Abstract
In marine ecology, dietary interpretations of faunal assemblages often rely on nitrogen isotopes as the main or only applicable trophic level tracer. We investigate the geographic variability and trophic level isotopic discrimination factors of bone zinc 66Zn/64Zn ratios (δ66Zn value) and compared it to collagen nitrogen and carbon stable isotope (δ15N and δ13C) values. Focusing on ringed seals (Pusa hispida) and polar bears (Ursus maritimus) from multiple Arctic archaeological sites, we investigate trophic interactions between predator and prey over a broad geographic area. All proxies show variability among sites, influenced by the regional food web baselines. However, δ66Zn shows a significantly higher homogeneity among different sites. We observe a clear trophic spacing for δ15N and δ66Zn values in all locations, yet δ66Zn analysis allows a more direct dietary comparability between spatially and temporally distinct locations than what is possible by δ15N and δ13C analysis alone. When combining all three proxies, a more detailed and refined dietary analysis is possible.
Similar content being viewed by others
Introduction
In ecology, archaeology and palaeontology, accurately reconstructing trophic levels can be challenging. Among others, these reconstructions are required for effective management and conservation strategies1, understanding changing predator-prey and foraging ecology related to climate change2 and studying long-term environmental changes through the analyses of modern to fossil faunal assemblages (and their bone collagen stable isotope values)3,4. Stable isotope analysis is an effective tool for analysing marine food webs, complementary to and often more reliable than non-stable isotope approaches5. Bone collagen and soft tissue bulk δ15N and δ13C values are the traditional geochemical proxies used for dietary and trophic level reconstructions6,7,8. These isotope analyses can now be conducted on single collagen amino acids and provide more refined information on the trophic level of animals (e.g., corrected for baseline variability)9,10, but these studies are still time consuming and expensive.
Only recently, studies of the bone’s mineral phase (bioapatite) non-traditional isotope systems such as calcium, magnesium and zinc (Zn) have shown potential as (palaeo)dietary and trophic level proxies in the terrestrial11,12,13 and marine realm14,15. Element and isotope ratios observed in a diagenetically more resistant mineral phase (e.g., enamel) can preserve dietary information beyond the scope of application of collagen, as recently demonstrated for 66Zn/64Zn ratios (expressed as the δ66Zn value)16. In addition, even when collagen is well preserved, combining traditional collagen δ15N and δ13C analyses with Zn isotope analyses of bioapatite may provide complementary dietary information as proven by the lack of correlation of those tracers within individuals of the same species, implying independent controlling mechanisms17. Therefore, analysing δ66Zn alongside traditional δ15N analyses may help verify δ15N results and provide a much-needed δ15N-independent additional geochemical trophic level and dietary indicator for ecological and archaeological studies.
Nitrogen-15 becomes relatively enriched in the tissues of aquatic consumers with successive trophic level18. The δ13C values behave much more conservatively with trophic level, increasing typically by <1‰ with trophic level for most tissues, compared to on average 3.4‰ higher δ15N values between a predator and its prey19,20. Carbon isotopes are therefore more commonly used to infer the source(s) of primary production at the base of the food web. For Zn, studies have shown a distinct 66Zn depletion in carnivore bioapatite relative to that of herbivores13,16. As muscles and most organs are typically 66Zn depleted relative to the animal’s diet and its bulk body δ66Zn composition21,22,23, bones of carnivores (and their bulk body composition) have lower δ66Zn values than their prey’s. Diet thus exerts control on the δ66Zn values of soft tissue and bioapatite. Most non-diet related factors, such as sex and age of an animal, have so far shown no effect on the isotope values and relative isotopic variability in body tissues23,24. Provenance, on the other hand, appears to affect the Zn isotope compositions of terrestrial vertebrates, though it remains unclear to what extent13,16,25. Provenance is known to play an important role for bulk δ15N and δ13C studies in marine food webs26 but has until now not been studied for δ66Zn.
In the marine realm, the δ15N and δ13C values of particulate organic matter (POM), consisting of phytoplankton, bacteria, microzooplankton and detritus, show a substantial spatial variation within and among ocean basins5,26,27,28. Variation in the isotopic composition at the base of the marine food web is passed along to higher trophic levels. Particularly for bone collagen, with its long turnover time29, trophic level reconstructions can be compromised when animals frequently migrate between areas of differing food web baseline isotopic composition, or when comparing animals from spatially and temporally distinct locations. Here, we aim at exploring Zn isotopes as a reliable tracer of marine trophic levels. To do so, we investigate combined bone δ66Zn, δ15N and δ13C from the same species across 13 locations (17 sites) in a large geographic area stretching across the Arctic from the Hudson Strait in the east to the Bering Strait in the west. We include 5 locations and two single samples with already published δ15N and δ13C values4,15,30,31, as well as one site with already published δ66Zn values15. For this study, we analysed 167 archaeological bones, concentrating on ringed seals Pusa hispida (Phoca hispida) and polar bears (Ursus maritimus). Focusing on these species allows us to investigate δ66Zn trophic level isotope discrimination factors between predator and prey geographically. Both species have a circumpolar Arctic distribution and are abundant throughout the Arctic today32,33. Particularly, P. hispida remains are frequently found in archaeological assemblages with a large temporal and spatial range30,34,35.
As we focus here on archaeological material, we have no direct food web baseline information for the isotope systems analysed herein. Therefore, we take advantage of multiple studies documenting P. hispida as the main prey species of U. maritimus and subsequently both species being close to one trophic level apart8,36,37,38,39 (Supplementary Note 1.1). We can therefore use these taxa to estimate species to species relative trophic level variability across the here studied sites. Studying the isotopic composition of high trophic level predators such as U. maritimus and P. hispida also has the advantage of their tissues’ isotopic composition dampening the effects of short-term environmental variation and integrating multiple food web channels. This effectively leads to less isotopic variability and “noise” in the animal’s tissues compared to those of lower trophic levels30. Therefore, these two species are prime targets to investigate the geographical variability of dietary proxies and trophic level isotopic discrimination factors. Anticipating an advantage of combining δ66Zn with δ15N and δ13C values, we also test in a series of bone dissolution experiments whether collagen-bound Zn influences bone δ66Zn values, which would preclude combining collagen extraction protocols with δ66Zn analysis of the mineral phase without resampling material.
Isotopic context
Marine phytoplankton from high latitudes shows particularly high variability in δ13C values40. Colder surface water temperatures lead to increasing aqueous CO2 content, and therefore a net transfer of isotopically light CO2 to the ocean and a depletion of 13C in the surface water41. Other factors influencing spatial POM δ13C values include phytoplankton growth rates, cell size and cell lipid content (see ref. 5,40 and ref. therein). Additional spatial variability may arise from the relative contribution of sea ice POM (sympagic-POM) and open water phytoplankton (pelagic-POM) to a food web. Coeval pelagic- and sympagic-POM have differing δ13C values, with the former being 13C depleted relative to the latter by 2–10‰37,42,43,44. Subtle shifts in Arctic consumers’ δ13C values for a specific area over time may occur with large-scale shifts in the relative importance of sympagic versus pelagic production related to changes in sea ice extent4,30.
A high variability in modern baseline δ15N and δ13C values is documented by the isotopic composition of POM, zooplankton, higher trophic level consumers, as well as filter feeders across the Arctic6,45,46,47,48,49,50. Pomerleau et al.48 documented a significant spatial variability in zooplankton δ15N values among the Labrador Sea, Baffin Bay and the Canadian Arctic Archipelago (CAA), but not for δ13C among these areas. Subsequent studies documented a higher variability for δ13C of POM, zooplankton and high trophic level consumers among and within these areas as well49,50. In addition, a pronounced west-east 13C depletion was observed throughout consumers from the Bering Sea (Bering Strait) through the Chukchi Sea to the Beaufort Sea6,45,46,49. A similar west-east trend was also found for sedimentary organic carbon accumulated along the Beaufort Shelf40. This eastward 13C depletion trend reaches its maximum in the south-eastern Beaufort Sea. Terrestrial organic matter derived from the Mackenzie River has δ13C values of ~−26 to −27‰ and dominates over autochthonous organic matter in the delta and at least parts of the Beaufort shelf51. Terrigenous 13C depleted carbon is also thought to play an important role for some animals (gammarid amphipods) of the Mackenzie shelf’s food web52. A similar variability in δ15N values between the Bering Sea, Chukchi Sea and Beaufort Sea is absent within animals of higher trophic levels6,53. However, geographic variations in δ15N values within these water bodies were observed for different zooplankton species6,46,49. Parson et al.52 explained high δ15N values in POM of the Mackenzie estuary instead of a low terrigenous signal as a potential bacterial recycling of nitrogen.
The eastward decrease of baseline δ13C values does not seem to continue into the CAA43,48, but significantly lower δ13C values have been reported in consumer tissues close to the Canadian mainland and in semi-enclosed basins54. In accordance, De La Vega et al.50 observed higher baseline δ13C values in inflow shelves connected to the Atlantic or Pacific Oceans (Barents Sea, Chukchi Sea) and the North Water Polynya (Northern Baffin Bay) compared to lower baseline values in the more freshwater-influenced Arctic shelves (Beaufort Sea, CAA, Hudson Bay). Lower baseline values for carbon in the more terrestrial-influenced areas are likely a result of terrigenous input and lower phytoplankton productivity. Higher stratification caused by inflowing freshwater hampers phytoplankton productivity on the interior shelves55. Indeed, Bering and Chukchi Sea annual primary production rates greatly exceed those of the Beaufort Sea6.
Zn isotopes are increasingly being used as tracers for past marine hydrochemistry56,57 and culture experiments have investigated Zn isotope fractionation in different planktonic species58,59. Still, there is hardly any data on the Zn isotope composition of natural marine planktonic organisms60,61. Indeed, data on food web baseline δ66Zn values and variability is non-existent for both the marine and continental realms. Because of biological uptake, dissolved Zn concentrations are highly depleted in marine surface waters, often much <1 nmol kg-1,62,63 and most oceans show a nutrient-like vertical distribution of dissolved Zn concentrations closely correlating with silicate concentrations64. The isotopic composition of dissolved Zn below 500 m seems to be globally homogenous with values close to +0.5‰, despite variable Zn concentrations65,66. The bulk isotopic composition of dissolved marine Zn is enriched in 66Zn relative to its major inputs from rivers and aeolian dust, which centre on the global crustal average of +0.3‰67.
Although most studies on cultured phytoplankton demonstrate a preferential uptake of light Zn into the cell relative to the bulk growth medium58,59, Atlantic and Pacific vertical dissolved Zn isotope profiles generally show lower δ66Zn values in surface waters compared to that of the deep water62,64,66,68,69. These studies demonstrate that the isotopic composition of Zn is most variable within the surface water (<500 m), often with higher values in the uppermost surface (<20 m). Surface water dissolved Zn isotope ratios vary across a North Atlantic transect from −1.1 to +0.9‰62 and across a North Pacific transect between −0.9 and +0.2‰69. Individual and combined mechanisms discussed to be responsible for this surface water δ66Zn variability include external inputs from rivers and aerosols67,69, scavenging of heavy Zn onto sinking organic matter64 and biological uptake and shallow remineralisation70.
We are unaware of any δ66Zn data from dissolved Zn in the Arctic. However, a recent study on Western Arctic dissolved Zn concentrations highlighted a deviation of Zn concentration vertical profiles from the nutrient-type Zn profiles observed in the Atlantic and Pacific71. These authors documented higher than global average surface Zn concentrations (~1.1 nmol kg-1) with a maximum concentration at 200 m and uniformly lower concentrations in the deep water. Jensen et al.71 hypothesises that Western Arctic surface water dissolved Zn originates primarily from incoming Pacific waters that are modified by shelf sediment fluxes from remineralised Zn-rich phytoplankton.
Results
Bone δ13C, δ15N and δ66Zn values
Bone collagen δ13C, δ15N and bone δ66Zn values of P. hispida, U. maritimus, harp seal (Pagophilus groenlandicus) and beluga whale (Delphinapterus leucas) are reported in Table 1 and Supplementary Data 1. All collagen samples had yields and elemental (wt% C, wt% N, C:Natomic) compositions characteristic of samples with isotopic compositions not altered by contaminant or degradation in the burial environment72,73 (Supplementary Data 1). Likewise, δ66Zn values do not indicate a modification due to diagenesis or contamination for the majority of samples, but we cannot exclude it as a possibility for outlier values (Supplementary Discussion 3.1, Supplementary Fig. 1). Within a site, we observe typically higher bulk collagen δ15N values and lower bone mineral δ66Zn values in U. maritimus relative to P. hispida and other prey species (Table 1, Fig. 1). Our results from applying different dissolution methods to reference materials and bone samples also indicate that the presence of collagen-bound Zn, and thereby collagen preservation, has no effect on the mineral phase δ66Zn values (Supplementary Methods 2.1). Mineral phase δ66Zn analyses can thus be coupled with collagen extraction protocols, provided precautions are taken to avoid Zn contamination (Supplementary Figs. 2–3, Supplementary Data 2, Supplementary Discussion 3.2).
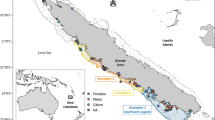
Pusa hispida (squares, n = 104) and U. maritimus (dots, n = 47) bone samples are colour coded as geographic groups. a Schematic map indicating the archaeological sites analysed and geographic colour coding: Light green for the Bering Strait; dark green for the Amundsen and Coronation Gulf; blue for the CAA; orange for the Hudson Bay; purple for the North Water Polynya; and red for sites influenced by the Labrador Sea in the Hudson Strait and Frobisher Bay. b δ15N versus δ13C plot for P. hispida samples (p-value < 0.05; R2 = 0.21; n = 104). c) δ15N versus δ66Zn plot for P. hispida samples (p-value < 0.05; R2 = 0.08; n = 104). d δ15N versus δ13C plot for U. maritimus samples (no correlation, p-value > 0.05; n = 47). e δ15N versus δ66Zn plot for U. maritimus samples (p-value < 0.05; R2 = 0.42; n = 47). We included already published δ15N and δ13C values4,15,30,31 and already published δ66Zn values from QjJx-115. The map is redrawn and modified using Adobe Illustrator CS6 after www.google.com/maps. Error bars represent the measurement uncertainty.
Statistical investigation of isotope values
Statistically significant differences between P. hispida populations was determined through ANOVA for δ13C (F(12, 91) = 24.4, p-value < 2e−16), and δ66Zn values (F(12, 91) = 5.867, p-value 1.93e−07), and through Welch ANOVA for δ15N (F(12, 32.2) = 71.8, p-value 1.00e−19). Post hoc Tukey pair-wise comparisons draw out the populations from Little Cornwallis (QjJx-1) and the North shore of Devon Island (QkHn-13) both part of the CAA, as well as eastern Ellesmere Island (near Skraeling Island, SfFk-4), linked to the North Water Polynya, as distinct from some of the other sites in regard to their δ66Zn values (Supplementary Figs. 4–5, Supplementary Data 3). Every other site, regardless of their broad geographic group, are not significantly different from one another.
Results for pair-wise comparisons of sites’ δ13C and δ15N P. hispida values show a higher degree of heterogeneity (Supplementary Figs. 6–7, Supplementary Data 3). However, most of the differences can be linked to geographic groups. Sites from the CAA are being drawn out as different in their δ13C and δ15N values to most of the other sites. The western sites of the Amundsen and Coronation Gulf, as well as the Bering Strait, differ in their δ15N values, but not for δ13C values. Finally, δ13C and δ15N values from the Eastern sites of the Hudson Bay and the Labrador Sea are identified as significantly different than those of western sites.
Levene’s tests for equal variance show that δ66Zn values are more homogeneous between P. hispida and U. maritimus (F(1, 125) = 3.43, p = 0.27) and across sites (F(8, 118) = 1.72, p = 0.40) than δ15N values (respectively F(1, 125) = 6.95, p = 0.04; and F(1, 118) = 2.62, p = 0.05).
Discussion
The maximum variability for inter-site mean P. hispida bone δ15N and δ13C values (3.55 and 3.40‰) exceeds the maximum intra-site (1.77 and 2.67‰, Fig. 1) and typical trophic level variability in δ15N between predator and prey (e.g., +3.4 to +3.8‰)8,18,19. The QjJx-1 site on Little Cornwallis Island is a notable exception with very high on-site P. hispida bone collagen δ15N variability (3.85‰)15. Post hoc Games-Howell and Tukey pair-wise comparisons demonstrate a large heterogeneity in δ15N and δ13C values between archaeological populations (Supplementary Figs. 6–7). Isotopic heterogeneity between populations is related to geographic location resulting in δ15N and δ13C values from populations of different regions plotting in distinct groups on a δ15N versus δ13C plot (Fig. 1b). Based on site proximity and sample isotopic composition, we grouped sites from the Bering/Chukchi Sea, Amundsen and Coronation Gulf, CAA, North Water Polynya, Hudson Bay, and sites influenced by the Labrador Sea (Hudson Strait and Frobisher Bay, Fig. 1a). The δ15N and δ13C variability between the archaeological sites is in good agreement in both spacing and amplitude with modern geographical variations observed from zooplankton28,45,46 and higher consumer soft tissue6,49 including P. hispida37,53,54,74,75,76 and U. maritimus33. While dietary differences between populations may have contributed to the geographic spacing of δ15N and δ13C values, integration of regional baseline isotopic patterns is likely the main factor controlling the observed inter-site isotopic variability in P. hispida and U. maritimus collagen (Supplementary Discussion 3.3).
In contrast to P. hispida bone δ15N and δ13C values, the highest variability for mean δ66Zn values between sites (0.23‰) does not exceed the maximum variability observed within a single site (0.36‰). In addition, mean δ66Zn values between sites never exceed the variability between mean U. maritimus and P. hispida bone δ66Zn values (Δ66ZnU. maritimus − P. hispida = −0.32‰). While ANOVA analysis of P. hispida δ66Zn values did reveal statistically significant differences between P. hispida populations, post hoc pair-wise comparison tests show considerably more homogeneity in δ66Zn values than for δ15N and δ13C values (Supplementary Figs. 4–7). Similarly, Levene’s tests for equal variance show that for all sites, P. hispida and U. maritimus δ66Zn values have an equal variance, whereas δ15N values are more heterogeneous. The low geographical δ66Zn variability in P. hispida and U. maritimus bones implies that Arctic food web baseline and/or low trophic level consumer δ66Zn values are more homogenous than for δ15N and δ13C values. This is remarkable considering the large surface water’s isotopic variability observed for dissolved Zn across the Atlantic and Pacific of −1.1 to +0.9‰ and −0.9 to +0.2‰, respectively62,69.
Based on post hoc Tukey pair-wise comparison, δ66Zn values from P. hispida populations from the sites QjJx-1 (Little Cornwallis Island), QkHn-13 (Devon Island) and SfFk-4 (eastern Ellesmere Island) were identified as statistically different from other populations (Fig. 2, Supplementary Fig. 4). Differences in the dietary Zn resources of these populations relative to others may have caused these statistical anomalies. Alternatively, they may reflect true variability in the δ66Zn regional food web baselines. QjJx-1 and QkHn-13 are located within the CAA. The CAA is composed of multiple channels and interconnected basins, in which water mass modification and transport are governed by its complex topography and shelf exchange processes77. Within this setting, baseline δ66Zn values are perhaps more variable on a regional scale than for the rest of the Arctic. For the SfFk-4 site, we observe in all three species analysed lower mean δ66Zn values compared to other sites indicating a regionally lower baseline δ66Zn value (Figs. 2, 3). The SfFk-4 site is located at the biologically highly productive78 northern edge of the North Water Polynya, a region in which the reduced ice-cover or ice-free conditions influence biological processes (e.g., by upwelling, increased nutrient renewal)79, which in turn may modify the δ66Zn baseline.
Range of δ13C (a), δ15N (b) and δ66Zn (c) values for P. hispida and U. maritimus bones for all locations. Site names are colour coded following Fig. 1. Dashed lines represent mean values when including all sites. We included already published δ15N and δ13C values from sites RbJu-1, PaJs-13, QkHn-13, KTZ and QjJx-1 sites4,15,30,31 and already published δ66Zn values from QjJx-115. Error bars represent the measurement uncertainty.
Isotopic composition (δ15N, δ13C versus δ66Zn) of U. maritimus (magenta circles), P. hispida (brown squares), P. groenlandicus (green triangle) and D. leucas (blue stars) bones for the SfFk-4 (a, c) and combined KkDo-1 and JfEl-4 sites (b, d). For (a) and (c) we present n = 5 U. maritimus, n = 8 P. hispida and n = 4 P. groenlandicus bone samples and for (b) and (d) n = 7 U. maritimus, n = 18 P. hispida, n = 7 P. groenlandicus and n = 2 D. leucas bone samples. Error bars represent the measurement uncertainty.
Both δ66Zn and δ15N values are controlled by diet but show a better correlation for U. maritimus samples across all sites than for P. hispida (Fig. 1c, e), perhaps related to the more specialised diet of U. maritimus38,39. As with δ15N values, bone δ66Zn values clearly demonstrate a trophic spacing between U. maritimus and P. hispida in all locations analysed (Fig. 2). The KkJg-1 site in Hudson Bay is an exception with two U. maritimus samples showing anomalously high δ66Zn values which may relate to non-dietary factors such as contamination, misidentification, diagenesis, or physiological effects (Supplementary Discussion 3.1). Even when including the KkJg-1 site, Levene’s tests for equal variance demonstrate an equal variance between P. hispida and U. maritimus δ66Zn values, whereas their δ15N values demonstrate heterogeneity. Because δ66Zn is more homogenous in its value for a specific taxon, and possibly diet, δ66Zn may more reliably reflect trophic levels than bulk δ15N values, when investigating multiple species across multiple sites, proving a better inter-site comparability.
Ursus maritimus bone δ66Zn values are on average 0.32‰ lower than those of P. hispida (mean Δ66ZnU. maritimus − P. hispida = −0.32‰). Because P. hispida is typically the primary prey species of U. maritimus for most locations today38,39, we predict this Δ66ZnU. maritimus − P. hispida value to be close to the Zn bone trophic level discrimination factor between a carnivore and its prey, when soft tissue is consumed. Previously, estimations of trophic discrimination factors between bioapatite of terrestrial mammalian carnivores and herbivores were between −0.6 and −0.4‰, respectively for the Tam Hay Marklot (THM) cave16 and the modern Koobi Fora region13. These studies, however, had a lower sample size and compared multiple carnivores and herbivores with varying diets. Predicted bone δ66Zn trophic level discrimination factors are between −0.36 and −0.38‰ when calculated using individual δ15N trophic levels8 from all marine mammal taxa with available δ66Zn data (Supplementary Discussion 3.4, Supplementary Tables 1–2, Supplementary Fig. 8). However, these estimations are oversimplified, not considering population-specific dietary differences, location-specific baseline variations and organism-specific trophic and tissue-type enrichment factors. We cannot exclude different δ66Zn trophic level discrimination factors between tissues of P. hispida relative to their prey (which could not be analysed herein). The δ66Zn values of different tissues vary within an organism21,22,23. It is therefore possible that when different tissues are consumed (e.g., consumption of soft tissue only versus consumption of the whole organisms), δ66Zn trophic level discrimination factors might vary as well. Nevertheless, bone δ66Zn trophic level discrimination factors calculated from δ15N trophic levels8 are close to the mean Δ66ZnU. maritimus − P. hispida value and should at least approximate true discrimination factors between bones of a carnivore and its prey.
Particularly for archaeological material, assigning a relative trophic level to multiple species when utilising δ15N values alone can be challenging, as shown by the large differences in mean δ15N trophic discrimination factors between U. maritimus and P. hispida (Δ15NU. maritimus − P. hispida) for individual sites (+2.2 to +7.0 ‰, Fig. 2b). Besides locally differing diet, Δ15NU. maritimus - P. hispida variability (and Δ66ZnU. maritimus − P. hispida) may be influenced by physiological effects or unknown archaeological assemblage effects related to human hunting and/or scavenging. Relative differences in the consumption of higher and lower trophic level prey alone are unlikely to explain the Δ15NU. maritimus − P. hispida variability. As P. hispida bones are the most abundant fauna remains in all archaeological sites analysed herein30,34,35, it stands to reason that they were a similarly important food item for the archaeological U. maritimus populations as they are today38,39. Therefore, we expect even substantial differences in P. hispida trophic levels among sites to have only a small effect on Δ15NU. maritimus − P. hispida. Feeding at substantially different trophic levels is incompatible with modern P. hispida and U. maritimus population trophic levels and diet variability38,39,54. In addition, most other U. maritimus prey species feed on lower or similar trophic levels relative to P. hispida8,80.
It is possible that due to the low intra-site sample size for both or either species, our site mean isotopic values do not capture the true means of the different populations. As the bones analysed are from individuals hunted or scavenged by humans, we cannot exclude differences in the segments of a P. hispida population hunted by humans and U. maritimus. For example, remains of P. hispida pups are very rare in archaeological assemblages34,81. Ursus maritimus, however, regularly preys on P. hispida pups and the contribution of pups to its diet may vary for different individuals, populations and with seal productivity82,83. As pups rely on their mother’s milk, they effectively feed on a different trophic level leading to higher collagen δ15N values in pups than adults84. Consequently, a higher consumption of P. hispida pups by U. maritimus relative to humans can lead to higher Δ15NU. maritimus − P. hispida values within an archaeological assemblage. Additional uncertainties for inter-site Δ15NU. maritimus − P. hispida values may arise from a higher contribution of migratory species such as D. leucas85 to the diet of certain U. maritimus populations39. Differences in the migratory behaviour of P. hispida86 and/or U. maritimus87 among sites could also lead to both species feeding along food chains with differing baseline δ15N values and therefore variable Δ15NU. maritimus − P. hispida values within the assemblages. It remains, as of yet, unclear if and how physiological effects may influence δ66Zn variability within a population. However, dietary differences as well as effects related to an archaeological assemblage (e.g., not capturing true population means) might have a similar effect on Δ66ZnU. maritimus − P. hispida as on Δ15NU. maritimus − P. hispida.
For δ66Zn, two sites from the same geographic area close to the Labrador Sea (KkDo-1 and JfEl-4) have a markedly lower trophic spacing between P. hispida and U. maritimus δ66Zn values of −0.22 and −0.24‰ (Fig. 2). Modern U. maritimus individuals from the area belong to the Davis Strait population88. In addition to P. hispida and contrary to most other U. maritimus populations, this one obtains a large percentage of its biomass from the consumption of harp seals (Pagophilus groenlandicus)38,39. However, bone δ66Zn values of P. hispida and P. groenlandicus from the same site are indistinguishable (Fig. 3, Supplementary Fig. 9). Instead of the consumption of P. groenlandicus, the lower trophic discrimination factor for these sites may arise from unknown dietary contributions, population-specific physiological effects or unknown archaeological assemblage effects.
Unlike P. hispida, most P. groenlandicus leave their Canadian Arctic summering grounds, ahead of the formation of local pack ice in autumn89,90. However, when sympatric with P. hispida, P. groenlandicus feeds at a similar trophic level, consuming many of the same prey species, and both species show no statistical difference in muscle and liver δ15N values91. Indeed, bones of both seal species cover the same δ66Zn range for the same location (Fig. 3). However, bones of P. groenlandicus from Hudson Strait (KkDo-1) have almost 1‰ lower mean δ15N and δ13C values than those of P. hispida, perhaps related to this species’ seasonal southwards migration (Fig. 3b, d). In contrast, some P. groenlandicus individuals remain in west Greenland waters during winter92,93, which may explain why bones of both seal species from eastern Ellesmere Island show a similar δ13C, δ15N and δ66Zn range (Fig. 3a, c). Due to its long turnover time, the bone collagen isotopic composition of P. groenlandicus likely represents an amalgamation of different food sources and local isotopic baseline values along their migration route and within their seasonal feeding grounds. P. groenlandicus bone δ66Zn values do not seem to record migratory signals, again arguing for lower baseline variability or Zn isotope homogenisation within low trophic level organisms. Despite a very low samples size (n = 2) beluga whale (Delphinapterus leucas) δ66Zn values fall within the same range as P. hispida and P. groenlandicus with slightly higher mean values (Fig. 3b, d). Indeed, all three species occupy a similar trophic level8,91. When sympatric with P. hispida, D. leucas typically has slightly lower soft tissue δ15N values likely due to migrating between areas with differing baselines or a more offshore/pelagic foraging8,76. Here, D. leucas δ15N values are higher than those of P. hispida and their δ13C values are highly variable (Fig. 3). Instead of only reflecting this species’ trophic level relative to P. hispida, their bulk collagen δ15N and δ13C values are likely influenced by the high mobility of this species94 and its foraging in locations with different isotopic baselines.
The trophic levels of U. maritimus, P. hispida and P. groenlandicus are reflected by their bone collagen δ15N and bone δ66Zn values across the Arctic. The analysis of Zn isotopes, however, offers additional advantages for studying marine trophic ecology, not only due to its greater preservation potential in fossil material16, but also due to the lower baseline controlled species-specific spatial isotopic gradients. The inclusion of δ66Zn analysis in ecological, archaeological and palaeontological studies may thus allow more robust interpretations of spatial and temporal trophic interactions. In addition, while both δ66Zn and δ15N generally record trophic levels, they do not record physiological and/or dietary effects equally, thus providing a strong incentive to combine δ66Zn with δ15N and δ13C analyses where possible.
In conclusion, this study compares archaeological bone δ66Zn values with traditional collagen δ15N and δ13C values for the same species across a large geographic area. Focussing on prey (P. hispida) and predator (U. maritimus) we investigate the baseline variability and trophic spacing of these dietary proxies. Our results show that:
-
(1)
Overall, δ66Zn values shows less site-specific variability within a species, likely due to a lower baseline variability than for δ15N and δ13C. As such, δ66Zn values are particularly valuable for dietary studies on highly mobile species (or consumers thereof) and for comparing geographically and temporally distinct populations.
-
(2)
We observe the expected trophic level spacing for collagen δ15N and bone δ66Zn values between U. maritimus and P. hispida. U. maritimus bone δ66Zn values are on average 0.32 ‰ lower than of its primary prey P. hispida.
-
(3)
Bone δ66Zn values of the migratory species P. groenlandicus and D. leucas are consistent with respect to their known trophic levels when compared with P. hispida and U. maritimus values. In contrast, their collagen δ15N (and δ13C) values appear to document their relative trophic levels less precisely, likely influenced by variations in baseline isotopic compositions along their migration routes.
In ecological, archaeological and palaeontological research, trophic level estimations often rely exclusively on the δ15N tracer, sometimes biased by physiological, habitat and baseline effects. We demonstrate that the inclusion of δ66Zn analysis can provide otherwise inaccessible supplementary dietary information and more robust trophic level estimations.
Methods
For this study, we compare δ66Zn, δ15N and δ13C values of 105 P. hispida, 47 U. maritimus, 11 P. groenlandicus and 2 D. leucas archaeological bone samples from across the Arctic (Supplementary Data 1). The data presented herein includes already-published δ66Zn values from an archaeological site (QjJx-1) on Little Cornwallis Island15. Additional δ66Zn values analysed for this study comprise 93 P. hispida bone samples from 13 archaeological sites (12 locations) and 37 U. maritimus bone samples from 11 archaeological sites (8 locations) as well as P. groenlandicus and D. leucas samples (2 sites, 1 site, respectively). For 6 of the 17 sites analysed here (RbJu-1, PaJs-13, QkHn-13, QjJx-1, KTZ sites), δ15N and δ13C values were already published elsewhere4,15,30. In addition, δ15N and δ13C values from one P. hispida sample from the NkRi-3 and OkRn-1 sites were already published elsewhere (Sample Nr. 4945, 9535)31. For the sites JfEl-4, KcFs-2, NkRi-3, seal bones analysed are identified as most likely P. hispida, but we cannot completely rule out that some samples may also come from other Phocidea (Supplementary Discussion 3.1). A single walrus (Odobenus rosmarus) bone and a potentially misidentified D. leucas bone from the JfEl-4 site were also measured and are compared to previously measured O. rosmarus bones from the QjJx-1 site15 and our δ66Zn data from other species and sites in the Supplementary Discussion 3.4. Additional information and references regarding the archaeological context of the samples and sites are provided in the Supplementary Note 1.2 and Supplementary Table 3.
Zinc analysis
All samples’ surfaces were mechanically abraded (cleaned) to avoid sediment contamination, using a dental drill equipped with a diamond-tipped burr. Approximately 10 to 50 mg chunks were then sampled using a diamond-tipped cutting wheel. The chunks were then ultrasonicated in ultrapure water (Milli-Q water) for 5 min and dried in a drying chamber for a few days at 50 °C. Bone samples and reference materials NIST SRM 1400 and NIST SRM 1486 were subjected to different dissolution methods (HCl and HNO3) to investigate the impact of the organic bone phases on its Zn isotope signal (Supplementary Methods 2.1, Supplementary Discussion 3.2). The column chromatography steps (3.1.2) for quantitative recovery of sample Zn95,96 was the same for all samples regardless of the dissolution methods used. Each column chromatography batch (n = 15) included up to 13 samples, one chemistry blank and at least one reference standard (SRM 1400 and/or 1486).
Zn purification was performed in two steps, following the modified ion exchange method adapted from Moynier et al.96, first described in Jaouen et al.13. Each step included AG-1 × 8 resin that was cleaned and conditioned prior to sample loading. One ml of AG-1 × 8 resin (200–400 mesh) was placed in 10 ml hydrophobic interaction columns (Macro-Prep® Methyl HIC). Resin cleaning involved 5 ml 3% HNO3 followed by 5 ml ultrapure water. These cleaning steps were repeated. The resin was then conditioned with 3 ml 1.5 M HBr. After loading, 2 ml HBr were added for matrix residue elution followed by Zn elution with 5 ml HNO3. Following the second column step, the solution was evaporated for 13 h at 100 °C and the residue re-dissolved in 1 ml 3% HNO3.
Zn isotope ratios were measured using a Thermo Fisher Neptune MC-ICP-MS at the Max Planck Institute for Evolutionary Anthropology (Leipzig, Germany) and a Thermo Fisher Neptune Plus MC-ICP-MS at the Géosciences Environnement Toulouse - Observatoire Midi-Pyrénées (Toulouse, France). Instrumental mass fractionation was corrected by Cu doping following the protocol of Maréchal et al.95 and Toutain et al.97. The in-house reference material Zn AA-MPI was used for standard bracketing. δ66Zn values are expressed relative to the JMC-Lyon reference material. Analysed sample solution Zn concentrations were close to 300 ppb as was the Zn concentration used for the standard mixture solution. Zn concentrations in the respective samples were estimated following a protocol adapted from one used for Sr by Copeland et al.98, applying a regression equation based on the 64Zn signal intensity (V) of three solutions with known Zn concentrations (150, 300 and 600 ppb). δ66Zn uncertainties were estimated from standard replicate analyses and ranged between ±0.01‰ and ±0.03‰ (1 SD). Additional reference materials SRM 1486 and SRM 1400 were analysed alongside the samples. SRM reference materials and samples show a normal Zn mass dependent isotopic fractionation, i.e., the absence of isobaric interferences, as the δ66Zn vs. δ67Zn and δ66Zn vs. δ68Zn values fall onto lines with slopes close to the theoretic mass fractionation values of 1.5 and 2, respectively.
Carbon and nitrogen isotope analysis
Bone surfaces were cleaned with a dental drill equipped with a diamond-cutting wheel. Subsamples of bone chunks (100–200 mg) were demineralised in 0.5 M HCl at 4 °C. After demineralisation, samples were rinsed to neutrality with Type I water (resistivity > 18.2 MΩ cm). Any bone samples with dark colouration were treated with 0.1 M NaOH for successive 30 min treatments under sonication at room temperature until the solution no longer changed colour. The samples rinsed to neutrality with Type I water and then the insoluble collagen residue was solubilised in ~8 ml of 0.01 M HCl at 75 °C for 48 h. The resulting solution containing the solubilised collagen was filtered through a 5–8 μm filter and then filtered using a Microsep® 30 kDa molecular weight cut-off (MWCO) ultrafilter (Pall Corporation, Port Washington, NY) to remove low molecular weight compounds99. The >30 kDa fraction was freeze-dried, and the collagen yield was calculated.
Carbon and nitrogen isotopic and elemental compositions were determined using an IsoPrime continuous flow isotope-ratio mass spectrometer (CF-IRMS) coupled to a Vario Micro elemental analyser (Elementar, Hanau, Germany). Carbon and nitrogen isotopic compositions were calibrated relative to the VPDB and AIR scales, respectively, using a two-point calibration anchored by USGS40 (accepted δ13C − 26.39 ± 0.04‰, δ15N − 4.52 ± 0.06‰) and USGS41 (accepted δ13C + 37.63 ± 0.05‰, δ15N + 47.57 ± 0.11‰)100. Standard uncertainty was determined to be ±0.20‰ for δ13C and ±0.25‰ for δ15N101. Additional details are provided in the Supplementary Methods 2.2 and Supplementary Tables 4–6.
Statistics and reproducibility
All 144 samples analysed herein for δ66Zn and all 102 samples analysed for δ13C and δ15N were measured when possible, at least in duplicate with a mean standard deviation for sample replicates of ±0.01‰, ±0.12‰ and ±0.14‰, respectively.
Analysis of variance (ANOVA) were performed across the dataset in order to determine statistical differences in δ13C, δ15N and δ66Zn values between P. hispida populations. A single P. hispida specimen from Little Cornwallis Island was excluded from the statistical analysis (δ66Zn = 1.00‰, from Jaouen et al.15), as it could disproportionately influence the analysis (see Supplementary Fig. 4 versus 5). It was singled-out as an extreme outlier lying more than three times the interquartile range above the third quartile, both within-site and for the whole P. hispida dataset. Where variance was found to be significant, post hoc Tukey pair-wise comparisons were carried out to determine which populations were significantly different from each other in terms of their δ13C, δ15N and δ66Zn values. To adhere to ANOVA’s assumptions, each P. hispida populations’ δ13C, δ15N and δ66Zn datasets underwent visual inspection to check for normally distributed and homogeneous residuals, as well as tested for equal variance using Levene’s test. Accordingly, we report the results of ANOVAs and post hoc Tukey pair-wise comparisons (Supplementary Figs. 4–7, Supplementary Data 3). As the δ15N dataset violated the equal variance assumption, an alternative Welch ANOVA was conducted instead, with post hoc Games-Howell pair-wise comparisons. In order to investigate the homogeneity of δ66Zn values within the Arctic relative to δ15N values, a series of Levene’s test for equal variance (with Bonferroni correction) was performed on Zn and N isotope values between P. hispida and U. maritimus, as well as between sites for which data are available for both species. All statistical analyses were conducted using the free program R software102.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
All data generated during this study are included in this published article (and Supplementary Data 1–3). Provenance information including sample ID, Bordon code, sampling location, feature and sample source are given in Supplementary Data 1, with additional site information in Supplementary Table 3.
Change history
23 June 2021
A Correction to this paper has been published: https://doi.org/10.1038/s42003-021-02343-3
References
Horstmann‐Dehn, L., Follmann, E. H., Rosa, C., Zelensky, G. & George, C. Stable carbon and nitrogen isotope ratios in muscle and epidermis of arctic whales. Mar. Mamm. Sci. 28, E173–E190 (2012).
Winder, M. & Schindler, D. E. Climate change uncouples trophic interactions in an aquatic ecosystem. Ecology 85, 2100–2106 (2004).
Misarti, N., Finney, B. P., Maschner, H. & Wooller, M. J. Changes in northeast Pacific marine ecosystems over the last 4500 years: evidence from stable isotope analysis of bone collagen from archaeological middens. Holocene 19, 1139–1151 (2009).
Szpak, P., Buckley, M., Darwent, C. M. & Richards, M. P. Long-term ecological changes in marine mammals driven by recent warming in northwestern Alaska. Glob. Chang. Biol. 24, 490–503 (2018).
Michener, R. H. & Kaufman, L. in Stable Isotopes in Ecology and Environmental Science (eds Michener, R. & Lajtha, K.), 238–282 (Oxford, 2007).
Dunton, K. H., Saupe, S. M., Golikov, A. N., Schell, D. M. & Schonberg, S. V. Trophic relationships and isotopic gradients among arctic and subarctic marine fauna. Mar. Ecol. Prog. Ser. 56, 89–97 (1989).
Ramsay, M. A. & Hobson, K. A. Polar bears make little use of terrestrial food webs: evidence from stable-carbon isotope analysis. Oecologia 86, 598–600 (1991).
Hobson, K. A. & Welch, H. E. Determination of trophic relationships within a high Arctic marine food web using δ13C and δ15N analysis. Mar. Ecol. Prog. Ser. 84, 9–18 (1992).
Evershed, R. P. et al. in Stable Isotopes in Ecology and Environmental Science (eds Michener, R. & Lajtha, K.) 480–540 (Oxford, 2007).
Jaouen, K. et al. Exceptionally high δ15N values in collagen single amino acids confirm Neandertals as high-trophic level carnivores. Proc. Natl Acad. Sci. USA 116, 4928–4933 (2019).
Heuser, A., Tütken, T., Gussone, N. & Galer, S. J. Calcium isotopes in fossil bones and teeth − Diagenetic versus biogenic origin. Geochim. Cosmochim. Acta 75, 3419–3433 (2011).
Martin, J. E., Vance, D. & Balter, V. Natural variation of magnesium isotopes in mammal bones and teeth from two South African trophic chains. Geochim. Cosmochim. Acta 130, 12–20 (2014).
Jaouen, K., Beasley, M., Schoeninger, M., Hublin, J. J. & Richards, M. P. Zinc isotope ratios of bones and teeth as new dietary indicators: results from a modern food web (Koobi Fora, Kenya). Sci. Rep. 6, 26281 (2016).
Martin, J. E., Tacail, T., Adnet, S., Girard, C. & Balter, V. Calcium isotopes reveal the trophic position of extant and fossil elasmobranchs. Chem. Geol. 415, 118–125 (2015).
Jaouen, K., Szpak, P. & Richards, M. P. Zinc isotope ratios as indicators of diet and trophic level in arctic marine mammals. PLoS ONE 11, e0152299 (2016).
Bourgon, N. et al. Zinc isotopes in Late Pleistocene fossil teeth from a Southeast Asian cave setting preserve paleodietary information. Proc. Natl Acad. Sci. USA 117, 4675–4681 (2020).
Jaouen, K. What is our toolbox of analytical chemistry for exploring ancient hominin diets in the absence of organic preservation? Quat. Sci. Rev. 197, 307–318 (2018).
Minagawa, M. & Wada, E. Stepwise enrichment of 15N along food chains: further evidence and the relation between δ15N and animal age. Geochim. Cosmochim. Acta 48, 1135–1140 (1984).
Vander Zanden, M. J. & Rasmussen, J. B. Variation in δ15N and δ13C trophic fractionation: implications for aquatic food web studies. Limnol. Oceanogr. 46, 2061–2066 (2001).
Post, D. M. Using stable isotopes to estimate trophic position: models, methods, and assumptions. Ecology 83, 703–718 (2002).
Moynier, F., Fujii, T., Shaw, A. S. & Le Borgne, M. Heterogeneous distribution of natural zinc isotopes in mice. Metallomics 5, 693–699 (2013).
Balter, V. et al. Contrasting Cu, Fe, and Zn isotopic patterns in organs and body fluids of mice and sheep, with emphasis on cellular fractionation. Metallomics 5, 1470–1482 (2013).
Mahan, B., Moynier, F., Jørgensen, A. L., Habekost, M. & Siebert, J. Examining the homeostatic distribution of metals and Zn isotopes in Göttingen minipigs. Metallomics 10, 1264–1281 (2018).
Jaouen, K. et al. Dynamic homeostasis modeling of Zn isotope ratios in the human body. Metallomics 11, 1049–1059 (2019).
Jaouen, K. et al. Zinc isotope variations in archeological human teeth (Lapa do Santo, Brazil) reveal dietary transitions in childhood and no contamination from gloves. PLoS ONE 15, e0232379 (2020).
McMahon, K. W., Hamady, L. L. & Thorrold, S. R. Ocean ecogeochemistry: a review. Oceanogr. Mar. Biol. 51, 327–374 (2013).
Rau, G. H., Sweeney, R. E. & Kaplan, I. R. Plankton 13C:12C ratio changes with latitude: differences between northern and southern oceans. Deep Sea Res. Part I Oceanogr. Res. 29, 1035–1039 (1982).
McMahon, K. W., Hamady, L. L. & Thorrold, S. R. A review of ecogeochemistry approaches to estimating movements of marine animals. Limnol. Oceanogr. 58, 697–714 (2013).
Hedges, R. E., Clement, J. G., Thomas, C. D. L. & O’Connell, T. C. Collagen turnover in the adult femoral mid‐shaft: modeled from anthropogenic radiocarbon tracer measurements. Am. J. Phys. Anthropol. 133, 808–816 (2007).
Szpak, P., Savelle, J. M., Conolly, J. & Richards, M. P. Variation in late Holocene marine environments in the Canadian Arctic Archipelago: evidence from ringed seal bone collagen stable isotope compositions. Quat. Sci. Rev. 211, 136–155 (2019).
Szpak, P. & Buckley, M. Sulfur isotopes (δ34S) in Arctic marine mammals: indicators of benthic vs. pelagic foraging? Mar. Ecol. Prog. Ser. https://doi.org/10.3354/meps13493 (2020).
Reeves, R. R. in Ringed Seals in the North Atlantic (eds Heide-Jørgensen, M. P. & Lydersen, C.) 9–45 (NAMMCO Scientific Publications, 1998).
Koehler, G., Kardynal, K. J. & Hobson, K. A. Geographical assignment of polar bears using multi-element isoscapes. Sci. Rep. 9, 9390 (2019).
Moody, J. F. & Hodgetts, L. M. Subsistence practices of pioneering Thule–Inuit: a faunal analysis of Tiktalik. Arct. Anthropol. 50, 4–24 (2013).
Dyke, A. S. et al. An assessment of marine reservoir corrections for radiocarbon dates on walrus from the Foxe Basin region of Arctic Canada. Radiocarbon 61, 67–81 (2019).
Derocher, A. E., Wiig, Ø. & Andersen, M. Diet composition of polar bears in Svalbard and the western Barents Sea. Polar Biol. 25, 448–452 (2002).
Hobson, K. A. et al. A stable isotope (δ13C, δ15N) model for the North Water food web: implications for evaluating trophodynamics and the flow of energy and contaminants. Deep Sea Res. Part II Top. Stud. Oceanogr. 49, 5131–5150 (2002).
Iverson, S. J., Stirling, I. & Lang, S. L. C. in Top Predators in Marine Ecosystems (eds Boyd, I. L., Wanless, S. & Camphuysen, C. J.) 98–117 (Cambridge University Press, 2006).
Thiemann, G. W., Iverson, S. J. & Stirling, I. Polar bear diets and arctic marine food webs: insights from fatty acid analysis. Ecol. Monogr. 78, 591–613 (2008).
Stein, R. & MacDonald, R. W. The Organic Carbon Cycle in the Arctic Ocean (Springer, 2004).
Lynch‐Stieglitz, J., Stocker, T. F., Broecker, W. S. & Fairbanks, R. G. The influence of air‐sea exchange on the isotopic composition of oceanic carbon: Observations and modeling. Glob. Biogeochem. Cycles 9, 653–665 (1995).
Hobson, K. A., Ambrose, W. G. Jr & Renaud, P. E. Sources of primary production, benthic-pelagic coupling, and trophic relationships within the Northeast Water Polynya: insights from δ13C and δ15N analysis. Mar. Ecol. Prog. Ser. 128, 1–10 (1995).
France, R., Loret, J., Mathews, R. & Springer, J. Longitudinal variation in zooplankton δ13C through the Northwest Passage: inference for incorporation of sea-ice POM into pelagic foodwebs. Polar Biol. 20, 335–341 (1998).
Søreide, J. E., Hop, H., Carroll, M. L., Falk-Petersen, S. & Hegseth, E. N. Seasonal food web structures and sympagic–pelagic coupling in the European Arctic revealed by stable isotopes and a two-source food web model. Prog. Oceanogr. 71, 59–87 (2006).
Saupe, S. M., Schell, D. M. & Griffiths, W. B. Carbon-isotope ratio gradients in western arctic zooplankton. Mar. Biol. 103, 427–432 (1989).
Schell, D. M., Barnett, B. A. & Vinette, K. A. Carbon and nitrogen isotope ratios in zooplankton of the Bering, Chukchi and Beaufort seas. Mar. Ecol. Prog. Ser. 162, 11–23 (1998).
Tamelander, T., Kivimäe, C., Bellerby, R. G., Renaud, P. E. & Kristiansen, S. Base-line variations in stable isotope values in an Arctic marine ecosystem: effects of carbon and nitrogen uptake by phytoplankton. Hydrobiologia 630, 63–73 (2009).
Pomerleau, C. et al. Spatial patterns in zooplankton communities across the eastern Canadian sub-Arctic and Arctic waters: insights from stable carbon (δ13C) and nitrogen (δ15N) isotope ratios. J. Plankton Res. 33, 1779–1792 (2011).
Pomerleau, C. et al. Pan-Arctic concentrations of mercury and stable isotope ratios of carbon (δ13C) and nitrogen (δ15N) in marine zooplankton. Sci. Total Environ. 551, 92–100 (2016).
De la Vega, C., Jeffreys, R. M., Tuerena, R., Ganeshram, R. & Mahaffey, C. Temporal and spatial trends in marine carbon isotopes in the Arctic Ocean and implications for food web studies. Glob. Chang. Biol. 25, 4116–4130 (2019).
Goni, M. A., Yunker, M. B., Macdonald, R. W. & Eglinton, T. I. Distribution and sources of organic biomarkers in arctic sediments from the Mackenzie River and Beaufort Shelf. Mar. Chem. 71, 23–51 (2000).
Parsons, T. R. et al. Autotrophic and heterotrophic production in the Mackenzie River/Beaufort Sea estuary. Polar Biol. 9, 261–266 (1989).
Dehn, L. A. et al. Feeding ecology of phocid seals and some walrus in the Alaskan and Canadian Arctic as determined by stomach contents and stable isotope analysis. Polar Biol. 30, 167–181 (2007).
Butt, C. M., Mabury, S. A., Kwan, M., Wang, X. & Muir, D. C. Spatial trends of perfluoroalkyl compounds in ringed seals (Phoca hispida) from the Canadian Arctic. Environ. Toxicol. Chem. 27, 542–553 (2008).
Dittmar, T. & Kattner, G. The biogeochemistry of the river and shelf ecosystem of the Arctic Ocean: a review. Mar. Chem. 83, 103–120 (2003).
Pons, M. L. et al. A Zn isotope perspective on the rise of continents. Geobiology 11, 201–214 (2013).
Isson, T. T. et al. Tracking the rise of eukaryotes to ecological dominance with zinc isotopes. Geobiology 16, 341–352 (2018).
Samanta, M., Ellwood, M. J. & Strzepek, R. F. Zinc isotope fractionation by Emiliania huxleyi cultured across a range of free zinc ion concentrations. Limnol. Oceanogr. 63, 660–671 (2018).
Köbberich, M. & Vance, D. Zn isotope fractionation during uptake into marine phytoplankton: implications for oceanic zinc isotopes. Chem. Geol. 523, 154–161 (2019).
Maréchal, C. N., Nicolas, E., Douchet, C. & Albarède, F. Abundance of zinc isotopes as a marine biogeochemical tracer. Geochem. Geophys. Geosyst. 1, 1015 (2000).
John, S. G. The Marine Biogeochemistry of Zinc Isotopes. [Doctoral Thesis]. (Massachusetts Institute of Technology, 2007).
Conway, T. M. & John, S. G. The biogeochemical cycling of zinc and zinc isotopes in the North Atlantic Ocean. Glob. Biogeochem. Cycles 28, 1111–1128 (2014).
Wyatt, N. J. et al. Biogeochemical cycling of dissolved zinc along the GEOTRACES South Atlantic transect GA10 at 40°S. Glob. Biogeochem. Cycles 28, 44–56 (2014).
John, S. G. & Conway, T. M. A role for scavenging in the marine biogeochemical cycling of zinc and zinc isotopes. Earth Planet. Sci. Lett. 394, 159–167 (2014).
Sieber, M. et al. Cycling of zinc and its isotopes across multiple zones of the Southern Ocean: insights from the Antarctic Circumnavigation Expedition. Geochim. Cosmochim. Acta 268, 310–324 (2020).
Samanta, M., Ellwood, M. J., Sinoir, M. & Hassler, C. S. Dissolved zinc isotope cycling in the Tasman Sea, SW Pacific Ocean. Mar. Chem. 192, 1–12 (2017).
Little, S. H., Vance, D., Walker-Brown, C. & Landing, W. M. The oceanic mass balance of copper and zinc isotopes, investigated by analysis of their inputs, and outputs to ferromanganese oxide sediments. Geochim. Cosmochim. Acta 125, 673–693 (2014).
Zhao, Y., Vance, D., Abouchami, W. & De Baar, H. J. Biogeochemical cycling of zinc and its isotopes in the Southern Ocean. Geochim. Cosmochim. Acta 125, 653–672 (2014).
Liao, W. H. et al. Zn isotope composition in the water column of the Northwestern Pacific Ocean: the importance of external sources. Glob. Biogeochem. Cycles 34, e2019GB006379 (2020).
Vance, D., de Souza, G. F., Zhao, Y., Cullen, J. T. & Lohan, M. C. The relationship between zinc, its isotopes, and the major nutrients in the North-East Pacific. Earth Planet. Sci. Lett. 525, 115748 (2019).
Jensen, L. T. et al. Biogeochemical cycling of dissolved zinc in the Western Arctic (Arctic GEOTRACES GN01). Glob. Biogeochem. Cycles 33, 343–369 (2019).
DeNiro, M. J. Postmortem preservation and alteration of in vivo bone collagen isotope ratios in relation to palaeodietary reconstruction. Nature 317, 806–809 (1985).
Ambrose, S. H. Preparation and characterization of bone and tooth collagen for isotopic analysis. J. Archaeol. Sci. 17, 431–451 (1990).
Muir, D. C. G. et al. Can seal eating explain elevated levels of PCBs and organochlorine pesticides in walrus blubber from eastern Hudson Bay (Canada)? Environ. Pollut. 90, 335–348 (1995).
Young, B. G. & Ferguson, S. H. Seasons of the ringed seal: pelagic open-water hyperphagy, benthic feeding over winter and spring fasting during molt. Wildl. Res. 40, 52–60 (2013).
Matley, J. K., Fisk, A. T. & Dick, T. A. Foraging ecology of ringed seals (Pusa hispida), beluga whales (Delphinapterus leucas) and narwhals (Monodon monoceros) in the Canadian High Arctic determined by stomach content and stable isotope analysis. Polar Res. 34, 24295 (2015).
Michel, C., Ingram, R. G. & Harris, L. R. Variability in oceanographic and ecological processes in the Canadian Arctic Archipelago. Prog. Oceanogr. 71, 379–401 (2006).
Tremblay, J. É., Gratton, Y., Carmack, E. C., Payne, C. D. & Price, N. M. Impact of the large‐scale Arctic circulation and the North Water Polynya on nutrient inventories in Baffin Bay. J. Geophys. Res. 107, 3112 (2002).
Ingram, R. G., Bâcle, J., Barber, D. G., Gratton, Y. & Melling, H. An overview of physical processes in the North Water. Deep Sea Res. Part II Top. Stud. Oceanogr. 49, 4893–4906 (2002).
Pauly, D., Trites, A. W., Capuli, E. & Christensen, V. Diet composition and trophic levels of marine mammals. ICES J. Mar. Sci. 55, 467–481 (1998).
Woollett, J. Oakes Bay 1: a preliminary reconstruction of a Labrador Inuit seal hunting economy in the context of climate change. Geogr. Tidsskr. 110, 245–259 (2010).
Stirling, I. & Archibald, W. R. Aspects of predation of seals by polar bears. J. Fish. Res. Board Can. 34, 1126–1129 (1977).
Pilfold, N. W., Derocher, A. E., Stirling, I. & Richardson, E. Polar bear predatory behaviour reveals seascape distribution of ringed seal lairs. Popul. Ecol. 56, 129–138 (2014).
Elorriaga-Verplancken, F., Aurioles-Gamboa, D., Newsome, S. D. & Martínez-Díaz, S. F. δ15N and δ13C values in dental collagen as a proxy for age-and sex-related variation in foraging strategies of California sea lions. Mar. Biol. 160, 641–652 (2013).
Hauser, D. D., Laidre, K. L., Suydam, R. S. & Richard, P. R. Population-specific home ranges and migration timing of Pacific Arctic beluga whales (Delphinapterus leucas). Polar Biol. 37, 1171–1183 (2014).
Harwood, L. A., Smith, T. G., Auld, J., Melling, H. & Yurkowski, D. J. Seasonal movements and diving of ringed seals, Pusa hispida, in the Western Canadian Arctic, 1999–2001 and 2010–11. Arctic 68, 193–209 (2015).
Ferguson, S. H., Taylor, M. K., Born, E. W., Rosing-Asvid, A. & Messier, F. Activity and movement patterns of polar bears inhabiting consolidated versus active pack ice. Arctic 54, 49–54. (2001).
Lunn, N. J. et al. Polar bear management in Canada 1997–2000. In: Proc. 13th Working Meeting of the IUCN/SSC Polar Bear Specialist Group, 23–28 June 2001, Nuuk, Greenland. Occasional Paper 26 (eds Lunn, N. J., Schliebe, S. & Born, E. W.) 41–52 (IUCN, 2002).
Ronald, K. & Dougan, J. L. The ice lover: biology of the harp seal (Phoca groenlandica). Science 215, 928–933 (1982).
Sergeant, D. E. Harp seals, man and ice. Can. Spec. Publ. Fish. Aquat. Sci. 114, (1991).
Ogloff, W. R., Yurkowski, D. J., Davoren, G. K. & Ferguson, S. H. Diet and isotopic niche overlap elucidate competition potential between seasonally sympatric phocids in the Canadian Arctic. Mar. Biol. 166, 103 (2019).
Mansfield, A. W. Seals of arctic and eastern Canada. Fish. Res. Board Canada Bull. 137 (1963).
Sergeant, D. E. Migrations of harp seals Pagophilus groenlandicus (Erxleben) in the Northwest Atlantic. J. Fish. Res. Board Can. 22, 433–464 (1965).
Richard, P. R., Heide-Jørgensen, M. P., Orr, J. R., Dietz, R. & Smith, T. G. Summer and autumn movements and habitat use by belugas in the Canadian High Arctic and adjacent areas. Arctic 54, 207–222 (2001).
Maréchal, C. N., Télouk, P. & Albarède, F. Precise analysis of copper and zinc isotopic compositions by plasma-source mass spectrometry. Chem. Geol. 156, 251–273 (1999).
Moynier, F., Albarède, F. & Herzog, G. F. Isotopic composition of zinc, copper, and iron in lunar samples. Geochim. Cosmochim. Acta 70, 6103–6117 (2006).
Toutain, J. P. et al. Evidence for Zn isotopic fractionation at Merapi volcano. Chem. Geol. 253, 74–82 (2008).
Copeland, S. R. et al. Strontium isotope ratios (87Sr/86Sr) of tooth enamel: a comparison of solution and laser ablation multicollector inductively coupled plasma mass spectrometry methods. Rapid Commun. Mass Spectrom. 22, 3187–3194 (2008).
Brown, T. A., Nelson, D. E., Vogel, J. S. & Southon, J. R. Improved collagen extraction by modified Longin method. Radiocarbon 30, 171–177 (1988).
Qi, H., Coplen, T. B., Geilmann, H., Brand, W. A. & Böhlke, J. K. Two new organic reference materials for δ13C and δ15N measurements and a new value for the δ13C of NBS 22 oil. Rapid Commun. Mass Spectrom. 17, 2483–2487 (2003).
Szpak, P., Metcalfe, J. Z. & Macdonald, R. A. Best practices for calibrating and reporting stable isotope measurements in archaeology. J. Archaeol. Sci. Rep. 13, 609–616 (2017).
R Core Team, R version 3.6.1 (R Foundation for Statistical Computing, Vienna, Austria, 2018).
Haug, T. et al. Trophic level and fatty acids in harp seals compared with common minke whales in the Barents Sea. Mar. Biol. Res. 13, 919–932 (2017).
Acknowledgements
The Max Planck Society funded the cost of Zn isotope analyses and salary. P.S. was supported by the SSHRC Insight Development grant (grant number 430 − 2014 − 00046). Salary support was provided to K.J., P.M. and N.B. by the ERC ARCHEIS project (Grant number 803676) and the DFG PALEODIET project (Grant number 378496604). We thank S. Steinbrenner and M. Trost (Department of Human Evolution, Max Planck Institute for Evolutionary Anthropology, Leipzig) for technical support.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
K.J. and J.M. designed the study. P.S., K.J. and J.M. selected the sample material. P.S., M.R. and C.H. performed the δ13C and δ15N analyses. J.M. and N.B. performed the δ66Zn analyses at the MPI EVA Leipzig. J.M., K.J. and P.M. performed the δ66Zn analyses at the CNRS in Toulouse. N.B. performed the statistical analysis. J.M., K.J., N.B., P.S., M.R. and J.-J.H., analysed and interpreted the data. J.M. wrote the initial manuscript. All authors contributed to editing the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
McCormack, J., Szpak, P., Bourgon, N. et al. Zinc isotopes from archaeological bones provide reliable trophic level information for marine mammals. Commun Biol 4, 683 (2021). https://doi.org/10.1038/s42003-021-02212-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-021-02212-z
This article is cited by
-
Zinc isotope composition of enameloid, bone and muscle of gilt-head seabreams (Sparus aurata) raised in pisciculture and their relation to diet
Marine Biology (2024)
-
Shark teeth zinc isotope values document intrapopulation foraging differences related to ontogeny and sex
Communications Biology (2023)
-
Tooth enamel nitrogen isotope composition records trophic position: a tool for reconstructing food webs
Communications Biology (2023)
-
Trophic position of Otodus megalodon and great white sharks through time revealed by zinc isotopes
Nature Communications (2022)
-
Combining collagen extraction with mineral Zn isotope analyses from a single sample for robust palaeoecological investigations
Archaeological and Anthropological Sciences (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.