Abstract
Aromaticity has been a central concept in chemistry since the discovery of benzene in the 19th century and has impacted the science of delocalized π-electron systems. The aromaticity of conventional aromatic compounds usually originates from electron delocalization through a single ring that consists of π-symmetric orbitals. Although double aromaticity, i.e. aromaticity composed of two circularly delocalized orbitals, has been theoretically predicted for over 20 years, the double aromaticity of a bench-stable compound is not well explored by experiment. Here we report the synthesis and isolation of the dication of hexakis(phenylselenyl)benzene, as well as its double aromaticity based on structural, energetic, and magnetic criteria. In this dication, cyclic σ-symmetric and π-symmetric delocalized orbitals are formally occupied by ten and six electrons, respectively, and the aromaticity thus follows the 4n + 2 (n = 1, 2,…) electron Hückel rule, regardless of the σ-orbital or π-orbital symmetry.
Similar content being viewed by others
Introduction
Since the discovery of benzene, aromaticity has become one of the most fundamental principles for the understanding of various phenomena in chemistry. Aromaticity is usually derived from conjugated cyclic orbitals that contain 4n + 2 (n = 1, 2,…) electrons, and aromatic compounds exhibit unusually high stability and unique structural characteristics that cannot be rationalized using chemical formulae based on the formalism of organic chemistry developed prior to the discovery of aromaticity. For a long time, aromaticity has been considered to emerge exclusively from interactions between π-symmetric orbitals; however, this symmetry constraint has meanwhile been expanded to include interactions of σ-type1,2,3, δ-type4,5,6, and φ-type symmetry7. Since double aromaticity arising from σ-orbital and π-orbital interactions has been proposed by Schleyer8, compounds exhibiting double aromaticity have been predicted theoretically9,10,11,12,13,14,15,16,17, and metal clusters and compounds with monocyclic carbon/boron rings that bear double aromaticity, generated in the gas phase, have already been characterized18,19,20,21,22,23. The concept of the σ-double and π-double aromaticity has also been discussed in the synthetic investigation of an anionic C2B3 ring compound24. However, the structural characterization of the double aromaticity of a bench-stable compound remains to be targeted.
Herein, we report the experimental evaluation of double aromaticity using the bench-stable hexakis(phenylselenyl)benzene dication, which exhibits double aromaticity caused by σ-orbital and π-orbital interactions that are consistent with the structural, energetic, and magnetic criteria based on the established definition of aromaticity. An X-ray diffraction analysis of this dication exhibited that the distances between adjacent selenium atoms converge to average values, and that simultaneously the C−C bond distances in the core benzene ring remain unaffected. We have also examined the ring currents due to the cyclic σ-orbitals and π-orbitals that contain 10 and 6 electrons, respectively: both induced ring currents decrease the external magnetic field. Our results demonstrate that the conventional Hückel rule, i.e., that aromaticity arises from π-electron delocalization, is also applicable to double aromaticity on account of the delocalization of σ-electrons and π-electrons. The experimental results presented herein may afford new arguments for the discussion on “what is aromaticity?”25,26,27,28. Currently, this issue, which has been ongoing since the nineteenth century, has become a hot topic, and should allow the chemical community to attain a better understanding of electronic structures. Moreover, the delocalization of electrons in orthogonal σ-orbitals and π-orbitals could become a strategy for the formation of multi-dimensional charge-transportation systems in the condensed phase.
Results
Molecular design for exhibiting double σ-aromaticity and π-aromaticity
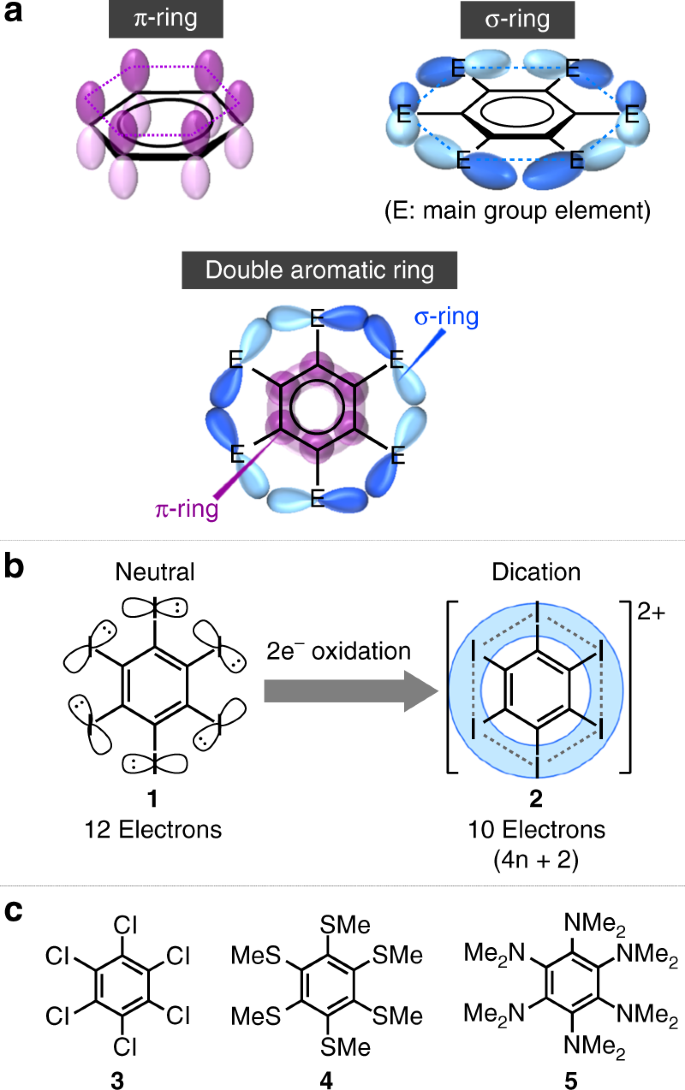
The introduction of six lone-pair-bearing heteroatom functionalities onto the benzene ring carbon atoms is of crucial importance for the creation of compounds that exhibit double σ-aromaticity and π-aromaticity. The central benzene ring provides π-orbitals that are circularly delocalized over the benzene platform, while the lone-pair electrons in the p-orbitals of the heteroatoms create σ-orbitals that are delocalized in the periphery of the benzene core (Fig. 1a). Martin has reported the synthesis of hexaiodobenzene dication 2 from the two-electron oxidation of hexaiodobenzene 129, which contains 12 electrons in the σ-orbitals of six lone pairs on the six iodine atoms (Fig. 1b)30. Dication 2 satisfies the Hückel rule as it contains 10 (4n + 2; n = 2) electrons in circularly delocalized σ-orbitals. However, on account of the prohibitively low solubility of 2, an X-ray diffraction analysis, which is one of the most informative experimental analysis techniques for the determination of the molecular structure, remains elusive. Alternative prospective precursors for double σ-aromatic and π-aromatic compounds are the dications of hexachlorobenzene (3) and hexakis(methylsulfanyl)benzene (4). However, triplet ground states have been predicted for these dications, and accordingly, their isolation as stable entities in the condensed phase should be relatively difficult (Fig. 1c)31,32,33. The two-electron oxidation of hexakis(dimethylamino)benzene (5) afforded the corresponding dication, which revealed a alternation of the C−C bonds in the central benzene ring34, indicative of a considerable contribution from a quinoidal resonance structure, which indicates the absence of double aromaticity.
Double aromaticity arising from σ-orbital and π-orbital interactions in hexaheteroatom-substituted benzenes. a Schematic illustration of a double aromatic ring and its π-aromatic and σ-aromatic rings, which consist of six vertically or perpendicularly arranged p-orbitals on a central benzene framework, respectively. b Synthesis of hexaiodobenzene dication 2 from the two-electron oxidation of the neutral precursor 1. c Chemical structures of 3, 4, and 5 as potential precursors for double σ-aromatic and π-aromatic compounds
A central design feature for the development of bench-stable double σ-aromatic and π-aromatic compounds that can be characterized by single-crystal X-ray diffraction analysis is the introduction of six arylselenyl groups on the benzene ring. Given the relatively large atomic radius of selenium, each selenium atom can interact with its adjacent selenium atoms to produce a circular array of σ-orbitals on the benzene periphery. The substituents on the selenium atoms should suppress molecular aggregation, and thus increase solubility, which would facilitate the generation of single crystals by recrystallization. Furthermore, electron-donating phenylselenyl groups should raise the energy of the highest occupied molecular orbital (HOMO), and thus facilitate the two-electron oxidation of the resulting molecule. The oxidation of hexakis(phenylselenyl)benzene (6) was independently investigated by Martin35. However, an experimental evaluation of the double aromaticity of 6 was not carried out due to a lack of structural information. The clarification of the double aromaticity of the dication of 6 (7) thus remains challenging and fundamentally important.
Preparation of dication 7 and its characterization
We synthesized 7 via a two-electron oxidation of precursor 636. Treatment of 6 with two equivalents of nitrosonium hexafluoroantimonate afforded the targeted dication 7 in 73% yield as a blue-green solid (Fig. 2a). The product is sufficiently stable to be stored in a vial under ambient conditions. Single crystals of 7 were obtained from the slow diffusion of hexane into a concentrated dichloromethane solution of 7. An X-ray diffraction analysis of these single crystals revealed that the molecular structure contains two hexafluoroantimonate counter anions, indicating a dicationic nature for the hexasubstituted benzene unit (Fig. 2b and Supplementary Data 1).
Based on structural criteria, we concluded that dication 7 exhibits double aromaticity. To elucidate the electronic structure of dication 7, four possible resonance forms were considered (Fig. 3a). The anticipated structural characteristics for dication 7 with double aromaticity should be: (i) the distances between adjacent selenium atoms should be comparable, and (ii) the benzene core should exhibit comparable C−C bonds (structure I). The open-shell structure with a triplet state (structure II) should possess two cationic selenium atoms that should be coordinated to their adjacent selenium atoms, resulting in alternating Se−Se distances. The quinoidal structure (structure III) should be characterized by a significant alternation of the C−C bonds in the benzene ring, resulting from enhanced double-bond character of the Se−C (benzene core) bonds. In the structure of the diselenide dication (structure IV), one of the pairs of two adjacent selenium atoms should form a single bond, leading to alternation of the distances between two adjacent selenium atoms. In the following section, the experimentally determined X-ray structure of 7 is compared to these four potential structures.
A structural perspective on the double aromaticity in dication 7. a Four conceivable resonance structures for 7: equalized double aromatic, open shell, and bond-alternating quinoidal structures, as well as a diselenide dication with a Se–Se single bond. b, c Crystal packing structures of 6 and 7; crystals of 6 and 7 contain two structurally independent molecules per unit cell (red: unit A; blue: unit B). d, e Molecular structures of neutral 6 and dication 7 with thermal ellipsoids set at 50% probability; all hydrogen atoms, solvent molecules, and counter anions ([SbF6]–) are omitted
Both neutral 6 and dicationic 7 contain two structurally independent molecules per unit cell (red: unit A; blue: unit B), which are slightly different from each other (Fig. 3b, c). The distances between adjacent selenium atoms fall into a relatively narrow range [3.240(1)–3.338(1) Å] (Table 1), and are much longer than standard Se−Se single bonds (2.29 Å)37. These structural features clearly refute a potential contribution from resonance structure IV to the electronic state of dication 7. The C−C bond distances [1.382(7)–1.399(7) Å] in the benzene core of 7 exhibit negligible alternation, even though they are slightly shorter than those of 6 [1.398(4)–1.405(5) Å]. The distances between the Se and C atoms on the central benzene ring of 7 [1.906(5)–1.912(5) Å] are also slightly shorter than those in 6 [1.916(3)–1.924(3) Å] but much longer than standard Se−C double bonds (1.82–1.86 Å)38. These structural features rule out a potential contribution from quinoidal resonance structure III. As electron paramagnetic resonance (EPR) signals were not observed in a dichloromethane solution of 7 at room temperature, 7 should not adopt a triplet state (structure II). This experimental result is consistent with our previous theoretical prediction that a singlet state of C6(SeH)62+ is more stable than that of a triplet state by 3 kcal mol−133, and is a sharp contrast with the triplet state in benzene dication (C6H62+) having four π-electrons, known as Baird-aromaticity39,40,41. Based on the entirety of the structural characteristics encountered, it is feasible to conclude that 7 exhibits double aromaticity with averaged C−C and Se−Se distances in and around the benzene core, respectively. Another characteristic structural feature that sets apart 6 and 7 are the dihedral angles C1–C2–Se2–CPh (Fig. 3d, e): in 6, angles of 48.4(3)° and 59.3(3)° avoid electronic repulsion between the phenyl groups and the lone pairs on the Se atoms, whereas those in 7 are almost perpendicular [83.1(5)°/96.6(5)°], maximizing the interactions between σ-symmetric p-orbitals on the Se atoms (Fig. 1a).
Evaluation of double σ-aromaticity and π-aromaticity
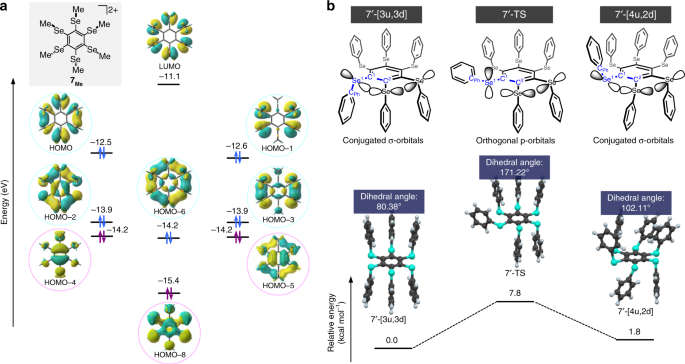
Theoretical calculations using a dispersion-corrected density functional theory (DFT) method on model dication 7Me, which contains six methyl instead of six phenyl groups, revealed that the double aromaticity in the dication is caused by five pairs of circularly arranged σ-type orbitals in the periphery of the benzene core and three pairs of π-type orbitals on the benzene core. The most stable π-type orbital (HOMO-8) does not exhibit any nodes and is delocalized over the entire benzene core, while the degenerate HOMO-4 and HOMO-5 contain one node, and thus represent the second most stable π-type orbitals (Fig. 4a). The arrangement of the molecular orbitals (MOs) clearly indicates π-aromaticity for the benzene core, similar to that in benzene. More interestingly, the σ-type orbitals are delocalized in a similar fashion on the periphery of the benzene core. The most stable σ-type orbital, derived from the 4p orbitals of the Se atoms (HOMO-6), corresponds to the π-orbital with no nodes in the π-aromatic compound. The second and third most stable MOs are nearly degenerate with one and two nodes, respectively. Based on the MO description of the π-aromaticity established by Hückel, it can be concluded that these σ-type orbitals produce σ-aromaticity with 10 (4n + 2; n = 2) electrons. It should be noted that the two types of aromaticity evolve independently, as the σ-type and the π-type orbitals described here are orthogonal with respect to each other.
Calculated molecular orbitals and resonance energies for the σ-aromaticity of dication 7. a Chemical structure of model compound 7Me and its calculated molecular orbitals. Blue and purple circles represent σ-symmetric and π-symmetric occupied orbitals, respectively. b Schematic drawings of the two conformers 7′-[3u,3d] and 7′-[4u,2d], as well as the transition state 7′-TS, together with the p-orbitals on their selenium atoms (top). Calculated structures for 7′-[3u,3d], 7′-[4u,2d], and 7′-TS, as well as relative electronic energies with zero-point corrections (bottom)
We estimated the resonance energy that arises from the σ-aromaticity in dication 7 based on the energy difference between the conformational isomers of the same molecule. To estimate the resonance energy derived from the σ-symmetric orbitals, a conformer that contains one p-orbital orthogonal to the original σ-symmetric conjugated p-orbitals should be an appropriate reference, as the σ-aromaticity should then collapse (Fig. 4b). Accordingly, we used DFT methods to calculate the electronic energies with zero-point corrections for three conformations. In conformer 7′-[3u,3d], three adjacent peripheral phenyl groups are located on the upper side of the central benzene ring, while the other three adjacent phenyl groups are located on the opposite side. A structural optimization of the cationic part (7′) encountered in the crystal structure of 7 suggested that this conformer is the most stable. Conformer 7′-[4u,2d] is obtained from 7′-[3u,3d] by bringing a fourth phenyl group to either side of the central benzene. These two conformers are separated by a small energy difference of 1.9 kcal mol–1. In their optimized structures, the phenyl rings are arranged almost perpendicular to the central benzene ring (7′-[3u,3d]: 80.38°; 7′-[4u,2d]: 102.11°), and their molecular orbitals are circularly delocalized through the σ-symmetric orbitals (Figure S1a,c). The third conformation is the transition state 7′-TS for the conformational transition from 7′-[3u,3d] to 7′-[4u,2d]. In the obtained TS structure, the C2–C1–Se1–Cph dihedral angle (171.22°) induces an almost coplanar arrangement with the central benzene ring, which results in partial delocalization due to the orthogonal p-orbitals on the Se atoms (Supplementary Figure 9b). The energy difference between 7′-[3u,3d] and 7′-TS (8.3 kcal mol–1) should thus correspond to the aromatic stabilization by the σ-symmetric orbitals. A similar energy was obtained from calculations on model compound 7Me and its corresponding conformers (Supplementary Figure 10). In contrast to the σ-system, it is very difficult to estimate the resonance stabilization in ten-electron delocalized π-systems such as [10]annulene derivatives, due to the presence of severe levels of strain in the carbon framework42. In dication 7, the σ-cyclic orbitals are, however, not located on such a covalently bound C–C framework, i.e., the resonance stabilization by σ-delocalization of ten electrons proceeds without increasing the intrinsic levels of strain. An atoms in molecule (AIM) analysis of 7′-[3u,3d] showed six bond critical points (BCPs) between all neighboring Se atoms (Supplementary Figure 11). The Wiberg bond indices (WBIs) of the neighboring Se atoms (Se1···Se2: 0.075; Se2···Se3: 0.075; Se3···Se1*: 0.073) indicate the presence of weak bonding interactions between them. In neutral 6, BCPs were not observed between neighboring Se atoms, and their WBIs (0.011–0.017) are smaller than those of 7′.
The double aromaticity of dication 7 is also corroborated by its magnetic behavior. The ring currents induced under an external magnetic field can be used as a diagnostic magnetic criterion for aromaticity. Assuming that dication 7 exhibits a double aromatic character, a double ring current should be induced by the delocalization of the 6 π-electrons and 10 σ-electrons (Fig. 5a). Based on the optimized geometry of the cationic part 7′, the zz component of the nucleus-independent chemical shift (NICS) was calculated at the center of the central benzene ring [NICS(0)zz]43 (Fig. 5b). The calculated NICS(0)zz value of 7′-[3u,3d] (–25.4 ppm) (Fig. 4b) is more negative than those of 6-[3u,3d] (–7.8 ppm) and benzene (−13.2 ppm). These results indicate that the levels of aromaticity in 7′ should be higher than those of compounds that exhibit only π-aromaticity. The NICS(0)zz value for the neutral transition state 6-TS (–8.6 ppm) was not remarkably different from that of 6-[3u,3d], whereas that of 7′-TS (–9.0 ppm) was significantly less negative than that of 7′-[3u,3d] and comparable to that of 6-TS. The conformational change in 7′ renders one of the σ-symmetric 4p orbitals orthogonal, resulting in a collapse of the σ-aromaticity. Upon further rotation, the σ-aromaticity recovers in 7′-[4u,2d] [NICS(0)zz: –24.0 ppm], which is comparable to that of the 7′-[3u,3d] conformer. To evaluate the contribution of the σ-symmetric MOs to NICS values clearly, we decomposed the NICS(0)zz of the dication 7Me (–31.1 ppm) into the contributions from each MO (CMO-NICS)44. The contribution from five σ-orbitals (HOMO, HOMO–1, HOMO–2, HOMO–3, and HOMO–6 shown in Fig. 4a), π-orbitals, and other σ-orbitals were –12.1 ppm (39%), –25.0 ppm (80%), and 6.1 ppm (–21%), respectively. Therefore, both π and σ aromaticities have comparable contributions to NICS(0)zz values. The ring currents of 6 and 7′ were also analyzed by the anisotropy of the current (induced) density (ACID) method (Fig. 5c, Supplementary Figure 12)45, wherein the applied external magnetic field was oriented perpendicular to the molecular plane defined by the carbon framework of the central benzene ring. Despite the fact that clockwise current density vectors were plotted at the central benzene ring of neutral 6, which usually indicates (diamagnetic) ring currents in aromatic systems, clockwise vectors through the six selenium atoms were not obtained. On the other hand, dication 7′-[3u,3d] showed clockwise vectors through the six Se atoms in addition to the central benzene ring, reflecting the double aromatic nature. These results are consistent with theoretical investigations of the analog C6(SeH)62+, which have been reported by Liégeois et al.46.
A magnetic perspective on the double aromaticity in dication 7. a A schematic illustration of the σ- and π-rings in 7. The π-ring on the central benzene ring contains six electrons, while the σ-ring hosts formally twelve (6) or ten electrons (7) on the Se atoms. b Calculated NICS(0)zz values for 6 and 7′ for the [3u,3d], TS, and [4u,2d] conformations. The NICS(0)zz values of benzene (red dotted line) are shown for comparison. c Molecular structures of 6 and 7′ used for the ACID calculations, and their current density vectors plotted onto the ACID isosurfaces (isosurface value: 0.025). Magnified images are shown in Supplementary Figure 12. d 13C CP/MAS solid-state NMR spectra of 6 (top) and 7 (bottom)
A 13C solid-state nuclear magnetic resonance (NMR) measurements using cross-polarization (CP) and magic-angle spinning (MAS) methods corroborated the double aromaticity of dication 7. In neutral 6, six signals (151.40, 150.02, 149.41, 148.78, 147.89, 145.53 ppm) were observed for the carbon nuclei on the central benzene ring. The signals for the 13C nuclei of the central benzene rings in 6 and 7 were assigned based on the signals of the independently synthesized 13C-enriched compounds 6-13C and 7-13C (Supplementary Figures 7 and 8). In contrast, dication 7 showed only three signals at higher magnetic field (145.84, 141.02, 139.93 ppm). Such an upfield shift would not be expected if the positive charges were delocalized over the π-system of the central C6Se6 skeleton. The decrease of the electron density on the C atoms should cause a downfield resonance of the C nuclei30. Removal of two electrons from the substrate induced a delocalization of the positive charges over the six Se atoms, resulting in the induction of the σ-ring current.
Methods
General considerations for synthesis, characterization and computations
See Supplementary Methods and Supplementary Figures 1–8.
Theoretical calculations on geometric optimization of the cationic part (7′)
See Supplementary Figure 9 and Supplementary Tables 1-3.
Theoretical calculations on 7Me and its conformers
See Supplementary Figure 10 and Supplementary Tables 4-6.
AIM analysis of neutral 6 and dicationic 7
See Supplementary Figure 11.
ACID calculations of 6 and 7′
See Supplementary Figure 12.
Comparison of the geometrical parameters of C6(SePh)6 2+ obtained using different DFT functionals
Data availability
The crystallographic information for this paper has been deposited at The Cambridge Crystallographic Data Center (http://www.ccdc.cam.ac.uk) under reference number CCDC1564760 for compound 7. All other data generated and analyzed during the current study are included in Supplementary Data 1, or are available from the corresponding authors upon reasonable request.
References
Zhang, X., Liu, G., Ganteför, G., Bowen, K. H. & Alexandrova, A. N. PtZnH5 –, A σ-aromatic cluster. J. Phys. Chem. Lett. 5, 1596–1601 (2014).
Zhu, C., Zhou, X., Xing, H., An, K., Zhu, J. & Xia, H. σ-Aromaticity in an unsaturated ring: osmapentalene derivatives containing a metallacyclopropene unit. Angew. Chem. Int. Ed. 54, 3102–3106 (2015).
Jiao, H., Schleyer, Pv. R. & Glukhovtsev, M. N. Are the D mh Symmetric Hx q Rings with 4n + 2 σ-Electrons and Hydrogen Clusters Aromatic? J. Phys. Chem. 100, 12299–12304 (1996).
Wannere, C. S. et al. Evidence for d orbital aromaticity in square planar coinage metal clusters. J. Am. Chem. Soc. 127, 5701–5705 (2005).
Wang, Y. et al. A simple synthesis of triangular all-metal aromatics allowing access to isolobal all-metal heteroaromatics. Chemistry 21, 12271–12274 (2015).
Zhai, H.-J., Averkiev, B. B., Zubarev, D. Y., Wang, L.-S. & Boldyrev, A. I. δ Aromaticity in [Ta3O3]−. Angew. Chem. Int. Ed. 46, 4277–4280 (2007).
Tsipis, A. C., Kefalidis, C. E. & Tsipis, C. A. The role of the 5f orbitals in bonding, aromaticity, and reactivity of planar isocyclic and heterocyclic uranium clusters. J. Am. Chem. Soc. 130, 9144–9155 (2008).
Chandrasekhar, J., Jemmis, E. D. & Schleyer, Pv. R. Double aromaticity: aromaticity in orthogonal planes. The 3,5-dehydrophenyl cation. Tetrahedron Lett. 20, 3707–3710 (1979).
Schleyer, Pv. R., Jiao, H., Glukhovtsev, M. N., Chandrasekhar, J. & Kraka, E. Double aromaticity in the 3,5-dehydrophenyl cation and in cyclo[6]carbon. J. Am. Chem. Soc. 116, 10129–10134 (1994).
Martín-Santamaría, S. & Rzepa, H. S. Double aromaticity and anti-aromaticity in small carbon rings. Chem. Commun. 1503–1504 (2000).
Hofmann, M. & Berndt, A. (π + σ)-Double aromatic and π,σ-mixed aromatic boron compounds with two electrons delocalized over three centers. Heteroat. Chem. 17, 224–237 (2006).
Ou, T. et al. On the structure and bonding in the B4O4 + cluster: a boron oxide analogue of the 3,5-dehydrophenyl cation with π and σ double aromaticity. Phys. Chem. Chem. Phys. 17, 29697–29706 (2015).
Romanescu, C., Galeev, T. R., Li, W.-L., Boldyrev, A. I. & Wang, L.-S. Transition-metal-centered monocyclic boron wheel clusters (M©Bn): a new class of aromatic borometallic compounds. Acc. Chem. Res. 46, 350–358 (2013).
Fowler, P. W., Mizoguchi, N., Bean, D. E. & Havenith, R. W. A. Double aromaticity and ring currents in all-carbon rings. Chemistry 15, 6964–6972 (2009).
Feixas, F., Matito, E., Poater, J. & Solà, M. Metalloaromaticity. WIREs Comput. Mol. Sci. 3, 105–122 (2012).
Zubarev, D. Y., Averkiev, B. B., Zhai, H.-J., Wang, L.-S. & Boldyrev, A. I. Aromaticity and antiaromaticity in transition-metal systems. Phys. Chem. Chem. Phys. 10, 257–267 (2008).
Wodrich, M. D., Corminboeuf, C., Park, S. S. & Schleyer, Pv. R. Double aromaticity in monocyclic carbon, boron, and borocarbon rings based on magnetic criteria. Chemistry 13, 4582–4593 (2007).
Li, X., Kuznetsov, A. E., Zhang, H.-F., Boldyrev, A. I. & Wang, L.-S. Observation of all-metal aromatic molecules. Science 291, 859–861 (2001).
Boldyrev, A. I. & Wang, L.-S. All-metal aromaticity and antiaromaticity. Chem. Rev. 105, 3716–3757 (2005).
Zhai, H.-J., Kiran, B., Li, J. & Wang, L.-S. Hydrocarbon analogues of boron clusters—planarity, aromaticity and antiaromaticity. Nat. Mater. 2, 827–833 (2003).
Zhai, H.-J., Alexandrova, A. N., Birch, K. A., Boldyrev, A. I. & Wang, L.-S. Hepta- and octacoordinate boron in molecular wheels of eight- and nine-atom boron clusters: observation and confirmation. Angew. Chem. Int. Ed. 42, 6004–6008 (2003).
Alexandrova, A. N., Zhai, H.-J., Wang, L.-S. & Boldyrev, A. I. Molecular wheel B8 2– as a new inorganic ligand. Photoelectron spectroscopy and ab initio characterization of LiB8 –. Inorg. Chem. 43, 3552–3554 (2004).
Van Orden, A. & Saykally, R. J. Small carbon clusters: spectroscopy, structure, and energetics. Chem. Rev. 98, 2313–2358 (1998).
Unverzagt, M., Subramanian, G., Hofmann, M., Schleyer, Pv. R., Berger, S., Harms, K., Massa, W. & Berndt, A. Carbene analogues of boron stabilized by neighboring B–B moieties: doubly aromatic bishomotriboriranides. Angew. Chem. Int. Ed. 36, 1469–1472 (1997).
Schleyer, Pv. R. & Jiao, H. What is aromaticity? Pure Appl. Chem. 68, 209–218 (2004).
Stanger, A. What is… aromaticity: a critique of the concept of aromaticity—can it really be defined? Chem. Commun. 1939–1947 (2009).
Hoffmann, R. The many guises of aromaticity. Am. Sci. 103, 18–22 (2015).
Ritter, S. K. Aromaticity for all. Chem. Eng. News 93, 37–28 (2015).
Sagl, D. J. & Martin, J. C. The stable singlet ground state dication of hexaiodobenzene: possibly a σ-delocalized dication. J. Am. Chem. Soc. 110, 5827–5833 (1988).
Martin, J. C. & Schaad, L. J. Sigma-delocalized aromatic species formed from cyclic arrays of hypervalent main-group element species. Pure Appl. Chem. 62, 547–550 (2005).
Wasserman, E., Hutton, R. S., Kuck, V. J. & Chandross, E. A. Dipositive ion of hexachlorobenzene. Ground-state triplet. J. Am. Chem. Soc. 96, 1965–1966 (1974).
Kobayashi, K., Takahashi, O., Namatame, K., Kikuchi, O. & Furukawa, N. σ-Delocalized hexakis(methylthio)benzene dication: a possible intermediate of pummerer-type rearrangement of monooxide of hexakis(methylthio)benzene. Chem. Lett. 27, 515–516 (1998).
Hatanaka, M., Saito, M., Fujita, M. & Morokuma, K. σ-Aromaticity in hexa-group 16 atom-substituted benzene dications: a theoretical study. J. Org. Chem. 79, 2640–2646 (2014).
Chance, J. M., Kahr, B., Buda, A. B., Toscano, J. P. & Mislow, K. Stereochemistry of hexakis(dimethylamino)benzene and its dication. J. Org. Chem. 53, 3226–3232 (1988).
Loop, C. K. The directed ortho-lithiation of lithium phenylsulfide and lithium phenylselenide plus Sigma-aromaticity and the hexakis(phenylseleno)benzene dication. University of Illinois Urbana-Champaign, PhD Thesis http://hdl.handle.net/2142/21941 (1990).
Saito, M. & Kanatomi, Y. Reinvestigation on the synthesis of hexakis(phenylseleno)benzene. J. Sulfur Chem. 30, 469–476 (2009).
Marsh, R. E. The crystal structure for diphenyl diselenide. Acta Cryst. 5, 458–462 (1952).
Otten, P. A., Gorter, S. & Gen, A. V. D. A structural study of selenobenzamides: crystal structures and dynamic 13C NMR. Eur. J. Inorg. Chem. 130, 49–54 (1997).
Ottosson, H. Exciting excited-state aromaticity. Nat. Chem. 4, 969–971 (2012).
Rosenberg, M., Dahlstrand, C., Kilsa, K. & Ottosson, H. Excited state aromaticity and antiaromaticity: opportunities for photophysical and photochemical rationalizations. Chem. Rev. 114, 5379–5425 (2014).
Baird, N. C. Quantum organic photochemistry. II. Resonance and aromaticity in the lowest “ππ“ state of cyclic hydrocarbons. J. Am. Chem. Soc. 94, 4941–4948 (1972).
Gribanova, T. N., Minyaev, R. M. & Minkin, V. I. Stabilization of non-standard conformations of the annulene rings in cyclobutadiene-framed [n]annulenes (n = 8, 10, 12, 14) and their beryllium sandwich-like complexes: a quantum chemical study. Struct. Chem. 27, 1229–1240 (2016).
Fallah-Bagher-Shaldael, H., Wannere, C. S., Corminboeuf, C., Puchta, R. & Schleyer, Pv. R. Which NICS aromaticity index for planar π rings is best? Org. Lett. 8, 863–866 (2006).
Heine, T., Schleyer, Pv. R., Corminboeuf, C., Seifert, G., Reviakine, R. & Weber, J. Analysis of aromatic delocalization: individual molecular orbital contributions to nucleus-independent chemical shifts. J. Phys. Chem. A 107, 6470–6475 (2003).
Geuenich, D., Hess, K., Köhler, F. & Herges, R. Anisotropy of the induced current density (ACID), a general method to quantify and visualize electronic delocalization. Chem. Rev. 105, 3758–3772 (2005).
Rauhalahti, M., Taubert, S., Sundholm, D. & Geois, V. L. X. Calculations of current densities for neutral and doubly charged persubstituted benzenes using effective core potentials. Phys. Chem. Chem. Phys. 19, 7124–7131 (2017).
Acknowledgements
This work was supported by a Grant-in-Aid for Scientific Research on Innovative Areas “π-System Figuration: Control of Electron and Structural Dynamism for Innovative Functions” from the Japan Society for the Promotion of Science (JSPS) (JP26102006 to M.S.) and “Stimuli-responsive Chemical Species for the Creation of Functional Molecules” (15H00918 to S.F. and 15H009382 to K.M.) from the Japanese Ministry of Education, Culture, Sports, Science, and Technology (MEXT). This work was also supported by a Challenging Research Exploratory JSPS KAKENHI grant (16K13945 to S.F.), a Grant-in-Aid for Scientific Research (C) (16K00175 to K.I.) and a Grant-in-Aid for Scientific Research (A) (18H03908 to M.S.). M.S. gratefully acknowledges a research grant from the Yamada Science Foundation. The authors thank Prof. Takashi Fujihara (Saitama Univ.) for assistance with solid-state NMR measurements.
Author information
Authors and Affiliations
Contributions
S.F., M.F., and Y.K. conducted the synthetic experiments and analyzed the data. M.H., K.I., and K.M. performed the computational investigations. M.M. carried out the X-ray diffraction analyses. S.F. and M.S. prepared the manuscript. M.S. conceived the project, which was directed by S.F. and M.S.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Furukawa, S., Fujita, M., Kanatomi, Y. et al. Double aromaticity arising from σ- and π-rings. Commun Chem 1, 60 (2018). https://doi.org/10.1038/s42004-018-0057-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42004-018-0057-4
This article is cited by
-
Theoretical interpretation of electron delocalization potential of the substituent in o-hydroxynaphthalidene-p-substituted anilines for prospective molecular electronics applications
Discover Molecules (2024)
-
A flat carborane with multiple aromaticity beyond Wade–Mingos’ rules
Nature Communications (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.