Abstract
Gag is the HIV structural precursor protein which is cleaved by viral protease to produce mature infectious viruses. Gag is a polyprotein composed of MA (matrix), CA (capsid), SP1, NC (nucleocapsid), SP2 and p6 domains. SP1, together with the last eight residues of CA, have been hypothesized to form a six-helix bundle responsible for the higher-order multimerization of Gag necessary for HIV particle assembly. However, the structure of the complete six-helix bundle has been elusive. Here, we determined the structures of both Gag in vitro assemblies and Gag viral-like particles (VLPs) to 4.2 Å and 4.5 Å resolutions using cryo-electron tomography and subtomogram averaging by emClarity. A single amino acid mutation (T8I) in SP1 stabilizes the six-helix bundle, allowing to discern the entire CA-SP1 helix connecting to the NC domain. These structures provide a blueprint for future development of small molecule inhibitors that can lock SP1 in a stable helical conformation, interfere with virus maturation, and thus block HIV-1 infection.
Similar content being viewed by others
Introduction
Gag is the structural precursor protein in HIV-1. It is a polyprotein synthesized from the unspliced viral mRNA, and is cleaved by the viral protease (PR) during the maturation stage of the replicative cycle into 6 proteins and peptides: matrix (MA), capsid (CA), spacer peptide 1 (SP1), nucleocapsid (NC), spacer peptide 2 (SP2) and p61. Gag plays a major role during viral particle assembly, with each of its protein domains performing distinct functions. MA is responsible for membrane targeting. NC is responsible for RNA encapsidation. The CA and SP1 region multimerizes forming the immature viral lattice, comprised of interconnected hexamers. The Gag hexamer is, therefore, the assembly unit of the immature lattice. Proteolytic cleavage of Gag results in the transition of the immature Gag lattice to a mature capsid formed by the CA2,3,4.
The SP1 segment of Gag is 14 amino acids long and is delimited by the very first (SP1|NC) and the very last (CA|SP1) cleavage sites processed during HIV-1 maturation. SP1 plays a major role in the higher-order multimerization of Gag during viral assembly and is essential for viral infectivity5,6,7. Molecular dynamics and circular dichroism studies indicated that at high local concentrations, the stretch of residues comprising the last 8 amino acids of CA and the SP1 form a six-helix bundle5,8. It has been hypothesized that the amphipathic nature of this region would lead to the association of six CA-SP1 molecules, thus burying the hydrophobic residues in the internal face of the bundle, thereby stabilizing the Gag hexamer8. An NMR study of this region indicated that it adopts an alpha-helical conformation in 30% TFE9, and at high protein concentrations in aqueous solutions8. Magic angle spinning (MAS) NMR of CA-SP1 tubular assemblies demonstrated that the SP1 peptide is highly dynamic10. The structures of immature CA lattice and the first 8 residues of SP1 were determined previously by cryoEM and X-ray crystallography11,12. Both structures show a six-helix bundle formed by a stretch of 16 residues, 8 from the CA C-terminal domain (CACTD) and 8 from SP1. However, the rest of SP1 and its connection to NC could not be resolved in these structures.
Other factors are considered to be important for immature lattice formation and stability, in particular the small molecule inositol hexakisphosphate (IP6)13,14,15,16. Above the CA-SP1 six-helix bundle there is a 2-tiered positively-charged ring formed by 12 lysines, K158 and K227, in the CACTD (K290 and K359 in Gag amino acid numbering). The cryoEM and X-ray crystal structures indicate that all the lysine side chains point toward the center of the Gag hexamer, creating a strong positive potential11,12. IP6 was found to neutralize the positive charges at the center of the Gag hexamer, thus acting as a co-factor stabilizing the Gag hexamer. Other polyanionic molecules may also fulfil this role17, but they remain to be identified and characterized.
The SP1 region is the target of multiple maturation inhibitors (MIs), such as Bevirimat (BVM) and PF-46396 (PF-96)18,19,20,21,22,23. MIs block CA|SP1 cleavage, presumably by stabilizing the CA-SP1 region, and/or obstructing protease binding, thus impairing viral infectivity11,12,24. Natural polymorphisms as well as escape mutations in the CACTD and SP1 can render MIs ineffective25,26. Compensatory mutations selected during the passage of MI-dependent viral mutants were shown to phenocopy the effect of MIs. One such compensatory mutation is the single amino acid change, SP1 T8I, which is particularly interesting. It was selected independently during the propagation of PF-96 and BVM-dependent viruses in the absence of these MIs22,25. The T8I mutation was found to promote the assembly of the Gag lattice and stabilize the alpha-helical conformation of CA-SP119,24.
Here we report the structures of two types of immature Gag assemblies bearing the T8I mutation: one from purified Gag assemblies made in bacterial cells where the Gag polyprotein was expressed; the other from purified VLPs produced in mammalian cells. We solved the structures of full-length CA-SP1 from both assemblies at 4.2 Å and 4.5 Å resolutions using cryo-electron tomography (cryoET) and subtomogram averaging (STA)27. Our maps show a fully extended six-helix bundle that connects the CACTD to the NC-RNA density layers and a possible organization of the NC-RNA. Intriguingly, densities were identified to coordinate lysine residues in a manner similar to IP6. We further investigated the effect of T8I mutation on Gag processing, demonstrating that PR-mediated cleavage at both SP1 boundaries was impaired.
Results
CryoET and subtomogram averaging of GagT8I assemblies and VLPs
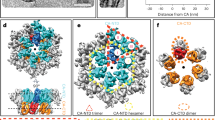
To resolve the structure of the full CA-SP1 six helix bundle, we utilized HIV-1 Gag proteins containing the T8I mutation at SP1 for stabilizing bundle assembly. SP1 T8I is a compensatory mutation that emerged in MI-dependent viruses subject to the selective pressure of replicating under sub-optimal concentrations of MIs. This mutation was shown to stabilize immature assembly22,24,25. We used two different Gag assemblies for structure analysis by cryoET: GagΔMAT8I protein assemblies expressed in E. coli (Fig. 1a and b) and GagT8I VLPs produced in HEK293T cells (Fig. 1a and d). The GagΔMAT8I construct lacks most of the globular domain of MA (residues 15 to 100) and p6 (Fig. 1a). Wild-type (WT) GagΔMA is a widely used construct for HIV-1 Gag lattice investigation11,13,28,29,30. Upon induction in E. coli, the WT GagΔMA protein was expressed, purified from bacterial lysates, and assembled in vitro into spherical particles bearing Gag immature lattices in the presence of oligonucleotides. The GagΔMAT8I protein, however, was not soluble when released from bacteria owing to its self-assembly into spherical particles inside the bacterial cells (Supplementary Fig. 1). We, therefore, purified GagΔMAT8I assemblies directly from lysed bacterial cells. The GagT8I VLPs were produced by transfection of a codon-optimized Gag expression vector into HEK293T cells and purified from the cell supernatant, similar to the WT Gag VLPs31 (Supplementary Fig. 2).
a Organization of the two constructs used for sample production: GagΔMAT8I, used for Gag assemblies made in E. coli cells; and GagT8I, used for VLP production in HEK293T cells. Both constructs bear the T8I mutation in the SP1 region (T371I in Gag sequence). b, c Purified GagΔMAT8I assemblies from E. coli cells are shown in a representative tomographic slice (b) and emClarity template matching result (c). d, e Purified GagT8I VLPs produced in HEK293T cells shown in a representative tomographic slice (d) and emClarity template matching result (e). f Subtomogram average of GagΔMAT8I assemblies in an extended lattice, shown in top and side views. The central GagΔMAT8I hexamer is highlighted in yellow and within which a monomer is highlighted in blue (CANTD) and orange (CACTD-SP1). Tomographic slices are 8 nm thick. Scale bars are 50 nm. Each point in (c) and (e) represents the position of a Gag hexamer.
A total of 90, 13 and 46 tilt series were collected for GagΔMAT8I assemblies in the absence and presence of IP6 and GagT8I VLPs, respectively. emClarity32 was used for subtomogram alignment and averaging. A low-pass filtered (25 Å) Gag hexamer density map (EMD-8403)33 was used for template-matching in emClarity, to automatically extract the position and initial orientation of the Gag hexamer in tomograms, as shown in Fig. 1c and e. Details of data acquisition and image processing are summarized in Table 1, and data processing workflow is illustrated in Supplementary Fig. 3. Upon iterative refinement, the final density maps of the Gag hexamer from GagΔMAT8I assemblies in the absence and presence of IP6 and GagT8I VLPs were obtained at 4.2 Å, 4.2 Å and 4.5 Å resolution, respectively (Supplementary Fig. 4c). The resulting maps confirm an immature Gag lattice from GagΔMAT8I assemblies and GagT8I VLPs, which is composed of interconnected hexamers formed by the CA N-terminal domain (CANTD), CACTD and an extended six-helix bundle formed by CACTD and SP1 peptide (Fig. 1f).
GagT8I assembles into a full six-helix bundle
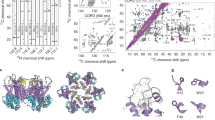
The subtomogram-averaged maps at 4.2 Å and 4.5 Å resolutions enabled refinement of atomic models of CA and SP1 (Fig. 2a, b). The refinement statistics are summarized in Table 1. Overall, the refined models of GagΔMAT8I and GagT8I VLPs are very similar, with an RMSD of 0.36 Å (Fig. 2a, b). The extended CA-SP1 helix and the NC-RNA density layer which SP1 connects are clearly resolved in both maps (Fig. 2a, b), which were not resolved in the previous CA and SP1 structures11,12. Nonetheless, interesting differences could be observed between the two. The GagT8I VLP subtomogram average shows a density that extends beyond the N-terminus of CA, where 5 residues in the junction region between MA and CA are resolved and modelled (Fig. 2c, bottom). The refined atomic model shows that the T8I mutation in SP1 stabilizes the Gag hexamer by enhancing the hydrophobicity in one face of the amphipathic CA-SP1 helix (Supplementary Fig. 5). This confirms the hypothesis that the Gag hexamer is formed by the association and burial of the hydrophobic CA-SP1 face in the internal channel of the six-helix bundle7,8.
a, b The density maps of immature Gag CA-SP1 from GagΔMAT8I assemblies (a) and GagT8I VLPs (b), overlaid with the refined respective atomic models shown in blue (CANTD) and orange (CACTD-SP1). Domain regions are labelled. c Enlarged views of CypA-binding loop (CypA-BL) and N-terminal MA-CA linker in GagΔMAT8I assemblies (top) and GagT8I VLPs (bottom). d Detailed views of the central densities within the six-helix bundle of GagΔMAT8I assemblies (left), GagT8I VLPs (middle) and GagΔMAT8I assemblies with 10 µM IP6 (right). Black arrows point to the lysine residues from CACTD, K158 and K227, forming two-rings to coordinate a central density (red arrows). Dashed lines mark the same height in both density maps.
Intriguingly, we observed a strong density above the six-helix bundle. This density is coordinated by two lysine rings formed by residues K158 and K227 (K290 and K359 in Gag amino acid numbering) near the bottom of the CACTD and above the bundle (Fig. 2d). This density is present when no symmetry is imposed throughout the process, when different B-factors are used for sharpening, and is within a confidence map set at 1% false discovery rate threshold (Supplementary Fig. 6, Supplementary Movie 1)34. A close inspection revealed that the profile and position of the internal density are different in the GagΔMAT8I and GagT8I VLPs maps (Fig. 2d, red arrows). For GagT8I VLPs produced in mammalian cells, this density is consistent with IP6, as suggested in previous studies13,14 (Fig. 2d, middle). IP6 is known to associate with the HIV-1 immature lattice and is present at cytoplasmic concentrations estimated at 20–50 nM in mammalian cells35. E. coli cells, however, lack the enzyme responsible for biosynthesis of the inositol ring and are thus thought to lack inositides36. Considering that GagΔMAT8I assemblies were produced in E. coli cells, it seems unlikely that the density in GagΔMAT8I map is IP6 (Fig. 2d, left). When the same GagΔMAT8I assembly is incubated with a buffer containing 10 μM IP6, this internal density is present at lower position resembling that seen in GagT8I VLPs (Fig. 2d. right). It is plausible that a small anionic molecule other than IP6 assists in the charge neutralization of the lysine rings and promoted assembly in E. coli-expressed GagΔMAT8I. Consistent with this, a similar density was also present at the same location in a previously published subtomogram average of bacterially expressed WT GagΔMA assemblies (Fig. 3c, blue)11. Further studies are needed to identify such small molecules. It is also possible that the density in E. coli-expressed GagΔMAT8I averaged map is a product of averaging hexamers with different molecules occupying the lysine ring.
a, b Comparison of the CA-SP1 hexamer in GagΔMAT8I (a) and GagΔMAWT (EMD-3782) (b), shown in a side-view. The density map is overlaid with the respective atomic model. The dashed arrows points to the position of the eighth aminoacid in SP1 in both maps. c An enlarged view of density overlay from the dashed box region in (a, b). The GagΔMAT8I density is in grey and the GagΔMAWT density is in blue. The red arrow points to the same central density shown in Fig. 2d. The green arrow points to BVM density in EMD-3782. d Overlay of the refined atomic models from GagΔMAT8I and GagΔMAWT (PDB 5L93). Arrow points to location of T8I mutation and CA|SP1 and SP1|NC cleavage sites. e, f In vitro protease cleavage assays. GagΔMA WT and GagΔMAT8I assemblies were incubated with recombinant HIV-1 PR at the indicated concentration (e) and for the indicated period of time (f), separated in an SDS-PAGE and stained with Coomassie Blue. For processing intermediates, bands are labelled by listing the first and last domains.
The T8I mutation reduces SP1 dynamics and impairs proteolytic cleavage
In the previous WT GagΔMA map (EMD-3782) and its corresponding atomic model (PDB 5L93), the SP1 region after T8 was not resolved11. The SP1 helix in the GagΔMAT8I map is complete and extends to the SP1 and NC interface (Fig. 3a, c), therefore allowing model building of the C-terminal portion of SP1, from T8I to M14 (Fig. 3d). The T8I mutation was shown to quench the dynamics of SP1 by MAS NMR24, and potentially stabilize a continuous helical conformation. It is worth noting that the WT GagΔMA map (EMD-3782) was determined in the presence of BVM. The density corresponding to BVM is visible in the WT GagΔMA map (Fig. 3c, green arrow), but not in GagΔMA T8I map (Fig. 2d, left panel). Yet, the observed unknown densities coordinated by K158 and K227 overlapped well in both WT GagΔMA and GagΔMAT8I maps (Fig. 3c, red arrow), even though the two proteins were expressed and assembled independently in different manners in the absence of IP6.
The SP1 peptide is flanked by the first (SP1|NC) and the last (CA|SP1) cleavage sites in the HIV-1 maturation cascade. If the T8I mutation stabilizes the extended six-helix bundle, this may affect PR processing of these cleavage sites. We thus tested the effect of the T8I mutation on PR-mediated cleavage of Gag assemblies using in vitro cleavage assays (Fig. 3e, f)37. We first analysed the concentration-dependent processing of WT GagΔMA and GagΔMAT8I by PR. WT GagΔMA processing showed the established ordering of cleavage as the PR concentration increased, with the first major cleavage products MA-CA-SP1 and NC-SP2 (cleaving at SP1|NC), followed by CA-SP1 (cleaving at MA|CA), and completed with CA and NC (Fig. 3e). In contrast, not only was a much higher PR concentration required to initiate processing of GagΔMAT8I, but also the order of cleavage was altered (Fig. 3e). The first cleavage occurred at MA|CA site, suggesting an impaired or delayed cleavage at the SP1|NC site. Furthermore, the CA|SP1 site was also protected from cleavage, such that very little CA was produced. The same trend was observed when we investigated the kinetics of PR processing in vitro (Fig. 3f). At the end point of PR processing (120 min), CA-SP1 was the major final product for GagΔMAT8I, instead of CA. Interestingly, the NC band appears before the NC-SP2 intermediate band in GagΔMAT8I, possibly resulting from the processing of the abnormal CA-SP1-NC-SP2 intermediate. Altogether, these results indicate that cleavage at both proteolytic sites flanking SP1 are impaired by the T8I mutation, which supports the formation of a stable and extended SP1 helix.
The NC-RNA layer is defined but variable in the SP1-NC domain organization
We next sought to investigate how the SP1 six-helix bundle connects to the NC-RNA region, which has been intractable previously. As shown in Fig. 4a, three layers of densities were observed in tomographic slices of the GagΔMAT8I particles. Radial density profiles from three different particles of similar size displayed three distinct peaks corresponding to the CANTD, CACTD and NC-RNA density layers (Fig. 4a)38. While CANTD, CACTD and SP1 are rigidly connected as seen in the GagΔMAT8I map, NC-RNA is flexibly linked to SP1, resulting in a smeared density in GagΔMAT8I subtomogram averages (Fig. 3a).
a Radial density profile of the Gag lattice. Top panel shows a tomographic slice of GagΔMAT8I lattice domain organization where CANTD, CACTD and NC-RNA density layers are coloured in blue, orange and pink, respectively. Graph shows the radial density profiles of GagΔMAT8I particles of similar size (n = 3). Black line indicates the average intensity while blue lines depict the individual particle profiles. Density profile peaks corresponding to CANTD, CACTD and NC-RNA are shaded with the same colour scheme as in the top panel. (b) Top view from density map of six-helix bundle and the NC-RNA layer, overlaid with two plausible NC-RNA (ѱ SL2 loop) models, in red and pink, fitted into the density, leaving the remaining NC-RNA density unmodeled. c, d Enlarged side view from density map in (b).
To gain structural information on the SP1-NC-RNA region, we carried out localized 3D alignment and classification, focusing on the SP1-NC-RNA region, using PCA and k-means subtomogram classification tools implemented in emClarity without imposing any symmetry (Supplementary Fig. 7a). Among 32 resulting 3D classes, we identified multiple classes that seemed to share common organization of the NC-RNA layer (Supplementary Fig. 7b, red boxes) and averaged 6747 particles from these classes to generate a density map for the SP1-NC-RNA region (Fig. 4b–c). A composite molecule of CACTD-SP1-NC was created based on the refined atomic model CA-SP1 and the published NC-RNA model (PDB 1F6U)39, which was then used separately for rigid body fitting of the NC and the RNA domains in the density map. We performed this for each CA monomer in the model, and illustrate two plausible models of SP1-NC-RNA organization (Fig. 4b, c). To bridge the space between the six-helix bundle density and the NC-RNA density layer, it is necessary to model the N-terminus of NC as a coil, up to residue Asn8. In both models, the NC-RNA complex is at the density layer below and surrounding the six-helix bundle (Fig. 4c). As HIV-1 NC is very flexible and the viral RNA can adopt many conformations, it is likely that there exist multiple modes of SP1 to NC connection, as exemplified by some other classes and other fitting possibilities. Taken together, a stretched coil conformation at the SP1 and NC junction and/or dynamic unfolding of this region could facilitate PR processing. As such, the helix stabilization imparted by the T8I mutation may explain the observed delays in SP1|NC cleavage.
Discussion
HIV-1 Gag is a multidomain and multifunctional protein responsible for membrane targeting, RNA encapsidation, particle assembly and budding. The CA and SP1 domains of Gag form the viral immature hexagonal lattice. Residues that make up the CA-SP1 helix are remarkably conserved and mutations in this region lead to loss of infectivity and/or assembly defects8,19,40,41.
The SP1-T8I mutation is a stabilizing compensatory mutation selected for during the passage of MI-dependent virus. Our refined atomic model shows that this mutation increases the hydrophobicity at the inner face of the six-helix bundle, thus stabilizing it (Supplementary Fig. 5). It has been shown that IP6 is also involved in Gag hexamer stabilization by neutralizing positive charges of lysine residues at the base of the CACTD13,14,15,16. Here, we show that E. coli Gag assemblies present density in a similar location as the one previously described for IP6 binding, even though E. coli lacks inosites. This suggests molecules other than IP6 may neutralize charged lysine rings to stabilize the immature lattice. More studies are necessary for the identification of this molecule (or molecules).
HIV-1 maturation is a highly regulated process. The sequence of cleavage events is thought to be determined by the affinity constants and processing kinetics between the different cleavage sites in Gag and HIV-1 PR42,43,44. Events that alter the kinetics or sequence of maturation invariably lead to loss of infectivity19,24,45,46. The T8I mutation was acquired as a second-site mutation that restores assembly and maturation to the BVM- and PF-96-dependent mutants, but the T8I single mutant exhibited a maturation defect with inefficient cleavage at CA-SP1. The inference is that the original resistant mutants exhibited unstable Gag lattices in the absence of the compound, which stabilizes the lattice, and T8I mutation phenocopies the compound by stabilizing the lattice, which then becomes hyper-stable in the absence of a destabilizing mutation. We show that the T8I mutation alters the cleavage sequence in HIV-1 maturation and impairs the processing of both SP1 boundaries, suggesting that a finely tuned SP1 stability is required to balance between lattice assembly, proper maturation and infectivity.
SP1 is critically important as one of the major targets for therapeutic drugs against HIV-1 and AIDS. The structure of full-length CA-SP1 in immature Gag assemblies derived from this study provides a blueprint for the future development of small molecule inhibitors that can lock SP1 in a stable helical conformation and thus interfere with PR-mediated processing and virus maturation, and block HIV-1 infection.
Methods
Particle production and purification
The GagΔMAT8I mutant was constructed from pET2 PRR400 by site-direct mutagenesis. Protein was expressed in E. coli, Rosetta 2 (DE3), cultured in Luria-Bertani medium, and induced with 0.5 mM isopropylthiogalactoside at 23 °C for 16 h. GagΔMAT8I assemblies were formed inside of bacterial cells and purified directly from cell lysate. The cell pellets were collected and resuspended in lysis buffer (25 mM Tris, pH 7.5, 0.5 M NaCl) and broken with a microfluidizer. Subsequently, the lysate was centrifuged at 5000 × g for 10 min to remove cell debris. The supernatant was collected and subjected to sucrose gradient centrifugation (30–70%) with a 15% sucrose cushion on the top. Gradient was spun at 210,000 × g for 18 h. Particles were collected from the pellet and resuspended in lysis buffer with and without 10 μM IP6 and used for CryoEM.
The GagT8I mutant VLPs were produced from HEK293T cells (ATCC CRL-3216) transfected with a codon-optimized full-length Gag expression plasmid (pCMV-Gag-opt)47. The culture supernatants were harvested approximately 40 h after transfection, filtered through a 0.45-micron pore-sized filter, and pelleted through a cushion of 20% sucrose (wt/vol) in STE buffer (20 mM Tris-HCl pH 7.4, 100 mM NaCl, 1 mM EDTA). The VLPs were gently resuspended in STE buffer and frozen at −80 °C.
CryoEM sample preparation
A homemade manual plunger was used for cryoEM grid preparation. Quantifoil R2/2 copper grids were freshly glow discharged at 30 mA for 30 s. For GagΔMAT8I assemblies, 2 µl sample was applied to the carbon-side of the grid, followed by 1 μl 10 nm fiducial gold bead solution, and blotted from the backside since samples are very fragile and sensitive to blotting paper. 1 μl sample buffer was then added on the grid backside to facilitate the filter paper blotting from the backside. For GagT8I VLPs, 1 μl 10 nm fiducial gold beads were applied to the grid first, followed by 1~2 μl sample, and blotted from the frontside. The grid after blotting was then quickly immersed into liquid ethane cooled by liquid nitrogen. Frozen grids were stored in liquid nitrogen until data collection.
CryoET data acquisition
Data for GagΔMAT8I assemblies and GagT8I VLPs were collected at the Electron Bio-Imaging Centre at Diamond Light Source (eBIC-DLS) in the United Kingdom. Tilt-series were acquired on a Gatan K2-Summit detector in super-resolution mode using a Thermo Fisher Scientific KriosG3i microscope operating at 300 kV equipped with a Gatan energy filter (slit width 20 eV; GIF Quantum LS, Gatan). Tilt-series were recorded at a nominal magnification of 105,000×, corresponding to a calibrated pixel size of 1.35 Å. A dose-symmetric scheme48 was used to collect tilt-series from −60° to 60° or −54° to 54° at a step size of 3° using SerialEM software49. At each tilt, a movie stack consisting of 5–10 frames was recorded with a set dose rate of 3–6 e−/Å2/s. Tilt-series were collected at a range of nominal defocus between −1.0 and −4.0 µm and a target total dose of 110–120 e−/Å2 was applied over the entire series. For GagΔMAT8I with 10 μM IP6 sample, data were collected at the Thermo Fisher Scientific RnD division in Eindhoven, The Netherlands. Tilt-series were acquired on a Falcon4 detector in EER (electron event representation)50,51 format using a Thermo Fisher Scientific KriosG4 operating at 300 kV equipped with a CFEG and SelectrisX energy filter (slit width 10 eV; Thermo Fisher Scientific). Tilt-series were recorded at a nominal magnification of ×105,000, corresponding to a calibrated pixel size of 1.196 Å. A dose-symmetric scheme was used to collect tilt-series from −60° to 60° at a step size of 3° using TEM Tomography software. The CFEG was automatically flashed every 8 h. At each tilt, a movie stack consisting of 217 EER frames was recorded with a dose rate of 4.6 e−/px/sec and a total dose of 2.98 e−/A2 per tilt. Tilt-series were collected at a nominal defocus range between −1.0 and −4.0 µm and a target total dose of 122 e−/Å2 was applied over the entire tilt-series. Further details are given in Table 1.
CryoET image processing
CryoET Toolbox (https://github.com/ffyr2w/cet_toolbox) was used for on-the-fly pre-processing of K2 datasets (freely available). In summary, movie frames were Fourier-cropped to a final pixel size of 1.35 Å and motion corrected by averaging frames for each tilt using program ‘alignframes’ implemented in the IMOD package52 or MotionCor253. For the Falcon4 data, 30 EER frames were grouped to create 7 dose fractions from 210 frames (last 7 frames were discarded) and motion corrected using Relion3.154. Tilt series were aligned using the default parameters in IMOD version 4.10.22 with the eTomo interface52, using gold-fiducial markers. The alignment parameters including the projection transformations, local alignments, and fitted tilt angles were then passed to emClarity32 framework. Sub-tomogram alignment and averaging were carried out in emClarity following the published protocol32. Briefly, the workflow was as follows: Particles were picked from 6× binned non-CTF-corrected tomograms by emClarity template matching function using EMD-840333 low-pass filtered to 25 Å as template. The template matching results were cleaned automatically on basis of geometrical restraints using ‘removeNeighbours’ function implemented in emClarity. Only particles that had at least three neighbours within 100 Å and oriented within 20° were retained. Subtomograms at the air-water interface were manually discarded using IMOD52. Following template matching, the data set was randomly split into two groups based on tomogram of origin, rather than randomly by sub-tomograms, which were processed independently for all subsequent steps. The initial positions and orientations of the first cycle averaging come from template matching results. C6 symmetry was applied throughout all sub-tomogram averaging procedures. The 3D alignment procedures were carried out gradually with binning of 5, 4, 3, 2 and 1. At each binning, duplicate particles were removed and the tilt-series geometry was refined using the positions of subtomograms as fiducial markers (TomoCPR). The Fourier Shell Correlation was calculated by the gold-standard method from even and odd data sets. Density maps were visualized in Chimera55 or PyMol (Schrödinger, Inc.). A diagram for the data processing is presented in Supplementary Fig. 3.
Model building and refinement
An atomic model was built and refined against the GagΔMAT8I density map. The crystal structure (PDB 4XFX)56 and cryoEM structure (PDB 5L93)11 of HIV-1 CA were used as an initial reference. At 4.2 Å resolution, positive and bulky side chains are clearly visible enabling reasonable positioning of the residues in the atomic models. Rotamers were generally not refined, unless there was good evidence for a different rotamer in our density map. For the SP1 region, a composite model was made by joining the SP1 residues from PDB 1U579 to the 5L93 model (lacking the last 6 SP1 residues) to serve as initial reference for the model refinement.
The model refinement procedure involved iterative manual refinement in Coot57 followed by three rounds of Phenix cryoEM real-space refinement58. After each round of real-space refinement, Ramachandran and rotamer outliers were manually refined in Coot, and another round of real-space refinement was performed until good model statistics were achieved. In order to ensure reasonable modelling in the SP1 region, the density map of GagΔMAT8I was sharpened with a B factor of 120 in combination with a 1% FDR confidence map34 and a locally sharpened B-factor map59 for the manual refinement in Coot.
Subtomogram classification and modelling of the NC-RNA layer
The classification was performed after 8 cycles of interative alignment in emClarity, at a pixel size of 4.1 Å (3× binned). Multi-scale PCA and k-means classification implemented in emClarity were used to identify common organization features in NC-RNA layer in the subtomograms. A 49 × 49 × 47 Å region that comprised the CA-CTD, the six-helix bundle and the NC-RNA layer was used for focused alignment and classification at the SP1-NC junction (Supplementary Fig. 7a). No symmetry was imposed. The multi-scale PCA was done using 3 resolution bands: 15, 20 and 40 Å. 25 eigenvolumes were generated for each resolution band. Of these, we selected the volumes which showed the most variance at the NC-RNA layer. Then we performed k-means clustering based on these eigenvolumes into 32 3D classes. Among those, 3 major classes showing similar NC-RNA organization were combined, comprising 6747 subvolumes (Supplementary Fig. 7b). The final resolution was calculated at 15.1 Å at 0.143 FSC.
A composite model was built in Coot57 using the refined GagΔMAT8I model and the published NC-ΨRNASL2 model (PDB 1F6U)39. UCSF Chimera55 was used for rigid body fitting of the NC and RNA region of the composite model into the NC-RNA density map. This was repeated for each CA monomer. Bond lengths were regularized in Coot by using the Regularize Zone from residue M14 in SP1 to residue Q9 in NC.
In vitro cleavage of Gag assemblies
Recombinant WT assemblies were produced following the protocol described previously33. Briefly, HIV-1 WT Gag was expressed in E. coli and purified by ion-exchange chromatography. In vitro assembly was done by mixing 4 mg/ml of protein with yeast transfer RNA overnight. PR digestion experiments with recombinant WT and T8I assemblies were performed by the addition of various concentrations of PR to the Gag WT assembly mixture and T8I cell lysate which was pre-treated and diluted to 2 mg/ml, and incubated at 37 °C for 2 h. For kinetic analysis of Gag WT and T8I cleavage, 3.3 μM of HIV-1 PR was added to the Gag assembly mixture and incubated at 37 °C, at different time points, 4ul of the digestion reaction mixture was taken out and mixed with NuPAGE® LDS Sample Buffer (Invitrogen) to stop the reaction, and then subjected to NuPAGE Novex 4–12% Bis-Tris gel (Invitrogen) for cleavage products analysis and visualized by Coomassie blue staining.
Statistics and reproducibility
In vitro cleavage experiments have been successfully reproduced, n = 3, representative gel is shown.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
All data are available from the corresponding author, Dr. Peijun Zhang (peijun.zhang@strubi.ox.ac.uk), upon request. The cryoEM density maps CA-SP1 hexamer from GagΔMAT8I assemblies in the absence and presence of IP6 and GagT8I VLPs were deposited in the EMDB under accession code EMD-11894, EMD-11899 and EMD-11897. The refined models were deposited in PDB under accession codes 7ASH and 7ASL. NC-RNA cryoEM map was deposited in EMDB under accession code EMD-12287.
References
Aiken, C. & Zhang, P. in Advances in HIV-1 Assembly and Release (ed. Freed, E. O.) 153–166 (Springer New York, 2013).
Byeon, I. J. et al. Structural convergence between Cryo-EM and NMR reveals intersubunit interactions critical for HIV-1 capsid function. Cell 139, 780–790 (2009).
Ni, T. et al. Intrinsic curvature of the HIV-1 CA hexamer underlies capsid topology and interaction with cyclophilin A. Nat. Struct. Mol. Biol. 27, 855–862 (2020).
Zhao, G. et al. Mature HIV-1 capsid structure by cryo-electron microscopy and all-atom molecular dynamics. Nature 497, 643–646 (2013).
Accola, M. A., Hoglund, S. & Gottlinger, H. G. A putative alpha-helical structure which overlaps the capsid-p2 boundary in the human immunodeficiency virus type 1 Gag precursor is crucial for viral particle assembly. J. Virol. 72, 2072–2078 (1998).
Krausslich, H. G., Facke, M., Heuser, A. M., Konvalinka, J. & Zentgraf, H. The spacer peptide between human immunodeficiency virus capsid and nucleocapsid proteins is essential for ordered assembly and viral infectivity. J. Virol. 69, 3407–3419 (1995).
Liang, C. et al. Characterization of a putative alpha-helix across the capsid-SP1 boundary that is critical for the multimerization of human immunodeficiency virus type 1 gag. J. Virol. 76, 11729–11737 (2002).
Datta, S. A. et al. On the role of the SP1 domain in HIV-1 particle assembly: a molecular switch? J. Virol. 85, 4111–4121 (2011).
Morellet, N., Druillennec, S., Lenoir, C., Bouaziz, S. & Roques, B. P. Helical structure determined by NMR of the HIV-1 (345-392)Gag sequence, surrounding p2: implications for particle assembly and RNA packaging. Protein Sci. 14, 375–386 (2005).
Han, Y. et al. Magic angle spinning NMR reveals sequence-dependent structural plasticity, dynamics, and the spacer peptide 1 conformation in HIV-1 capsid protein assemblies. J. Am. Chem. Soc. 135, 17793–17803 (2013).
Schur, F. K. et al. An atomic model of HIV-1 capsid-SP1 reveals structures regulating assembly and maturation. Science 353, 506–508 (2016).
Wagner, J. M. et al. Crystal structure of an HIV assembly and maturation switch. Elife 5, e17063 (2016).
Dick, R. A. et al. Structures of immature EIAV Gag lattices reveal a conserved role for IP6 in lentivirus assembly. PLoS Pathog. 16, e1008277 (2020).
Dick, R. A. et al. Inositol phosphates are assembly co-factors for HIV-1. Nature 560, 509–512 (2018).
Mallery, D. L. et al. Cellular IP6 levels Limit HIV production while viruses that cannot efficiently package IP6 are attenuated for infection and replication. Cell Rep. 29, 3983–3996.e3984 (2019).
Mallery, D. L. et al. IP6 is an HIV pocket factor that prevents capsid collapse and promotes DNA synthesis. Elife 7, e35335 (2018).
Campbell, S. et al. Modulation of HIV-like particle assembly in vitro by inositol phosphates. Proc. Natl Acad. Sci. USA 98, 10875–10879 (2001).
Blair, W. S. et al. New small-molecule inhibitor class targeting human immunodeficiency virus type 1 virion maturation. Antimicrob. Agents Chemother. 53, 5080–5087 (2009).
Fontana, J. et al. Identification of an HIV-1 mutation in spacer peptide 1 that stabilizes the immature CA-SP1 lattice. J. Virol. 90, 972–978 (2016).
Li, F. et al. PA-457: a potent HIV inhibitor that disrupts core condensation by targeting a late step in Gag processing. Proc. Natl Acad. Sci. USA 100, 13555–13560 (2003).
Nguyen, A. T. et al. The prototype HIV-1 maturation inhibitor, bevirimat, binds to the CA-SP1 cleavage site in immature Gag particles. Retrovirology 8, 101 (2011).
Waki, K. et al. Structural and functional insights into the HIV-1 maturation inhibitor binding pocket. PLoS Pathog. 8, e1002997 (2012).
Zhou, J. et al. Small-molecule inhibition of human immunodeficiency virus type 1 replication by specific targeting of the final step of virion maturation. J. Virol. 78, 922–929 (2004).
Wang, M. et al. Quenching protein dynamics interferes with HIV capsid maturation. Nat. Commun. 8, 1779 (2017).
Adamson, C. S. et al. In vitro resistance to the human immunodeficiency virus type 1 maturation inhibitor PA-457 (Bevirimat). J. Virol. 80, 10957–10971 (2006).
Adamson, C. S., Sakalian, M., Salzwedel, K. & Freed, E. O. Polymorphisms in Gag spacer peptide 1 confer varying levels of resistance to the HIV- 1 maturation inhibitor bevirimat. Retrovirology 7, 36 (2010).
Zhang, P. Advances in cryo-electron tomography and subtomogram averaging and classification. Curr. Opin. Struct. Biol. 58, 249–258 (2019).
Bartonova, V. et al. Residues in the HIV-1 capsid assembly inhibitor binding site are essential for maintaining the assembly-competent quaternary structure of the capsid protein. J. Biol. Chem. 283, 32024–32033 (2008).
Gross, I., Hohenberg, H., Huckhagel, C. & Krausslich, H. G. N-Terminal extension of human immunodeficiency virus capsid protein converts the in vitro assembly phenotype from tubular to spherical particles. J. Virol. 72, 4798–4810 (1998).
Gross, I. et al. A conformational switch controlling HIV-1 morphogenesis. EMBO J. 19, 103–113 (2000).
Martin, J. L., Cao, S., Maldonado, J. O., Zhang, W. & Mansky, L. M. Distinct particle morphologies revealed through comparative parallel analyses of retrovirus-like particles. J. Virol. 90, 8074–8084 (2016).
Himes, B. A. & Zhang, P. emClarity: software for high-resolution cryo-electron tomography and subtomogram averaging. Nat. Methods 15, 955–961 (2018).
Ning, J. et al. In vitro protease cleavage and computer simulations reveal the HIV-1 capsid maturation pathway. Nat. Commun. 7, 13689 (2016).
Beckers, M., Jakobi, A. J. & Sachse, C. Thresholding of cryo-EM density maps by false discovery rate control. IUCrJ 6, 18–33 (2019).
Barker, C. J., Wright, J., Hughes, P. J., Kirk, C. J. & Michell, R. H. Complex changes in cellular inositol phosphate complement accompany transit through the cell cycle. Biochem J. 380, 465–473 (2004).
Das-Chatterjee, A. et al. Introgression of a novel salt-tolerant L-myo-inositol 1-phosphate synthase from Porteresia coarctata (Roxb.) Tateoka (PcINO1) confers salt tolerance to evolutionary diverse organisms. FEBS Lett. 580, 3980–3988 (2006).
Meng, X. et al. Protease cleavage leads to formation of mature trimer interface in HIV-1 capsid. PLoS Pathog. 8, e1002886 (2012).
Zhou, L. et al. Low-dose phase retrieval of biological specimens using cryo-electron ptychography. Nat. Commun. 11, 2773 (2020).
Amarasinghe, G. K. et al. NMR structure of the HIV-1 nucleocapsid protein bound to stem-loop SL2 of the psi-RNA packaging signal. Implications for genome recognition. J. Mol. Biol. 301, 491–511 (2000).
Melamed, D. et al. The conserved carboxy terminus of the capsid domain of human immunodeficiency virus type 1 gag protein is important for virion assembly and release. J. Virol. 78, 9675–9688 (2004).
Pettit, S. C. et al. The p2 domain of human immunodeficiency virus type 1 Gag regulates sequential proteolytic processing and is required to produce fully infectious virions. J. Virol. 68, 8017–8027 (1994).
Louis, J. M., Weber, I. T., Tozser, J., Clore, G. M. & Gronenborn, A. M. HIV-1 protease: maturation, enzyme specificity, and drug resistance. Adv. Pharm. 49, 111–146 (2000).
Pettit, S. C., Clemente, J. C., Jeung, J. A., Dunn, B. M. & Kaplan, A. H. Ordered processing of the human immunodeficiency virus type 1 GagPol precursor is influenced by the context of the embedded viral protease. J. Virol. 79, 10601–10607 (2005).
Pettit, S. C., Lindquist, J. N., Kaplan, A. H. & Swanstrom, R. Processing sites in the human immunodeficiency virus type 1 (HIV-1) Gag-Pro-Pol precursor are cleaved by the viral protease at different rates. Retrovirology 2, 66 (2005).
de Marco, A. et al. Role of the SP2 domain and its proteolytic cleavage in HIV-1 structural maturation and infectivity. J. Virol. 86, 13708–13716 (2012).
Ludwig, C., Leiherer, A. & Wagner, R. Importance of protease cleavage sites within and flanking human immunodeficiency virus type 1 transframe protein p6* for spatiotemporal regulation of protease activation. J. Virol. 82, 4573–4584 (2008).
Huang, Y., Kong, W. P. & Nabel, G. J. Human immunodeficiency virus type 1-specific immunity after genetic immunization is enhanced by modification of Gag and Pol expression. J. Virol. 75, 4947–4951 (2001).
Hagen, W. J. H., Wan, W. & Briggs, J. A. G. Implementation of a cryo-electron tomography tilt-scheme optimized for high resolution subtomogram averaging. J. Struct. Biol. 197, 191–198 (2017).
Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005).
Guo, H. et al. Electron-event representation data enable efficient cryoEM file storage with full preservation of spatial and temporal resolution. IUCrJ 7, 860–869 (2020).
Nakane, T. et al. Single-particle cryo-EM at atomic resolution. Nature 587, 152–156 (2020).
Kremer, J. R., Mastronarde, D. N. & McIntosh, J. R. Computer visualization of three-dimensional image data using IMOD. J. Struct. Biol. 116, 71–76 (1996).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Scheres, S. H. W. Amyloid structure determination in RELION-3.1. Acta Crystallogr D. Struct. Biol. 76, 94–101 (2020).
Pettersen, E. F. et al. UCSF Chimera–a visualization system for exploratory research and analysis. J. Comput Chem. 25, 1605–1612 (2004).
Gres, A. T. et al. STRUCTURAL VIROLOGY. X-ray crystal structures of native HIV-1 capsid protein reveal conformational variability. Science 349, 99–103 (2015).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr D. Biol. Crystallogr 60, 2126–2132 (2004).
Afonine, P. V. et al. Real-space refinement in PHENIX for cryo-EM and crystallography. Acta Crystallogr D. Struct. Biol. 74, 531–544 (2018).
Jakobi, A. J., Wilmanns, M. & Sachse, C. Model-based local density sharpening of cryo-EM maps. Elife 6, e27131 (2017).
Acknowledgements
We thank Dr. Teresa Brosenitsch for critical reading of the manuscript. We are grateful to Drs Alistair Siebert, Kyle Dent, Andy Howe and Lingbo Yu for technical support. We thank Reint Boer Iwema for support with TEM Tomography software. We acknowledge Diamond for access and support of the CryoEM facilities at the UK national electron bio-imaging centre (eBIC, proposal NR18477, NR21005 and NT21004), funded by the Wellcome Trust, MRC and BBSRC. We also thank Thermo Fisher Scientific for microscope access. The computational aspects of this research were supported by the Wellcome Trust Core Award Grant Number 203141/Z/16/Z and the NIHR Oxford BRC. This work was supported by the National Institutes of Health P50 grant AI150481 (P.Z.), the UK Wellcome Trust Investigator Award 206422/Z/17/Z (P.Z.), and the UK Biotechnology and Biological Sciences Research Council grant BB/S003339/1 (P.Z.).
Author information
Authors and Affiliations
Contributions
P.Z. conceived the research and designed the experiments. J.N. purified GagΔMAT8I assemblies and performed in vitro protease cleavage experiments. J.Z. and C.A. purified GagT8I VLPs. X.F prepared cryoEM samples. J.L. and A.K. collected cryoET data. T.F. developed CryoET Toolbox used for pre-processing. L.M., D.S. and B.H. performed subtomogram averaging using emClarity. L.M. and M.O. refined the structural models. A.B.K. performed functional assays. L.M., E.O.F., D.S. and P.Z. analysed data and wrote the paper with support from all the authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare the following competing interests: A.K. is an employee of Thermo Fisher Scientific. The other authors have no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mendonça, L., Sun, D., Ning, J. et al. CryoET structures of immature HIV Gag reveal six-helix bundle. Commun Biol 4, 481 (2021). https://doi.org/10.1038/s42003-021-01999-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-021-01999-1
This article is cited by
-
Comparative analysis of retroviral Gag-host cell interactions: focus on the nuclear interactome
Retrovirology (2024)
-
The HIV-1 gag p6: a promising target for therapeutic intervention
Retrovirology (2024)
-
Learning structural heterogeneity from cryo-electron sub-tomograms with tomoDRGN
Nature Methods (2024)
-
nextPYP: a comprehensive and scalable platform for characterizing protein variability in situ using single-particle cryo-electron tomography
Nature Methods (2023)
-
Molecular architecture and conservation of an immature human endogenous retrovirus
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.