relay through the iodide bridge from one palladium sphere to the other palladium sphere, which facilitates dissociation of the stable six-membered chelating ring in palladium intermediate and accelerates the catalytic cycle. Such a trans effect-relay represents a bimetallic cooperation mode and may open an avenue to design and develop multinuclear catalysts.
Similar content being viewed by others
Introduction
The prevalence of the metalloenzymes containing binuclear or polynuclear transition metal complexes in their active sites has motivated the interest of chemists in the binuclear or polynuclear homogeneous catalysts for organic transformations1,2,3,4,5,6,7,8,9,10. A number of well-defined binuclear catalysts have been reported with unique activity and selectivity accessible through the cooperation between two metal centres1,4,5,6,8,9,10,11,12. Two types of bimetallic cooperation are commonly encountered in the catalytic processes of these binuclear catalysts: one is the simultaneous activation of two reaction partners by the binuclear catalysts in which multidentate ligands hold two metal centres in close proximity to create two electronically discrete, conformationally advantageous active sites for binding the corresponding substrate molecules1,4,5,13,14, as illustrated by olefin polymerization13 or enantioselective binuclear catalysts5,14; the other is that two metal centres of the metal–metal bond-containing binuclear catalyst synergistically participate in elemental redox steps in catalytic pathways6,9,10,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32, as exemplified by Pd(III) dimer-catalysed oxidative C–H functionalization17,18,19 and dirhodium-catalysed reactions for carbene20,21,22 or nitrene insertion23 into C–H bond. In many of the metalloenzymes, on the other hand, metal centres at the active site are held together by μ2-bridging ligands, with the metal–metal bond absent, implicating that the catalysis therein may invoke different bimetallic or polymetallic cooperation in which μ2-bridging ligands likely play key roles. Disclosing the bimetallic or polymetallic cooperation mechanism for catalysis would provide platforms to rationally design the binuclear or polynuclear metal catalysts with the unique activity and selectivity. Due to the rapid equilibration of the multinuclear complexes with other metal species in the catalytic conditions, however, it has been highly challenging to identify the active species and reveal the responsible cooperation mechanism in a definitive manner7, which is the reason why the promising binuclear or multinuclear metal catalysts are relatively underdeveloped compared with the mononuclear metal catalysts that dominate in homogenous catalysis.
Here, we report an iodide-bridged binuclear palladium catalyst generated in situ from palladium acetate, azobenzene and tetra-n-butyl ammonium iodide (TBAI), which accelerates C–H nitrosation/annulation reaction and significantly enhances its yields compared with palladium acetate alone as a catalyst (Fig. 1). This binuclear palladium species, according to the kinetic studies, retains the integrity of its iodide-bridged binuclear core structure during the catalytic cycle. Computational studies further reveal that a strongly σ-donating η1 phenyl ligand around one palladium centre of this binuclear complex is able to exert a trans effect33, through the bridging iodide ligand, on the ligand at the other palladium centre and therefore labilize the coordination bond trans to this bridging iodide34,35. Consequently, two palladium centres within the binuclear cluster cooperatively decrease the activation barriers of dissociation of the chelating product fragment from catalyst and accelerate the whole catalytic cycle. The binuclear metal catalyst that features the trans effect-relay through bridging ligand may provide a solution to metal-catalysed efficient syntheses of the chelating compounds that often impede catalytic cycle.
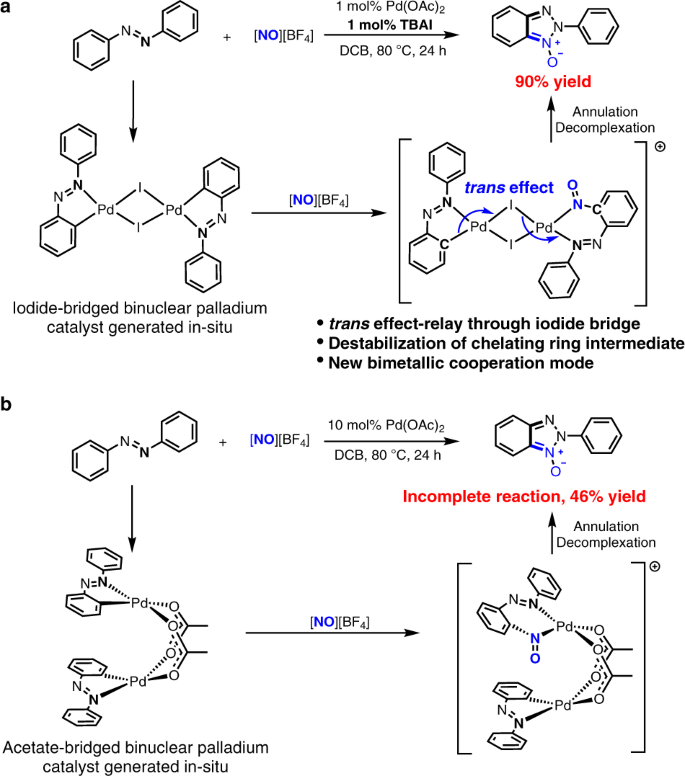
a The equimolar combination of (n-Bu4N)I with Pd(OAc)2 leads to enhancement of palladium catalysis via formation of the iodide-bridged binuclear palladium catalyst that is able to destabilize six-membered chelating ring of reaction intermediate due to trans effect-relay through iodide bridge, and therefore facilitate the reaction. The trans effect-relay through iodide bridge represents a bimetallic cooperation mode for catalysis. b The reaction catalyzed by 10 mol% Pd(OAc)2 alone affords lower yield than the reaction with combination of 1 mol% (n-Bu4N)I with 1 mol% Pd(OAc)2. The difficult in decomplexation of six-membered chelating ring of reaction intermediate retards the reaction catalysed by Pd(OAc)2 alone and results in incomplete reaction.
Results and discussion
Discovery of beneficial effect of iodide
Our interest in the binuclear palladium catalyst stemmed from a discovery during exploration of palladium-catalysed ortho C–H bond nitrosation reaction of azobenzenes. The development of this Pd-catalysed aryl C–H nitrosation was aimed at achieving a straightforward approach to nitrosoarenes that are a class of versatile synthetic intermediates utilized in a variety of transformations36. Importantly, the metal-catalyzed aryl C–H nitrosation method has a potential to get over substrate limitation, poor regioselectivity37 and substrate pre-activation36 problems encountered in the existing methods for syntheses of nitrosoarenes. To this end, we started our investigation by examining the reaction of azobenzene (1a) with two equivalents of nitrosonium tetrafluoroborate ([NO][BF4]) (2) conducted in 1,2-dicholobenzene (DCB) of 1.5 mL at 80 °C for 24 h in the presence of 1 mol% Pd(OAc)2 as a catalyst (Table 1). The initial reaction conditions enabled the desired ortho C–H nitrosation but generated 2H-benzotriazole N-oxide product (2-phenyl-2H-benzo[d][1,2,3]triazole 1-oxide 3a) as a final product in only 7% yield via a C–H nitrosation/annulation sequence (entry 1 in Table 1).
The fact that 2H-benzotriazole heterocyclic N-oxides are the privileged structural motifs in biologically active compounds, pharmaceuticals, and functional materials38,39,40 prompted us to identify the optimal conditions for this Pd-catalysed reaction of azobenzene with [NO][BF4]. Increasing catalyst loading to 10 mol% offered a 46% yield of the desired 3a with azobenzene recovered (entry 2 in Table 1). Screening a variety of additives revealed that 10 mol% tetra-n-butyl ammonium chloride (TBAC), in combination with 10 mol% Pd(OAc)2, enhanced the yield of 3a to 76% (entry 3 in Table 1). Interestingly, TBAI exhibited a more beneficial effect on the reaction than TBAC. 1 mol% TBAI allowed using 1 mol% Pd(OAc)2 to achieve a 90% yield (entry 4 in Table 1). Considering that p-toluenesulfonic acid (TsOH) may facilitate cyclopalladation of azobenzene, 2 mol% TsOH was introduced into the reaction system with 1 mol% TBAI and 1 mol% Pd(OAc)2, but still afforded a 90% yield (entry 5 in Table 1). Other protonic acids did not show beneficial effect on the reaction yield as well (see Supplementary Table 1). Control experiment showed that Pd(OAc)2 was indispensable for this reaction (entry 6 in Table 1). The beneficial effect of iodide on the Pd-catalysed C–H nitrosation/annulation sequence is to some extent surprising because it is at variance of the previous findings that iodide anion tends to retard late transition metal-catalysed C–H functionalization reactions by weakening electrophilicity of transition metal ions41,42. Although halide anions including iodide enhance Pd-catalysed traditional cross-coupling reactions of aryl halides with nucleophilic organometallics43, these Pd-catalysed traditional cross-coupling reactions via Pd(0)/Pd(II) cycle are distinctly different from the Pd-catalysed oxidative C–H nitrosation/annulation reaction that would begin with the electrophilic cyclopalladation of Pd(II) species and involves Pd(II)/Pd(III)17,18,19 or Pd(II)/Pd(IV)18,44,45 cycle.
Identification of iodide-bridged binuclear palladium complex as a catalytically active intermediate
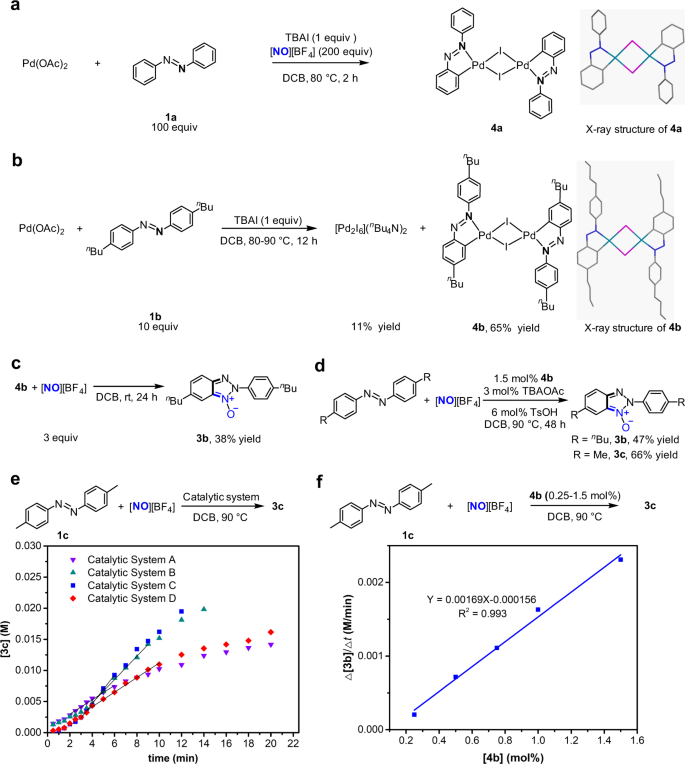
Intrigued by this beneficial effect of iodide, we performed mechanistic investigation to identify its origin. Initially, we tried to isolate a palladium intermediate lying on catalytic cycle. Under the conditions mimicking the aforementioned Pd(OAc)2/TBAI-catalysed reaction, the reaction of 0.2 mmol Pd(OAc)2 with one equivalent of TBAI, 100 equivalents of azobenzene and 200 equivalents of [NO][BF4] afforded an iodide-bridged binuclear palladium complex bearing cyclopalladated azobenzene ligand as dark red solid (4a) (Fig. 2a). Single-crystal X-ray diffraction analysis revealed that the molecule of 4a comprises a twofold iodide-bridged binuclear palladium core with each palladium atom chelated by an azobenzene ligand through nitrogen and carbon atoms (Fig. 2a)46, and square planar coordination environments around these two palladium atoms are identical and lie in the same plane because they are related by centrosymmetry. Within 4a, the I–Pd bond trans to Pd–C bond is 0.12 Å longer than the other I–Pd bond between the iodide atom in question and the other palladium centre owing to strong trans influence of phenyl ligand, leading to unsymmetrical iodide bridges. Because the poor solubility of crystalline 4a in 1,2-dichlorobenzene impeded the investigations of reactivity of 4a, we turned our attention to synthesis of highly soluble congeners of 4a. The treatment of Pd(OAc)2 with one equivalent of TBAI and 12 equivalents of 4,4′-di-n-butyl-azobenzene (1b) in DCB at 80 °C for 12 h afforded a soluble analogue of 4a, an iodide-bridged binuclear palladium complex of cyclopalladated 4,4′-di-n-butyl-azobenzene ligand (4b) in 65% yield with concomitant formation of [Pd2I6](n-Bu4N)247 in 11% yield (Fig. 2b). The cyclopalladated azobenzene ligands in 4a and 4b indicate that Pd-introduced C–H bond cleavage can occur in the presence of iodide anion, though iodide anion was thought to tend to form stable Pd–I bond and thus weaken the Pd electrophilicity that is required for C–H metallation41.
a Isolation of iodide-bridged binuclear palladium complex bearing cyclopalladated azobenzene ligand (4a) from the reaction under conditions mimicking catalysis process. b Preparation of iodide-bridged binuclear palladium complex bearing cyclopalladated 4,4′-di-n-butyl-azobenzene ligand 4b from self-assembly of 4,4′-di-n-butyl-azobenzene ligand and Pd(OAc)2. c Stoichiometric reaction of 4b with [NO][BF4] at room temperature to generate 2H-benzotriazole N-oxide. d 4b-catalysed reaction of 4,4′-di-substituted azobenzenes with [NO][BF4]. e Comparison of activity of catalyst systems by determining the initial rates on the basis of plot of product 3c concentration (M) versus time (min). The reaction of 4,4′-dimethyl-azobenzene with [NO][BF4] was used a model, which was analysed by HPLC with 3,4-dichlorotoluene as the internal standard. Catalyst system A is 3 mol% Pd(OAc)2 and 6 mol% TsOH; catalyst system B is 3 mol% Pd(OAc)2, 3 mol% TBAI and 6 mol% TsOH; catalyst system C is 1.5 mol% 4b; catalyst system D is 1.5 mol% 5b and 6 mol% TsOH. f A first-order dependence of the initial rate Δ[3c]/Δt (M/minute) on the 4b concentration [4b] (mol%) catalyst, using the reaction of 4,4′-dimethyl-azobenzene with [NO][BF4] as a model.
Using soluble 4b as a model compound, reactivity and kinetics of the iodide-bridged binuclear palladium complexes were then examined to identify their roles in the catalytic process. The reaction of 4b with 3 equivalents of [NO][BF4] at room temperature for 24 h showed that cyclopalladated 4,4′-di-n-butyl-azobenzene ligand in 4b underwent nitrosation and subsequent annulation to form 2H-benzotriazole N-oxide derivative bearing two n-butyl groups (3b) in 38% yield (Fig. 2c), which raised the possibility that such an iodide-bridged binuclear palladium complex was a reaction intermediate in the catalysis cycle. Furthermore, 1.5 mol% 4b, together with 3 mol% tetra-n-butyl ammonium acetate (n-Bu4NOAc) as an additive, served as a catalyst to effect the nitrosylation/annulation reactions of both 4,4′-di-n-butyl-azobenzene (1b) and 4,4′-dimethyl-azobenzene (1c) in the yields (Fig. 2d) comparable with those obtained with the combination of 3 mol% Pd(OAc)2 and 3 mol% TBAI (see below). The use of 3 mol% n-Bu4NOAc as an additive in these 4b-catalysed reactions is because the reaction of azobenzene catalysed by Pd(OAc)2 and TBAI might generate in situ the iodide-bridged binuclear palladium complex along with 2 equivalents of n-Bu4NOAc and 2 equivalents of acetic acid. In the 4b-catalysed transformation of azobenzenes to the corresponding 2H-benzotriazole N-oxide products, 1.5 mol% 4b provided slightly higher initial reaction rate compared with the combination of 3 mol% Pd(OAc)2, 3 mol% TBAI and 6 mol% TsOH, indicating that this binuclear palladium complex 4b is a kinetically competent catalyst (Fig. 2e). In the experiments determining the initial rate for reaction with 3 mol% Pd(OAc)2 and 3 mol% TBAI, 6 mol% TsOH was introduced as an additive into the reaction system to neutralize OAc− anion from Pd(OAc)2 and rule out the influence of OAc− when comparing with the reaction with 4b. The initial rates of the nitrosylation/annulation of 1c exhibited a first-order dependence on the concentration of 4b (Fig. 2f), supporting that the iodide-bridged binuclear palladium complex 4b and its congeners are catalytically active species, and retain integrity of its [Pd2I2] core structure during catalysis. The first-order dependence of the initial reaction rate on the concentration of 4b may also account for the observation that the reaction catalysed by 1.5 mol% 4b was slightly faster than the reaction catalysed by the combination of 3 mol% Pd(OAc)2 and 3 mol% TBAI since the concentration of the binuclear palladium complex generated in situ from self-assembly of 3 mol% Pd(OAc)2, 3 mol% TBAI and azobenzene is likely less than 1.5 mol%.
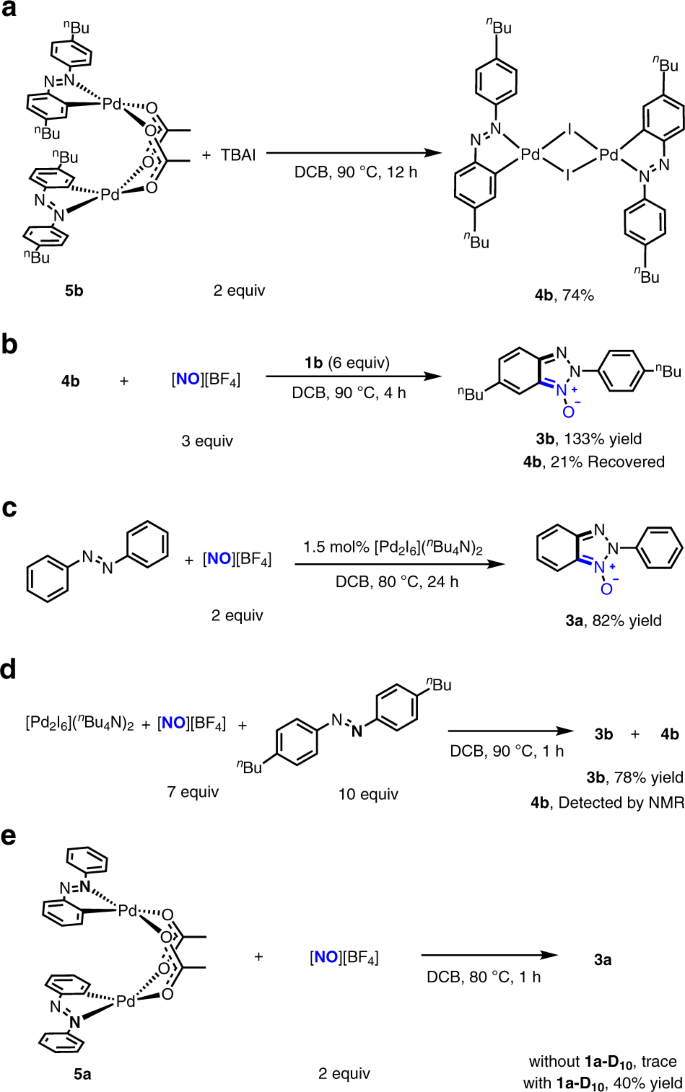
To confirm that 4b and its congeners are the catalytically active intermediates lying on catalytic cycle, we need to preclude the possibility that 4b and its congeners are the off-cycle precatalysts. The acetate-bridged binuclear palladium complex bearing a cyclopalladated 4,4′-di-n-butyl-azobenzene ligand on each palladium centre (5b)48 was observed to react with 2 equivalents of TBAI to lead to replacement of acetate bridge by iodide bridge and afford 4b in 74% yield (Fig. 3a). The conversion of 5b to 4b in turn implicated that the iodide-bridged binuclear palladium complexes were stable against the Pd–I bond cleavage caused through the ligand substitution, which is consistent with the fact that 4b was generated from the reaction of Pd(OAc)2 with excessive coordinating azobenzenes. Moreover, treatment of 0.1 mmol of 4b with 3 equivalents of [NO][BF4] and 6 equivalents of 4,4′-di-n-butyl-azobenzene in 5 mL DCB at 90 °C for 4 h gave rise to formation of 2H-benzotriazole N-oxide derivative 3b in 133% yield relative to 4b with 21% of 4b recovered (Fig. 3b), which supported that 4b could be re-generated during catalysis and that [NO][BF4] did not fragment this binuclear palladium complex via oxidation of bridging iodide. [Pd2I6](n-Bu4N)2 (1.5 mol%), a side-product in the preparation of 4b (Fig. 2b), was observed to catalyse the reaction of 1a with [NO][BF4] to afford 3a in 82% yield (Fig. 3c). However, in the stoichiometric reaction of [Pd2I6](n-Bu4N)2 with 7 equivalents of [NO][BF4] and 10 equivalents of 1b (Fig. 3d), 4b was detected with 3b formed, indicating that the catalytic behaviour of [Pd2I6](n-Bu4N)2 likely originated from its in situ conversion to the congener of 4b via oxidation of terminal iodide ligand by [NO][BF4]49. The experimentally demonstrated stability of 4b relative to other Pd species under the conditions similar to those for 4b-catalysis supported that the robust [Pd2I2] core structure of 4b retained integrity in the 4b-catalysed reactions.
a The reaction of acetate-bridged binuclear palladium complex bearing cyclopalladated 4,4′-di-n-butyl-azobenzene ligand (5b) with 2 equivalents of TBAI to generate 4b. b Stoichiometric reaction of 4b with 3 equivalents of [NO][BF4] and 6 equivalents of 4,4′-di-n-butyl-azobenzene. The reaction formed 3b in 133% yield relative to 4b with 21% of 4b recovered. c Catalytic reaction of azobenzene with [NO][BF4] using 1.5 mol% [Pd2I6](n-Bu4N)2 as a catalyst. d Stoichiometric reaction of [Pd2I6](n-Bu4N)2 with 7 equivalents of [NO][BF4] and 10 equivalents of 4,4′-di-n-butyl-azobenzene. The reaction formed 3b and 4b. e The reaction of acetate-bridged binuclear palladium complex bearing cyclopalladated azobenzene ligand 5a with 3 equivalents of [NO][BF4] with or without fully deuterated azobenzene (1a-D10).
As shown in Fig. 2e, the reaction catalysed by the combination of 3 mol% Pd(OAc)2 and 3 mol% TBAI was two times as fast as the reaction catalysed by 3 mol% Pd(OAc)2, revealing that 4a as a catalyst is more active than Pd(OAc)2. 1.5 mol% 5b provided the similar initial rate to 3 mol% Pd(OAc)2, supporting the reaction catalyzed by Pd(OAc)2 proceeds via cyclopalladation of azobenzenes to form analogues of 5b. In line with this surmise, an acetate-bridged binuclear palladium complex bearing cyclopalladated unsubstituted azobenzene ligand (5a)48, an analogue of 5b, reacted with [NO][BF4], in the presence of one equivalent of deuterated azobenzene (1a-D10) at 80 °C for 1 h, to produce the expected 3a in 40% yield (Fig. 3e). In contrast, the stoichiometric reaction of 5a with [NO][BF4] gave none of 3a in the absence of 1a-D10, which may implicate the reason why the reaction of 1a with [NO][BF4] catalyzed by 10 mol% Pd(OAc)2 alone did not go to completion.
Computational studies on the origin of the high activity of iodide-bridged binuclear palladium catalyst
The density functional theory (DFT) calculations on the mechanism for the nitrosylation/annulation reaction of azobenzene with [NO][BF4] catalysed by 4a. For comparison, DFT studies of this process with Pd(OAc)2 alone as the precatalyst were also carried out, in which acetate-bridged binuclear palladium complex 5a is assumed to be the catalyst generated in situ on the basis of aforementioned investigations.
Figure 4a presents the computed reaction pathways with catalyst 4a (blue line) and catalyst 5a (red line). These two pathways comprise similar elemental step. Reactions begin with oxidative addition of nitrosonium to one of two palladium atoms in 4a and 5a to produce higher oxidation state nitrosyl-palladium intermediates (LM1′ and LM1) (see Supplementary Fig. 16), which are followed by reductive elimination step to construct C–N bond and form six-membered chelation ring complexes (LM2′ and LM2). Then, assisted by the coordination of azobenzene ligand to palladium centre, dechelation of such six-membered chelation rings via TS2′ and TS2, and subsequent N–N bond formation for annulation proceed to release the target product 2H-benzotriazole N-oxide, respectively. Finally, with azobenzene as a base, azobenzene coordinating to palladium centre undergoes C–H palladation via a concerted metalation–deprotonation pathway to re-generate catalysts (4a and 5a) from the corresponding σ-complexes LM4′ and LM4. In the pathway of 4a, redox steps involve the change of the formal oxidation state of only one palladium atom from II to IV18, while in the pathway of 5a, both palladium atoms synergistically participate in redox steps via switch between Pd(II)-Pd(II) and Pd(III)-Pd(III) oxidation states17,18,19. The rate-determining step in the pathway of 4a is the C–H activation step with the activation barrier of 24.8 kcal/mol, in accordance with the experimentally observed intermolecular kinetic isotope effect (KIE) value of 4.4 (see Supplementary Fig. 14). In contrast, the step for dechelation of the palladium intermediate containing six-membered chelating ring is the rate-determining step in the pathway of 5a with the activation barrier of 27.5 kcal/mol. The lower activation barrier in the pathway of 4a is consistent with the observation that the 4b-catalysed reaction is faster than 5b-catalysed reaction (Fig. 2e).
a Free energy profiles for reaction catalyzed by 4a (blue line) and 5a (red line). b Optimized structures (bond length in Å) of iodide-bridged (LM2′) and acetate-bridged (LM2) binuclear palladium intermediates bearing six-membered chelating rings, and activation barriers for the dechelation/N–N bond formation steps from LM2′ via TSA′ transition state and LM2 via TSA transition state in the absence of azobenzene. c Dissociation curves along Pd1–N1 bond for LM2 (red line) and LM2′ (blue line).
Figure 4b shows the calculated structures of two palladium intermediates containing six-membered chelating rings (LM2 and LM2′). As reflected by bond lengths of the structure of LM2′, phenyl part of cyclopalladated azobenzene weakens the trans Pd2–I2 bond due to its strong trans influence, and therefore strengthens the Pd1–I2 bond, which enhances the trans influence of I2 on Pd1–N1 bond to labilize Pd1–N1 bond for dechelation. As a result, the calculated bond length for Pd1–N1 bond of iodide-bridged binuclear palladium species is longer by 0.045 Å than the corresponding bond of acetate-bridged species. In line with the bond lengths, the calculated Pd1–N1 bond strength in 4a-derived LM2′ is weaker than that of the 5a-derived LM2 (Fig. 4c). The further calculations (Fig. 4b) on the dechelation step without aid of azobenzene illustrate that the dechelation/N–N bond formation step of 5a-derived LM2 is infeasible in kinetics with a high free energy barrier of 34.3 kcal/mol (TSA), while the same step of 4a-derived LM2′ only needs to experience a lower barrier of 25.5 kcal/mol (TSA′). Such a conclusion is supported by the experimental observations that in the absence of free deuterated azobenzene, the reaction of 5a with [NO][BF4] at 80 °C did not produce 3a (Fig. 3e), while without any free azobenzene, 4b reacted with [NO][BF4] at room temperature to give the expected product (Fig. 2c), and that the 10 mol% Pd(OAc)2-catalysed reaction of 1a with [NO][BF4] did not go completion (entry 2, Table 1). As such, the trans effect-relay through bridging iodide in the 4a-derived binuclear palladium intermediate make the dechelation step easier and accelerates the reaction.
Substrate scope
This Pd-catalyzed C–H nitrosylation/annulation reaction of azobenzenes with [NO][BF4] is quite general. To achieve the satisfied yields, some of reactions were conducted at 90 °C for 48 h with 3 mol% Pd(OAc)2, 3 mol% TBAI and 6 mol% TsOH. In some cases, the combination of 0.5 mol% Pd(OAc)2 and 0.5 mol% TBAI afforded good yields. As shown in Fig. 5a, a spectrum of 4,4′-di-substituted symmetrical azoarenes bearing n-butyl, methyl, cyclohexyl, chloro, bromo, iodo and ester substituents could participate in the reaction to generate the corresponding products in good-to-excellent yields (3b-k) with the exception of 3e and 3f. The low yield of 3e and 3f may result from the steric hindrance from ortho-substitutes, which impeded the ligation of azobenzene nitrogen atoms to palladium centre.
a Substrate scope with respect to symmetrical azoarenes. b Substrate scope with respect to symmetrical azoarenes. Reaction conditions: azobenzene (0.2 mmol), [NO][BF4] (3 equiv), Pd(OAc)2 (3 mol%), TBAI (3 mol%), TsOH (6 mol%), DCB (1.5 mL), 90 °C, 48 h. Yields are isolated yields. iThe reaction was run at 100 °C for 24 h. iiThe ratio of product isomers was determined by NMR. iiiThe reaction was run in nitrobenzene (1.5 mL) at 100 °C for 24 h. iv0.6 mmol scale reaction was run at 80 °C for 24 h, with [NO][BF4] (2 equiv), Pd(OAc)2 (0.5 mol%), TBAI (0.5 mol%). vThe reaction was run in DCB (1.0 mL) and nitrobenzene (0.5 mL) at 90 °C for 48 h.
As shown in Fig. 5b, electronically unsymmetric azobenzenes containing an array of functional groups preferentially underwent reactions at more electron-rich benzene moiety with good-to-excellent yields obtained (3l–3s). For unsymmetric azobenzenes containing ortho-substituted benzene moieties, ortho-substituents led reactions to selectively occur at ortho-substituted benzene rings in good yields (3t–3z, 3aa–3ae). Ortho-chloro group, an electron-withdrawing group, favoured reaction at chloro-containing benzene moiety (3ab), which illustrated that the steric effect overrode the electronic effect in the control of reaction selectivity. The regioselectivity controlled by steric factor was also observed in 3,5-dimethyl azobenzene that favoured the reaction at less sterically hindered moiety with 18.5:1 selectivity (3ae).
In summary, an iodide-bridged binuclear palladium complex has been identified as an efficient catalyst to enhance both yield and rate of the nitrosylation/annulation reaction of azobenzenes with [NO][BF4]. The good performance of this binuclear catalyst arises from the bimetallic cooperation by which the strongly σ-donating η1 phenyl ligand on the “spectator” metal centre exerts a trans effect on the chelating product fragment on the catalytic metal centre through a bridging iodide ligand, and facilitates product release to re-generate catalytically active species. The trans effect-relay through bridging ligand within a binuclear complex represents a new bimetallic cooperation mode for catalysis and opens an avenue to design and develop multinuclear catalysts, especially for syntheses of the chelating products that often impede re-generation of active metal catalysts and therefore retard catalytic cycles.
Methods
General procedure for the reaction of azobenzene with [NO][BF4]
In a glove box, a 25 mL of the Schlenk tube equipped with a stir bar was charged with Pd(OAc)2 (3 mol%, 0.0014 g), TBAI (3 mol%, 0.0022 g), p-toluenesulfonic acid (6 mol%, 0.0021 g), azobenzene (0.2 mmol), [NO][BF4] (0.6 mmol, 0.0701 g) and DCB (1.5 mL). The tube was sealed and removed out of the glove box. The reaction mixture was stirred at 90 °C for 48 h. Upon completion, the reaction mixture was diluted with 10 mL of ethyl acetate, filtered through a pad of silica gel, followed by washing the pad of the silica gel with the ethyl acetate (20 mL). The filtrate was concentrated under reduced pressure. The residue was then purified by chromatography on silica gel to provide the corresponding product.
Optimization studies
See Supplementary Methods and Supplementary Table 1.
Identification of iodide-bridged binuclear palladium complex as a catalytically active species
See Supplementary Methods for details.
Kinetic experiments
See Supplementary Methods, Supplementary Tables 2–5 and Supplementary Figs. 1–5.
Determination of the order in 4b
See Supplementary Methods, Supplementary Tables 6–10 and Supplementary Figs. 6–11.
KIE experiments
See Supplementary Methods, Supplementary Table 11 and Supplementary Figs. 12–14.
Typical procedure for the preparation of substrates
See Supplementary Methods for details.
Computational studies
See Supplementary Methods, Supplementary Eq. 1 and Supplementary Figs. 15, 16. DFT computed Cartesian coordinates of important structures are provided in Supplementary Data 1.
Crystallography
The CIF files for compounds 3a, 4a, 4b and 5b are available in Supplementary Data 2–5, respectively. The check CIF/PLATON report file is available in Supplementary Data 6. Crystal data and structure refinement for compounds 3a, 4a, 4b and 5b are shown in Supplementary Tables 12–15.
NMR spectra
1H NMR13, C NMR and 19F NMR spectra of products, 4b and 5b. See Supplementary Figs. 17–103.
Data availability
All data generated and analysed during this study are included in the article and its Supplementary Information file, or from the corresponding authors on request. Crystallographic data have been deposited at the Cambridge Crystallographic Data Centre (CCDC) as CCDC 1428672 (3a), CCDC 1858247(4a), CCDC 1858251(4b) and CCDC 1858266 (5b), and can be obtained free of charge from the CCDC via http://www.ccdc.cam.ac.uk/structures.
References
Shibasaki, M., Sasai, H. & Arai, T. Asymmetric catalysis with heterobimetallic compounds. Angew. Chem. Int. Ed. 36, 1236–1256 (1997).
van den Beuken, E. K. & Feringa, B. L. Bimetallic catalysis by late transition metal complexes. Tetrahedron 54, 12985–13011 (1998).
Wheatley, N. & Kalck, P. Structure and reactivity of early-late heterobimetallic complexes. Chem. Rev. 99, 3379–3419 (1999).
Delferro, M. & Marks, T. J. Multinuclear olefin polymerization catalysts. Chem. Rev. 111, 2450–2485 (2011).
Park, J. & Hong, S. Cooperative bimetallic catalysis in asymmetric transformations. Chem. Soc. Rev. 41, 6931–6943 (2012).
Kornecki, K. P., Berry, J. F., Powers, D. C. & Ritter, T. Metal-metal bond-containing complexes as catalysts for C-H functionalization. in Progress in Inorganic Chemistry, Vol 58 (ed Karlin, K. D.) (John Wiley & Sons Inc, Hoboken, 2014) pp. 225–302.
Buchwalter, P., Rose, J. & Braunstein, P. Multimetallic catalysis based on heterometallic complexes and clusters. Chem. Rev. 115, 28–126 (2015).
Serrano-Plana, J., Garcia-Bosch, I., Company, A. & Costas, M. Structural and reactivity models for copper oxygenases: cooperative effects and novel reactivities. Acc. Chem. Res. 48, 2397–2406 (2015).
Powers, I. G. & Uyeda, C. Metal-metal bonds in catalysis. ACS Catal. 7, 936–958 (2017).
Mankad, N. P. Diverse bimetallic mechanisms emerging from transition metal Lewis acid/base pairs: development of co-catalysis with metal carbenes and metal carbonyl anions. Chem. Commun. 54, 1291–1302 (2018).
Ammal, S. C., Yoshikai, N., Inada, Y., Nishibayashi, Y. & Nakamura, E. Synergistic dimetallic effects in propargylic substitution reaction catalyzed by thiolate-bridged diruthenium complex. J. Am. Chem. Soc. 127, 9428–9438 (2005).
Choy, S. W. S., Page, M. J., Bhadbhade, M. & Messerle, B. A. Cooperative catalysis: large rate enhancements with bimetallic rhodium complexes. Organometallics 32, 4726–4729 (2013).
McInnis, J. P., Delferro, M. & Marks, T. J. Multinuclear group 4 catalysis: olefin polymerization pathways modified by strong metal-metal cooperative effects. Acc. Chem. Res. 47, 2545–2557 (2014).
Shibasaki, M., Kanai, M., Matsunaga, S. & Kumagai, N. Recent progress in asymmetric bifunctional catalysis using multimetallic systems. Acc. Chem. Res. 42, 1117–1127 (2009).
Bonney, K. J., Proutiere, F. & Schoenebeck, F. Dinuclear Pd(I) complexes-solely precatalysts? Demonstration of direct reactivity of a Pd(I) dimer with an aryl iodide. Chem. Sci. 4, 4434–4439 (2013).
Aufiero, M., Sperger, T., Tsang, A. S. K. & Schoenebeck, F. Highly efficient C-SeCF3 coupling of aryl iodides enabled by an air-stable dinuclear Pd-I catalyst. Angew. Chem. Int. Ed. 54, 10322–10326 (2015).
Powers, D. C. & Ritter, T. Bimetallic Pd(III) complexes in palladium-catalysed carbon-heteroatom bond formation. Nat. Chem. 1, 302–309 (2009).
Deprez, N. R. & Sanford, M. S. Synthetic and mechanistic studies of Pd-catalyzed C-H arylation with diaryliodonium salts: evidence for a bimetallic high oxidation state Pd intermediate. J. Am. Chem. Soc. 131, 11234–11241 (2009).
Powers, D. C., Xiao, D. Y., Geibel, M. A. L. & Ritter, T. On the mechanism of palladium-catalyzed aromatic C-H oxidation. J. Am. Chem. Soc. 132, 14530–14536 (2010).
Nakamura, E., Yoshikai, N. & Yamanaka, M. Mechanism of C-H bond activation/C-C bond formation reaction between diazo compound and alkane catalyzed by dirhodium tetracarboxylate. J. Am. Chem. Soc. 124, 7181–7192 (2002).
Doyle, M. P., Duffy, R., Ratnikov, M. & Zhou, L. Catalytic carbene insertion into C-H Bonds. Chem. Rev. 110, 704–724 (2010).
Davies, H. M. L. & Lian, Y. J. The combined C-H functionalization/cope rearrangement: discovery and applications in organic synthesis. Acc. Chem. Res. 45, 923–935 (2012).
Roizen, J. L., Harvey, M. E. & Du Bois, J. Metal-catalyzed nitrogen-atom transfer methods for the oxidation of aliphatic C-H bonds. Acc. Chem. Res. 45, 911–922 (2012).
Broussard, M. E. et al. A bimetallic hydroformylation catalyst: high regioselectivity and reactivity through homobimetallic cooperativity. Science 260, 1784–1788 (1993).
Mazzacano, T. J. & Mankad, N. P. Base metal catalysts for photochemical C-H borylation that utilize metal-metal cooperativity. J. Am. Chem. Soc. 135, 17258–17261 (2013).
Nishibayashi, Y., Wakiji, I. & Hidai, M. Novel propargylic substitution reactions catalyzed by thiolate-bridged diruthenium complexes via allenylidene intermediates. J. Am. Chem. Soc. 122, 11019–11020 (2000).
Hostetler, M. J. & Bergman, R. G. Synthesis and reactivity of Cp2Ta(CH2)2Ir(CO)2: an early-late heterobimetallic complex that catalytically hydrogenates, isomerizes and hydrosilates alkenes. J. Am. Chem. Soc. 112, 8621–8623 (1990).
Rudd, P. A., Liu, S. S., Gagliardi, L., Young, V. G. & Lu, C. C. Metal-alane adducts with zero-valent nickel, cobalt, and iron. J. Am. Chem. Soc. 133, 20724–20727 (2011).
Baxter, R. J., Knox, G. R., Pauson, P. L. & Spicer, M. D. Synthesis of dicarbonyl(eta(4)-tricarbonylcobaltacyclopentadiene)cobalt complexes from Co-2(CO)(8). A general route to intermediates in cobalt carbonyl mediated alkyne trimerization. Organometallics 18, 197–205 (1999).
Yamanaka, M. & Nakamura, E. Density functional studies on the Pauson-Khand reaction. J. Am. Chem. Soc. 123, 1703–1708 (2001).
Pal, S. & Uyeda, C. Evaluating the effect of catalyst nuclearity in Ni-catalyzed alkyne cyclotrimerizations. J. Am. Chem. Soc. 137, 8042–8045 (2015).
Steiman, T. J. & Uyeda, C. Reversible substrate activation and catalysis at an intact metal-metal bond using a redox-active supporting ligand. J. Am. Chem. Soc. 137, 6104–6110 (2015).
Coe, B. J. & Glenwright, S. J. Trans-effects in octahedral transition metal complexes. Coord. Chem. Rev. 203, 5–80 (2000).
Ye, M. C., Gao, G. L. & Yu, J. Q. Ligand-promoted C-3 selective C-H olefination of pyridines with Pd catalysts. J. Am. Chem. Soc. 133, 6964–6967 (2011).
Kwak, J., Ohk, Y., Jung, Y. & Chang, S. Rollover cyclometalation pathway in rhodium catalysis: dramatic NHC effects in the C-H bond functionalization. J. Am. Chem. Soc. 134, 17778–17788 (2012).
Molander, G. A. & Cavalcanti, L. N. Nitrosation of Aryl and heteroaryltrifluoroborates with nitrosonium tetrafluoroborate. J. Org. Chem. 77, 4402–4413 (2012).
Bosch, E. & Kochi, J. K. Direct nitrosation of aromatic hydrocarbons and ethers with the electrophilic nitrosonium cation. J. Org. Chem. 59, 5573–5586 (1994).
Nicholas, G. M., Blunt, J. W. & Munro, M. H. G. Cortamidine oxide, a novel disulfide metabolite from the New Zealand basidiomycete (mushroom) Cortinarius species. J. Nat. Prod. 64, 341–344 (2001).
De Moor, O. et al. Discovery and SAR of 2-arylbenzotriazoles and 2-arylindazoles as potential treatments for Duchenne muscular dystrophy. Bioorg. Med. Chem. Lett. 21, 4828–4831 (2011).
Kuila, D. et al. Tris(hydroxyphenyl)ethane benzotriazole: a copolymerizable UV light stabilizer. Chem. Mater. 11, 109–116 (1999).
Lebrasseur, N. & Larrosa, I. Room temperature and phosphine free palladium catalyzed direct C-2 arylation of indoles. J. Am. Chem. Soc. 130, 2926–2927 (2008).
Daugulis, O., Do, H. Q. & Shabashov, D. Palladium- and copper-catalyzed arylation of carbon-hydrogen bonds. Acc. Chem. Res. 42, 1074–1086 (2009).
Phan, N. T. S., Van Der Sluys, M. & Jones, C. W. On the nature of the active species in palladium catalyzed Mizoroki-Heck and Suzuki-Miyaura couplings—homogeneous or heterogeneous catalysis, a critical review. Adv. Synth. Catal. 348, 609–679 (2006).
Ye, Y. D., Ball, N. D., Kampf, J. W. & Sanford, M. S. Oxidation of a cyclometalated Pd(II) dimer with “CF3+“: formation and reactivity of a catalytically competent monomeric Pd(IV) aquo complex. J. Am. Chem. Soc. 132, 14682–14687 (2010).
Wang, X. S., Truesdale, L. & Yu, J. Q. Pd(II)-catalyzed ortho-trifluoromethylation of arenes using TFA as a promoter. J. Am. Chem. Soc. 132, 3648–3649 (2010).
Crispini, A., Ghedini, M. & Neve, F. Cyclopalladated compounds. Structural studies on dinuclear azobenzene complexes. J. Organomet. Chem. 448, 241–245 (1993).
Chan, S., Lee, S.-M., Lin, Z. & Wong, W.-T. Syntheses, structures and reactivities of [Os6Pd(CO)18(bipy)] and [{(bipy)Pd}2Os3(CO)12]: crystal and molecular structures of [{(bipy)Pd}2(μ-H)(μ-CO)][H3Os4(CO)12] and [(C4H9)4N]2[Pd2I6]. J. Organomet. Chem. 510, 219–231 (1996).
Thompson, J. M. & Heck, R. F. Carbonylation reactions of ortho-palladation products of. alpha.-arylnitrogen derivatives. J. Org. Chem. 40, 2667–2674 (1975).
Giri, R., Chen, X. & Yu, J. Q. Palladium-catalyzed asymmetric iodination of unactivated C-H bonds under mild conditions. Angew. Chem. Int. Ed. 44, 2112–2115 (2005).
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2017YFA0206801), National Natural Science Foundation of China Grants (21602221, 21931011, 21431008, 21973094), the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB20000000), the Key Research Program of Frontier Sciences of the Chinese Academy of Sciences (QYZDJ-SSW-SLH024) and the Natural Science Foundation of Fujian Province (2019J01131).
Author information
Authors and Affiliations
Contributions
Y.Z. and W.S. conceived and designed the project. Y.Z., X.Z. and X.D. performed experiments and analysed the data. Z.-N.C. and W.Z. performed computational studies. W.S., Y.Z., Z.-N.C. and W.Z. wrote and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, Y., Chen, ZN., Zhang, X. et al. Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex. Commun Chem 3, 41 (2020). https://doi.org/10.1038/s42004-020-0287-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42004-020-0287-0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.