Abstract
Dynamic nuclear polarization (DNP) coupled with 15N magnetic resonance imaging (MRI) provides an opportunity to image quantitative levels of biologically important metal ions such as Zn2+, Mg2+ or Ca2+ using appropriately designed 15N enriched probes. For example, a Zn-specific probe could prove particularly valuable for imaging the tissue distribution of freely available Zn2+ ions, an important known metal ion biomarker in the pancreas, in prostate cancer, and in several neurodegenerative diseases. In the present study, we prepare the cell-permeable, 15N-enriched, d6-deuterated version of the well-known Zn2+ chelator, tris(2-pyridylmethyl)amine (TPA) and demonstrate that the polarized ligand had favorable T1 and linewidth characteristics for 15N MRI. Examples of how polarized TPA can be used to quantify freely available Zn2+ in homogenized human prostate tissue and intact cells are presented.
Similar content being viewed by others
Introduction
Nitrogen is one of the four most abundant elements in the human body yet there are very few spectroscopic methods available to monitor the various forms of nitrogen in biological molecules. Nitrogen-15 (0.37% natural abundance) has a favorable nuclear spin of ½ and a wide chemical shift range (900 ppm), yet 15N NMR is much less widely used as a tool in biological systems compared to 13C and 1H because of its poor sensitivity1. However, 15N NMR in combination with hyperpolarization techniques such as dissolution dynamic nuclear polarization (d-DNP) or parahydrogen induced polarization (PHIP) is beginning to find its way into the biological realm2. DNP refers to technologies that enhance the NMR signal-to-noise ratio by amplifying nuclear spin polarization via microwave driven transfer of high electron spin polarization to coupled nuclear spins at low temperatures and high magnetic fields3. The frozen HP sample is then rapidly dissolved with a superheated solvent to produce a solution of the hyperpolarized compound. The hyperpolarized spin state is not persistent and decays to thermodynamic equilibrium by spin-lattice (longitudinal) relaxation. Hence, unlike in conventional NMR experiments, exceedingly long T1 values are advantageous for HP studies. The sensitivity improvements offered by HP makes it feasible to perform molecular/functional MR imaging of nuclei other than 1H.
The most widely studied NMR-active nucleus for hyperpolarized metabolic imaging is 13C. HP-13C-labeled tracers not only allow one to detect biological products from a starting HP substrate but also provide the opportunity to perform dynamic measurements to determine flux through specific enzyme catalyzed reactions in vivo by 13C MRS/MRI4,5,6,7. The 15N nucleus has several favorable properties for HP studies including narrow linewidths, relatively long T1 values, and a much larger chemical shift range compared to 13C. HP-15N-labeled compounds are particularly well suited for creating newer types of sensors that can potentially offer molecular information with high spectral resolution, low background interference, and minimal invasiveness. Successful application of hyperpolarized 15N probes in biological systems requires 15N-labeled compounds that have long spin-lattice (T1) relaxation time and large chemical shift dispersion in response to changes in physiological parameters such as pH, redox, and concentration of reactive oxygen species or biologically relevant metal ions8,9.
Diamagnetic Zn2+ is an important target for imaging because these ions participate in various biochemical processes such as enzyme catalysis, neurotransmission, intracellular signaling, and antibiotic activity. It has been shown that cellular Zn2+ plays an important role in the progression of diabetes and prostate cancer as well as in the development of various neurodegenerative disorders10,11. In the pancreas, Zn2+ is essential for proper storage of insulin β-cell granules12. In the brain, Zn2+ is released along with glutamate from presynaptic vesicles via calcium-dependent exocytosis. Healthy prostate tissue stores and secretes large quantities of Zn2+ ions while malignant cells have reduced levels of Zn2+13,14,15,16,17,18. For this reason alone, quantitative imaging of Zn2+ in vivo could be a very useful diagnostic tool for the detection of malignant tissues. Although there are many cell-based optical methods for monitoring intracellular and extracellular Zn2+, nondestructive detection of Zn2+ levels in vivo remains a major challenge. Optical methods have potential for quantitative in vivo detection of Zn2+ but the limited penetration depth due to attenuation and scattering of light in tissues is a major obstacle19. Two recent MRI studies have demonstrated that a gadolinium-based Zn-responsive contrast agent can be used to monitor Zn2+ secretion from pancreatic islets20,21 and prostate in mice22. In both tissues, secretion of Zn2+ from intracellular stores into the extracellular space was initiated after a bolus injection of glucose. Given that the Zn2+ content of prostate tissue is known to be much lower in prostate cancer22, this technology has the potential to differentiate between healthy versus malignant tissue in a simple MRI exam in combination with a Zn2+-sensitive contrast agent. However, it should be pointed out that this test does not detect total Zn2+ content of prostate tissue but rather only the Zn2+ ions released in response to glucose. Hence, an imaging method that provided a direct measure of total Zn2+ content could perhaps be even more informative. The detection limit of a typical Gd-based MRI contrast agent is also limited to around 50 μM so this could become a limiting factor in some tissues11.
There have been previous reports of HP-based sensors for detection of metal ions including 13C-EDTA23 and 129Xe-based Zn2+ sensors24. However, these do have limitations for in vivo use such as significant line broadening of the ligand 13C signals after binding to Zn2+23 and the lack of sufficient chemical shift differences between the Zn2+, Mg2+, and Ca2+ complexes24. A recent study demonstrated that HP [1-13C]cysteine can detect Zn2+ in biological samples at physiological concentrations25. In that system, the 13C resonance of [1-13C]cysteine shifted downfield and broadened upon addition of Zn2+ so quantitative detection of Zn2+ could be problematical because the signal position is not only dependent upon freely available Zn2+ levels but also the sensor concentration as well. It was also not clear in these previous studies whether these reporter systems would report total tissue Zn2+ or specifically extracellular Zn2+.
Here we report a HP-15N based probe, tris(2-pyridylmethyl)amine (TPA) (Fig. 1) for the detection and quantification of total freely available Zn2+ in biological samples. TPA is a tripodal ligand that has excellent selectivity for Zn2+ over other common biological cations. Most optical and Gd-based Zn2+ responsive agents contain a sensing moiety structurally derived from TPA22,26,27. This compound is known to distribute across cell membranes27 so in principle should detect freely available tissue Zn2+ in all compartments and not just Zn2+ ions released from cells in response to a biological stimulus22.
Results
15N-enriched TPA design
To evaluate TPA as a prototype Zn2+ imaging sensor, we first performed NMR experiments on HP TPA containing natural abundance level of 15N. There are two types of nitrogen atoms in TPA, the pyridine N-atoms and the central aliphatic tertiary N-atom. 15N NMR spectra showed that there is a substantial chemical shift difference in both the pyridine (50 ppm upfield) and the central 15N (20 ppm upfield) signals after addition of Zn2+ ions (Supplementary Fig. 1). However, the pyridine 15N signals relaxed more rapidly (the T1 was not measured) than the central 15N signal so, based on these observations, the tertiary nitrogen was chosen as the optimal site for 15N enrichment.
The 15N-labeled version of TPA was synthesized starting from 15N-labeled ammonium acetate and 2-chloromethyl pyridine or 1-(chloromethyl-d2) pyridine as outlined in Fig. 2 with an overall yield of 25% and 61%, respectively. As nearby protons can provide an efficient relaxation mechanism for 15N by dipole–dipole interaction, we also prepared a 15N-TPA derivative in which deuterium was substituted for each proton in the proximity of the 15N nucleus (Fig. 2). This modification is expected to significantly increase the 15N T1 relaxation time.
Dynamic nuclear polarization of the 15N probes
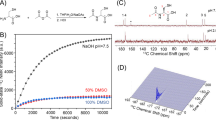
The compounds polarized well in a HyperSense polarizer using standard DNP conditions with OX063 trityl radical as polarizing agent to a level of 8 ± 2% (23,795 ± 6950) and 6 ± 2% (18,164 ± 6237) polarization for [15N]TPA and [15N]TPA-d6, respectively, after dissolution in PBS buffer (Supplementary Fig. 2). Both ligands showed an identical 20 ppm upfield shift in their 15N resonances upon addition of Zn2+ (Fig. 3a and Supplementary Fig. 2). The T1 of the non-deuterated [15N]-TPA and Zn2+-[15N]TPA species were 25.9 ± 1.6 s and 17.9 ± 0.1 s28,29, respectively (Fig. 3b). However, as expected, deuteration of the methylene groups resulted in a substantially prolonged T1values for [15N]TPA-d6 and Zn2+-[15N]TPA-d6: 70.9 ± 1.1 (p < 0.05 compared to T1 values of [15N]TPA) and 57.0 ± 2.3 (p < 0.05 compared to T1 values of Zn2+-[15N]TPA) at 9.4 T, respectively. The detection limits of these polarized samples were evaluated by varying the [Zn2+] in samples of HP-[15N]TPA-d6 and observing the 15N signals in buffered solutions. These experiments showed that the detection threshold of Zn2+ using HP-[15N]TPA-d6 was ~5 μM (Fig. 3c).
a 15N NMR chemical shift of [15N]TPA-d6 in the absence and presence of Zn2+ (0.25 eq). b Sequential [15N]TPA and [15N]TPA-d6 spectrum decay and T1 relaxation of free ligand a) and Zn2+ complexes b) measured at 9.4 T, 298 K, pH 6.8. c Single scan of 15N NMR spectra of HP [15N]TPA-d6 (1.2 mM) with various concentration of Zn2+.
MRSI of HP 15N-TPA-d6
In a proof of principle MR imaging experiment, 15N CSI of phantoms containing HP-[15N]TPA-d6 in the absence and presence of Zn2+ was collected to evaluated the feasibility of spectroscopic imaging of Zn2+ (Fig. 4). HP-[15N]TPA-d6, polarized for 2 h, was injected into three NMR tubes containing different concentrations of [Zn2+] (Fig. 4a). Axial 15N chemical shift images of these tubes were acquired using a standard CSI sequence at 9.4 T. The 15N resonances of the free and Zn-bound [15N]TPA-d6 were observed at 40 ppm and 20 ppm reflecting [15N]TPA-d6 (Fig. 4b, c) and Zn2+-[15N]TPA-d6 (Fig. 4b, d), respectively. The 15N data were reconstructed and analyzed using MATLAB (Mathworks, Natick MA, USA).
HP-[15N]TPA-d6 to measure freely available Zn2+ in biological sample
We next tested the feasibility of using 15N-NMR and [15N]TPA-d6 to quantify Zn2+ levels in tissue and intact cell samples. First, known amounts of HP-[15N]TPA-d6 (1 mM) were added to HEPES buffered samples containing physiologically relevant concentration of Zn2+ ranging from 0 to 200 μM and 15N spectra were recorded using a single 45 degree pulse. Given that the total concentration of [15N]TPA-d6 was known, the area of the Zn2+-[15N]TPA-d6 signal relative to the total 15N signal provides a direct readout of total Zn2+ (Supplementary Eq. 1). Figure 5a shows there is an excellent correlation between [Zn2+] as measured by HP-15N NMR versus the analytically determined Zn2+ levels (R2 = 0.99). To illustrate the feasibility of using [15N]TPA-d6 as a sensor of free tissue Zn2+, an HP sample of [15N]TPA-d6 was mixed with a fresh surgically resected human prostate tissue homogenate. The tissue was homogenized as described in the Methods section to facilitate rapid mixing prior to the addition of freshly prepared HP-[15N]TPA-d6 (final concentration of 4.2 mM). Sequential 15N NMR spectra were then recorded every 2 s to monitor decay of the HP signals (Supplementary Fig. 4). Figure 5b shows the first 15N NMR spectrum collected within seconds after mixing HP-[15N]TPA-d6 with the tissue sample demonstrating that the ligand is in excess with respect to the total amount of Zn2+ present in the sample. Quantification of Zn2+ as described in the Supplementary Methods indicated that the total concentration of freely available Zn2+ in this particular tissue sample was 66 μM. This value is in good agreement with the Zn concentration (71 μM) measured by ICP-MS. It should be noted, however, that homogenization destroys all cellular compartments and thus, this experiment does not demonstrate that HP-[15N]TPA-d6 can be used to measure intracellular Zn2+. To examine whether HP-[15N]TPA-d6 can detect the free Zn2+ in intact cell, we performed HP experiments in which HP-[15N]TPA-d6 was added to Zn2+ spiked human prostate epithelial (PNT1A) cells after the extracellular zinc was washed away30. Note that spiking is necessary to achieve Zn levels in cultured cells that mimic in vivo situation. Figure 5c, d shows the time-dependent 15N spectra and the first spectrum, respectively, after HP-[15N]TPA-d6 was mixed with intact PNT1A cells. Based on the area of the Zn2+-[15N]TPA-d6 signal relative to the total 15N signal and the known total concentration of [15N]TPA-d6 (2.8 mM), the Zn2+ concentration in these samples were calculated to be 118 and 123 μM (n = 2) (Supplementary Methods). These values agree well with the result of ICP-MS analysis (151 and 146 μM, respectively).
a A plot of [Zn2+] as determined by HP versus the analytically known [Zn2+] in each sample. b–d quantitative detection of [Zn2+] in a surgical specimen of human benign prostatic hyperplasia (BPH) tissue and human prostate epithelial PNT1A cells. Spectra of free ligand (40 ppm) and Zn2+ complexes (20 ppm) were recorded every 2 s using 30° and 45° flip angle for BPH tissue and PNT1A cells. b the first 15N spectrum collected after dissolution and mixing on a 500 mg sample of BPH tissue after adding 4.2 mM HP-[15N]TPA-d6. (9.4 T, 298 K and pH 7.0). c Time-dependent 15N spectra collected on a sample of PNT1A cells (~65 × 106 cells) after adding 2.8 mM HP TPA-d6 and d the first 15N spectrum. (9.4 T, 298 K and pH 7.4).
Discussion
In this study, we demonstrate that 15N-labeled TPA can be used as hyperpolarized magnetic resonance Zn2+ probe to detect the freely available Zn2+. In general, hyperpolarized chemical probes are designed as a combination of sensing and signaling moieties8. In this case, the signaling part is the HP-15N-enriched nucleus and the sensing part consists of a tetradentate tripodal ligand structure that ensures high selectivity for the analyte of interest, Zn2+. We have chosen tris(2-pyridylmethyl) amine (TPA) for Zn-sensing moiety because it forms stable complexes with Zn2+ and displays excellent selectivity over most endogenous metal ions. A deuterated version of [15N]TPA was also synthesized to prolong the life-time of the hyperpolarized 15N reporter atom. While the chemistry involved in the synthesis is fairly straightforward, special attention was given in the synthesis design to finding suitable starting materials for the introduction of the 15N and 2H label and selecting optimal reaction conditions that give reasonable yields in each step to maximize the overall yield of the final, 15N-enriched and deuterated product. 15N-labeled phthalimide, conveniently synthesized by melting together 15N ammonium acetate and phthalimide, satisfied these requirements to afford the final product [15N]TPA and [15N]TPA-d6 in reasonable overall yield.
It is well-known that all four nitrogen donor atoms of TPA coordinate to Zn2+ and this results in an upfield shift of the 15N chemical resonance26. The fact that separate signals were observed for the free ligand and the Zn2+ complex throughout a TPA/Zn2+ titration shows that exchange between the two species is slow on the NMR timescale (Fig. 3a). This is an advantage over HP-[1-13C]cysteine for detection of Zn2+ because the ratio of the bound versus unbound resonance areas provide a direct readout of total Zn2+(Fig. 3a). Like [1-13C]cysteine, [15N]TPA is also selective for Zn2+, but unlike cysteine, its pKa values are below 7, which eliminates interference from protonation of the free unbound ligand. In addition, while 15N has lower NMR sensitivity compared to 13C because of its lower gyromagnetic ratio, 15N typically displays much longer T1 values and sharper NMR resonances than 13C. Moreover, deuterated version of [15N]TPA further elongated the 15N T1. In general, the dominant mechanism for spin-lattice relaxation of 15N includes both dipolar and chemical shift anisotropy (CSA) contributions. The major source of dipolar relaxation is the dipole–dipole interaction of 15N with 1H spins present in the molecule. The dipolar relaxation rate (Rd) is independent of the B0 field strength (\(R_d\sim \frac{{\gamma _N^2X\gamma _{1H}^2}}{{r^6}}\), where γ is gyromagnetic ratio and r is the distance between the 15N nucleus and the dipolarly coupled 1H), while the CSA relaxation rate (RCSA) increases with B0 (\(R_{CSA}\sim \gamma _N^2{\Delta}\sigma ^2B_0^2\), where B0 is the magnetic field and Δσ is the magnetic shielding anisotropy)31,32,33,34,35,36. The Rd can be minimized by substituting 2H (deuterium), which has much lower γ than 1H, for all nearby protons33,37 The contribution of RCSA can be reduced by increasing the molecular symmetry and/or performing the HP experiments at low field38. The significant increase in T1 upon deuteration indicated that the dominant relaxation mechanism for 15N-TPA was dipolar relaxation. Measurements of the 15N T1 at other field strengths (1, 3, and 9.4 T) yielded similar T1 values confirming that the chemical shift anisotropy contribution was indeed negligible as expected for a such a symmetrical molecule (TPA and related tripodal ligands have a C3 symmetry). (Supplementary Table 1)39.
Chemical shift imaging (CSI), widely used in HP-13C studies, provides both spectral and spatial information40. Here, 15N CSI experiments were performed on phantoms in a vertical bore high-resolution spectroscopy magnet equipped with gradients. [15N]TPA-d6 and its Zn2+-complex were easily distinguished with good spatial resolution and sensitivity due to the large, 20 ppm 15N NMR chemical shift separation between the free ligand and the Zn complex. This result clearly demonstrates that HP-[15N]TPA-d6 has the potential to map the distribution of freely available Zn2+ (defined as all Zn2+ except that tightly bound to metalloproteins). We also demonstrated that the Zn2+ concentration in phantoms can be assessed using the integrals of the free and Zn-bound [15N]TPA-d6 signals. In principle, protonation of TPA would alter the effective binding constant for Zn2+ (the conditional stability of the complex) but, fortunately, the pKa values of TPA are 2.55, 4.35, 6.1741, so the ligand is largely deprotonated at pH 7.4 and little proton competition exists. This was also evident in 15N NMR spectra of TPA, whose chemical shift did not change between pH 6.4 and 8.0. The TPA binding unit is commonly used in optical probe designs because of its high affinity and specificity for Zn2+ (log KZnTPA = −11.0)26,27,42.
HP studies confirmed that there is no interference from Ca2+ ions (2 mM) at biologically relevant pH values (Supplementary Fig. 3a). In addition, a 1:1 binding stoichiometry was confirmed by thermal 15N NMR spectroscopy (Supplementary Fig. 3b) and a detection limit of 1~5 μM Zn2+ (Fig. 3c) is well-below the intracellular concentration of Zn2+ in secretory cells (μM to mM)14,43,44. The feasibility of using the [15N]TPA-d6 as a Zn2+ sensor in biological samples was demonstrated using freshly isolated human benign prostate hyperplasia (BPH) homogenates prepared from biopsy samples as well as human prostate epithelial (PNT1A) cells. ICP-MS measurements of the Zn concentration verified that [15N]TPA-d6 could accurately report Zn2+ levels in these biological samples. The successful Zn2+ detection in intact PNT1A cells also demonstrated that the cellular uptake of HP-[15N]TPA-d6, a known membrane permeable Zn chelator27, fast compared to HP-[15N] signal decay.
In general, hyperpolarized 15N for in vivo imaging has remained largely unexplored at this point primarily due to the technical challenges associated with 15N. First, conventional preclinical and clinical MR scanners are typically not equipped for imaging low gamma nuclei. Hyperpolarization of exogenous 15N-labeled substrates overcomes the MR sensitivity problem, but other scanner-associated requirements in hardware (RF coils and frequency synthesizer) and software remain for 15N imaging. Moreover, the low Larmor frequency requires stronger gradient coil performance to achieve sharp localization (e.g., slice selection) and high-resolution image acquisition.
In conclusion, we have developed hyperpolarized 15N-labeled TPA derivatives for use as an imaging sensor of freely available Zn2+in a biological sample. The tertiary 15N atom in deuterated TPA was found to have favorable properties including a long T1 value and a sharp 15N resonance that displays a large chemical shift difference upon complexation of Zn2+. It was shown that HP-[15N]TPA-d6 can detect Zn2+ in the low μM range with no interference from protons or other endogenous metal ions. The probe was successfully used to quantify free Zn levels in human prostate tissue homogenate and intact human prostate epithelial cells. Since total Zn2+ in the prostate is known to drop dramatically in malignant tissue, the current results demonstrate that the use of HP-[15N]TPA-d6 to measure freely available Zn2+ in prostate tissues in vivo during progression of prostate cancer would likely be diagnostically informative. The cytotoxicity of TPA due to its strong binding to Zn2+, which can lead to zinc depletion, could limit its application for in vivo studies27. This, however, could be ameliorated by the design of second generation agents with weaker Zn-binding constant.
Methods
General remarks
All reagents and solvents were purchased from commercial sources and used as received without further purification unless stated otherwise. Zinc analyses (ICP-MS) were performed by Galbraith Laboratories, Inc. 1H, 13C, and 15N NMR spectra were recorded using a 9.4 T Varian/Agilent VNMRS400 NMR spectrometer operating at 400, 100, and 40 MHz, respectively. LCMS experiments were carried out on a Waters Alliance LC system with a Atlantis T3 column, (C18, 5 μ, 250 × 4.6 mm), connected to a Waters diode array UV detector, and an electrospray ionization mass spectrometer (ESI MS) using a Waters Qtof-MS-XEVO ESI positive mode for detection.
General procedures for dynamic nuclear polarization (DNP) NMR studies
DNP was performed using a HyperSense commercial polarizer (3.35 T, Oxford Instruments Molecular Biotools, UK). The TPA ligands were dissolved in glycerol-water matrix (50:50 w/w) containing 2 mM Gd chelate (ProHance®) and 15 mM trityl radical polarizing agent (OX063, GE Healthcare, UK). The final concentrations of the substrates in the glassing matrix was ~0.5 M. Sample volumes for DNP varied from 18 µL to 50 µL The polarization was performed at 1.05 K with 94.112 GHz microwave irradiation for 2 h and the frozen polarized samples were subsequently rapidly dissolved in a phosphate buffered solution (10 mM phosphate, pH~7) or HEPES buffered solution in a 10-mm NMR tube. 15N NMR spectra were acquired with a 9.4 T Varian vertical bore microimager (Varian, USA) using flip angle of 5° to 45° depending on the experiment with repetition time (TR) of 2 s. All 15N chemical shifts were externally referenced to [15N]ammonium chloride (0 ppm). The 15N spectra were processed using ACD/SpecManager (ACD Labs, Canada). The binding stoichiometry experiment was performed using thermal 15N NMR spectrometer (45° flip angle with repetition time (TR) of 250 s. For chemical structure characterization, 1H, 13C, and 15N NMR spectra were recorded on Varian VNMRS direct drive console spectrometer operating at 400, 100, and 40 MHz, respectively. The 15N spin-lattice relaxation times (T1) were measured by following the decay of the HP-15N magnetization over time by applying a small flip angle pulse. The T1 values were calculated using the following equation.
Where, I(t) is the signal intensity at time t, I0 is the initial magnetization, TR is repetition time, and θ is the flip angle of radiofrequency (rf) pulse used to monitor the hyperpolarization signal.
Synthesis of 15N-labeled tris(2-pyridylmethyl)amine, [15N]TPA
[15N]Phthalimide (1): [15N]Ammonium acetate (6.5 mmol) was mixed with phthalic anhydride (5.4 mmol) and stirred for 4 h at 160 °C. After the reaction mixture cooled to room temperature, the white solid was washed with cold water, filtered, and dried in vacuum to afford [15N]phthalimide as white powder (0.73 g, 91% yield). 1H NMR (DMSO-d6, 400 MHz) δ (ppm) = 11.27 (1H, br s, NH), 7.81 (4H, s, phthalic); 13C NMR (DMSO-d6, 100 MHz) δ (ppm) = 169.21 (d, 1JCN = 13 Hz, C=O), 134.28, 132.56 (d, 2JCN = 7 Hz, CC=O), 122.91; 15N NMR (DMSO-d6, 40 MHz) δ = 153.51; LCMS(ESI): m/z calc. for [M + H]+ = 149.0369, found [M + H]+ = 149.0351.
[15N](Pyridine-2-ylmethyl)phthalimide (2)45,46: [15N]Phthalimide (645 mg, 4.3 mmol) was dissolved in dimethylformamide (10 mL) and potassium carbonate (1.5 g, 10.9 mmol) and 2-(chloromethyl)pyridine hydrochloride (749 mg, 4.6 mmol) were added. The reaction mixture was stirred for 12 h at 50 °C. After removal of the solvent by rotary evaporation, water was added to precipitate the product and to remove inorganic salts. The crude product was filtered and dried in vacuum to afford [15N](pyridine-2-ylmethyl)phthalimide as a white solid (1.03 g, 98% yield). 1H NMR (DMSO-d6, 400 MHz) δ (ppm) = 8.43 (1H, d, J = 4 Hz, pyridyl), 7.90 (2H, s, phthalic), 7.87 (2H, s, phthalic), 7.76 (1H, t, J = 8 Hz, pyridyl), 7.40 (1H, d, J = 8 Hz, pyridyl), 7.26 (1H, t, J = 4 Hz, pyridyl), 4.91 (2H, s, NCH2); 13C NMR (DMSO-d6, 100 MHz) δ = 166.78 (d, 1JCN = 13 Hz, C=O), 155.11, 149.07, 136.88, 134.56, 131.69 (d, 2JCN = 8 Hz, CC=O), 123.23, 122.52, 121.28, 42.11 (d, 2JCN = 10 Hz, CH215N-); 15N NMR (DMSO-d6, 40 MHz) δ = 154.45; LCMS(ESI): m/z calc. for [M + H]+ = 240.0791, found [M + H]+ = 240.0600.
[15N]-2-Aminomethyl pyridine (3): [15N](Pyridine-2-ylmethyl)phthalimide (850 mg, 3.6 mmol) was dissolved in 10% hydrochloric acid (30 mL) and the solution was stirred for 12 h at 80 °C. The hydrochloric acid was removed by rotary evaporation and the residue was dissolved in 20% sodium hydroxide (5 mL) and extracted with dichloromethane (50 mL). The organic layer was dried over anhydrous sodium sulfate, filtered, and concentrated by rotary evaporation. The residue was dried in vacuum to afford [15N]-2-aminomethyl pyridine as a yellowish oil (349 mg, 89% yield). 1H NMR (CDCl3, 400 MHz) δ (ppm) = 8.52 (1H, d, J = 4 Hz, pyridyl), 7.61 (1H, t, J = 8 Hz, pyridyl), 7.24 (1H, d, J = 8 Hz, pyridyl), 7.12 (1H,t, J = 4 Hz, pyridyl), 3.95 (2H, s, NCH2); 13C NMR (CDCl3, 100 MHz) δ (ppm) = 161.67, 149.32, 136.62, 121.90, 121.31, 47.70 (d, 1JCN = 3 Hz, C-15NH2); 15N NMR (CDCl3, 40 MHz) δ (ppm, referenced to [15N]NH4Cl) = 98.90; LCMS(ESI): m/z calc. for [M + H]+ = 110.0736, found [M + H]+= 110.0750.
[15N]Tris(2-pyridylmethyl)amine (4): [15N]-2-Aminomethyl pyridine (348 mg, 3.2 mmol) was dissolved in acetonitrile (15 mL) and potassium carbonate (1.8 g, 12.8 mmol) and 2-(chloromethyl)pyridine hydrochloride (1.1 g, 6.4 mmol) were added. The reaction mixture was stirred for 12 h at ambient temperature. After filtration, the crude product was dissolved in dichloromethane and filtered to remove potassium carbonate. The organic layer was dried over anhydrous sodium sulfate, filtered, and dried in vacuum. The oily residue was purified by column chromatography on silica gel with dichloromethane and methanol (97:3) as eluent followed by recrystallization from ethyl ether/hexane to afford [15N]-tris(2-pyridylmethyl)amine as light yellowish solid (291 mg, 31% yield). 1H NMR (CDCl3, 400 MHz) δ (ppm) = 8.51 (3H, d, J = 4 Hz, pyridyl), 7.61 (3H, t, J = 8 Hz, pyridyl), 7.55 (3H, d, J = 8 Hz, pyridyl), 7.11 (3H, t, J = 4 Hz, pyridyl), 3.87 (6H, s, NCH2); 13C NMR (CDCl3, 100 MHz) δ (ppm) = 159.38, 149.17, 136.51, 123.05, 122.08, 60.22 (d, 1JCN = 5 Hz, –CH2-15N-); 15N NMR (CDCl3, 40 MHz) δ (ppm, referenced to [15N]NH4Cl) = 39.71; LCMS(ESI): m/z calc. for [M + 1]+ = 292.1580, found [M + H]+= 292.1456.
Synthesis of 15N-labeled tris(2-pyridylmethyl-d2)amine, [15N]TPA-d6
2-Pyridinemethan-d2-ol (5): Sodium borodeuteride (4.2 g, 99.2 mmol) was added slowly to a deuterated methanol solution (18 mL) of ethyl picolinate (5.0 g, 33.1 mmol) at 0 °C for 40 min. The reaction mixture was stirred for 12 h at ambient temperature. After removal of the solvent by rotary evaporation, the residue was stirred with saturated potassium carbonate solution (100 mL) for 1 h and then extracted with chloroform. The organic layer was dried over anhydrous sodium sulfate, filtered, and dried in vacuum to afford the product as white solid (3.22 g, 88% yield). 1H NMR (CDCl3, 400 MHz) δ (ppm) = 8.26 (1H, d, J = 4 Hz, pyridyl), 7.47 (1H, t, J = 8 Hz, pyridyl), 7.24 (1H, d, J = 8 Hz, pyridyl), 6.96 (1H, t, J = 8 Hz, pyridyl), 5.72 (s, br, –OH); 13C NMR (CDCl3, 100 MHz) δ = 160.20, 148.05, 136.71, 121.93, 120.61, 63.53 (p, JCD = 21 Hz, CD2); 2H NMR (CDCl3, 61 MHz) δ = 4.57 (2D, CD2OH). LCMS(ESI): m/z calc. for [M + H]+ = 112.0731, found = 112.0596.
2-(Chloromethyl-d2)pyridine (6): 2-(Chloromethyl-d2)pyridine was prepared as previously described47. Briefly, 2-pyridinemethan-d2-ol (3.2 g, 28.0 mmol) was added dropwise to a solution of thionyl chloride (16.7 g, 140.2 mmol) in dichloromethane (20 mL) in an ice bath. The reaction mixture was refluxed for 3 h and then stirred at room temperature for 12 h. The product was precipitated by adding ethyl ether (20 mL). The precipitation was filtered and dried in vacuum to afford 2-(chloromethyl-d2)pyridine hydrochloride salt as pale yellow powder (3.56 g, 77% yield as the HCl salt). 1H NMR (CDCl3, 400 MHz) δ (ppm) = 8.66 (1H, d, J = 4 Hz, pyridyl), 8.44 (1H, t, J = 8 Hz, pyridyl), 8.02 (1H, d, J = 8 Hz, pyridyl), 7.89 (1H, t, J = 8 Hz, pyridyl); 13C NMR (CDCl3, 100 MHz) δ (ppm) = 150.71, 146.68, 140.85, 127.36, 126.61, 38.77 (p, JCD = 24 Hz, CD2); 2H NMR (CDCl3, 61 MHz) δ (ppm) = 5.00 (2D, CD2Cl). LCMS(ESI): m/z calc. for [M + H]+ = 130.0393, found = 130.0232.
[15N](2-Pyridinelmethyl-d2)phthalimide (7): It was synthesized from 2-(chloromethyl-d2)pyridine hydrochloride as described for 15N-(pyridine-2-ylmethyl)phthalimide. [15N]phthalimide (1.5 g, 10.1 mmol) was dissolved in dimethylformamide (18 mL) and potassium carbonate (3.5 g, 25.3 mmol) and 2-(chloromethyl-d2) pyridine (1.8 g, 11.1 mmol) was added and the reaction mixture was stirred for 12 h at 50 °C. After removal of the solvent, water was added to the crude product to remove the inorganic salts. The precipitated product was filtered and dried in vacuum to afford [15N](2-pyridinemethyl-d2)phthalimide as white powder (2.39 g, 98% yield). 1H NMR (CDCl3, 400 MHz) δ (ppm) = 8.51 (1H, d, J = 4 Hz, pyridyl), 7.86 (1H, s, phthalic), 7.85 (1H, s, phthalic), 7.72 (1H, s, phthalic), 7.71 (1H, s, phthalic), 7.61 (1H, t, J = 8 Hz, pyridyl), 7.26 (1H, d, J = 8 Hz, pyridyl), 7.14 (1H, t, J = 4 Hz, pyridyl); 13C NMR (CDCl3, 100 MHz) δ (ppm) = 168.20 (d, 1JCN = 13 Hz, C=O), 155.35, 149.75, 136.74, 134.12, 132.27, 123.56, 122.58, 121.67, 42.64 (m, J = 25.5 Hz, -15NCD2); 2H NMR (CDCl3, 61 MHz) δ (ppm) = 4.95 (2D, -15NCD2); 15N NMR (CDCl3, 40 MHz) δ (ppm, referenced to [15N]NH4Cl) = 154.14; LCMS(ESI): m/z calc. for [M + H]+ = 242.0916, found = 242.0682.
[15N]-2-(Aminomethyl-d2)pyridine (8): This intermediate was synthesized from [15N](2-pyridinemethyl-d2)phthalimide as described for 15N-2-aminomethyl pyridine. A solution of [15N](2-pyridinemethyl-d2)phthalimide (2.2 g, 9.3 mmol) in 10% hydrochloride (45 mL) was stirred for 12 h at 80 °C. After removal of the hydrochloric acid, the residue was dissolved in 20% sodium hydroxide (13 mL) and extracted with dichloromethane. The organic layer was dried with anhydrous sodium sulfate, filtered, and dried in vacuum to afford [15N]-2-(aminomethyl-d2)pyridine as a pale yellow oil (877 mg, 85 % yield). 1H NMR (CDCl3, 400 MHz) δ = 8.20 (1H, d, J = 4 Hz, pyridyl), 7.26 (1H, t, J = 8 Hz, pyridyl), 6.91 (1H,d, J = 8 Hz, pyridyl), 6.77 (1H, t, J = 8 Hz, pyridyl); 13C NMR (CDCl3, 100 MHz) δ (ppm) = 161.45, 148.60, 135.84, 121.11, 120.53, 47.70 (m, J = 20 Hz, -15NH2CD2); 2H NMR (CDCl3, 61 MHz) δ (ppm) = 3.56 (2D, -15NH2CD2); 15N NMR (CDCl3, 40 MHz) δ (ppm, referenced to [15N]NH4Cl) = 106.98; LCMS(ESI): m/z calc. for [M + H]+ = 112.0862, found = 112.0730.
[15N]Tris(2-pyridylmethyl-d2)amine (9): [15N]-2-(Aminomethyl-d2)pyridine (535 mg, 4.9 mmol) was dissolved in acetonitrile (15 mL) and potassium carbonate (2.7 g, 19.6 mmol) and 2-(chloromethyl-d2)pyridine (1.6 g, 9.8 mmol) were added. The reaction mixture was stirred for 12 h at 50 °C. The inorganic salts were removed by filtration and organic layer was evaporated by rotary evaporation. The residue was dissolved in hot ethyl acetate and filtered to remove insoluble impurities. The solution was concentrated by rotary evaporation and the oily residue was purified by recrystallization in ethyl ether to afford 15N-tris(2-pyridylmethyl-d2)amine as pale yellow solid (920 mg, 81% yield). 1H NMR (CDCl3, 400 MHz) δ = 8.35 (3H, d, J = 4 Hz, pyridyl), 7.47 (3H, t, J = 8 Hz, pyridyl), 7.42 (3H, d, J = 4 Hz, pyridyl), 6.95 (3H,t, J = 4 Hz, pyridyl); 13C NMR (CDCl3, 100 MHz) δ = 159.05, 148.60, 136.12, 122.73, 121.74, 59.02 (pd, JCD = 21 Hz, -15NH2CD2); 2H NMR (CDCl3, 61 MHz) δ = 3.73 (2D, -15NCD2); 15N NMR (CDCl3, 40 MHz) δ = 40.47; LCMS(ESI): m/z calc. for [M + H]+ = 298.1957, found = 298.1693.
Hyperpolarized 15N MRS studies with human benign prostatic hyperplasia (BPH) tissue samples
Human PBH tissue (approximately 500 mg) was homogenized in 1 mL 10% aqueous solution of NP-40 (nonyl phenoxypolyethoxylethanol) using a tissue homogenizer (PowerGen 500, Fisher Scientific) in an ice bath. The resulting mixture was further lysed using Ultrasonic Homogenizer (3 × 10 s). The pH was adjusted to pH~3 and allowed to stand for 10 min in an ice bath. It was then centrifuged for 10 min at 12,000 rpm. The supernatant was collected and the pH was adjusted to around 6 for the HP experiment. The homogenized prostate tissue (1 mL) was placed in 10 mm NMR tube. Hyperpolarized [15N]TPA-d6 was injected into the tube within 10 s after dissolution with PBS (final concentration of 4.2 mM, pH 7.0). HP 15N MRS studies were performed 9.4 T using 30° flip angle with repetition time (TR) of 2 s.
Hyperpolarized 15N MRS studies with human prostate epithelial cells (PNT1A-WT)
Normal prostate epithelial cells (PNT1A) were cultured in a 150 mm culture dish with RPMI-1640 medium (Sigma, R8758) supplemented with 10% of fetal bovine serum (Sigma, F2442) and 20 units of penicillin-streptomycin (Sigma, P0781). When the cells reached ~90% confluence, they were washed twice with PBS (5 mL) and supplemented with zinc. Zn-supplementation was performed with 15 µM zinc pyrithione (Sigma, H6377) in assay buffer (114 mM NaCl, 4.7 mM KCl, 1.2 mM KH2PO4, 2.5 mM CaCl2, 1.16 mM MgSO4, 3 mM glucose and 20 mM HEPES, pH 7.4). After 10 min incubation with zinc pyrithione at 37 °C, the buffer was removed and the cells were washed three times with 5 mL PBS. Assay buffer (4 mL) was added to cell culture plate and the cells (~65 × 106) were collected using a cell scraper (Sigma, CLS3008). After centrifuging at 1000 rpm for 5 min, the supernatant was removed and the cell pellets were resuspended in assay buffer to achieve 1 m L of final volume. The cells were suspended in 1 mL of assay buffer and placed in 10 mm NMR tube. Hyperpolarized [15N]TPA-d6 was injected into the tube within 10 sec following the dissolution with assay buffer (final concentration of 2.8 mM, pH 7.4). HP 15N MRS studies in cells were performed 9.4 T using 45° flip angle with repetition time (TR) of 2 s.
15N chemical shift imaging (CSI)
Phantom imaging with [15N]TPA-d6 was performed on a 9.4 T Agilent (Varian) vertical bore microimager (Varian, USA). The phantom consisted of three 8-mm NMR tubes each containing different concentrations of zinc chloride inserted into a 22-mm NMR tube containing deionized water (10 mL). The axial imaging plane was positioned near the center of the phantom. The acquisition of 15N CSI started 10 s after the HP sample transfer was completed. CSI parameter: CSI2 sequence (Agilent VnmrJ 4 Imaging, USA), field of view (FOV) = 40 × 40 mm; TR = 200 ms; TE = 1.30 ms; flip angle = 30°; NA = 1, matrix = 16 × 16. The 15N CSI imaging data were reconstructed and analyzed using MATLAB (Mathworks, Natick MA, USA).
Statistical analysis
Statistical significance was evaluated by an unpaired t-test (α = 0.05, two-tailed, homoscedastic) using GraphPad Prism version 8.0 (GraphPad Software, Inc., La Jolla CA, USA). All data are presented as mean ± SD.
Associated content
The Supplementary Information is available for general experimental considerations, detailed synthetic procedures, characterization of [15N]TPA derivatives, and additional supporting data (PDF).
Data availability
The authors declare that all data supporting the findings of this study are available within the paper and Supplementary Information.
Code availability
The MATLAB scripts used for image reconstruction in the study will be available from the author upon request.
References
Witanowski, M. Nitrogen n.m.r. spectroscopy. Pure Appl. Chem. 37, 225–233 (1974).
Gunther, U. L. Dynamic nuclear hyperpolarization in liquids. Top. Curr. Chem. 335, 23–69 (2013).
Ardenkjær-Larsen, J. H. et al. Increase in signal-to-noise ratio of >10,000 times in liquid-state NMR. Proc. Natl Acad. Sci. USA 100, 10158–10163 (2003).
Golman, K. in’t Zandt, R. & Thaning, M. Real-time metabolic imaging. Proc. Natl Acad. Sci. USA 103, 11270–11275 (2006).
Gallagher, F. A., Kettunen, M. I. & Brindle, K. M. Biomedical applications of hyperpolarized 13C magnetic resonance imaging. Prog. Nucl. Magn. Reson. Spectrosc. 55, 285–295 (2009).
Kurhanewicz, J. et al. Analysis of cancer metabolism by imaging hyperpolarized nuclei: prospects for translation to clinical research. Neoplasia 13, 81–97 (2011).
Singh, J. et al. Probing carbohydrate metabolism using hyperpolarized 13C-labeled molecules. NMR Biomed. 32, e4018 (2019).
Nonaka, H. et al. A platform for designing hyperpolarized magnetic resonance chemical probes. Nat. Commun. 4, 2411 (2013).
Jiang, W. et al. Hyperpolarized 15N-pyridine derivatives as pH-sensitive MRI agents. Sci. Rep. 5, 9104 (2015).
Frederickson, C. J., Koh, J. Y. & Bush, A. I. The neurobiology of zinc in health and disease. Nat. Rev. Neurosci. 6, 449–462 (2005).
De Leon-Rodriguez, L., Lubag, A. J. M. & Sherry, A. D. Imaging free zinc levels in vivo—what can be learned? Inorg. Chim. Acta 393, 12–23 (2012).
Li, Y. V. Zinc and insulin in pancreatic beta-cells. Endocrine 45, 178–189 (2014).
Costello, L. C., Feng, P., Milon, B., Tan, M. & Franklin, R. B. Role of zinc in the pathogenesis and treatment of prostate cancer: critical issues to resolve. Prostate Cancer Prostatic Dis. 7, 111 (2004).
Costello, L. C. & Franklin, R. B. Zinc is decreased in prostate cancer: an established relationship of prostate cancer! J. Biol. Inorg. Chem. 16, 3–8 (2011).
Ghosh, S. K. et al. A novel imaging approach for early detection of prostate cancer based on endogenous zinc sensing. Cancer Res. 70, 6119–6127 (2010).
Franklin, R. B., Milon, B., Feng, P. & Costello, L. C. Zinc and zinc transporters in normal prostate and the pathogenesis of prostate cancer. Front. Biosci. 10, 2230–2239 (2005).
Ho, E. & Song, Y. Zinc and prostatic cancer. Curr. Opin. Clin. Nutr. Metab. Care 12, 640–645 (2009).
Cortesi, M. et al. Clinical assessment of the cancer diagnostic value of prostatic Zinc: a comprehensive needle-biopsy study. Prostate 68, 994–1006 (2008).
Rice, B. W., Cable, M. D. & Nelson, M. B. In vivo imaging of light-emitting probes. J. Biomed. Opt. 6, 432–440 (2001).
Martins, A. F. et al. Imaging insulin secretion from mouse pancreas by MRI is improved by use of a zinc-responsive MRI sensor with lower affinity for Zn2+ ions. J. Am. Chem. Soc. 140, 17456–17464 (2018).
Yu, J. et al. Amplifying the sensitivity of zinc(II) responsive MRI contrast agents by altering water exchange rates. J. Am. Chem. Soc. 137, 14173–14179 (2015).
Clavijo Jordan, M. V. et al. Zinc-sensitive MRI contrast agent detects differential release of Zn(II) ions from the healthy vs. malignant mouse prostate. Proc. Natl Acad. Sci. USA 113, E5464–E5471 (2016).
Mishra, A., Pariani, G., Oerther, T., Schwaiger, M. & Westmeyer, G. G. Hyperpolarized multi-metal 13C-sensors for magnetic resonance imaging. Anal. Chem. 88, 10790–10794 (2016).
Naoko, K. et al. A sensitive zinc‐activated 129Xe MRI probe. Angew. Chem. Int. Ed. 51, 4100–4103 (2012).
Wang, S. et al. Amino acid-derived sensors for specific Zn2+ detection using hyperpolarized 13C magnetic resonance spectroscopy. Chem. Eur. J. 25, 11842–11846 (2019).
Dai, Z. & Canary, J. W. Tailoring tripodal ligands for zinc sensing. N. J. Chem. 31, 1708–1718 (2007).
Huang, Z., Zhang, X.-a, Bosch, M., Smith, S. & Lippard, S. J. Tris(2-pyridylmethyl)amine (TPA) as a membrane-permeable chelator for interception of biological mobile zinc. Metallomics 5, 648–655 (2013).
Patyal, B. R. et al. Longitudinal relaxation and diffusion measurements using magnetic resonance signals from laser-hyperpolarized129Xe nuclei. J. Magn. Reson. 126, 58–65 (1997).
Lumata, L. et al. DNP by thermal mixing under optimized conditions yields >60 000-fold enhancement of 89Y NMR signal. J. Am. Chem. Soc. 133, 8673–8680 (2011).
Zalewski, P. D., Forbes, I. J. & Giannakis, C. Physiological role for zinc in prevention of apoptosis (gene-directed death). Biochem. Int. 24, 1093–1101 (1991).
Nonaka, H. et al. Design of a 15N molecular unit to achieve long retention of hyperpolarized spin state. Sci. Rep. 7, 40104 (2017).
Mishkovsky, M. et al. Measuring glucose cerebral metabolism in the healthy mouse using hyperpolarized 13C magnetic resonance. Sci. Rep. 7, 11719 (2017).
Allouche-Arnon, H., Lerche, M. H., Karlsson, M., Lenkinski, R. E. & Katz-Brull, R. Deuteration of a molecular probe for DNP hyperpolarization—a new approach and validation for choline chloride. Contrast Media Mol. Imaging 6, 499–506 (2011).
Allouche-Arnon, H. et al. In vivo magnetic resonance imaging of glucose—initial experience. Contrast Media Mol. Imaging 8, 72–82 (2013).
Qu, W. et al. Facile synthesis [5-13C-4-2H2]-L-glutamine for hyperpolarized MRS imaging of cancer cell metabolism. Acad. Radiol. 18, 932–939 (2011).
Barb, A. W., Hekmatyar, S. K., Glushka, J. N. & Prestegard, J. H. Probing alanine transaminase catalysis with hyperpolarized 13CD3-pyruvate. J. Magn. Reson. 228, 59–65 (2013).
Chiavazza, E., Viale, A., Karlsson, M. & Aime, S. 15N-permethylated amino acids as efficient probes for MRI-DNP applications. Contrast Media Mol. Imaging 8, 417–421 (2013).
Meier, S., Jensen, P. R., Karlsson, M. & Lerche, M. H. Hyperpolarized NMR probes for biological assays. Sensors (Basel) 14, 1576–1597 (2014).
Sudmeier, J. L., Anderson, S. E. & Frye, J. S. Calculation of nuclear spin relaxation times. Concepts Magn. Reson. 2, 197–212 (1990).
Yen, Y.-F. et al. Imaging considerations for in vivo 13C metabolic mapping using hyperpolarized 13C-pyruvate. Magn. Reson. Med. 62, 1–10 (2009).
Chiu, Y. H. & Canary, J. W. Stability and acidity constants for ternary ligand-zinc-hydroxo complexes of tetradentate tripodal ligands. Inorg. Chem. 42, 5107–5116 (2003).
Hahn, F. E., Jocher, C., Lügger, T. & Pape, T. Coordination chemistry of aliphatic, tripodal ligands with zinc salts. Z. Anorg. Allg. Chem. 629, 2341–2347 (2003).
Costello, L. C. & Franklin, R. B. The clinical relevance of the metabolism of prostate cancer; zinc and tumor suppression: connecting the dots. Mol. Cancer 5, 17–17 (2006).
Frederickson, C. J. et al. Concentrations of extracellular free zinc (pZn)e in the central nervous system during simple anesthetization, ischemia and reperfusion. Exp. Neurol. 198, 285–293 (2006).
Mikata, Y., Kawata, K., Iwatsuki, S. & Konno, H. Zinc-specific fluorescent response of tris(isoquinolylmethyl)amines (isoTQAs). Inorg. Chem. 51, 1859–1865 (2012).
Weingarth, M. et al. Revealing molecular self-assembly and geometry of non-covalent halogen bonding by solid-state NMR spectroscopy. Chem. Commun. 7, 5981–5983 (2008).
Zhang, J., Cui, H., Hojo, M., Shuang, S. & Dong, C. Synthesis and spectral studies of 2-[(N-ethyl carbazole)-3-sulfonyl ethylenediamine]-1-N,N-2-(2-methypyridy) as a fluorescence probe for Zn2+. Bioorg. Med. Chem. Lett. 22, 343–346 (2012).
Acknowledgements
We thank Dr. Douglas Strand and Dr. Jer-Tsong Hsieh at the Department of Urology, the University of Texas Southwestern Medical Center, Dallas, TX for providing the human prostate tissue samples and PNT1A-WT cell. We acknowledge partial financial support for this work from the National Institutes of Health (R37-HR034557 and P41-EB015908), the Texas Institute for Brain Injury and Repair, the Robert A. Welch Foundation (AT-584, AT-1877-03242018 and I-2009-20190330), and Collaborative Biomedical Research Award (CoBRA).
Author information
Authors and Affiliations
Contributions
Z.K. conceived the idea. E.H.S., A.D.S., and Z.K. designed the research; E.H.S. and L.L. performed the preliminary HP-stubiodies. E.H.S. and J.M.P. performed the hyperpolarized 15N imaging experiment; E.H.S. and Z.K. performed the synthesis TPA derivatives and performed hyperpolarized 15N spectroscopy in tissue; E.H.S., A.D.S., and Z.K. performed the HP studies in biological samples. E.H.S. A.D.S., and Z.K. analyzed the 15N NMR spectra data; E.H.S., J.M.P., A.D.S., and Z.K. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Suh, E.H., Park, J.M., Lumata, L. et al. Hyperpolarized 15N-labeled, deuterated tris(2-pyridylmethyl)amine as an MRI sensor of freely available Zn2+. Commun Chem 3, 185 (2020). https://doi.org/10.1038/s42004-020-00426-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42004-020-00426-6
This article is cited by
-
Opportunities and challenges with hyperpolarized bioresponsive probes for functional imaging using magnetic resonance
Nature Chemistry (2023)
-
Prediction of multiple pH compartments by deep learning in magnetic resonance spectroscopy with hyperpolarized 13C-labelled zymonic acid
EJNMMI Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.