Abstract
The development of precise folding techniques for synthetic polymer chains that replicate the unique structures and functions of biopolymers has long been a key challenge. In particular, spiro-type (i.e., 8-, trefoil-, and quatrefoil-shaped) polymer topologies remain challenging due to their inherent structural complexity. Herein, we establish a folding strategy to produce spiro-type multicyclic polymers via intramolecular ring-opening metathesis oligomerization of the norbornenyl groups attached at predetermined positions along a synthetic polymer precursor. This strategy provides easy access to the desired spiro-type topological polymers with a controllable number of ring units and molecular weight while retaining narrow dispersity (Ɖ < 1.1). This effective strategy marks an advancement in the development of functionalized materials composed of specific three-dimensional nanostructures.
Similar content being viewed by others
Introduction
Precise folding of a biopolymer chain is an essential process to attain sophisticated higher-ordered structures, such as DNA packing and three-dimensional (3D) protein structures, which is responsible for their outstanding functions in living systems1,2,3. Inspired by the folding process of biopolymers, significant efforts have been dedicated to the folding of synthetic polymers. The synthesis of topologically unique polymers from linear polymers can be regarded as mimicking biopolymer folding processes4,5,6. One successful example of this approach is the intramolecular crosslinking of linear polymers to afford single-chain nanoparticles (SCNPs) that feature a densely packed single-chain globule with a 3D nanostructure7,8. However, the resulting SCNP is a statistical mixture of undefined-shape chains since the intramolecularly crosslinked formations randomly occur along the main chain.
Another remarkable approach that has been demonstrated is the programmed folding of polymer chains into predetermined cyclic-type topologies6,9,10. The simplest case involves intramolecular coupling between the chain ends of a linear polymer to form a monocyclic polymer with unique properties attributed to the lack of chain ends9,10,11,12. In addition, multicyclic topological polymers (i.e., fused- (such as θ-shaped), bridged- (such as manacle-shaped), and spiro-multicyclic polymers (such as 8-shaped)) that consist of multiple macromolecular rings, have also been intriguing synthetic targets due to their interesting 3D structures13,14. Among these multicyclic topological polymers, effective construction of spiro-multicyclic topologies remains the most challenging due to inherently complicated architectures consisting of multiple cyclic units tethered at a single junction point.
Several synthetic strategies have been developed over the past decade to prepare spiro-multicyclic polymers: (i) intermolecular coupling of cyclic constituents15,16, (ii) intermolecular cyclization of linear polymers with a multifunctionalized linker17, and (iii) intramolecular cyclization of linear/star polymer precursors, in which functional groups are placed at the chain ends and/or chain center for multiple bond-forming reactions (such as the electrostatic self-assembly and covalent fixation (ESA-CF) protocol)18,19,20,21. More specifically, the intermolecular coupling of monocyclic constituents, i.e., strategy (i), can produce a series of spiro-multicyclic polymers with varied number of cyclic units; however, it requires the elaborated synthesis of cyclic polymers having a reactive functional group, as well as a tedious purification process to remove the excessive monocyclic reactant15,16. Strategy (ii) is a simple way to access an 8-shaped topology although it is limited by the formation of many possible byproducts17. On the other hand, strategy (iii) is advantageous in terms of suppressing by-product formation because the reaction is essentially concluded in a single polymer chain under a high-dilution condition18,19,20,21. However, this strategy does not exhibit synthetic versatility, such as control over the size and number of cyclic units, because it requires sophisticated preparation of highly functionalized precursors. Thus, a precise yet universal folding strategy to spiro-multicyclic polymers has remained elusive because increasing the number of constitutional cyclic units leads to synthetic difficulties. Owing to lack of an effective synthetic platform, only few comprehensive studies have been attempted to control the size and number of cyclic units, and thus, the structure–property relationships associated with this folded topology are not well-defined22,23.
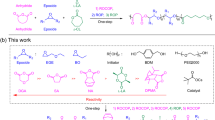
Our group recently reported a robust and precise strategy for constructing a variety of cage-shaped multicyclic topologies based on intramolecular consecutive cyclization (i.e., intramolecular ring-opening metathesis oligomerization (ROMO) of an exo-norbornenyl group attached to each terminus of star-shaped polymers), which enabled systematic synthesis and characterization24. A unique feature of our strategy is that polymerizable exo-norbornenyl groups at the chain termini are immediately transformed for intramolecular chain-growth upon addition of excess Grubbs’ 3rd catalyst (G3). We envisaged that the aforementioned challenges in spiro-multicyclic polymer synthesis, i.e., synthetic simplicity and versatility to allow access to a series of polymers with varied size and number of cyclic units, can be overcome by applying this novel strategy. Herein, we demonstrate the utility of intramolecular ROMO to accomplish programmed folding into spiro-multicyclic polymer topologies. This is the first example, to our knowledge, of the synthesis of inherently complex spiro-type multicyclic architectures through the intramolecular chain reaction to facilitate cyclic unit formation.
Results and discussion
Construction of an 8-shaped polymer
For synthesis of an 8-shaped polymer, we initially designed a poly(ε-caprolactone) (PCL)-based linear precursor with norbornenyl groups at each ω-chain-end and chain center (P2; Fig. 1). To introduce the norbornenyl groups at not only the chain ends but also the chain center, a diol initiator possessing a protected hydroxyl group was employed for the polymerization. The precursor of P2 [P2-a; molecular weight from 1H nuclear magnetic resonance (NMR) (Mn,NMR) = 6200 g mol−1, molecular weight from size-exclusion chromatography (SEC; Mn,SEC) = 9970 g mol−1, dispersity (Ð) = 1.06] was successfully synthesized by the ring-opening polymerization of ε-caprolactone using the initiator, followed by deprotection and condensation with (±)-exo-5-norbornene carboxylic acid.
Subsequently, the intramolecular ROMO was carried out under very dilute conditions in CH2Cl2 (final concentration = 0.02 mM) in the presence of excess Grubbs’ 3rd generation catalyst ([precursor]0/[G3] = 1/6) with slow addition of the polymer precursor. After removing the catalyst residue, the product was obtained in high yield (91.0%).1H NMR analysis of the obtained product revealed that signals arising from the exo-norbornenyl groups of the precursor completely disappeared, while new signals due to the oligonorbornene backbone appeared at 1.6‒3.4 and 4.8‒6.5 p.p.m. (Supplementary Fig. 1e). The SEC trace of the product showed a monomodal peak, and the elution peak was observed in a lower molecular weight region (Mn,SEC = 7340 g mol−1, Ð = 1.08) than that of the linear precursor (P2-a; Mn,SEC = 9970 g mol−1, Ð = 1.06, Fig. 2a and Supplementary Fig. 2). The dramatic shift in the SEC elution peak maximum indicates that intramolecular cyclization occurred to produce a hydrodynamically smaller polymer (i.e., MC2-a), rather than intermolecular side reactions. By comparing the SEC peak-top molecular weights of the precursors and cyclized polymers, the compaction parameter <G> was calculated as 0.67, which agrees with the reported value of <G> for 8-shaped polymers of comparable molecular weights (<G> = 0.67‒0.81; Table 1)16,18,19,25,26,27. The matrix-assisted laser desorption/ionization-time of flight mass spectrometry (MALDI-TOF MS) spectrum of the obtained product showed two series of peaks with a regular m/z interval of 114.09, corresponding to the mass of the repeating ε-caprolactone unit (Fig. 2b). The major series of peaks (denoted with •) was assigned to the expected chemical structure of MC2-a; for example, the peak at m/z = 5184.84 agrees well with the calculated mass of the desired MC2-a with a total degree of polymerization of 40 ([M + Na]+ = 5184.12). The minor series of peaks (denoted with ▪) was assigned to cyclic or tadpole-shaped PCLs (calculated [M + Na]+ = 5174.11, n = 39) formed by reaction with two G3 molecules (Supplementary Fig. 3). The amount present was determined to be 7.9% by peak deconvolution. Two topological isomers of cyclic- and tadpole-forms are expected to be produced by a side reaction, with the preferred isomer expected to have the tadpole topology due to the folding process being primarily directed by the spatial distance of crosslinking points21. Other possible byproducts (e.g., linear PCL; calculated [M + Na]+ = 5278.17, n = 39) were not detected. Overall, successful folding into the 8-shaped topology (MC2-a) was achieved by intramolecular ROMO of the linear precursor in a precise manner.
a, c, e SEC traces of the precursors (P2-a, P3-a, and P4-a; blue curves) and the obtained spiro-multicyclic polymers (MC2-a, MC3-a, and MC4-a; red curves) (RI detection; PS standards; eluent, THF). b, d, f MALDI-TOF MS spectra of MC2-a, MC3-a, and MC4-a (reflector mode; n denotes number of monomer units).
Construction of trefoil- and quatrefoil-shaped polymers
To further extend this strategy, intramolecular ROMO was applied to the synthesis of trefoil- (MC3) and quatrefoil-shaped PCLs (MC4), where the number of cyclic units are three and four, respectively. For the synthesis of MC3-a and MC4-a, the well- defined three- and four-armed star-shaped PCLs bearing an exo-norbornenyl group at the chain center and each terminus (P3-a and P4-a; Mn,NMR = ~6500 g mol−1) were subjected to ROMO conditions (Supplementary Figs. 4–9). 1H NMR analysis confirmed quantitative consumption of the exo-norbornenyl groups despite an increase in the number of groups in the precursors (Supplementary Figs. 5 and 8). Importantly, in both cases, the SEC elution peak clearly shifted to the lower molecular weight region while the peak shape remained monomodal with narrow dispersity (Ɖ = 1.07‒1.09; Fig. 2c, e). The estimated <G> values for MC3-a and MC4-a (0.57‒0.58) were much lower than that of MC-2a (0.67), confirming the much smaller volume. Notably, each MALDI-TOF MS spectrum showed only one series of peaks assignable to the expected structures of MC3-a and MC4-a (Fig. 2d, f). The SEC and MALDI-TOF MS analyses strongly support that ROMO proceeds in an intramolecular manner without side reactions regardless of the number of cyclic units. A question therefore arises with respect to the reaction order of the norbornenes. According to a recent report by Tezuka and co-workers21, it can be reasonably expected that the norbornenes should react with those that are closer. However, to fully understand the folding process, further investigation is necessary, which is currently ongoing in our laboratory.
Functionalization of topological polymers
Chain-end functionalization of topological polymers is essential to facilitate higher-order functions with a combination of diverse molecular designs. Typically, the α- and ω-chain ends of the polynorbornene backbone produced by ROMO can be readily transformed to the desired reactive groups by using functional Ru initiators and end-capping agents, respectively28,29. Indeed, the α-/ω-end-functionalized trefoil-shaped PCLs with hydroxyl groups were precisely synthesized while retaining narrow dispersity (Ɖ = 1.12‒1.13, Supplementary Figs. 10‒14). This allows facile access to surface-modified metal/semiconductor nanoparticles and substrates with topological polymers, which could be used to increase colloidal stability and to prepare bioinert and superlubricating coatings30,31,32.
Size control of multicyclic polymers
For systematic characterization of the folded polymers, size control of the cyclic unit and control over the number of cyclic units is indispensable. By simply changing the degree of polymerization of the precursor, the molecular weight of each multicyclic (8-, trefoil-, and quatrefoil-shaped) polymer was successfully controlled from ~6000 to 12,000 g mol−1 (Table 1). Note that the suffix on the name of each polymer sample represents its molecular weight (-a for ~6,000 g mol−1, -b for ~9,000 g mol−1, and -c for ~12,000 g mol−1).
Applicability of intramolecular ROMO to diverse polymer species
Furthermore, to verify the applicability of intramolecular ROMO to other polymer backbones, we applied this approach to the synthesis of spiro-multicyclic poly(L-lactide) (PLLA) and poly(2-ethylhexyl glycidyl ether) (PEHGE) (Supplementary Figs. 15‒21 and Supplementary Table 4). Specifically, trefoil-shaped PLLA and PEHGE were synthesized by the ring-opening polymerization of the corresponding monomers with I3 as the initiator using 1,8-diazabicyclo[5.4.0]-7-undecene (DBU) and t-Bu-P4 catalysts33,34, respectively, followed by the deprotection reaction, installation of norbornene groups, and ROMO under the optimized conditions. The targeted folded structures were confirmed in both synthesized products through the comprehensive characterization by 1H NMR, SEC, and MALDI-TOF MS, which suggested that the presented method is applicable for the synthesis of a broad range of polymer species.
Programmed folding into a predetermined topology via intramolecular ROMO under different conditions
Here, it is important to note that the present folding strategy affords a polymer with a predetermined topology, as opposed to the synthesis of SCNPs, in which the size and conformation of the resulting product are considerably affected by the solvent quality35. To provide a proof-of-concept of our programmed folding, we performed intramolecular ROMO under different conditions that could affect the polymer chain dimension during the reaction (Fig. 3). For example, to attain the complicated multicyclic folding of MC4-a, different solvents and/or elevated temperatures were used to determine whether the folded structure of the resulting product is affected. Notably, the products obtained from the intramolecular ROMO of P4-a in CH2Cl2/n-hexane showed exactly the same peak-top as MC4-a in the SEC, even in the case of n-hexane-rich media with up to 60% n-hexane (Fig. 3c‒e), indicating successful folding into the same architecture. By switching the solvent to toluene, successful formation of MC4-a was also observed upon heating (Fig. 3f, g). Thus, these results suggest that the precursor can be unambiguously folded into the predetermined topology.
Systematic investigation of structure–property relationships
Hydrodynamic diameter (Dh) and intrinsic viscosity ([η]), which are correlated to the polymer chain dimensions, are good measures to understand the folded polymer structure. To get information about the polymer chain dimensions, the obtained spiro-multicyclic PCLs and their related linear and cyclic counterparts were subjected to online SEC measurement combined with light scattering, viscosity, and reflective index detectors (SEC-MALS-Visco) in THF. Note that the previously synthesized linear and monocyclic counterparts with comparable molecular weights were subjected to these analyses for comparison24. As shown in Supplementary Fig. 22, both the Dh and [η] values decreased in the order of linear > cyclic > 8-shaped (MC2) > trefoil-shaped (MC3) > quatrefoil-shaped (MC4) when comparing the topologically different polymers with comparable molecular weights. This demonstrates that the polymer chain dimensions decrease with increasing number of cyclic units when the total molecular weight remains the same. Such a trend matches very well with the theoretically predicted one; the radius of gyration decreased in the same order as reported by Deguchi and colleagues36. Interestingly, the aforementioned [η] values of spiro-multicyclic polymers were smaller than those of the topologically related cage-shaped PCLs (7.4‒27.7 mg mL−1 for cyclic, three-arm cage-shaped, and four-arm cage-shaped)20. The spiro-multicyclic polymer is assumed to be a topological analog of the cage-shaped polymer, whereby an additional constraint at the focal point of the spiro-multicyclic polymer exists, further decreasing the chain dimension.
While the solution properties are important for understanding the folded structure, understanding the bulk state properties also provides new insight into topological effects. Initially, we performed thermogravimetric analysis (TGA) for the spiro-multicyclic PCLs with Mn,NMR of ~6000 g mol−1 to examine the structure–thermal degradation relationship. The TGA results revealed a negligible difference in the degradation temperature (Td) for 10% weight loss among the spiro-type multicyclic PCLs and the corresponding linear and cyclic counterparts (384‒390 °C), which suggested that the polymer topology has no significant impact on the thermal degradation of PCL (See Supplementary Fig. 30 and Supplementary Table 5). A similar conclusion was drawn in a previous report by Grayson, in which a cyclic PCL and its precursor were compared37. Among a number of cyclic polymers reported thus far, cyclic PCLs have been a target of investigation to understand the effect of cyclic topology on polymer crystallization behavior (e.g., melting point, crystallite size, crystallinity, and crystallization kinetics). For example, cyclic PCLs are known to possess a higher melting point and crystallinity than their linear counterparts, albeit the reason remains unclear38,39,40. With a series of well-defined spiro-type PCLs in hand, we thus investigated the melting temperature (Tm) and crystallinity (XWAXD) using differential scanning calorimetry (DSC) and wide-angle X-ray diffraction (WAXD), respectively. Note that previously synthesized linear and monocyclic PCLs with comparable molecular weight were subjected to these analyses for comparison24. Figure 4 shows the Tm and XWAXD for each architecture, which seem to correlate with both the number of cyclic units and total molecular weight. The important finding here is that 8-shaped PCLs exhibit higher melting points and crystallinity than cyclic PCLs (Fig. 4a, b). More specifically, MC2-a with molecular weight of ~6000 g mol−1 exhibited dramatically enhanced Tm (53.8 °C) and XWAXD (41.8%) compared with its linear and cyclic counterparts (Tm = 43.2‒51.1 °C and XWAXD = 35.2‒40.3%). This can be explained by the topological confinement of the non-crystallizable segments (Supplementary Fig. 23). The non-crystallizable segments, such as the initiator moiety and oligonorbornene backbone in the cyclic PCL have mobility, which suppresses the ordered packing of the PCL chains, resulting in lesser crystallization ability. However, the initiator moiety and oligonorbornene backbone in the 8-shaped PCL are all constrained at the focal point, which successfully reduces the random placing of non-crystallizable segments, thus resulting in better crystallization ability. Further increase in the number of cyclic units in MC3 and MC4 apparently lowers the Tm and crystallinity. This can be attributed to suppressed molecular mobility and chain-packing ability due to strong constraints induced with increasing the number of arms. Small-angle X-ray scattering (SAXS) further supports this unique trend in lamellae thickness that appears to be correlated with Tm, while the long period (~12 nm) exhibits almost no change (Supplementary Note 1 and Supplementary Fig. 24), which is consistent with a previous report41. Compared to its counterparts, MC2-b showed the longest lamellae thickness of 5.1 nm, which is approximately 25% of the theoretical extended length of 20 nm for each of its arms (see the SI for details). This suggests that the spiro-type topological chain is likely further folded in the crystalline domain multiple times.
a DSC thermograms during the 2nd heating of the multicyclic polymers (MC2-a, MC3-a, and MC4-a) and their counterparts with molecular weights of ~6000 g mol−1. The melting temperature (Tm) was determined as the peak-top of the transition marked with a triangle. b, c Plots of Tm and XWAXD for the linear, monocyclic, and multicyclic polymers (MC2s, MC3s, and MC4s) with different molecular weights.
In summary, we demonstrated the programmed folding of linear and star-shaped polymer precursors into spiro-multicyclic polymers via intramolecular ROMO of the norbornenyl groups at predetermined positions. With the present strategy, diverse spiro-type multicyclic topologies with different amounts of cyclic units and total molecular weight were successfully constructed, demonstrating the versatility of this strategy as an effective means to synthesize topological polymers. Remarkably, this comprehensive study on the structure–property relationships of the folded PCLs revealed enhanced crystallization ability in the 8-shaped topology. Polymer folding into a spiro-type topology plays a crucial role in rendering higher-ordered functions to biomacromolecules. Furthermore, the results of this study can be applied to other polymer species for the development of bio-inspired materials with specific 3D nanostructures.
Data availability
All data are available from the authors upon reasonable request.
References
Teif, V. B. & Bohinc, K. Condensed DNA: condensing the concepts. Prog. Biophys. Mol. Biol. 105, 208–222 (2011).
Seeman, N. C. DNA in a material world. Nature 421, 427–431 (2003).
Craik, D. J. Seamless proteins tie up their loose ends. Science 311, 1563–1564 (2006).
Ouchi, M., Badi, N., Lutz, J. F. & Sawamoto, M. Single-chain technology using discrete synthetic macromolecules. Nat. Chem. 3, 917–924 (2011).
Gonzalez-Burgos, M., Latorre-Sanchez, A. & Pomposo, J. A. Advances in single chain technology. Chem. Soc. Rev. 44, 6122–6142 (2015).
Tezuka, Y. Topological polymer chemistry designing complex macromolecular graph constructions. Acc. Chem. Res. 50, 2661–2672 (2017).
Harth, E. et al. A facile approach to architecturally defined nanoparticles via intramolecular chain collapse. J. Am. Chem. Soc. 124, 8653–8660 (2002).
Foster, E. J., Berda, E. B. & Meijer, E. W. Metastable supramolecular polymer nanoparticles via intramolecular collapse of single polymer chains. J. Am. Chem. Soc. 131, 6964–6966 (2009).
Polymeropoulos, G. et al. 50th anniversary perspective: polymers with complex architectures. Macromolecules 50, 1253–1290 (2017).
Laurent, B. A. & Grayson, S. M. Synthetic approaches for the preparation of cyclic polymers. Chem. Soc. Rev. 38, 2202–2213 (2009).
Zhang, K., Lackey, M. A., Cui, J. & Tew, G. N. Gels based on cyclic polymers. J. Am. Chem. Soc. 133, 4140–4148 (2011).
Honda, S., Yamamoto, T. & Tezuka, Y. Topology-directed control on thermal stability: Micelles formed from linear and cyclized amphiphilic block copolymers. J. Am. Chem. Soc. 132, 10251–10253 (2010).
Yamamoto, T. & Tezuka, Y. Topological polymer chemistry: a cyclic approach toward novel polymer properties and functions. J. Polym. Sci. Part A Polym. Chem. 2, 1930–1941 (2011).
Oike, H. et al. Designing unusual polymer topologies by electrostatic self-assembly and covalent fixation. J. Am. Chem. Soc. 122, 9592–9599 (2000).
Lonsdale, D. E. & Monteiro, M. J. Various polystyrene topologies built from tailored cyclic polystyrene via CuAAC reactions. Chem. Commun. 46, 7945–7947 (2010).
Ko, Y. S., Yamamoto, T. & Tezuka, Y. Click construction of spiro‐ and bridged‐quatrefoil polymer topologies with kyklo‐telechelics having an azide group. Macromol. Rapid Commun. 35, 412–416 (2014).
Lee, T. et al. Figure-eight-shaped and cage-shaped cyclic polystyrenes. Macromolecules 49, 3672–3680 (2016).
Schmidt, B. V. K. J., Fechler, N., Falkenhagen, J. & Lutz, J. F. Controlled folding of synthetic polymer chains through the formation of positionable covalent bridges. Nat. Chem. 3, 234–238 (2011).
Isono, T. et al. Synthesis of star-and figure-eight-shaped polyethers by t-Bu-P4-catalyzed ring-opening polymerization of butylene oxide. Macromolecules 46, 3841–3849 (2013).
Satoh, Y. et al. Synthesis of well-defined three- and four-armed cage-shaped polymers via “topological conversion” from trefoil- and quatrefoil-shaped polymers. Macromolecules 50, 97–106 (2017).
Kyoda, K., Yamamoto, T. & Tezuka, Y. Programmed polymer folding with periodically positioned tetrafunctional telechelic precursors by cyclic ammonium salt units as nodal points. J. Am. Chem. Soc. 141, 7526–7536 (2019).
Hossain, M. D. et al. Influence of constraints within a cyclic polymer on solution properties. Biomacromolecules 19, 616–625 (2018).
Pipertzis, A., Hossain, M. D., Monteiro, M. J. & Floudas, G. Segmental dynamics in multicyclic polystyrenes. Macromolecules 51, 1488–1497 (2018).
Mato, Y. et al. A versatile synthetic strategy for macromolecular cages: intramolecular consecutive cyclization of star-shaped polymers. Chem. Sci. 10, 440–446 (2019).
Oike, H., Hamada, M., Eguchi, S., Danda, Y. & Tezuka, Y. Novel synthesis of single-and double-cyclic polystyrenes by electrostatic self-assembly and covalent fixation with telechelics having cyclic ammonium salt groups. Macromolecules 34, 2776–2782 (2001).
Antonietti, M. & Fölsch, K. J. Synthesis and characterization of “eight‐shaped” polystyrene. Makromol. Chem. Rapid Commun. 9, 423–430 (1988).
Schappacher, M. & Deffieux, A. Controlled synthesis of bicyclic “eight-shaped” poly(chloroethyl vinyl ether)s. Macromolecules 28, 2629–2636 (1995).
Hilf, S. & Kilbinger, A. F. M. Functional end groups for polymers prepared using ring-opening metathesis polymerization. Nat. Chem. 1, 537–546 (2009).
Bielawski, C. W., Benitez, D., Morita, T. & Grubbs, R. H. Synthesis of end-functionalized poly(norbornene)s via ring-opening metathesis polymerization. Macromolecules 34, 8610–8618 (2001).
Morgese, G. et al. Next‐generation polymer shells for inorganic nanoparticles are highly compact, ultra‐dense, and long‐lasting cyclic brushes. Angew. Chem. Int. Ed. 56, 4507–4511 (2017).
Ramakrishna, S. N., Morgese, G., Zenobi-Wong, M. & Benetti, E. M. Comblike polymers with topologically different side chains for surface modification: assembly process and interfacial physicochemical properties. Macromolecules 52, 1632–1641 (2019).
Morgese, G., Cavalli, E., Rosenboom, J. G., Zenobi-Wong, M. & Benetti, E. M. Cyclic polymer grafts that lubricate and protect damaged cartilage. Angew. Chem. Int. Ed. 57, 1621–1626 (2018).
Brown, H. A., De Crisci, A. G., Hedrick, J. L. & Waymouth, R. M. Amidine-mediated zwitterionic polymerization of lactide. ACS Macro Lett. 1, 1113–1115 (2012).
Misaka, H. et al. Synthesis of end-functionalized polyethers by phosphazene base-catalyzed ring-opening polymerization of 1,2-butylene oxide and glycidyl ether. J. Polym. Sci. Part A Polym. Chem. 50, 1941–1952 (2012).
Watanabe, K. et al. Intramolecular olefin metathesis as a robust tool to synthesize single-chain nanoparticles in a size-controlled manner. Polym. Chem. 7, 4782–4792 (2016).
Uehara, E. & Deguchi, T. Mean-square radius of gyration and the hydrodynamic radius for topological polymers expressed with graphs evaluated by the method of quaternions revisited. React. Funct. Polym. 133, 93–102 (2018).
Hoskins, N. J. & Grayson, M. S. Synthesis and degradation behavior of cyclic poly(ε-caprolactone). Macromolecules 42, 6406–6413 (2009).
Shin, E. J. et al. Crystallization of cyclic polymers: synthesis and crystallization behavior of high molecular weight cyclic poly(ε-caprolactone)s. Macromolecules 44, 2773–2779 (2011).
Schäler, K. et al. Influence of chain topology on polymer dynamics and crystallization. Investigation of linear and cyclic poly(ε-caprolactone)s by 1H solid-state NMR methods. Macromolecules 44, 2743–2754 (2011).
Xiang, L., Ryu, W., Kim, H. & Ree, M. Precise synthesis, properties, and structures of cyclic poly(ε-caprolactone)s. Polymers 10, 577–606 (2018).
Takizawa, K., Tang, C. & Hawker, C. J. Molecularly defined caprolactone oligomers and polymers: synthesis and characterization. J. Am. Chem. Soc. 130, 1718–1726 (2008).
Zhang, Y., Gao, A., Zhang, Y., Xu, Z. & Yao, W. Aluminum complexes with benzoxazolphenolate ligands: synthesis, characterization and catalytic properties for ring-opening polymerization of cyclic esters. Polyhedron 112, 27–33 (2016).
Acknowledgements
We gratefully acknowledge financial support from the MEXT Grant-in-Aid for Challenging Exploratory Research (16K14000 and 19K22209), Grant-in-Aid for Scientific Research (B) (19H02769), Grant-in-Aid for Scientific Research on Innovative Areas “Hybrid Catalysis” (18H04639 and 20H04798), and JST CREST (Grant Number JPMJCR19T4). This work was, in part, performed under the approval of the Photon Factory Program Advisory Committee (Proposal No. 2017G589 and 2019G579).
Author information
Authors and Affiliations
Contributions
T.I. and T.S. designed the experiment. Y.M. wrote the manuscript. Y.M. and K.H. synthesized and characterized the polymers and analyzed all the data. B.J.R., T.Y., T.D, and K.T. contributed to discussion. K.T., T.I., and T.S. supervised the work. The manuscript was written through the contributions of all authors. All authors have given their approval to the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mato, Y., Honda, K., Ree, B.J. et al. Programmed folding into spiro-multicyclic polymer topologies from linear and star-shaped chains. Commun Chem 3, 97 (2020). https://doi.org/10.1038/s42004-020-00355-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42004-020-00355-4
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.