Abstract
Chemical reactions via radical cation intermediates are of great interest in photoredox catalysis and electrosynthesis, while their reactivities are not clearly understood. For example, how the counter anions correlate with the reactivity of radical cations is still ambiguous. Here we report the effect of anions and fluorinated alcohols on the reactivity of organic radical cations in hole catalysis. The addition of salts in a radical cation Diels–Alder reaction under photoredox catalysis demonstrates that common anions significantly decease the efficiency of hole catalysis. The use of 1,1,1,3,3,3-hexafluoroisopropanol (HFIP) restores the reaction efficiency in the presence of salts, presumably due to solvation of the anions by HFIP to reduce their nucleophilicity. These findings enable hole catalysis under electrolytic conditions with greatly improved efficiency. The effect of anions and fluorinated alcohol described in this paper gives important insights on the fundamental understanding for the reactivity of arene radical cations.
Similar content being viewed by others
Introduction
Radical cations, a free radical species generated by one-electron removal from a closed-shell organic molecule, are playing an important role in chemistries involving single-electron transfer (SET) processes. Migration of radical cations (hole) has been a great interest in charge transfer in DNA for the application in nanotechnology1 and in organic devices for photovoltaics2. The recent explosive growth of photoredox catalysis3 and electrosynthesis4 has motivated synthetic organic chemists to understand the reactivity of organic radical cations. Although the radical cation itself is not a newly discovered intermediate5,6, it is still much less established compared to other common intermediate, such as carbocations, carboanions, radicals, and carbenes, presumably due to its high reactivity and short lifetime. In other words, harnessing reactivity of radical cations will open a door to developing novel and unique reactions.
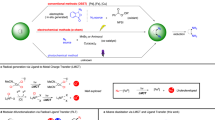
Among various SET-related organic reactions, redox neutral reactions via radical cation intermediates, namely, hole catalysis (Fig. 1)7,8,9, have emerged as an attractive research target owing to their nature of using holes as the “catalyst,”10,11,12 as well as their resulting unique transformations. In these reactions, a radical cation is generated by SET to react with a neutral molecule to form a C–C bond, followed by another SET to give the desired neutral molecule and regenerate the starting radical cation. Hole catalysis is known in peri-cyclization reactions including [2+1]13,14, [2+2]15,16,17,18,19,20, and [3+2]21 cycloadditions and Diels–Alder reactions22,23,24. Olefin metathesis reactions25 and polymerizations26 mediated by radical cations have also been reported. Recently, researchers proposed that radical cations from styrene derivatives react with alcohols and carbonyl groups to realize radical addition via hole catalytic manner, where carbonyl group works as a intermolecular radical acceptor for the first time27. Hole catalytic transformations of molecular switches, such as diarylethene or azobenzene, are also been reported28,29.
In such reactions, radical cations are generated by various methods, including photoredox catalysts14,17,19,21,23, semiconducting nanomaterials30,31, chemical oxidants7,20 and electrochemistry16,24,25. Due to the high reactivity of the radical cation intermediate, the efficiency in hole catalysis is highly dependent on the reaction media. For example, the radical cation-mediated Diels–Alder reaction has been studied intensively with photoredox catalysts and chemical oxidants. In these reactions, non-coordinating solvents such as dichloromethane (DCM) and nitromethane (NM) are commonly preferred, presumably to avoid interaction between the solvent and the electrophilic radical cation intermediate.
In addition to the solvents, anion, which is essential counterpart of radical cations, also should have some effects on the reactivity of the radical cations. To our surprise, there is no precedent of the comprehensive study on the effect of anions for the reactivity of radical cations. Considering that the increasing number of researchers are working on redox-involved organic synthesis, studying the effect of anions will be of great help to ultimately control the reactivity of radical cations.
Based on these ideas, here we report the effect of anions on the reactivity of radical cations in hole catalysis. Anions are found to slow down the reaction depending on their donor numbers. Also, the use of fluorinated alcohols is found to cancel the effect of anions and also increase the reactivity of radical cations.
Results
Radical cation Diels–Alder reaction under various oxidation conditions
We first examined the Diels–Alder reaction of trans-anethol (1) and isoprene (2) with various oxidation methods, that is, photoredox catalysis, chemical oxidant, and electrochemistry (Fig. 2). Intriguingly, our trial of an electrochemical Diels–Alder reaction in DCM afforded the desired product 3 in quite low yield, on the contrary to the high-yielding results in photoredox and chemical oxidation systems. This difference in reaction efficiency between electrochemical and other oxidation systems caught our attention. We attributed this to the supporting electrolyte, which is one of the most significant differences between these systems. Considering that an electron-deficient radical cation is the key intermediate in hole catalysis, the effect of the anion in the electrolyte, which is usually regarded as an inert component in the electrosynthesis, could be significant.
Radical cation Diels–Alder reaction with various oxidation methods. a Mechanism for radical cation-mediated Diels–Alder reaction. b Radical cation Diels–Alder reactions by photoredox catalyst (data from ref. 23), chemical oxidant, and electrochemical oxidation (original data). BArF24 = tetrakis[3,5-bis(trifluoromethyl)phenyl]borate]. CF = carbon felt
Radical cation Diels–Alder reaction with salts and fluorinated alcohols
To determine the effect of anions in hole catalysis, we performed radical cation Diels–Alder reactions of 1 and 2 using a Ru-based photoredox catalyst in the presence of various Bu4NX salts (X = TsO−, TfO−, ClO4−, BF4−, PF6−, BArF20−, in order of reported donor numbers32,33) [BArF20− = tetrakis(pentafluoro)phenyl borate]. Based on the report by Yoon and co-workers23, we chose the [Ru(bpz)3](PF6)2 (bpz = 2,2′-bipyrazine) complex as the photoredox catalyst and NM as the solvent (Fig. 3a). We could not use DCM here due to the poor solubility of the Ru complex with the counter anions we employed, with the exception of BArF20−.
Photoredox radical cation Diels–Alder reaction with salts and 1,1,1,3,3,3-hexafluoroisopropanol (HFIP) additives. a Scheme of radical cation-mediated Diels–Alder reaction using Ru(bpz)3(PF6)2 photoredox catalyst. Additive: 0.1 M Bu4NX (X = TsO−, TfO−, ClO4−, BF4−, PF6−, and BArF20−) or 0.5 M of HFIP or both. b Summary of the yield of 3 in the presence and absence of salt and HFIP. Ru catalyst precipitated out when Bu4NTsO was added in the absence of HFIP. c Relationship between yield of 3 and log10([HFIP]) in the presence of 0.1 M Bu4NTfO
In the absence of the salt, 3 was obtained in 86% yield. When various salts were added to the reaction mixture, the product yield decreased in all cases (Fig. 3b, red bars). In the case of TsO− salt, the Ru salt precipitated out of the NM solution such that no reaction occurred. All other systems were homogeneous, and the decrease in the product yield was roughly aligned with the order of the reported donor numbers32,33. These results suggest that the more donating the anions are, the more strongly they interact with the generated radical cation to inhibit the hole catalysis.
To improve the reaction efficiency in the presence of additional anions, one can imagine that the solvation of the anions could suppress their interactions with the radical cations. Based on this idea, 1,1,1,3,3,3-hexafluoroisopropanol (HFIP) was chosen as an additive due to its strong ability as hydrogen bonding donor for solvating anions, its extremely low nucleophilicity, thus leaving radical cations non-coordinated, and its high polarity for dissolving the supporting electrolyte34,35.
Five equivalents (vs. salt) of HFIP was added to all the systems tested above. The yields of 3 were remarkably improved in the presence of anions, while no change was observed in the absence of additional salt (Fig. 3b, green bars). Interestingly, the TsO− system became homogeneous upon the addition of HFIP, presumably due to the solvation of the TsO anion by HFIP such that the Ru catalyst no longer formed insoluble TsO salts.
The use of MeCN as a coordinating solvent dramatically decreased the reaction efficiency, and the addition of HFIP was not helpful to restore the efficiency to the level of the NM system (Supplementary Fig. 7). This result suggests that both coordinating anions and solvents can diminish the reactivity of arene radical cations to decrease the efficiency of the radical cation chain cycle.
Effect of HFIP concentration on the product yield
Although HFIP was employed to solvate anions, it could also interact with the Ru catalyst (via hydrogen bonding to bipyrazine ligand) or 1. To clarify the role of HFIP in this reaction, the yield dependence on the concentration of HFIP was investigated under photoredox conditions (Fig. 3c). When 0.1 M Bu4NTfO was added as a salt, the yield of 3 increased slightly upon the addition of HFIP in the range of 0.2–1.6eq. (vs. salt). At over 1.6eq., the product yield dramatically increased up to quantitative conversion at 12.4eq. Considering that the drastic change in yield was observed around the range of concentrations of the Bu4NTfO salt (0.1 M) and 1 (0.08 M), we could reasonably rule out the activation of the Ru catalyst by HFIP as the dominant reason for this phenomenon. A control experiment with 0.05 M of salt resulted in a lower threshold concentration, indicating that this phenomenon correlates with the concentration of salt (Supplementary Fig. 8). Hence, one role of HFIP is likely the solvation of anions. Additionally, based on the result that the increase in the product yield with more than equimolar quantity of HFIP, it was suggested that higher order aggregate of HFIP contribute to decrease the nucleophilicity of TfO anions. Similar solution phase aggregate is proposed for the activation of hydrogen peroxide36.
Radical cation Diels–Alder reaction with various dienophiles
Diels–Alder reactions with other combinations of dienophiles and dienes were also investigated (Fig. 4). The results showed that the addition of salts decreased the reaction efficiency, whereas they were improved by the addition of HFIP in general. Interestingly, in these cases, HFIP improved the yield even in the absence of additional salts. This result was attributed to the solvation of PF6 anions in the Ru catalyst by HFIP. It is also possible that HFIP stabilize arene radical cations, as often mentioned in the redox chemistry using HFIP34,35.
Summary of the yield of 4–7 in the presence and absence of salt and 1,1,1,3,3,3-hexafluoroisopropanol (HFIP). Reaction conditions were varied depending on the substrates (for details, see Supplementary Table 1). Additive: 0.1 M Bu4NX (X = TfO−, ClO4−, BF4−, and PF6−) or 0.5 M of HFIP or both
Electrochemical analysis
To gain additional insights, the electrochemistry of the arenes was investigated by cyclic voltammetry (CV). Since the radical cation of all the arenes used for Diels–Alder reactions were too short-lived to observe a reversible redox on the time scale of the CV measurement37, 1,4-dimethoxybenzene (8) was employed for this study (Fig. 5). DCM was used as a non-coordinating solvent. The voltammograms of 8 with various supporting electrolytes are shown in Fig. 5a. The oxidation waves of 8 were irreversible in the presence of TfO− and ClO4−. When less donating anions, that is, BF4− and PF6−, were used, the oxidation peak currents decreased and reduction currents appeared. In particular, 8 exhibited a quasi-reversible voltammogram in the presence of PF6−. This result suggests that the stability of the radical cation of 8 strongly depends on the nucleophilicity of the co-existing counter anions. The higher oxidation current and irreversible nature of the oxidation wave in the presence of donating anions was attributed to an oxidative oligomerization of 8 and further oxidation of oligomers.
Cyclic voltammograms of 4 (2 mM) recorded at a scan rate of 100 mV/s. a In 0.1 M Bu4NX/DCM, X = TfO−, ClO4−, BF4−, and PF6−. b In 0.1 M Bu4NTfO/DCM + 1,1,1,3,3,3-hexafluoroisopropanol (HFIP). Dotted line indicates CV of 4 in neat HFIP for comparison. c In 0.1 M Bu4NX/MeCN, X = TfO−, ClO4−, BF4−, and PF6−. d In 0.1 M Bu4NTfO/MeCN + HFIP. In b, d), 1–15eq. of HFIP was successively added to the supporting electrolyte
Cyclic voltammograms of 8 recorded in 0.1 M Bu4NTfO/DCM with successive addition of HFIP are shown in Fig. 5b, along with that recorded in neat HFIP for comparison. As expected, upon the addition of HFIP to an electrolytic solution, oxidation peak currents decreased and reduction waves appeared. This result indicates that HFIP solvates TfO− to lower its nucleophilicity. Over 5eq. of HFIP to the supporting electrolyte was sufficient to restore the quasi-reversible voltammogram. However, unlike the experiment in Fig. 5a, the oxidation onset potential shifted in the negative direction upon increasing the amount of HFIP. Such a shift in oxidation potential in the presence of HFIP is usually translated as a result of the stabilization of the radical cation species17, but how the radical cations are stabilized in HFIP is still ambiguous. Our CV measurements suggests that HFIP stabilizes the radical cation in two different ways, by trapping the anions and suppressing the formation of the ion pair, resulting in the quasi-reversible redox of 8, and also by interacting with the substrate to energetically stabilize the radical cation, which induces a shift in oxidation potential.
The CV measurement of 8 was also performed in MeCN (Fig. 5c, d). In contrast to the case of DCM, successive addition of HFIP to 8 and 0.1 M Bu4NTfO/MeCN did not improve the reversibility nor affect the oxidation onset potential (Fig. 5d). This result suggests that a coordinating solvent such as MeCN interacts with the radical cation such that the stabilization of the radical cation by HFIP is negated.
Although a reversible redox of the dienophiles was not observed on the time scale of the CV under any conditions, the oxidation behavior of 1 changed depending on the solvent and addition of HFIP, in accordance with the CV result of 8 (Fig. 6). The use of less coordinating anions as supporting electrolyte did not change the redox chemistry of 1, but the addition of HFIP lowered the oxidation potential (Fig. 6a, b). In MeCN, the oxidation potential of 1 did not shift as much as in DCM (Fig. 6c, d). Same effect by HFIP in DCM was also observed for other dienophiles (Supplementary Fig. 4). Interestingly, even the dienophile with a hydrogen bond acceptor, an acetoxy group, resulted in a lowered oxidation potential in the presence of HFIP. Another fluorinated alcohol, 2,2,2-trifluoroethanol (TFE), also showed a similar effect on the redox chemistry of 1 (Supplementary Figs. 5, 6).
Cyclic voltammograms of 1 (2 mM) recorded at a scan rate of 100 mV/s. a In 0.1 M Bu4NX/DCM, X = TfO−, ClO4−, BF4−, and PF6−. b In 0.1 M Bu4NTfO/DCM + HFIP. c In 0.1 M Bu4NX/MeCN, X = TfO−, ClO4−, BF4−, and PF6−. d In 0.1 M Bu4NTfO/MeCN + HFIP. In b, d, 1–15eq. of HFIP as the supporting electrolyte was successively added
DFT calculations
Computational simulations based on density functional theory (DFT) were performed. 1·+ with corresponding anions were optimized under UB3LYP/6–31G(d) basis set. Single-point calculation was carried out with UB3LYP/6–311 + G(2df, 2p) basis set. We obtained the most stable geometries for each combinations of 1·+, and anions were further optimized under UB3LYP/6–31 + G(d, p) basis set to simulate the potential mapping and orbital energy levels. Comparison of energy levels obtained from 6–31G(d) and 6–31 + G(d, p) level basis sets is described in Supplementary Fig. 15. Figure 7a shows optimized structures and static potential mapping of the optimized conformations of 1·+ with the corresponding anions. In optimized structure, more donating anions such as TsO−, TfO−, and ClO4− strongly interacted with β-carbon of 1·+, while BF4− and PF6− left 1·+ intact. This resulted in more pronounced charge separation in the latter case. Similar tendency was observed in the analysis on molecular orbitals (Fig. 7b). Molecular orbital analysis suggested that the singly occupied molecular orbital (SOMO) and the lowest unoccupied molecular orbital (β-LUMO) were significantly affected by the donor number of the anions, resulting in lowered SOMO and β-LUMO energy levels with less donating anions (Fig. 4b). These results suggest that the more donating anions are, the more influence they have on the electronic structure of 1·+. It should be noted that the actual state of ion pair of 1·+ and anion in solution must be dynamic unlike simulation, so that the orbital energy will be a statistical average of all those states. However, electronic structure of optimized geometry should be a largest portion of that average, so estimating electronic structure of ion pair by DFT calculation is still meaningful.
Density functional theory (DFT) calculation of isoprene or 1+ with various counter anions. a Optimized structure of 1·+ with anions overlaid with static potential mapping (ISO value = 0.004). Red and blue indicate negative and positive charge density, respectively. A common scale was used so that the surfaces can be compared visually. b Molecular orbitals for 2 and 1·+ with anions. For 1·+ with anions, left indicates the α- orbital, and right indicates the β-orbital. Calculations were conducted B3LYP/6–31 + G(d, p) level basis set
Hole catalysis by electrochemistry
Finally, we carried out an electrochemical radical cation Diels–Alder reaction using fluorinated alcohols (Fig. 8, Table 1). As mentioned at the beginning of this paper, the electrochemical Diels–Alder reaction was not successful when 0.1 M Bu4NTfO/DCM was used as an electrolyte (Table 1, entry 1). When TFE was used as the solvent, 3 was obtained in high yields in the presence of TfO− or TsO− anion (Table 1, entries 2, 3). The use of HFIP as a solvent gave the desired product in high yield, where lower charge was required when less donating anions were used (Table 1, entries 4–6). HFIP was also used as an additive in DCM. Compared to the result without HFIP, the product yield was significantly improved (Table 1, entries 1, 9).
Electrochemical radical cation Diels–Alder reaction. Full conditions are given in Table 1
The radical cation catalyzed [2+2] reaction in fluorinated solvent was also examined (Supplementary Table 2). Our previous study showed that a larger amount of charge was required for the completion of the [2+2] cyclization reaction compared to the radical cation Diels–Alder reaction, presumably due to the slower kinetics of cyclobutane ring formation9. [2+2] Cycloaddition reactions in TFE gave the desired product with a maximum yield of 66%, but much lower yields were observed in most cases (Supplementary Table 2, entries 1–4). On the other hand, the reaction in HFIP with Bu4NOTf or Bu4NClO4 gave 90% yields after 1 F/mol of charge was passed (Supplementary Table 2, entries 5, 6). The use of HFIP as an additive in DCM did not give desired product (Supplementary Table 2, entry 7). The difference in product yield between HFIP and TFE was attributed to the difference in the degree of radical cation stabilities, implied in the CV measurements (Supplementary Figs. 5, 6).
Discussion
We have demonstrated the effect of anions and fluorinated alcohols on the efficiency of hole catalysis. The effect of anions was analyzed by CV measurement and DFT calculation. CV measurement of model compound 8 demonstrated that the more donating anions were, the shorter the lifetimes of radical cations were. DFT calculation indicated that donating anions are more influential on the electronic structure of 1·+. Both of these were supposedly responsible for the diminished reaction efficiency in hole catalysis. It was also determined that fluorinated alcohols, such as HFIP, can negate the effect of the anions, presumably by decreasing their nucleophilicity via hydrogen bonding interactions. Finally, we demonstrated hole catalytic Diels–Alder reaction and [2 + 2] cycloaddition reaction under electrochemical condition. The supporting salt is an essential component in electrochemistry, thus the idea of using fluorinated alcohols in order to suppress the effect of anions to preserve the inherent reactivity of the organic radical cations can bridge the gap between electrosynthetic chemistry and other oxidation chemistries. It is noteworthy that recent development of electrosynthesis achieved various valuable chemical transformation by the aid of fluorinated alcohols, while its role is not clearly understood38,39,40,41,42,43,44. This work shed light on the effect of anions and fluorinated alcohols to the radical cation intermediate, and we believe that our findings will give new direction to develop novel reaction systems.
Methods
General considerations
All reagents and solvents were purchased from commercial sources and used without further purification. (E)-tert-butyldimethyl(4-(prop-1-en-1-yl)phenoxy)silane and 1-acetoxy-4-propenylbenzene were synthesized from trans-anethol (1) in two steps. Ru(bpz)3(PF6)2 was synthesized according to reported procedure45. Proton nuclear magnetic resonance (1H-NMR) spectra were collected on 600 or 400 MHz NMR spectrometers using the deuterated solvent as an internal deuterium reference. All cyclic voltammograms were recorded using a glassy carbon working electrode, a platinum counter electrode, and an Ag/AgCl reference electrode at a scan rate of 100 mV/s. Synthetic procedures are given in the Supplementary Methods. For 1H-NMR of synthesized materials, see Supplementary Figs. 1–3. CVs recorded in various conditions are shown in Supplementary Figs. 4–6.
General procedure for radical cation mediated Diels–Alder reaction using Ru photoredox catalyst
In a vial, 1 (0.08 M), 2 (0.24 M), and Ru catalyst (0.5 mol% vs. 1) were dissolved in NM (4.2 mL). Bu4NX (0.1 M) (X = TsO−, TfO−, ClO4−, BF4−, PF6−, BArF20−) [BArF20− = tetrakis(pentafluoro)phenyl borate]) and/or 0.5 M HFIP were also added for comparison. The reaction mixture was irradiated with 23 W visible light and reacted for 15 min under stirring (1000 rpm). After the reaction, the solvent was removed in vacuo, and the crude mixture was dissolved into CDCl3. The product yield was determined by 1H-NMR using benzaldehyde as an internal standard, according to the previously reported 1H-NMR spectrum of 323.
Photoredox Diels–Alder reaction in MeCN with salts and HFIP were performed under the same conditions mentioned above, except the solvent. The summary of the reaction is shown in Supplementary Fig. 7.
Photoredox radical cation Diels–Alder reactions with various concentration of HFIP were perfomed under the above-mentioned conditons with Bu4NTfO (0.1 or 0.05 M) and 0.2–12.4 of HFIP (vs. Bu4NTfO). The product yields under these conditions are summarized in Fig. 3c and Supplementary Fig. 8.
Radical cation Diels–Alder reaction with other dienophile and dienes were performed under the similar conditions. Specific conditions for each reaction are described in Supplementary Table 1.
1H-NMR data for photoredox catalysis are shown in Supplementary Figs. 9–13.
Hole catalysis under by electrochemical oxidation
All reactions were carried out in 10 mL of electrolyte solution with two electrode system at room temperature under a constant potential electrolysis using carbon felt as working and counter electrodes. Yields were determined by 1H-NMR using benzaldehyde as an internal standard. Conditions and yields for Diels–Alder reactions and [2 + 2] reaction are summarized in Table 1 and Supplementary Table 2, respectively.
DFT calculations
All calculations were performed using the Gaussian 13 software. Geometry optimizations and frequency calculations were performed for all of the tellurophene derivatives at the UB3LYP level of theory using 6–31G(d) basis set for all atoms. Frequency calculations were performed with the same basis set with scaling factor of 0.977. Single-point calculation was conducted using UB3LYP/6–311 + (2df, 2p) basis set. Optimized geometry of 1·+ with anions were obtained by using four deferent initial geometries for optimization (Supplementary Fig. 14), and compared the structural energy after taking zero-point energy in account. The most stable geometries were β-carbon bound form for more coordinating anions (TsO, TfO, and ClO4). For these anions, second most stable geometries were found when anions stayed near methoxy group, which were ca. 4 kJ/mol unstable. For less coordinating anions (BF4 and PF6), anions stayed outer sphere of 1·+. We obtained the most stable geometries for each combinations of 1·+, and anions were further optimized under UB3LYP/6–31 + G(d, p) level basis set to simulate the potential mapping and orbital energy levels. Comparison of orbital energy diagram calculated with UB3LYP/6–31G(d) and UB3LYP/6–31 + G(d, p) basis sets are shown in Supplementary Fig. 15. Cartesian coordinates and structural images of optimized molecules are shown in Supplementary Tables 3–14 and Supplementary Figs. 16–27, respectively.
Data availability
All other relevant source data are available from the corresponding author upon reasonable request.
References
Genereux, J. C. & Barton, J. K. Mechanisms for DNA charge transport. Chem. Rev. 110, 1642–1662 (2010).
Lin, Y., Li, Y. & Zhan, X. Small molecule semiconductors for high-efficiency organic photovoltaics. Chem. Soc. Rev. 41, 4245–4272 (2012).
Prier, C. K., Rankic, D. A. & MacMillan, D. W. Visible light photoredox catalysis with transition metal complexes: applications in organic synthesis. Chem. Rev. 113, 5322–5363 (2013).
Yoshida, J. I., Kataoka, K., Horcajada, R. & Nagaki, A. Modern strategies in electroorganic synthesis. Chem. Rev. 108, 2265–2299 (2008).
Kita, Y., Tohma, H., Inagaki, M., Hatanaka, K. & Yakura, T. A novel oxidative azidation of aromatic compounds with hypervalent iodine reagent, phenyliodine(III) bis(trifluoroacetate) (PIFA) and trimethylsilyl azide. Tetrahedron Lett. 32, 4321–4324 (1991).
Kita, Y. et al. Hypervalent iodine-induced nucleophilic substitution of para-substituted phenol ethers. generation of cation radicals as reactive intermediated. J. Am. Chem. Soc. 116, 3684–3691 (1994).
Bauld, N. L. et al. Cation radical pericyclic-reactions. Acc. Chem. Res. 20, 371–378 (1987).
Ischay, M. A. & Yoon, T. P. Accessing the synthetic chemistry of radical ions. Eur. J. Org. Chem. 2012, 3359–3372 (2012).
Okada, Y. & Chiba, K. Redox-tag processes: Intramolecular electron transfer and its broad relationship to redox reactions in general. Chem. Rev. 118, 4592–4630 (2018).
Studer, A. & Curran, D. P. The electron is a catalyst. Nat. Chem. 6, 765–773 (2014).
Studer, A. & Curran, D. P. Organocatalysis and C-H activation meet radical- and electron-transfer reactions. Angew. Chem. Int. Ed. 50, 5018–5022 (2011).
Luca, O. R., Gustafson, J. L., Maddox, S. M., Fenwick, A. Q. & Smith, D. C. Catalysis by electrons and holes: formal potential scales and preparative organic electrochemistry. Org. Chem. Front. 2, 823–848 (2015).
Stufflebeme, G., Lorenz, K. T. & Bauld, N. L. Cation radical chain cyclopropanation. J. Am. Chem. Soc. 108, 4234–4235 (1986).
Sarabia, F. J. & Ferreira, E. M. Radical cation cyclopropanations via chromium photooxidative catalysis. Org. Lett. 19, 2865–2868 (2017).
Bauld, N. L. & Pabon, R. Cation radical catalyzed olefin cyclodimerization. J. Am. Chem. Soc. 105, 633–634 (1983).
Chiba, K., Miura, T., Kim, S., Kitano, Y. & Tada, M. Electrocatalytic intermolecular olefin cross-coupling by anodically induced formal [2+2] cycloaddition between enol ethers and alkenes. J. Am. Chem. Soc. 123, 11314–11315 (2001).
Ischay, M. A., Lu, Z. & Yoon, T. P. [2+2] cycloadditions by oxidative visible light photocatalysis. J. Am. Chem. Soc. 132, 8572–8574 (2010).
Perkowski, A. J. & Nicewicz, D. A. Direct catalytic anti-Markovnikov addition of carboxylic acids to alkenes. J. Am. Chem. Soc. 135, 10334–10337 (2013).
Riener, M. & Nicewicz, D. A. Synthesis of cyclobutane lignans via an organic single electron oxidant-electron relay system. Chem. Sci. 4, 1–10 (2013).
Colomer, I., Coura Barcelos, R. & Donohoe, T. J. Catalytic hypervalent iodine promoters lead to styrene dimerization and the formation of tri- and tetrasubstituted cyclobutanes. Angew. Chem. Int. Ed. 55, 4748–4752 (2016).
Sarabia, F. J., Li, Q. & Ferreira, E. M. Cyclopentene annulations of alkene radical cations with vinyl diazo species using photocatalysis. Angew. Chem. Int. Ed. 57, 11015–11019 (2018).
Pabon, R. A., Bellville, D. J. & Bauld, N. L. Cation radical Diels–Alder reactions of electron-rich dienophiles. J. Am. Chem. Soc. 105, 5158–5159 (1983).
Lin, S., Ischay, M. A., Fry, C. G. & Yoon, T. P. Radical cation Diels–Alder cycloadditions by visible light photocatalysis. J. Am. Chem. Soc. 133, 19350–19353 (2011).
Okada, Y., Yamaguchi, Y., Ozaki, A. & Chiba, K. Aromatic “Redox tag”-assisted Diels–Alder reactions by electrocatalysis. Chem. Sci. 7, 6387–6393 (2016).
Miura, T., Kim, S., Kitano, Y., Tada, M. & Chiba, K. Electrochemical enol ether/olefin cross-metathesis in a lithium perchlorate/nitromethane electrolyte solution. Angew. Chem. Int. Ed. 45, 1461–1463 (2006).
Ogawa, K. A., Goetz, A. E. & Boydston, A. J. Metal-free ring-opening metathesis polymerization. J. Am. Chem. Soc. 137, 1400–1403 (2015).
Pitzer, L., Sandfort, F., Strieth-Kalthoff, F. & Glorius, F. Intermolecular radical addition to carbonyls enabled by visible light photoredox initiated hole catalysis. J. Am. Chem. Soc. 139, 13652–13655 (2017).
Goulet-Hanssens, A. et al. Hole catalysis as a general mechanism for efficient and wavelength-independent Z→E azobenzene isomerization. Chem 4, 1740–1755 (2018).
Peters, A. & Branda, N. R. Electrochromism in photochromic dithienylcyclopentenes. J. Am. Chem. Soc. 125, 3404–3405 (2003).
Zhao, Y. & Antonietti, M. Visible-light-irradiated graphitic carbon nitride photocatalyzed Diels–Alder reactions with dioxygen as sustainable mediator for photoinduced electrons. Angew. Chem. Int. Ed. 56, 9336–9340 (2017).
Okada, Y., Maeta, N., Nakayama, K. & Kamiya, H. TiO2 photocatalysis in aromatic “redox tag”-guided intermolecular formal [2+2] cycloadditions. J. Org. Chem. 83, 4948–4962 (2018).
Schmeisser, M., Illner, P., Puchta, R., Zahl, A. & Van Eldik, R. Gutmann donor and acceptor numbers for ionic liquids. Chem. Eur. J. 18, 10969–10982 (2012).
Geiger, W. E. & Barrière, F. Organometallic electrochemistry based on electrolytes containing weakly-coordinating fluoroarylborate anions. Acc. Chem. Res. 43, 1030–1039 (2010).
Colomer, I., Chamberlain, A. E. R., Haughey, M. B. & Donohoe, T. J. Hexafluoroisopropanol as a highly versatile solvent. Nat. Rev. Chem. 1, 88 (2017).
Eberson, L., Hartshorn, M. P., Persson, O. & Radner, F. Making radical cations live longer. Chem. Commun. 2105–2112 (1996).
Berkessel, A. & Adrio, J. A. Dramatic acceleration of olefin epoxidation in fluorinated alcohols: Activation of hydrogen peroxide by multiple H-bond networks. J. Am. Chem. Soc. 128, 13412–13420 (2006).
Colomer, I., Batchelor-Mcauley, C., Odell, B., Donohoe, T. J. & Compton, R. G. Hydrogen bonding to hexafluoroisopropanol controls the oxidative strength of hypervalent iodine reagents. J. Am. Chem. Soc. 138, 8855–8861 (2016).
Kirste, A., Schnakenburg, G., Stecker, F., Fischer, A. & Waldvogel, S. R. Anodic phenol-arene cross-coupling reaction on boron-doped diamond electrodes. Angew. Chem. Int. Ed. 49, 971–975 (2010).
Elsler, B. et al. Source of selectivity in oxidative cross-coupling of aryls by solvent effect of 1,1,1,3,3,3-hexafluoropropan-2-ol. Chem. Eur. J. 21, 12321–12325 (2015).
Kawamata, Y. et al. Scalable, electrochemical oxidation of unactivated C-H bonds. J. Am. Chem. Soc. 139, 7448–7451 (2017).
Wiebe, A., Lips, S., Schollmeyer, D., Franke, R. & Waldvogel, S. R. Single and twofold metal- and reagent-free anodic C−C cross-coupling of phenols with thiophenes. Angew. Chem. Int. Ed. 56, 14727–14731 (2017).
Mei, R., Sauermann, N., Oliveira, J. C. A. & Ackermann, L. Electroremovable traceless hydrazides for cobalt-catalyzed electro-oxidative C–H/N–H activation with internal alkynes. J. Am. Chem. Soc. 140, 7913–7921 (2018).
Zhao, H.-B., Liu, Z.-J., Song, J. & Xu, H.-C. Reagent-free C−H/N−H cross-coupling: regioselective synthesis of N-heteroaromatics from biaryl aldehydes and NH3. Angew. Chem. Int. Ed. 56, 12732–12735 (2017).
Sanjose-Orduna, J., Sarria Toro, J. M. & Perez-Temprano, M. H. HFIP-Assisted C-H functionalization by Cp*Co(III): access to key reactive cobaltacycles and implication in catalysis. Angew. Chem. Int. Ed. 57, 11369–11373 (2018).
Schultz, D. M., Sawicki, J. W. & Yoon, T. P. An improved procedure for the preparation of Ru(bpz)3(PF6)2 via a high-yielding synthesis of 2,2′-bipyrazine. Beilstein. J. Org. Chem. 11, 61–65 (2015).
Acknowledgements
This research was supported in part by JSPS KAKENHI through a Grant-in-Aid for Scientific Research (B; 15H04494) to K.C. N.S. is grateful for a Grant-in-Aid for JSPS Research Fellows (No. 16J07350).
Author information
Authors and Affiliations
Contributions
K.C. conceived and directed the project. N.S., Y.I. and S.N. designed and performed the experiments. K.C., Y.O. discussed results. K.C. and N.S. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shida, N., Imada, Y., Nagahara, S. et al. Interplay of arene radical cations with anions and fluorinated alcohols in hole catalysis. Commun Chem 2, 24 (2019). https://doi.org/10.1038/s42004-019-0125-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42004-019-0125-4
This article is cited by
-
Unimolecular net heterolysis of symmetric and homopolar σ-bonds
Nature (2024)
-
Pushing the boundaries of C–H bond functionalization chemistry using flow technology
Journal of Flow Chemistry (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.