Abstract
Transmembrane protein 135 (TMEM135) is thought to participate in the cellular response to increased intracellular lipids yet no defined molecular function for TMEM135 in lipid metabolism has been identified. In this study, we performed a lipid analysis of tissues from Tmem135 mutant mice and found striking reductions of docosahexaenoic acid (DHA) across all Tmem135 mutant tissues, indicating a role of TMEM135 in the production of DHA. Since all enzymes required for DHA synthesis remain intact in Tmem135 mutant mice, we hypothesized that TMEM135 is involved in the export of DHA from peroxisomes. The Tmem135 mutation likely leads to the retention of DHA in peroxisomes, causing DHA to be degraded within peroxisomes by their beta-oxidation machinery. This may lead to generation or alteration of ligands required for the activation of peroxisome proliferator-activated receptor a (PPARa) signaling, which in turn could result in increased peroxisomal number and beta-oxidation enzymes observed in Tmem135 mutant mice. We confirmed this effect of PPARa signaling by detecting decreased peroxisomes and their proteins upon genetic ablation of Ppara in Tmem135 mutant mice. Using Tmem135 mutant mice, we also validated the protective effect of increased peroxisomes and peroxisomal beta-oxidation on the metabolic disease phenotypes of leptin mutant mice which has been observed in previous studies. Thus, we conclude that TMEM135 has a role in lipid homeostasis through its function in peroxisomes.
Similar content being viewed by others
Introduction
Dysregulation of lipid metabolism is a hallmark of many pathological conditions including diabetes1, atherosclerosis2, Alzheimer’s disease3, and age-related macular degeneration4. To maintain adequate regulation of lipid metabolism, cells depend on multiple organelles to balance the synthesis and breakdown of lipids. Notably, mitochondria serve as the regulatory hub for lipid metabolism5. Mitochondria possess the enzymatic machinery for beta-oxidation of fatty acids and conversion of these substrates to energy6. In conditions of excess energy production, mitochondria can transport excess metabolites to the cytosol for fatty acid synthesis that can curb fatty acid oxidation7. Furthermore, peroxisomes and endoplasmic reticulum (ER) aid mitochondria in their lipid metabolic duties. For example, peroxisomes exclusively degrade very long-chain and branched-chain fatty acids through beta- and alpha-oxidation8 whereas the ER can degrade medium-chain fatty acids by omega-oxidation9. Peroxisomes can also generate 1-O-alkyl glycerol-3-phosphates for plasmalogen synthesis10 and docosahexaenoic acid (DHA) from essential dietary fatty acids8 while the ER produces membrane lipids11. Emerging evidence suggests that signaling occurs between organelles to preserve lipid homeostasis12. Thus, it is important to elucidate how organelles participate in lipid metabolism, which may lead to better treatments for diseases with disrupted lipid homeostasis.
Transmembrane protein 135 (TMEM135) is a 52 kilodalton protein with five transmembrane domains that is important for murine retinal and cardiac health13,14,15. Multiple proteomic studies have identified TMEM135 as a key component of peroxisomes16,17,18,19, but it is also present in other organelles including mitochondria13,20 and lipid droplets20,21. While no study has defined the molecular function of TMEM135 on these organelles, it has been speculated that TMEM135 may play a role in the cellular stress response to increased intracellular lipids20. This cellular stress response may impinge on mitochondrial dynamics13,15,22,23,24,25, energy expenditure20, and cholesterol degradation26 as these pathways are affected in cells with altered TMEM135 function. In support of the hypothesis that TMEM135 participates in maintaining lipid homeostasis, we have recently found that a mutation in the murine Tmem135 gene increased the expression of genes involved in lipid metabolism in the murine retina27, an organ with unique lipid demands28.
In this study, we investigated the role of TMEM135 in lipid metabolism. We found that a mutation in the Tmem135 gene modified the lipid profiles of the livers, retinas, hearts, and plasmas of mice. Strikingly, we detected reductions in DHA in all tissues of the Tmem135 mutant (Tmem135FUN025/FUN025) mice. Along with the decrease of DHA, we observed increased peroxisomal fatty acid oxidation enzymes and proliferation of peroxisomes that resulted from the activation of peroxisome proliferator-activated receptor a (PPARa) signaling in Tmem135 mutant mice. Furthermore, we confirmed the protective effect of the increases in peroxisomes and their beta-oxidation enzymes by observing amelioration of genetically-induced obesity, dyslipidemia, and fatty liver in mice due to the Tmem135 mutation. In summary, we conclude that TMEM135 is an important protein involved in lipid homeostasis through its function in peroxisomes.
Results
Tmem135 mutation reduces DHA concentrations in mice
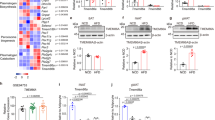
Previous studies have indicated TMEM135 has a role in lipid metabolism20,26,27,29 but how TMEM135 contributes to maintaining lipid homeostasis remains unknown. In this study, we performed a high-throughput and semi-quantitative lipidomics analysis on tissues collected from mice with modified TMEM135 function. We evaluated the lipid profiles of four tissues (livers, hearts, retinas, and plasmas) from 2.5-month-old male wildtype (WT) mice, Tmem135FUN025/FUN025 mutant mice, and mice over-expressing Tmem135 (Tmem135 TG). Using principal components analysis, we found Tmem135FUN025/FUN025 tissues have distinct lipid profiles from WT and Tmem135 TG tissues (Fig. 1a). We focused on investigating the types of lipids altered in Tmem135FUN025/FUN025 tissues and observed many lipid classes were affected by the Tmem135 mutation (Supplementary Fig. 1, Supplementary Table 1). One common result in the Tmem135FUN025/FUN025 tissues was decreased membrane lipids classified as phosphatidylcholine (PC) and phosphatidylethanolamine (PE) (Supplementary Fig. 1, Supplementary Table 1). Remarkably, the Tmem135FUN025/FUN025 livers had accumulations of triglycerides (Supplementary Fig. 1a) while the Tmem135FUN025/FUN025 plasmas had reductions of triglycerides (Supplementary Fig. 1b) that appeared to be unique features of these Tmem135FUN025/FUN025 tissues. Next, we studied the acyl side chains of the lipids that were significantly different in Tmem135FUN025/FUN025 tissues (Supplementary Table 2). We found a large proportion of lipids containing DHA (C22:6n3) decreased across all the Tmem135FUN025/FUN025 tissues used in this study (Fig. 1b). We also observed modifications of lipids containing other fatty acids including C16:0, C16:1, C18:0, C18:1, C18:2, C20:3, C20:4, and C22:5 in the Tmem135FUN025/FUN025 tissues (Supplementary Table 2). However, the changes in these lipids appeared to occur in a tissue-specific manner (Supplementary Table 2). In summary, our lipidomics analysis revealed significant alterations in tissue lipid profiles and reductions of DHA-containing lipids as a common consequence of the Tmem135 mutation in mice.
a Principal component analysis of lipids detected in the positive (+) ion mode and negative (−) ion mode of male WT, Tmem135 TG (TG), and Tmem135FUN025/FUN025 (FUN025) livers, retinas, hearts, and plasmas based on log10 lipid concentrations. Number in parentheses represent the N of independent mouse samples per genotype used in the experiment. b Pie graphs of docosahexaenoic acid (DHA, C22:6n3)-containing lipids that were significantly upregulated (dark green) or downregulated (medium green) as well as unchanged (light green) in Tmem135FUN025/FUN025 tissues compared to WT. Numbers denote the total of lipids within the category. All altered lipid species can be found in Supplementary Tables 6–9. Significance was determined by one-way ANOVA with post hoc Tukey’s test between WT and Tmem135FUN025/FUN025 livers (P < 0.05).
To confirm the decreases of DHA-containing lipids in Tmem135FUN025/FUN025 tissues, we quantified the concentrations of fatty acids in the livers, retinas, hearts, and plasmas of 2.5-month-old WT and Tmem135FUN025/FUN025 mice. We detected significant alterations in the fatty acid composition of the different tissues from the Tmem135FUN025/FUN025 mice (Supplementary Table 3). We found that DHA was robustly decreased in Tmem135FUN025/FUN025 livers (Fig. 2a), retinas (Fig. 2b), hearts (Fig. 2c), and plasmas (Fig. 2d) relative to WT controls. Thus, results from our fatty acid analysis confirmed that the Tmem135 mutation reduces DHA concentrations in multiple murine tissues.
Bar graphs depicting relative moles of docosahexaenoic acid (DHA, C22:6n3) quantified by gas chromatography mass spectrometry (GC-MS) in four 2.5-month-old wildtype (WT) (3 male/1 female) and Tmem135FUN025FUN025 (FUN025) (2 male/2 female) livers (a), retinas (b), hearts (c), and plasmas (d). All fatty acid data can be found in Table S3. **** indicates P < 0.0001 significance by two-way Student’s t test. Number in parentheses represents the N of independent mouse samples per genotype used in the experiment. Dots represent individual data points. Data are presented as mean ± SD.
Reduced DHA in Tmem135 mutant mice is not due to defects in the Sprecher pathway of DHA synthesis
Because reductions in DHA were a common observation in the Tmem135FUN025/FUN025 tissues, we focused on investigating the role of TMEM135 in DHA metabolism. In vivo, DHA is derived from either the conversion of alpha-linolenic acid to DHA within cells via the “Sprecher pathway” or through the consumption of foods enriched in DHA30. Since the diets fed to the mice used in this study do not contain DHA, all DHA present in these mice must originate from endogenous DHA biosynthesis. Although all tissues have the capacity to generate DHA, the main producer of this PUFA species within the mouse is the liver31. To decipher the role of TMEM135 in cellular DHA metabolism, we harvested livers from 2.5-month-old WT and Tmem135FUN025/FUN025 mice and determined the expression level of key components of the Sprecher pathway of DHA synthesis (Fig. 3a)32. We performed qPCR for the desaturases [fatty acid desaturase 1 (Fads1) and 2 (Fads2)] and elongases [elongation of very-long-chain fatty acids-like 2 (Elovl2) and 5 (Elovl5)] required for the desaturation and elongation of dietary essential fatty acid 18:3n3 to generate C24:6n3 in the ER33. We found no significant changes in the expression levels of Fads1, Fads2, Elovl2, and Elovl5 in Tmem135FUN025/FUN025 livers relative to WT (Fig. 3b).
a Schematic of the Sprecher pathway of docosahexaenoic acid (C22:6n3) synthesis from alpha-linolenic acid (C18:3n3) through a series of elongation and desaturation steps in the endoplasmic reticulum (ER) that is finished within peroxisomes via their beta-oxidation machinery. b Gene expression analysis of the ER-localized Fatty acid desaturase 1 (Fads1) and 2 (Fads2) and Elongation of very-long-chain fatty acids-like 2 (Elovl2) and 5 (Elovl5) involved in the Sprecher pathway in the livers of three 2.5-month-old female WT and Tmem135FUN025/FUN025 (FUN025) mice. c Gene expression analysis of ATP binding cassette subfamily D member 2 (Abcd2) in the livers of three 2.5-month-old female WT and Tmem135FUN025/FUN025 mice. Ribosomal protein lateral stalk subunit P0 (Rlplp0) served as the housekeeping gene in these studies. d Western blot analysis of peroxisomal beta-oxidation enzymes including acyl-CoA oxidase 1 (ACOX1), D-bifunctional protein (DBP), acetyl-Coenzyme A acyltransferase 1 (ACAA1), and sterol carrier protein x (SCPx) using liver lysates from 2.5-month-old WT, Tmem135 TG (TG), and Tmem135FUN025/FUN025 mice. ACTB served as the loading control for these experiments. 4 WT (2 males/2 females), 4 Tmem135 TG (2 males/2 females), and 5 Tmem135FUN025/FUN025 (3 males/2 females) were used in these experiments. Asterisks (** and ****) indicates post hoc Tukey test for a P < 0.01 and P < 0.0001 significance following a significant difference detected by one-way ANOVA. Dots represent individual data points. The number in parentheses represents the N of independent mouse samples per genotype used in the experiment. The protein size next to the immunoblot images denotes the size of the immunoband measured for this analysis. These experiments were repeated twice to ensure reproducibility. Data are presented as mean ± SD.
We also performed qPCR for ATP binding cassette subfamily D member 2 (Abcd2) which encodes for the importer of C24:6n3 into the peroxisome34. However, we detected no significant difference in Abcd2 expression in Tmem135FUN025/FUN025 livers relative to WT (Fig. 3c). Next, we utilized Western blot analysis to quantitate peroxisomal beta-oxidation enzymes required for generating C22:6n3 from C24:6n3: acyl-Coenzyme A oxidase 1 (ACOX1), D-bifunctional protein (DBP), acetyl-Coenzyme A acyltransferase 1 (ACAA1), and sterol carrier protein x (SCPx)35. We found a significant increase in ACOX1, DBP, and ACAA1 but no significant change in SCPx between Tmem135FUN025/FUN025 and WT livers (Fig. 3d). Our data shows that the peroxisomal beta-oxidation enzymes of the Sprecher pathway of DHA synthesis are increased in the Tmem135FUN025/FUN025 livers. Taken together, our data suggest that reduced DHA in the Tmem135 mutant mice does not result from a defect in previously-identified components of the Sprecher pathway of DHA synthesis.
Tmem135 mutation increases peroxisomes in mice
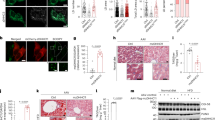
Next, we investigated if the upregulation of peroxisomal beta-oxidation enzymes in Tmem135FUN025/FUN025 livers was due to an increase in peroxisomes. We labeled WT, Tmem135FUN025/FUN025, and Tmem135 TG liver cryosections with an anti-peroxisome biogenesis factor 14 (PEX14) antibody to visualize hepatic peroxisomes (Fig. 4a). After quantifying peroxisomes using a previously described method36, we found an increase of peroxisomes in the Tmem135FUN025/FUN025 livers while there was a decrease of peroxisomes in the Tmem135 TG livers (Fig. 4b). We validated our immunohistochemistry results through Western blot analysis probing for PEX14 (Fig. 4c). We also confirmed these findings by examining the expression of two additional peroxisome markers, peroxisome membrane protein 70 (PMP70) (Supplementary Fig. 2) and catalase (CAT) (Supplementary Fig. 3), in the livers of WT, Tmem135FUN025/FUN025, and Tmem135 TG mice. Next, we investigated if TMEM135 regulates peroxisomal proteins in other tissues of Tmem135FUN025/FUN025 and Tmem135 TG mice. We examined the expression level of PMP70 in the neural retinas, eyecups, and hearts of Tmem135FUN025/FUN025 and Tmem135 TG mice since these tissues displayed decreases of DHA in Tmem135FUN025/FUN025 mice. We detected a significant increase of PMP70 in the Tmem135FUN025/FUN025 neural retinas, eyecups, and hearts while there were decreases of PMP70 in the Tmem135 TG neural retinas, eyecups, and hearts (Supplementary Fig. 4). We further confirmed that the peroxisome number is increased by the Tmem135 mutation using cultured fibroblasts from WT, Tmem135FUN025/FUN025, and Tmem135 TG mice. We performed immunocytochemistry on these cells using an antibody targeting PEX14 to count the number of peroxisomes per cell (Fig. 5a). We found more peroxisomes in Tmem135FUN025/FUN025 fibroblasts and fewer peroxisomes in Tmem135 TG fibroblasts compared to WT fibroblasts (Fig. 5b). We also detected similar changes in PMP70 protein levels between cultured fibroblasts from these genotypes (Supplementary Fig. 5). Our results show an important role of TMEM135 function in maintaining the number of peroxisomes in multiple mouse tissues and cell types.
a Representative 100x immunohistochemical images of PEX14 labeled (green) and DAPI stained (blue) WT, Tmem135 TG (TG), and Tmem135FUN025/FUN025 (FUN025) livers. The white boxes in these images were expanded to highlight differences in PEX14-positive peroxisome staining between these genotypes. Scale bar for images = 50 microns. b Quantitation of PEX14-positive peroxisomes from the 100x images of WT (2 males/2 females), Tmem135 TG (1 male/3 females), and Tmem135FUN025/FUN025 (2 males/2 females) livers using the Analyze Particles function in ImageJ. c Western blot analysis of peroxisome biogenesis factor 14 (PEX14) using livers from 2.5-month-old WT (2 males/2 females), Tmem135 TG (2 males/2 females), and Tmem135FUN025/FUN025 (3 males/2 females) mice. ACTB served as the loading control for this Western blot experiments. Asterisks (* and ***) indicates post hoc Tukey test for a P < 0.05 and P < 0.001 significance following a significant difference detected by one-way ANOVA. Number in parentheses represent the N of independent mouse samples per genotype used in the experiment. Dots represent individual data points. The protein size next to the immunoblot images denotes the size of the immunoband measured for this analysis. Data are presented as mean ± SD.
a Representative 60x immunohistochemical images of peroxisome biogenesis factor 14 (PEX14) labeled (green) and DAPI stained (blue) WT, Tmem135 TG (TG), and Tmem135FUN025/FUN025 (FUN025) fibroblasts. The white boxes in these images were expanded to show differences in the peroxisome number between these cells. Scale bar for images = 50 microns. b Quantitation of PEX14-positive peroxisomes from WT, Tmem135 TG, and Tmem135FUN025/FUN025 livers using the Analyze Particles function in ImageJ. Asterisks (* and ***) indicates post hoc Tukey test for a P < 0.05 and P < 0.001 significance following a significant difference detected by one-way ANOVA. Number in parentheses represent the N of individual fibroblasts per genotype assessed in this experiment. Dots represent individual data points. Data are presented as mean ± SD.
Tmem135 mutation increases PPARa signaling in the murine liver
The increase in peroxisomes observed in the Tmem135FUN025/FUN025 tissues and cells could result from the activation of a peroxisome proliferator-activated receptor (PPAR)37. To determine the contribution of PPAR signaling on the peroxisomal changes in the Tmem135FUN025/FUN025 mice, we crossed Tmem135FUN025/FUN025 mice with peroxisome proliferator-activated receptor alpha knockout (Ppara−/−) mice to generate Tmem135FUN025/FUN025/Ppara−/− mice that do not express one of the main PPAR proteins in the liver38. We performed immunohistochemistry on liver sections from WT, Ppara−/−, Tmem135FUN025/FUN025, and Tmem135FUN025/FUN025/Ppara−/− mice for quantification of the number of PEX14-positive peroxisomes in order to determine the effect of PPARa on peroxisome proliferation due to the Tmem135 mutation (Fig. 6a). Our quantification indicated reduced peroxisomes in Tmem135FUN025/FUN025/Ppara−/− livers compared to Tmem135FUN025/FUN025 livers (Fig. 6b), indicating that activation of PPARa is responsible for peroxisome proliferation in Tmem135FUN025/FUN025 mice. We also examined the expression of additional peroxisomal proteins (PMP70, ACOX1, DBP, and ACAA1) using liver lysates from WT, Ppara−/−, Tmem135FUN025/FUN025, and Tmem135FUN025/FUN025/Ppara−/− mice by Western blot (Fig. 6c). We found that the concentrations of PMP70, ACOX1, DBP, and ACAA1 were lower in Tmem135FUN025/FUN025/Ppara−/− livers than in Tmem135FUN025/FUN025 livers (Fig. 6c). Interestingly, the levels of PMP70 and ACOX1 in Tmem135FUN025/FUN025/Ppara−/− livers remained significantly higher than WT livers (Fig. 6c), suggesting PPARa-independent pathways may also contribute to the increases of PMP70 and ACOX1 in the Tmem135FUN025/FUN025 livers. To further confirm the activation of PPARa signaling in the Tmem135FUN025/FUN025 livers, we measured the level of cytochrome P450, family 4, subfamily a, polypeptide 10 (CYP4A10), a protein whose expression is contingent on PPARa39, in these same liver lysates (Supplementary Fig. 6). We detected a significant increase of CYP4A10 in Tmem135FUN025/FUN025 livers compared to WT controls, while the CYP4A10 level in Tmem135FUN025/FUN025/Ppara−/− livers is comparable to WT controls (Supplementary Fig. 6), supporting the activation of PPARa signaling in the mouse liver due to the Tmem135 mutation. In summary, our data shows the increase of peroxisomes and their proteins in the Tmem135 mutant liver is at least partly mediated by PPARa signaling.
a Representative 100x immunohistochemical images of PEX14 labeled (green) and DAPI stained (blue) 3-month-old WT, Ppara−/−, Tmem135FUN025/FUN025 (FUN025), and Tmem135FUN025/FUN025/Ppara−/− (FUN025/Ppara−/−) livers. The white boxes in these images were expanded to highlight differences of the PEX14-positive peroxisome staining between these genotypes. Scale bar for images = 50 microns. b Quantitation of PEX14-positive peroxisomes from the 100x images of WT (1 male/ 2 females), Ppara−/− (1 male/2 females), Tmem135FUN025/FUN025 (1 male/2 females), and Tmem135FUN025/FUN025/Ppara−/− (1 male/ 3 females) livers using the Analyze Particles function in ImageJ. c Western blot analysis of peroxisomal proteins in the livers of 3-month-old WT (3 males/1 female), Ppara−/− (2 males/2 females), Tmem135FUN025/FUN025 (2 males/2 females), and Tmem135FUN025/FUN025/Ppara−/− (3 males/2 females) mice. HSC70 served as the loading control for these experiments. Asterisks (*, **, ***, and ****) indicates post hoc Tukey test for a P < 0.05, P < 0.01, P < 0.001, and P < 0.0001 significance following a significant difference detected by one-way ANOVA. PMP70, peroxisomal membrane protein 70. ACOX1, acyl-CoA oxidase 1. DBP, D-bifunctional protein. ACAA1, acetyl-Coenzyme A acyltransferase 1. Dots represent individual data points. Number in parentheses represents N of independent mouse samples per genotype used in the experiment. The protein size next to the immunoblot images denotes the size of the immunoband measured for this analysis. Data are presented as mean ± SD.
Tmem135 mutation reduces obesity and dyslipidemia phenotypes in leptin mutant mice
Having established an important role of TMEM135 in peroxisomal homeostasis, we sought to determine the physiological significance of peroxisomal changes due to the Tmem135 mutation in a mouse model with dysregulated lipid metabolism. We chose leptin mutant (Lepob/ob) mice because modifications in peroxisomal function are known to modulate the phenotypes of Lepob/ob mice40,41. In order to test whether the peroxisomal changes caused by the Tmem135 mutation have an effect on any of the disease phenotypes in the Lepob/ob mice, we crossed Tmem135FUN025/FUN025 mice with Lepob/ob mice. We found that 3-month-old male and female Tmem135FUN025/FUN025/Lepob/ob mice had smaller body weights (Fig. 7a), liver weights (Fig. 7b), and gonadal fat pad weights (Fig. 7c) than age-matched Lepob/ob mice. Plasma cholesterol levels were also significantly reduced in male and female Tmem135FUN025/FUN025/Lepob/ob mice compared to Lepob/ob controls (Fig. 7d), but no changes in plasma triglycerides (Fig. 7e) and non-fasting glucose (Fig. 7f) were observed between these genotypes. We measured the amounts of plasma apolipoproteins in Tmem135FUN025/FUN025/Lepob/ob mice to determine if the decreased plasma cholesterol in this genotype resulted from altered lipoprotein concentrations. We found decreases in apolipoprotein B100 (APOB100) in the plasmas of Tmem135FUN025/FUN025/Lepob/ob male (Fig. 7g) and female mice (Supplementary Fig. 7). No significant change in plasma apolipoprotein B48 (APOB48) (Fig. 7g) and apolipoprotein A1 (APOA1) (Fig. 7h) were observed between these male genotypes. APOB100 is a marker for very-low-density lipoprotein (VLDL) and low-density lipoprotein (LDL), both of which are major carriers of cholesterol from the liver to peripheral tissues42. Thus, the decreased plasma cholesterol in Tmem135FUN025/FUN025/Lepob/ob mice can be explained by their decreased plasma APOB100-containing lipoproteins. In summary, the Tmem135 mutation reduces obesity and improves dyslipidemia in male and female Lepob/ob mice.
a Body, b liver, and c gonadal fat pad weights as well as d plasma cholesterol, e plasma triglyceride, and f plasma non-fasting glucose levels of 3-month-old WT, Tmem135FUN025/FUN025 (FUN025), Lepob/ob (ob), and Tmem135FUN025/FUN025/Lepob/ob (FUN025/ob) male and female mice. Western blot analysis of plasmas from 3-month-old WT, Tmem135FUN025/FUN025, Lepob/ob, and Tmem135FUN025/FUN025/Lepob/ob male mice for g apolipoprotein B100 (APOB100) and B48 (APOB48) and h apolipoprotein A1 (APOA1). Transferrin served as a loading control for these experiments. Number in parentheses represent the N of independent mice or mouse samples per genotype used in the experiment. Dots represent individual data points. The protein size next to the immunoblot images denotes the size of the immunoband measured for this analysis. Asterisks (*, **, and ****) indicate a P < 0.05, P < 0.01, and P < 0.0001 significance by post hoc Tukey test following a significant difference detected by one-way ANOVA. Data are presented as mean ± SD.
Tmem135 mutation ameliorates the liver disease of leptin mutant mice
Lepob/ob mice develop non-alcoholic fatty liver disease (NAFLD), a liver condition categorized by excessive fat accumulation that can progress to inflammation and fibrosis43. We examined the liver sections of 3-month-old Tmem135FUN025/FUN025/Lepob/ob and Lepob/ob mice and found that Tmem135FUN025/FUN025/Lepob/ob mice had less severe NAFLD pathologies relative to Lepob/ob mice (Fig. 8a). We also observed less oil red o staining of liver sections from Tmem135FUN025/FUN025/Lepob/ob mice compared to Lepob/ob mice, indicating less neutral lipid accumulation in Tmem135FUN025/FUN025/Lepob/ob mice (Fig. 8a). We found no signs of a histological phenotype in the livers of Tmem135FUN025/FUN025 mice (Fig. 8a). Next, we calculated NAFLD scores based on the incidence of microvesicles, macrovacuoles, and hypertrophy. Both male and female Tmem135FUN025/FUN025/Lepob/ob mice had lower NAFLD scores than Lepob/ob mice (Fig. 8b). To assess if these hepatic histological results equated to liver injury changes, we measured the activity of alanine transaminase (ALT), a liver enzyme that is leaked into the plasma after liver injury44 and increased in Lepob/ob plasmas45. Plasma ALT activities were significantly reduced in both male and female Tmem135FUN025/FUN025/Lepob/ob mice compared to Lepob/ob mice (Fig. 8c). These results indicate that the Tmem135 mutation ameliorates NAFLD in Lepob/ob mice.
a Representative 20x images of hematoxylin and eosin (H&E) and oil red o (ORO) stained liver sections from 3-month-old male WT, Tmem135FUN025/FUN025 (FUN025), Lepob/ob (ob), and Tmem135FUN025/FUN025/Lepob/ob (FUN025/ob) mice. Scale Bar = 100 microns. b Non-alcoholic fatty liver disease (NAFLD) severity scores. c Plasma ALT activity. Number in parentheses represent the N of independent mouse samples per genotype used in the experiment. Dots represent individual data points. Asterisks (*, ***, and ****) indicates a P < 0.05, P < 0.01, P < 0.001, and P < 0.0001 significance by post hoc Tukey test following a significant difference detected by one-way ANOVA. Data are presented as mean ± SD.
Tmem135 mutation augments peroxisomal proteins in leptin mutant mice
The absence of leptin leads to increased lipid synthesis and decreased lipid oxidation, setting the stage for obesity, dyslipidemia, and NAFLD development in Lepob/ob mice46. We speculate that the Tmem135 mutation ameliorates these phenotypes in Lepob/ob mice by increasing peroxisomal beta-oxidation. We sought to confirm if the Tmem135 mutation caused lipid and peroxisomal changes in Lepob/ob mice that we had observed in Tmem135 mutant mice. We evaluated the lipid profiles of 3-month-old male Lepob/ob and Tmem135FUN025/FUN025/Lepob/ob plasmas. Principal component analysis revealed a lipid profile in Tmem135FUN025/FUN025/Lepob/ob plasmas that was different from Lepob/ob plasmas (Fig. 9a). There were many lipid species modified in Tmem135FUN025/FUN025/Lepob/ob plasmas (Supplementary Fig. 8, Supplementary Table 4) but lipids containing DHA were significantly decreased in Tmem135FUN025/FUN025/Lepob/ob plasmas relative to Lepob/ob plasmas (Fig. 9b, Supplementary Table 5). We confirmed the increases of peroxisomal beta-oxidation enzymes ACOX1, DBP, and ACAA1 in male (Fig. 9c) and female (Supplementary Fig. 9a) Tmem135FUN025/FUN025/Lepob/ob liver lysates compared to gender-matched Lepob/ob liver lysates. Additionally, peroxisome markers PEX14, PMP70, and CAT were significantly increased in male (Fig. 9d) and female (Supplementary Fig. 9b) Tmem135FUN025/FUN025/Lepob/ob liver lysates. This data verified that the Tmem135 mutation reduces DHA-containing lipids but increases peroxisomal proteins in Lepob/ob mice.
a Principal component analysis of lipids detected in the positive (+) ion mode and negative (–) ion mode of 3-month-old male Lepob/ob (ob) (N = 8) and Tmem135FUN025/FUN025/Lepob/ob (FUN025/ob) (N = 4) plasmas based on log10 concentrations. b Pie graph of docosahexaenoic acid (DHA, C22:6n3)-containing lipids that were significantly downregulated (medium green) and unchanged (light green) in Tmem135FUN025/FUN025/Lepob/ob plasmas compared to Lepob/ob. Numbers denote the total of lipids within the category. All altered lipid species including DHA-containing lipids can be found in Supplementary Table 10. Significance was determined by two-way Student’s t test (P < 0.05). c Western blot analysis of peroxisomal beta-oxidation enzymes including acyl-CoA oxidase 1 (ACOX1), D-bifunctional protein (DBP), acetyl-Coenzyme A acyltransferase 1 (ACAA1), and sterol carrier protein x (SCPx) using liver lysates from 3-month-old male Lepob/ob (N = 3), Tmem135 TG/Lepob/ob (TG/ob) (N = 3) and Tmem135FUN025/FUN025/Lepob/ob (N = 3) mice. d Western blot analysis of peroxisome biogenesis factor 14 (PEX14), peroxisome membrane protein 70 (PMP70), and catalase (CAT) using livers from 3-month-old male Lepob/ob (N = 3–6), Tmem135 TG/Lepob/ob (N = 3) and Tmem135FUN025/FUN025/Lepob/ob (N = 3) mice. HSC70 served as the loading control for these experiments. Asterisks (*, **, ***, and ****) indicates post hoc Tukey test for a P < 0.05, P < 0.01, P < 0.001, and P < 0.0001 significance following a significant difference detected by one-way ANOVA. Dots represent individual data points. The protein size next to the immunoblot images denotes the size of the immunoband measured for this analysis. Data are presented as mean ± SD.
Discussion
Maintaining lipid metabolism is an important task for cells to sustain their health. Here, we sought to identify the role of TMEM135 in lipid metabolism. We found a mutation in Tmem135 significantly altered the lipid profiles of multiple murine tissues including the liver, retina, heart, and plasma. Remarkably, a major discovery from our lipidomics analysis was severely diminished concentrations of DHA-containing lipids in all the Tmem135 mutant (Tmem135FUN025/FUN025) tissues evaluated in this study. We confirmed the results from our lipidomic analysis by quantifying fatty acids and detecting substantial decreases of DHA in the livers, retinas, hearts, and plasmas from Tmem135 mutant mice. DHA is an important modulator of disease47. For instance, DHA can be used to synthesize anti-inflammatory eicosanoids required for immune responses30 and mediate fatty acid synthesis48. DHA is also an abundant component of photoreceptor membranes required for their fluidity and permeability49. The decreased DHA in Tmem135 mutant mice may explain their global lipid profile changes and severe retinal degeneration13,15,27 that phenocopies other mice with DHA deficiencies such as elongation of very-long-chain fatty acids-like 2 (Elovl2) mutant50,51, major facilitator superfamily domain containing 2A (Mfsd2a) knockout52,53,54, and adiponectin receptor 1 (Adipor1) knockout mice55,56. Altogether, this evidence supports the role of TMEM135 in cellular DHA homeostasis.
The origin of DHA in mice stems from the contributions of dietary intake and cellular production of DHA. Since there is no DHA present in the food available to the mice used in this study, the reduction of DHA in Tmem135 mutant mice must originate from a defect in its endogenous production. However, we did not observe any decreases in the components required for DHA synthesis in the Tmem135 mutant livers that could explain their decreased DHA concentrations. The remaining step that could be affected is the export of DHA synthesized in peroxisomes. While little is known about how DHA leaves the peroxisome, it has been postulated that there is a protein responsible for the export of DHA from the peroxisome57. Since TMEM135 is present on peroxisomal membranes16,17,18,19,20,26, we hypothesize that TMEM135 exports DHA from peroxisomes for its use by cells (Fig. 10a). TMEM135 may either modulate the activity of the peroxisomal DHA exporter or function as the peroxisomal DHA exporter. We predict that the Tmem135 mutation leads to the retention of DHA within the peroxisome, causing DHA to be degraded by peroxisomal beta-oxidation (Fig.10b). There lies the key difference between Tmem135 mutant mice and other mouse models displaying DHA deficiencies from peroxisomal abnormalities. Mice with peroxisomal biogenesis defects that are unable to produce functional peroxisomes such as peroxisome biogenesis factor 258 and peroxisome biogenesis factor 5 knockout mice59 have reduced DHA concentrations. Also, mice with peroxisomal beta-oxidation defects such as Acox160 and multifunctional protein 2 (also known as DBP) knockout mice61 have decreased levels of DHA. However, these mice obviously lack peroxisome functions in general or peroxisomal beta-oxidation capacity, while Tmem135 mutant mice retain them. These unique characteristics may underlie the increase in peroxisomes that occurs in Tmem135 mutant mice as described below.
a Cells depend on the interaction between the endoplasmic reticulum (ER) and peroxisomes to generate docosahexaenoic acid (C22:6n3). (i) The ER synthesizes C24:6n3 from C18:3n3 through a process of sequential desaturation and elongation steps. (ii) The ER transfers C24:6n3 to the cytosol for uptake by peroxisomes through ABCD2. (iii) One round of peroxisomal beta-oxidation converts C22:6n3 from C24:6n3. (iv) C22:6n3 leaves the peroxisome and migrates back to the ER for lipid incorporation. Since we observed reductions in DHA and no decreases of the components involved in steps i through iii in Tmem135 mutant mice, we hypothesized TMEM135 functions by exporting C22:6n3 from the peroxisome. b The consequence of the Tmem135 mutation may direct C22:6n3 away from the ER and toward peroxisomal beta-oxidation for its catabolism, thus accounting for the reduction of DHA in these mice. c The increased peroxisomal beta-oxidation of DHA within peroxisomes of Tmem135 mutant mice may generate or alter ligands required for the activation of peroxisome proliferator-activated receptors (PPARs). The activation of PPARs can lead to the augmentation of peroxisomes and their beta-oxidation enzymes as we observed in Tmem135 mutant mice. We hypothesized that the increased peroxisomes and their beta-oxidation enzymes due to the Tmem135 mutation may confer protection against the development of leptin mutant mouse phenotypes.
In addition to their role in DHA biogenesis through the Sprecher pathway, peroxisomes perform other unique anabolic and catabolic lipid metabolic functions that are critical for cellular homeostasis8,62,63. In this study, we found that the number of peroxisomes in murine livers and fibroblasts was negatively correlated with the amount of functional TMEM135. The mutation in Tmem135 augments the number of peroxisomes while over-expression of Tmem135 reduces the number of peroxisomes, indicating an important role of TMEM135 function in maintaining the number of peroxisomes. Additionally, the increased number of peroxisomes due to the Tmem135 mutation is consistent with augmented peroxisomal beta-oxidation enzymes including ACOX1, DBP, and ACAA1 in the Tmem135 mutant livers. We posit that the increased beta-oxidation of DHA retained in peroxisomes of Tmem135 mutant mice may alter the concentrations of peroxisome-derived metabolites known to drive peroxisomal biogenesis or division potentially through PPAR signaling (Fig. 10c)35,64. Consistent with this notion, we found that peroxisomes and their protein contents were decreased in Tmem135 mutant livers with the genetic ablation of Ppara, demonstrating that PPAR signaling is at least partly mediating peroxisome levels in the Tmem135 mutant liver. A ligand that could be activating PPAR signaling in the Tmem135 mutant liver is ether phosphatidylethanolamines (EtherPEs)10, which are produced by peroxisomes10 and are higher in Tmem135 mutant tissues (Supplementary Table 1). In fact, EtherPE16:1e_18:1 and EtherPE16:1e_18:2 are commonly increased across Tmem135 mutant tissues (Supplementary Tables 6–9). Further investigation is necessary to identify the exact ligands that modify the number of peroxisomes in Tmem135 mutant mice as well as to determine whether these ligands are responsible for peroxisome proliferation observed in mouse models of peroxisomal beta-oxidation deficiencies65,66,67.
Our study also uncovered the effect of the Tmem135 mutation on the phenotypes of leptin mutant mice. The leptin mutant mouse is a well-characterized mouse model of dysregulated lipid metabolism often used to evaluate key pathways involved in metabolic diseases43,68. We found leptin mutant mice that are homozygous for the Tmem135 mutation have reduced body weight, smaller gonadal fat pads, lower plasma cholesterol, and decreased plasma APOB compared to leptin mutant mice. We also observed that the Tmem135 mutation ameliorates fatty liver disease in leptin mutant mice. We postulate that the protection provided by the Tmem135 mutation on leptin mutant phenotypes can be explained by enhanced peroxisomal beta-oxidation from the activation of PPARa signaling due to the Tmem135 mutation (Fig. 10c). We support this idea through a study evaluating wildtype and Tmem135 mutant mice in metabolic phenotyping cages that found the Tmem135 mutation increased fatty acid oxidation in mice (Supplementary Fig. 10). It was previously shown that the level of peroxisomal beta-oxidation can affect the phenotypes of leptin mutant mice. For example, leptin mutant mice treated with fenofibrate, a PPARa agonist69, decreased body weight, reduced plasma cholesterol, lessened fat accumulation, and improved their fatty liver phenotypes41. In contrast, mice with the genetic ablation of Ppara on the leptin mutant background showed worsened obesity, increased fat accumulation, and more severe hepatic steatosis compared to leptin mutant mice40. In support of increased PPARa activation in the double Tmem135 and leptin mutant mice, we confirmed the higher concentrations of hepatic peroxisomal markers and beta-oxidation enzymes in these mice compared to leptin mutant mice.
In summary, we investigated a role of TMEM135 in lipid metabolism. We found TMEM135 has a critical role in maintaining cellular DHA levels in mice that is abolished by a mutation in the murine Tmem135 gene. Since we did not observe any decreases in the components involved in cellular DHA synthesis, we hypothesized that the Tmem135 mutation prevents the export of DHA from peroxisomes, leading to the retention and degradation of DHA within peroxisomes in the Tmem135 mutant tissues. We also discovered TMEM135 is important for maintaining peroxisomal number and function through the regulation of PPARa signaling. Lastly, we established that the Tmem135 mutation is protective against leptin mutant-induced mouse phenotypes that can be explained by the increased peroxisomal beta-oxidation induced by the Tmem135 mutation, Together, we conclude TMEM135 has a role in maintaining lipid homeostasis in mice through its function in peroxisomes.
It is important to note that there were limitations of this study concerning the origin of protection of the Tmem135 mutation on leptin mutant mouse phenotypes. For example, the impact of the Tmem135 mutation on other organelles known to harbor TMEM135 such as mitochondria13,20 and lipid droplets20,21 could possibly explain the decreased severity of leptin mutant mouse phenotypes. We detected changes in mitochondrial proteins carnitine palmitoyltransferase 1a (CPT1a) (Supplementary Fig. 11) and carnitine-acylcarnitine translocase (CACT) (Supplementary Fig. 11) but no differences in perilipin 2 (PLIN2) (Supplementary Fig. 12), the main protein constituent of hepatic lipid droplets70, in Tmem135 mutant livers. Interestingly, hepatic CPT1a protein levels were decreased in Tmem135 mutant livers (Supplementary Fig. 11). Cpt1a is a known target of PPARa signaling71 that we found to be activated in Tmem135 mutant livers. The decreased CPT1a could result from a PPARa-independent regulatory mechanism involving epigenetic and posttranslational modifications that would affect its protein levels71. Alternatively, the decreased CPT1a could reflect an impaired function of TMEM135 on mitochondrial membranes since the Tmem135 mutation has profound effects on mitochondrial homeostasis13,14,15,22,23. CPT1a is the rate-limiting step for mitochondrial fatty acid oxidation that converts acyl-CoA esters to acylcarnitines for their import through the mitochondrial outer membrane71. It has been shown that liver-specific Cpt1a knockout mice were protected against diet-induced weight gain72. Thus, decreased hepatic CPT1a concentrations in Tmem135 mutant mice may explain the smaller body weight of these mice and leptin double mutant mice compared to leptin mutant controls. Future studies are warranted to investigate whether the mitochondrial dysfunction caused by the Tmem135 mutation is from its role on peroxisomes or mitochondria as this may shed insight into the mechanisms underlying its protection against the leptin mutant mouse phenotypes.
It is also possible that the protection of the Tmem135 mutation on leptin mutant mouse phenotypes is from its effect on another tissue. We have shown in our study that multiple tissues in Tmem135 mutant mice exhibit similar changes in DHA concentrations and peroxisomal proteins. Therefore, additional experiments are required to determine the contribution of specific tissues towards the protection of the Tmem135 mutation on the phenotypes of leptin mutant mice.
Methods
Mice
Tg(CAG-Tmem135)#Aike (Tmem135 TG) mice congenic on the C57BL/6J background and Tmem135FUN025/FUN025 mice were used in this study13,14,15. B6;129S4-Pparatm1Gonz/J (Ppara−/−) (Stock #008154) and B6.Cg-Lepob/J (Lepob) (Stock #000632) and were obtained from The Jackson Laboratory (Bar Harbor, ME) and bred in the animal facility at the University of Wisconsin-Madison. C57BL/6J mice served as WT controls for this study. All mice were fed a global soy protein-free extruded rodent diet (#2016, Envigo, Madison, WI) and housed in the Medical Sciences Center Vivarium at the University of Wisconsin-Madison. Both males and females were used in this study. All numbers of mice used in experiments are provided within the figures and their legends. All mouse procedures were performed in accordance with the protocols approved by the Animal Care and Use Committee at the University of Wisconsin-Madison. We adhere to the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines in reporting our animal research.
Sample preparation for lipidomics analysis
Livers, retinas (neural retinas and eyecups), hearts, and plasmas were collected and stored at −80 °C prior to lipid extraction. Prior to extraction, all solutions were pre-chilled on ice. Samples were transported and cut on a dry ice-cooled stainless-steel plate and weighed on a Mettler (XSR205) analytical balance to the nearest hundredth of a milligram. This weight would be used to normalize samples for analysis. Fifty microliter aliquots of plasma from each mouse were used in this study. No normalization occurred with the plasma samples since the same volume for each sample was used in this study. Tissues were then placed into Qiagen PowerBead tubes (P/N 13113-50) for homogenization and extraction. Lipids were extracted in a solution of 250 µL PBS, 225 µL methanol containing internal standards (Avanti SPLASH LipidoMix (Lot#3307-07) at 10 µL per sample), and 750 µL MTBE (methyl tert-butyl ether)73. The sample was homogenized in three 30-second cycles alternated with a 5-minute rest on ice using a Qiagen TissueLyzer II operated at 30 Hz. The final rest on ice was 15 minutes. After centrifugation at 16,000 × g for 5 minutes at 4 °C, 500 µL of the upper phase was collected in 1.5 ml centrifuge tubes and evaporated to dryness in a Savant speedvac concentrator. Lipid samples were reconstituted in 150 µL of isopropanol. A process blank was prepared in parallel with the tissue samples during extraction and analyzed concurrently with the samples. A pooled sample, used for lipid identification and quality control, was prepared by taking equal volumes from each sample of a given tissue after final resuspension in IPA.
LC-MS methods for lipidomics
Sample analysis was done at different dilution factors for different tissues and ionization polarities. For each injection samples were diluted in isopropanol by adding a given sample volume to isopropanol in an LC-MS vial with deactivated glass insert (Agilent P/N 5182-0554 and 5183-2086), vortexed to mix, and spun at low speed to collect the liquid prior to placing in the autosampler for analysis. Lipid extracts were separated on a Waters Acquity UPLC BEH C18 1.7 µm 2.1 × 100 mm column coupled in tandem with a Waters Acquity UPLC BEH C18 1.7 µm VanGaurd pre-column 2.1 × 5 mm and maintained at 50 °C. These columns in turn were connected to an Agilent HiP 1290 Multisampler, Agilent 1290 Infinity II binary pump, and column compartment connected to an Agilent 6546 Accurate Mass Q-TOF dual ESI mass spectrometer. For positive ion mode, the source gas temperature was set to 250 °C, with a gas flow of 12 L/min and a nebulizer pressure of 35 psig. VCap voltage was set at 4000 V, fragmentor at 145 V, skimmer at 45 V, and Octopole RF peak at 750 V. For negative ion mode, the source gas temperature was set to 350 °C, with a drying gas flow of 12 L/min and a nebulizer pressure of 25 psig. VCap voltage is set at 5000 V, fragmentor at 200 V, skimmer at 45 V, and Octopole RF peak at 750 V. Reference masses in positive mode (m/z 121.0509 and 922.0098) and negative mode (m/z 966.0007, and 112.9856) were delivered to the second emitter in the dual ESI source by isocratic pump at 15 uL/min. Samples were analyzed in a randomized order in both positive and negative ionization modes in separate experiments acquiring with the scan range m/z 100–1500. Mobile phase A consisted of acetonitrile:water (60:40 v/v) containing 10 mM ammonium formate and 0.1% formic acid, and mobile phase B consists of isopropanol: acetonitrile:water (90:9:1 v/v) containing 10 mM ammonium formate and 0.1% formic acid. The chromatography gradient for both positive and negative modes started at 15% mobile phase B then increases to 30% B over 2.4 min, then increased to 48% B from 2.4–3.0 min, then increased to 82% B from 3–13.2 min, then increased to 99% B from 13.2–13.8 min where it was held until 15.4 min and then returned to the initial conditions and equilibrated for 4 min. Flow was 0.5 mL/min throughout the gradient. Injection volumes were 2 µL for positive mode and 5 µL for negative mode MS1 acquisitions, and 4 µL for positive mode, and 7 µL for negative mode MS/MS acquisitions. Tandem mass spectrometry (MS/MS) was conducted using the same LC gradient as above with isolation width set to “narrow” (~1.3 m/z) and collision energy of 25 V. MS/MS data were collected on the pooled sample per tissue in iterative mode, in which the sample is analyzed five times with different precursors selected upon each injection. This permits access to lower-abundance lipid species in a background of highly abundant lipids.
Data analysis
Pooled MS/MS and individual sample MS data were analyzed using a combination of Agilent and web-based applications. Lipid identification was achieved from the pooled MS/MS data via Lipid Annotator (Agilent). This software utilizes accurate mass m/z values for the intact lipid and experimental and theoretical fragment ion m/z values to assign lipid class and alkyl chain identities where possible. In some cases, specific alkyl chain identities cannot be assigned, but the sum composition of those alkyl chains (i.e., carbon number and degree of unsaturation) can be determined and that is reported instead. The output from Lipid Annotator is a database of lipid species with m/z values and retention times for each. This process was performed independently for positive and negative ion modes. Quantitation of lipids in individual samples used MS data alone. In this analysis, data for each sample was collected separately. The abundance of each lipid in each sample was tabulated using the Profinder application (Agilent). In this analysis, the database of lipid identity, m/z values (for protonated, deprotonated, ammonium or sodium adducted, or formate adducted, depending on ionization mode), and retention time was used to extract an ion chromatogram for each lipid species. These chromatograms were then integrated to produce an area for that lipid in each sample. These abundance values were then exported as a .csv file for statistical analysis using the web-based tool MetaboAnalyst 5.074. This software was used for principal component analysis and statistical testing. All significantly changed lipids in Tmem135 mutant tissues identified in our study are provided in Supplementary Tables 6–10. All lipidomics data from this study is available on the Dryad public database (accession code: doi:10.5061/dryad.vx0k6djvm).
Fatty acid composition analysis
Total lipids from retina and plasma (100 µL) were extracted following the method of Bligh and Dyer75 with modifications76. Total lipids from heart and liver were extracted following the method of Folch et al.77. To each lipid extract were added 25 nanomoles of 15:0 and 17:0 as internal standards. The lipid extracts were subjected to acid hydrolysis/methanolysis by heating at 100 °C for 2 h in 16% v/v concentrated HCl in methanol to generate fatty acid methyl esters (FAMEs). FAMEs were extracted into hexane and purified via TLC78. FAMEs were quantified using an Agilent Technologies 7890B gas chromatograph with flame ionization detector79. Data are represented as the relative mole percent of each fatty acid.
Quantitative PCR
Livers were collected from mice and kept at −80 °C degrees. RNA was extracted using a RNeasy lipid tissue mini kit (#74804, Qiagen, Germantown, MD) according to the manufacturer’s instructions. RNA concentrations were determined using the Nanodrop 2000 UV-Vis Spectrophotometer (Thermo Fisher Scientific, Waltham, MA). One microgram of RNA was used to make cDNA using an Oligo d(T)18 primer (#S1316S, NEB, Ipswich, MA) and the ProtoScript II Reverse Transcriptase (#M0368L, NEB, Ipswich, MA) following the manufacturer’s protocol. Expression of each gene was assessed in triplicate reactions in the Roche Lightcycler 480 system using cDNA, 200 nmol/L of each primer, and Lightcycler 480 SYBR Green I Supermix (#507203180, Roche). Relative mRNA expression was normalized to the ribosomal protein lateral stalk subunit P0 (Rlpl0) using the quantitative 2−ΔΔCT method80. Primer sequences used in this study can be found in Supplementary Table 11.
Western blot analysis
Tissues were isolated from mice and stored at −80 °C. Liver and heart lysates were made with an Ultra-Turrax T8 tissue homogenizer in RIPA buffer (#P189901, Thermo Fisher Scientific, Waltham, MA) containing protease inhibitors (#11836170001, Thermo Fisher Scientific, Waltham, MA), respectively. Neural retina and eyecup (RPE/choroid/sclera) samples were homogenized using a Bel-Art Homogenizer system motor with RIPA buffer containing protease inhibitors. Protein concentrations were quantified using a BCA Protein Assay Kit (#P123228, Thermo Fisher Scientific, Waltham, MA). Equal protein amounts were aliquoted, reduced using XT Reducing Agent (#1610792, Biorad, Hercules, CA) for seven minutes at 105 °C, and loaded onto 10% Bis-Tris Criterion XT gels (#3450112, Biorad, Hercules, CA) in MOPS buffer (#1610788, Biorad, Hercules, CA) and transferred to nitrocellulose membranes (#102673-324, Biorad, Hercules, CA). Membranes were blocked with milk and probed overnight with their respective primary antibody at 4 °C. For Westerns using plasma samples, equal volumes of each sample were loaded in the gel but were not reduced with XT Reducing Agent. Also, these blots were blocked with bovine serum albumin diluted in PBS.
The primary antibodies and their dilutions used in this study can be found in Supplementary Table 12. Blots were washed with TBST buffer the next day and incubated with their corresponding secondary antibody. Secondary antibodies used in this study included donkey anti-rabbit IgG 680RD (#926-68073, LI-COR), donkey anti-rabbit IgG 800CW (#926-32213, LI-COR), donkey anti-guinea pig IgG 800CW (#925-32411, LI-COR), donkey anti-goat IgG 680RD (#926-68074, LI-COR), goat anti-mouse IgG1 800CW (#926-32350, LI-COR), goat anti-mouse IgG2a 800CW (#926-32351, LI-COR), and goat anti-mouse IgM 800CW (#925-32280, LI-COR). Blots were washed again with TBST and imaged using the Odyssey Imaging System (LI-COR Biosciences, Lincoln, NE) and analyzed using NIH’s ImageJ (Bestheda, MD). Blots were stripped with Newblot Stripping Buffer (LI-COR Biosciences, Lincoln, NE) according to the manufacturer’s protocol and reprobed with another primary antibody in this study. All immunobands were normalized to the loading control on their respective immunoblot.
Immunohistochemistry for peroxisomal markers
Tissues were fixed in 4% paraformaldehyde overnight at 4 °C, processed for cryoprotection, and embedded in Tissue-Tek O.C.T. Compound. Sections were cut at 8-micron thickness on a cryostat. We labeled peroxisomes with rabbit anti-PEX14 (#10594-1-AP, Proteintech, 1:200 dilution) or anti-PMP70 (#ab3421, Abcam, 1:200 dilution) and stained nuclei with 4′,6-Diamidine-2′-phenylindole dihydrochloride (DAPI) (#D9542, Sigma Aldrich, 1:1000 dilution). Secondary donkey anti-rabbit IgG antibodies either conjugated with an Alexafluor 488 (#A21206, Invitrogen) or 568 tag (#A10042, Invitrogen) were used for this study. The sections were imaged with a Nikon A1RS confocal microscope at the University of Wisconsin-Madison Optical Imaging Core. For each sample in this study, we collected seven images and analyzed these images using the ‘Analyze Particles’ application in NIH’s ImageJ program as detailed in Darwisch et al.36.
Fibroblast cultures
Primary fibroblasts were isolated from one-month-old mouse ears by our published protocol13. To briefly summarize the protocol, ears were collected from the mice into 1.5 ml microcentrifuge tubes containing 70% ethanol and rinsed with PBS containing penicillin and streptomycin antibiotics (Thermo Fisher Scientific, Waltham, MA). The tissues were cut into smaller pieces, and placed in a new tube with 0.5 ml Trypsin-EDTA (0.25% Trypsin, 0.1% EDTA) (Thermo Fisher Scientific, Waltham, MA) and 0.5 ml Dispase (5 U/ml) (STEMCELL Technologies, Vancouver, Canada), and incubated at 37 °C for 30 min. Preparations were centrifuged, and the supernatant was discarded. Tissues were washed with Hank’s Balanced Salt Solution (Gibco) and centrifuged again, where the supernatant was discarded. Trypsin-EDTA was added to the tissue and allowed to incubate at 37 °C for 20 min. Following incubation, the samples were centrifuged, and the supernatant was removed from them. The remaining pellet was resuspended in Dulbecco’s Modified Eagle’s Medium (DMEM, ATCC, Manassas, VA) with 10% Fetal Bovine Serum (FBS, ATCC, Manassas, VA) and 1% Penicillin Streptomycin (Thermo Fisher Scientific, Waltham, MA). The pellet was triturated and then plated into a tissue culture dish and placed in a cell culture hood set at 37 °C with 5% CO2. Fibroblasts were tested for mycoplasma contamination and the results were negative. Cultures were prepared for lysates for Western blot analysis or immunohistochemistry for PMP70. Lysates were made from the same number of cells using RIPA buffer containing protease inhibitors and quantified using a BCA Protein Assay Kit. For PEX14 and PMP70 immunohistochemistry, fibroblasts were allowed to grow on collagen-coated coverslips and then fixed with 4% PFA. The same western blot and immunohistochemistry protocol described above was used.
Plasma parameter measurements
Blood samples were collected from non-fasted mice by a submandibular bleed in EDTA capillary tubes and spun for 10 minutes at 1200 × g for 10 minutes. Plasma fraction was collected from the tube, placed into a separate microcentrifuge tube, and stored at −80 °C. Plasma cholesterol, triglycerides, and glucose concentrations were assayed and calculated with a Total Cholesterol Assay Kit (#STA-384, Cell Biolabs Inc, San Diego, CA), Triglyceride Assay Kit (#10010303, Cayman Chemical, Ann Arbor, MI), and Glucose Assay Kit (#10009582, Cayman Chemical, Ann Arbor, MI). Plasma ALT activity was quantified using an ALT Activity Assay Kit (#700620, Cayman Chemical, Ann Arbor, MI). We followed the protocols provided by the manufacturer.
Liver histology
3-month-old mice were perfused using a 2% glutaraldehyde and 2% paraformaldehyde mixture before harvesting the largest liver lobe for paraffin sectioning. For livers collected for oil red o (ORO) staining, mice were perfused using a 4% paraformaldehyde fixative prior to subjecting the liver to a sucrose gradient and cryopreservation in Tissue-Tek O.C.T. Compound. The lobe was incubated in their respective fixative overnight and rinsed with PBS the following day. Samples were submitted to the University of Wisconsin–Madison’s Translational Research Initiatives in Pathology (TRIP) core for processing and sectioning. Paraffin liver sections were stained with hematoxylin and eosin (H&E) and liver cryosections were stained with ORO using standard protocols.
NAFLD phenotype scoring
Five random different areas of each H&E-stained liver section were imaged using the tiling function of the Axio Imager 2 microscope (Carl Zeiss Microscopy LLC, White Plains, NY). In total, there were 125 images taken from each sample for scoring. We modified a scoring strategy based on the criteria for non-alcoholic fatty liver disease from the Pathology Committee of the NASH Clinical Research Network81. In this study, we focused on the presence of and total area affected by microvesicles, macrovacuoles, and hypertrophy. We scored a hepatic section for microvesicles as 1, macrovacuoles as 1, and hypertrophy as 1 if the pathology was present. We also scored if the pathologies encompassed 25–50% of the image as 1, 51–75% of the image as 2, and 76–100% of the image as 3. We summed the scores for each image and calculated the average of the image scores to generate the NALFD score for each mouse in this study. Two independent scorers evaluated liver images in this study.
Metabolic cage phenotyping
Male mice were acclimated to individual housing within metabolic phenotyping cages (Promethion Core, Sable Systems) with bedding and conditions identical to their home-cage environment. Stable body weight was confirmed 1 week prior to the metabolic phenotyping. For the study, mice were provided ad lib chow (#2016, Envigo, Madison, WI) and water. The environment was maintained at a constant temperature of 23 °C with a 12-hour light cycle that started at 7 A.M for 2.5 days. Oxygen, food, and water consumption as well as carbon dioxide production were measured continuously for the duration of the experiment. Energy expenditure was calculated using the Weir equation82. Body weight was measured at the start and end of the feeding trial. Body composition was measured using a Minispec LF-90 NMR machine (Bruker). Metabolic data were processed using the OneClick Macro (Sable Systems) to exclude outliers and non-physiological values. Data from only the final 48 hours (i.e. starting at 7 AM the morning after handling the mice) were used to avoid artifacts of animal handling. Hourly and daily average data were generated using CalR83. T tests and linear regression were performed using Prism 9.4 (GraphPad) to determine significant differences between the genotypes.
Statistics and reproducibility
One-way analysis of variance was performed in experiments using more than two groups and post hoc Tukey’s honest significance test was utilized to determine which groups were significantly different from each other. A two-way Student’s t test was used when there were only two experimental groups. All sample sizes are included within the figures and their legends. We used the G*Power application (RRID:SCR_013726) to determine sample sizes that would give us statistical power for 95% confidence with 80% power with an average standard deviation of 25%. We assumed all groups followed a normal distribution and had equal standard deviations. All statistical tests were performed using Prism Software (GraphPad, San Diego, CA). All statistical parameters can be found within the Supplementary Data file. We ensure the reproducibility of our results by obtaining similar conclusions from at least two independent experiments in this study.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All lipidomic source data can be obtained through the Dryad public database under the accession code, doi:10.5061/dryad.vx0k6djvm. The raw mass spectrometry data will be given to those who are interested upon request to the corresponding author. Source data for figures and Supplementary Table 3 can be found in the Supplementary Data file.
References
Athyros, V. G. et al. Diabetes and lipid metabolism. Hormones (Athens) 17, 61–67 (2018).
Gupta, M. et al. Novel emerging therapies in atherosclerosis targeting lipid metabolism. Expert Opin. Investig. Drugs 29, 611–622 (2020).
Chew, H., Solomon, V. A. & Fonteh, A. N. Involvement of lipids in Alzheimer’s disease pathology and potential therapies. Front. Physiol. 11, 598 (2020).
van Leeuwen, E. M. et al. A new perspective on lipid research in age-related macular degeneration. Prog. Retin Eye Res. 67, 56–86 (2018).
Aon, M. A., Bhatt, N. & Cortassa, S. C. Mitochondrial and cellular mechanisms for managing lipid excess. Front. Physiol. 5, 282 (2014).
Houten, S. M., Violante, S., Ventura, F. V. & Wanders, R. J. The biochemistry and physiology of mitochondrial fatty acid β-oxidation and its genetic disorders. Annu. Rev. Physiol. 78, 23–44 (2016).
Martínez-Reyes, I. & Chandel, N. S. Mitochondrial TCA cycle metabolites control physiology and disease. Nat. Commun. 11, 102 (2020).
Lodhi, I. J. & Semenkovich, C. F. Peroxisomes: a nexus for lipid metabolism and cellular signaling. Cell Metab. 19, 380–392 (2014).
Miura, Y. The biological significance of ω-oxidation of fatty acids. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 89, 370–382 (2013).
Dean, J. M. & Lodhi, I. J. Structural and functional roles of ether lipids. Protein Cell 9, 196–206 (2018).
Jacquemyn, J., Cascalho, A. & Goodchild, R. E. The ins and outs of endoplasmic reticulum-controlled lipid biosynthesis. EMBO Rep. 18, 1905–1921 (2017).
Bravo-Sagua, R. et al. Organelle communication: signaling crossroads between homeostasis and disease. Int. J. Biochem. Cell Biol. 50, 55–59 (2014).
Lee, W. H. et al. Mouse Tmem135 mutation reveals a mechanism involving mitochondrial dynamics that leads to age-dependent retinal pathologies. Elife 5, 1–29 (2016).
Lewis, S. A. et al. The effect of Tmem135 overexpression on the mouse heart. PLoS One 13, 1–29 (2018).
Landowski, M. et al. Modulation of Tmem135 leads to retinal pigmented epithelium pathologies in mice. Invest. Ophthalmol. Vis. Sci. 61, 16 (2020).
Islinger, M., Luers, G. H., Li, K. W., Loos, M. & Volkl, A. Rat liver peroxisomes after fibrate treatment. a survey using quantitative mass spectrometry. J. Biol. Chem. 282, 23055–23069 (2007).
Islinger, M. et al. Peroxisomes from the heavy mitochondrial fraction: isolation by zonal free flow electrophoresis and quantitative mass spectrometrical characterization. J. Proteome Res. 9, 113–124 (2010).
Kikuchi, M. et al. Proteomic analysis of rat liver peroxisome: presence of peroxisome-specific isozyme of Lon protease. J. Biol. Chem. 279, 421–428 (2004).
Mi, J., Kirchner, E. & Cristobal, S. Quantitative proteomic comparison of mouse peroxisomes from liver and kidney. Proteomics 7, 1916–1928 (2007).
Exil, V. J. et al. Stressed-induced TMEM135 protein is part of a conserved genetic network involved in fat storage and longevity regulation in Caenorhabditis elegans. PLoS One 5, 1–11 (2010).
Bosch, M. et al. Mammalian lipid droplets are innate immune hubs integrating cell metabolism and host defense. Science 370 https://doi.org/10.1126/science.aay8085 (2020).
Lee, W. H. et al. Metabolic alterations caused by the mutation and overexpression of the Tmem135 gene. Exp. Biol. Med. (Maywood) 245, 1571–1583 (2020).
Muench, N. A. et al. The influence of mitochondrial dynamics and function on retinal ganglion cell susceptibility in optic nerve disease. Cells 10 https://doi.org/10.3390/cells10071593 (2021).
Someya, S. & Ikeda, A. in Handbook of the Biology of Aging, Ch. 14, 297–321 (Elsevier, 2021).
Beasley, H. K., Rodman, T. A., Collins, G. V., Hinton, A. & Exil, V. TMEM135 is a novel regulator of mitochondrial dynamics and physiology with implications for human health conditions. Cells 10 https://doi.org/10.3390/cells10071750 (2021).
Maharjan, Y. et al. TMEM135 regulates primary ciliogenesis through modulation of intracellular cholesterol distribution. EMBO Rep. 21, 1–16 (2020).
Landowski, M. et al. A mutation in transmembrane protein 135 impairs lipid metabolism in mouse eyecups. Sci. Rep. 12, 756 (2022).
Lewandowski, D. et al. Dynamic lipid turnover in photoreceptors and retinal pigment epithelium throughout life. Prog. Retin Eye Res. 89, 101037 (2022).
Chu, B. B. et al. Cholesterol transport through lysosome-peroxisome membrane contacts. Cell 161, 291–306 (2015).
Saini, R. K. & Keum, Y. S. Omega-3 and omega-6 polyunsaturated fatty acids: dietary sources, metabolism, and significance - A review. Life Sci. 203, 255–267 (2018).
Domenichiello, A. F., Kitson, A. P. & Bazinet, R. P. Is docosahexaenoic acid synthesis from α-linolenic acid sufficient to supply the adult brain? Prog. Lipid Res. 59, 54–66 (2015).
Metherel, A. H. & Bazinet, R. P. Updates to the n-3 polyunsaturated fatty acid biosynthesis pathway: DHA synthesis rates, tetracosahexaenoic acid and (minimal) retroconversion. Prog. Lipid Res. 76, 101008 (2019).
Agbaga, M. P., Mandal, M. N. & Anderson, R. E. Retinal very long-chain PUFAs: new insights from studies on ELOVL4 protein. J. Lipid Res. 51, 1624–1642 (2010).
Fourcade, S. et al. A key role for the peroxisomal ABCD2 transporter in fatty acid homeostasis. Am. J. Physiol. Endocrinol. Metab. 296, E211–E221 (2009).
Ferdinandusse, S. et al. Identification of the peroxisomal beta-oxidation enzymes involved in the biosynthesis of docosahexaenoic acid. J. Lipid Res. 42, 1987–1995 (2001).
Darwisch, W. et al. Cerebellar and hepatic alterations in ACBD5-deficient mice are associated with unexpected, distinct alterations in cellular lipid homeostasis. Commun. Biol. 3, 713 (2020).
Bentley, P. et al. Hepatic peroxisome proliferation in rodents and its significance for humans. Food Chem. Toxicol. 31, 857–907 (1993).
Wang, Y., Nakajima, T., Gonzalez, F. J. & Tanaka, N. PPARs as metabolic regulators in the liver: lessons from liver-specific PPAR-null mice. Int. J. Mol. Sci. 21 https://doi.org/10.3390/ijms21062061 (2020).
Rakhshandehroo, M., Knoch, B., Müller, M. & Kersten, S. Peroxisome proliferator-activated receptor alpha target genes. PPAR Res. 2010 https://doi.org/10.1155/2010/612089 (2010).
Gao, Q. et al. PPARα-deficient ob/ob obese mice become more obese and manifest severe hepatic steatosis due to decreased fatty acid oxidation. Am. J. Pathol. 185, 1396–1408 (2015).
Carmona, M. C. et al. Fenofibrate prevents Rosiglitazone-induced body weight gain in ob/ob mice. Int. J. Obes. (Lond.) 29, 864–871 (2005).
Kim, E. & Young, S. G. Genetically modified mice for the study of apolipoprotein B. J. Lipid Res. 39, 703–723 (1998).
Anstee, Q. M. & Goldin, R. D. Mouse models in non-alcoholic fatty liver disease and steatohepatitis research. Int. J. Exp. Pathol. 87, 1–16 (2006).
Peng, C., Stewart, A. G., Woodman, O. L., Ritchie, R. H. & Qin, C. X. Non-alcoholic steatohepatitis: a review of its mechanism, models and medical treatments. Front. Pharm. 11, 603926 (2020).
Kristiansen, M. N. et al. Obese diet-induced mouse models of nonalcoholic steatohepatitis-tracking disease by liver biopsy. World J. Hepatol. 8, 673–684 (2016).
Reidy, S. P. & Weber, J. Leptin: an essential regulator of lipid metabolism. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 125, 285–298 (2000).
Shahidi, F. & Ambigaipalan, P. Omega-3 polyunsaturated fatty acids and their health benefits. Annu. Rev. Food Sci. Technol. 9, 345–381 (2018).
Hishikawa, D. et al. Hepatic levels of DHA-containing phospholipids instruct SREBP1-mediated synthesis and systemic delivery of polyunsaturated fatty acids. iScience 23, 101495 (2020).
SanGiovanni, J. P. & Chew, E. Y. The role of omega-3 long-chain polyunsaturated fatty acids in health and disease of the retina. Prog. Retin. Eye Res. 24, 87–138 (2005).
Chen, D. et al. The lipid elongation enzyme ELOVL2 is a molecular regulator of aging in the retina. Aging Cell 19, e13100 (2020).
Pauter, A. M. et al. Elovl2 ablation demonstrates that systemic DHA is endogenously produced and is essential for lipid homeostasis in mice. J. Lipid Res. 55, 718–728 (2014).
Lobanova, E. S. et al. Disrupted blood-retina lysophosphatidylcholine transport impairs photoreceptor health but not visual signal transduction. J. Neurosci. 39, 9689–9701 (2019).
Nguyen, L. N. et al. Mfsd2a is a transporter for the essential omega-3 fatty acid docosahexaenoic acid. Nature 509, 503–506 (2014).
Wong, B. H. et al. Mfsd2a is a transporter for the essential ω-3 fatty acid docosahexaenoic acid (DHA) in eye and is important for photoreceptor cell development. J. Biol. Chem. 291, 10501–10514 (2016).
Sluch, V. M. et al. ADIPOR1 is essential for vision and its RPE expression is lost in the Mfrp. Sci. Rep. 8, 14339 (2018).
Osada, H. et al. ADIPOR1 deficiency-induced suppression of retinal ELOVL2 and docosahexaenoic acid levels during photoreceptor degeneration and visual loss. Cell Death Dis. 12, 458 (2021).
Wanders, R. J. A., Waterham, H. R. & Ferdinandusse, S. Peroxisomes and their central role in metabolic interaction networks in humans. Subcell. Biochem. 89, 345–365 (2018).
Faust, P. L., Su, H. M., Moser, A. & Moser, H. W. The peroxisome deficient PEX2 Zellweger mouse: pathologic and biochemical correlates of lipid dysfunction. J. Mol. Neurosci. 16, 289–297 (2001).
Janssen, A. et al. Docosahexaenoic acid deficit is not a major pathogenic factor in peroxisome-deficient mice. Lab. Invest. 80, 31–35 (2000).
Vluggens, A. et al. Reversal of mouse Acyl-CoA oxidase 1 (ACOX1) null phenotype by human ACOX1b isoform [corrected]. Lab. Invest. 90, 696–708 (2010).
Das, Y. et al. Peroxisomal multifunctional protein 2 deficiency perturbs lipid homeostasis in the retina and causes visual dysfunction in mice. Front. Cell Dev. Biol. 9, 632930 (2021).
Kohlwein, S. D., Veenhuis, M. & van der Klei, I. J. Lipid droplets and peroxisomes: key players in cellular lipid homeostasis or a matter of fat–store ’em up or burn ’em down. Genetics 193, 1–50 (2013).
Wanders, R. J., Waterham, H. R. & Ferdinandusse, S. Metabolic interplay between peroxisomes and other subcellular organelles including mitochondria and the endoplasmic reticulum. Front. Cell Dev. Biol. 3, 83 (2015).
Grygiel-Górniak, B. Peroxisome proliferator-activated receptors and their ligands: nutritional and clinical implications–a review. Nutr. J. 13, 17 (2014).
Fan, C. Y. et al. Steatohepatitis, spontaneous peroxisome proliferation and liver tumors in mice lacking peroxisomal fatty acyl-CoA oxidase. Implications for peroxisome proliferator-activated receptor alpha natural ligand metabolism. J. Biol. Chem. 273, 15639–15645 (1998).
Seedorf, U. et al. Defective peroxisomal catabolism of branched fatty acyl coenzyme A in mice lacking the sterol carrier protein-2/sterol carrier protein-x gene function. Genes Dev. 12, 1189–1201 (1998).
Jia, Y. et al. Overexpression of peroxisome proliferator-activated receptor-alpha (PPARalpha)-regulated genes in liver in the absence of peroxisome proliferation in mice deficient in both L- and D-forms of enoyl-CoA hydratase/dehydrogenase enzymes of peroxisomal beta-oxidation system. J. Biol. Chem. 278, 47232–47239 (2003).
Lindström, P. The physiology of obese-hyperglycemic mice [ob/ob mice]. ScientificWorldJournal 7, 666–685 (2007).
Lakhia, R. et al. PPARα agonist fenofibrate enhances fatty acid β-oxidation and attenuates polycystic kidney and liver disease in mice. Am. J. Physiol. Ren. Physiol. 314, F122–F131 (2018).
Sztalryd, C. & Brasaemle, D. L. The perilipin family of lipid droplet proteins: gatekeepers of intracellular lipolysis. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 1862, 1221–1232 (2017).
Schlaepfer, I. R. & Joshi, M. CPT1A-mediated fat oxidation, mechanisms, and therapeutic potential. Endocrinology 161 https://doi.org/10.1210/endocr/bqz046 (2020).
Sun, W. et al. Hepatic CPT1A facilitates liver-adipose cross-talk via induction of FGF21 in mice. Diabetes https://doi.org/10.2337/db21-0363 (2021).
Matyash, V., Liebisch, G., Kurzchalia, T. V., Shevchenko, A. & Schwudke, D. Lipid extraction by methyl-tert-butyl ether for high-throughput lipidomics. J. Lipid Res. 49, 1137–1146 (2008).
Pang, Z. et al. MetaboAnalyst 5.0: narrowing the gap between raw spectra and functional insights. Nucleic Acids Res. 49, W388–W396 (2021).
BLIGH, E. G. & DYER, W. J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 37, 911–917 (1959).
Li, F. et al. DHA does not protect ELOVL4 transgenic mice from retinal degeneration. Mol. Vis. 15, 1185–1193 (2009).
Folch, J., Lees, M. & Sloane Stanley, G. H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 226, 497–509 (1957).
Agbaga, M. P. et al. Role of Stargardt-3 macular dystrophy protein (ELOVL4) in the biosynthesis of very long chain fatty acids. Proc. Natl. Acad. Sci. USA 105, 12843–12848 (2008).
Agbaga, M. P. et al. Differential composition of DHA and very-long-chain PUFAs in rod and cone photoreceptors. J. Lipid Res. 59, 1586–1596 (2018).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25, 402–408 (2001).
Kleiner, D. E. et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 41, 1313–1321 (2005).
WEIR, J. B. New methods for calculating metabolic rate with special reference to protein metabolism. J. Physiol. 109, 1–9 (1949).
Mina, A. I. et al. CalR: a web-based analysis tool for indirect calorimetry experiments. Cell Metab. 28, 656–666.e651 (2018).
Acknowledgements
The authors would like to thank Satoshi Kinoshita and the University of Wisconsin (UW) Translational Research Initiatives in Pathology laboratory (TRIP), supported by the UW Department of Pathology and Laboratory Medicine, UWCCC (P30 CA014520) and the Office of The Director- NIH (S10OD023526) for the use of facilities and services, as well as Randall Massey and the University of Wisconsin Electron Microscope Core for tissue processing, sectioning, and assistance for this study. Confocal microscopy was performed at the University of Wisconsin-Madison Biochemistry Optical Core, which was established with support from the University of Wisconsin-Madison Department of Biochemistry Endowment. The authors want to recognize the laboratories of Dr. Freya Mowat and Janis Eells for their advice and feedback on this work. The authors would also like to extend their gratitude to Dr. Gregory Barrett-Wilt, Timothy Shriver, and the UW Biotechnology Center’s Advanced Lipidomics Platform for their time and efforts in optimizing the protocols for our lipidomics experiments. The lipidomics work was supported in part by the UW Comprehensive Diabetes Center Core Services Pilot Award UWCDC-CSPA-20-7 and the UW Office of the Vice Chancellor for Research Graduate Education with funding from the Wisconsin Alumni Research Foundation. This work was also supported by grants from the National Eye Institute (R01EY022086 to A. Ikeda; P30EY016665 to the Department of Ophthalmology and Visual Sciences at the University of Wisconsin-Madison; NIH T32EY027721 to M. Landowski; F32EY032766 to M. Landowski; R01EY030513 to M-P Agbaga), Timothy William Trout Chairmanship (A. Ikeda), Research to Prevent Blindness Unrestricted grant to Dean McGee Eye Institute (M-P. Agbaga), and NIH grants S10OD028739, R01DK131742, and R01DK124696 (C.L.E.Yen).
Author information
Authors and Affiliations
Contributions
Conceptualization—M.L., S.I., and A.I. Data curation—M.L, V.J.B., S.G., Z.H., Y.K.G., M.T., R.S.B., L.J.M., D.W.N., C.R.D., and S.I. Formal analysis—M.L., V.J.B., Z.H., Y.K.G., M.T., R.S.B., L.J.M., D.W.N., and C.R.D. Funding acquisition—M.L., C.L.E.Y., M.P.A., and A.I. Investigation—M.L., C.L.E.Y., S.I., M.P.A., and A.I. Methodology—M.L., V.J.B., S.G., M.T., R.S.B., D.W.N., C.R.D., C.L.E.Y., S.I., M.P.A., and A.I. Project administration—C.L.E.Y., S.I., M.P.A., and A.I. Resources—M.L., M.T., R.S.B., D.W.N., C.L.E.Y., S.I., M.P.A., and A.I. Supervision—A.I. Validation—M.L., V.J.B., S.G., Z.H., Y.K.G., M.T., R.S.B., L.J.M., D.W.N., C.R.D., S.I., M.P.A., and A.I. Visualization—M.L., V.J.B., S.G., D.W.N., S.I., and A.I. Writing—original draft—M.L., S.I., and A.I. Writing—review and editing—all authors have contributed to the review and editing of this manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications Biology thanks Vernat Exil and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Karli Montague-Cardoso and Gene Chong. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Landowski, M., Bhute, V.J., Grindel, S. et al. Transmembrane protein 135 regulates lipid homeostasis through its role in peroxisomal DHA metabolism. Commun Biol 6, 8 (2023). https://doi.org/10.1038/s42003-022-04404-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-022-04404-7
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.