Abstract
APOBEC3A (A3A) and APOBEC3B (A3B) enzymes drive APOBEC-mediated mutagenesis. Identification of factors affecting the activity of these enzymes could help modulate mutagenesis and associated clinical outcomes. Here, we show that canonical and alternatively spliced A3A and A3B isoforms produce corresponding mutagenic and non-mutagenic enzymes. Increased expression of the mutagenic A3B isoform predicted shorter progression-free survival in bladder cancer. We demonstrate that the production of mutagenic vs. non-mutagenic A3B protein isoforms was considerably affected by inclusion/skipping of exon 5 in A3B. Furthermore, exon 5 skipping, resulting in lower levels of mutagenic A3B enzyme, could be increased in vitro. Specifically, we showed the effects of treatment with an SF3B1 inhibitor affecting spliceosome interaction with a branch point site in intron 4, or with splice-switching oligonucleotides targeting exon 5 of A3B. Our results underscore the clinical role of A3B and implicate alternative splicing of A3B as a mechanism that could be targeted to restrict APOBEC-mediated mutagenesis.
Similar content being viewed by others
Introduction
Enrichment of C-to-T or C-to-G mutations within the TCA and TCT motifs, attributed to APOBEC-mediated mutagenesis, has been implicated in cancer susceptibility1, tumor evolution2,3, metastatic progression4,5, treatment response3,6, and survival1. Thus, understanding and harnessing the mechanisms regulating this mutational process is of clinical importance. Among the seven members of the APOBEC3 enzyme family, A3A7,8, A3B9, and an allelic variant of APOBEC3H (A3H)10 have been linked with APOBEC mutagenesis in tumors.
The intrinsic and extrinsic factors that regulate the expression levels of APOBEC3s might explain some of the differences in a load of APOBEC-signature mutations within and between tumors of different types. These intrinsic factors include common germline variants—a single-nucleotide polymorphism (SNP) rs1014971 and the polymorphic A3AB deletion1,11, or their correlated proxies, SNPs rs17000526 and rs126284031. The same genetic variants have also been associated with A3B expression. Specifically, rs1014971 regulates A3B expression through an allele-specific effect on an enhancer upstream of the APOBEC3 gene cluster1 and the A3AB deletion—through the elimination of one or both copies of the A3B gene11,12. A haplotype represented by a missense SNP rs139297 (Gly105Arg) that creates an A3H protein isoform with nuclear localization (A3H-I) has been associated with APOBEC-signature mutations in A3A and A3B-null breast and lung tumors10. Extrinsic factors that induce expression of specific APOBEC3s include viral infections1,13 and exposure to environmental or chemotherapeutic DNA-damaging agents1,2,14. Most conclusions about the role of A3A and A3B in mutagenesis were drawn based on RNA-seq studies despite the poor ability to resolve and confidently quantify mRNA expression of these highly homologous APOBECs by short sequencing reads10.
Alternative splicing (AS) of pre-mRNA can produce functionally distinct isoforms or regulate gene expression through downstream mechanisms, such as nonsense-mediated decay (NMD)15. Alternatively spliced isoforms of other APOBEC3 genes (APOBEC3H and APOBEC3F) have been reported to generate enzymes with variable activity16,17,18, but the effects of AS in A3A and A3B on functional activities of these enzymes and relevant clinical outcomes have not been explored. Here, we characterized AS in A3A and A3B in relation to APOBEC-mediated mutagenesis and explored the mechanisms of its regulation and possible therapeutic modulation.
Results
Expression profiling of A3A and A3B transcripts: quantification of exon–exon junctions vs. total gene expression
According to the human reference genome annotation (hg19, UCSC), A3A and A3B genes have two and three alternative isoforms, respectively (Supplementary Table 1 and Supplementary Fig. 1a). Among these, only canonical isoforms, which we designate as A3A1 and A3B1, but not alternatively spliced isoforms (A3A2, A3B2, and A3B3), have been studied. First, we confirmed the existence of all these isoforms by analyzing RNA-seq reads in the TCGA dataset (see Supplementary Table 2 for samples used in the analysis) and identifying exon–exon junction reads specific to each of these isoforms (Fig. 1a and Supplementary Fig. 1b). The alternative A3A and A3B isoforms were expressed in 3.49% and ~50% samples with mean read counts of 15.4 and 5.2, respectively (Fig. 1b, c). In Supplementary Note 1, we show that due to the high homology between A3A and A3B, quantification based on specific exon–exon junctions (by RNA-seq or qRT–PCR) is more reliable than based on total gene expression analysis (by RNA-seq). The misaligned RNA-seq reads would incorrectly represent the expression of both genes, affecting downstream analyses and biological interpretations.
a Nucleotide sequences of exon–exon junctions specific to A3A and A3B isoforms. “X” – mismatch between A3A and A3B sequences. b Schematics of A3A exons and splicing junctions. The bar graphs show the numbers of samples (y axis) in relation to RNA-seq read counts grouped in 5 sub-categories (x axis) for the total A3A expression and exon–exon junction-based expression of A3A1 and A3A2 isoforms. ‘E’ refers to exon and ‘aE’ refers to an alternative exon. Based on exon junction reads (≥1 read/sample), A3A1 (E1–E2 junction) and A3A2 (E1-aE2 junction) is expressed in 6979 (63.10%) and 386 (3.49%) TCGA samples, respectively. c Schematics of A3B exons and splicing junctions and comparison of gene expression based on the total and exon–exon junction reads corresponding to A3B isoforms. A3B1 is expressed in 10,572 (95.8%) TCGA samples, but A3B2 and A3B3 are expressed in 53.55% (5922 of 11,058) and 51.84% (5733 of 11,058) of TCGA samples, respectively. The TCGA RNA-seq set includes 10,328 tumors and 730 adjacent normal tissue samples.
Specifically, based on total RNA-seq reads, A3B was undetectable only in 0.16% (18 of 11,058) of TCGA samples (Fig. 1b). However, based on exon–exon junction reads for the canonical isoform A3A1 (E1–E2 junction), expression was undetectable in 36.9% (4079 of 11,058) of TCGA samples (Fig. 1b). Similarly, A3B expression was undetectable by total RNA-seq reads only in 0.07% (8 of 11,058) of TCGA samples, but this number was 4.2% (468 of 11,058) based on exon–exon junction reads for the canonical A3B1 isoform (E5–E6 junction reads, Fig. 1c).
Alternative protein isoforms of A3A and A3B are non-mutagenic
To better understand the functional properties of these alternative isoforms, we performed computational analysis of their corresponding protein sequences. Compared to A3A1, A3A2 lacks 18 aa (residues 10–28) due to AS between exons 1 and 2, including residues His11 and His16, which are important for deamination activity19 (Fig. 2a). A3B2 is produced due to AS in exon 6, resulting in the loss of 25 aa (residues 242–266, Fig. 2b), including His253 that stabilizes the zinc cofactor, and Glu255 that directly participates in a nucleophilic attack on cytosine during deamination20.
Clustal Omega alignment of protein sequences for a canonical A3A1 and alternative A3A2 and b canonical A3B1 and alternative A3B2 and A3B3 isoforms. A3A2 lacks 18 N-terminal aa (R10 - G27), including functional residues H11 and H1619, and A3B2 lacks a fragment with functional residues E255 and H25320. Skipping of exon 5 generates an A3B3 transcript that encodes a truncated protein without the catalytic domain, in which a frameshift at position L191 results in the replacement of a 132 aa fragment in the C terminus of A3B by 62 aa of an aberrant frame-shifted sequence. c Due to a premature termination codon (PTC) in the penultimate exon 6, A3B3 might be targeted by nonsense-mediated decay (NMD). The effect of NMD demonstrated by analysis of mRNA expression of ATF4 (positive control) and A3B isoforms with TaqMan expression assays indicated by arrows in HT-1376 cells treated with DMSO (vehicle) or digoxin, an NMD inhibitor. d Outline of the in vitro deamination assays testing conversion of a 48-nucleotide (nt) ssDNA substrate (S) into a 25-nt product (P) by the recombinant C-terminally Myc-DDK tagged protein isoforms e A3A and f A3B. Negative control reactions (Neg ctrl) lack A3A and A3B proteins; positive control probe (Pos ctrl) is completely converted by the UDG enzyme. Western blot (WB) analysis with an anti-DDK antibody shows the amounts of A3A and A3B proteins in reactions. f Asterisk indicates a non-specific band resulting from deamination of ssDNA probe by A3B3. Because DNA binding domain is intact in A3B3, the higher molecular weight (MW) band could indicate a probe bound by A3B3. g HIV-1 infectivity restriction assays show no inhibitory effects of A3B2 on the activity of the A3B1 protein isoform measured for A3G/A3B protein fusions (G/B). Shown one of three representative experiments with Western blot for corresponding recombinant proteins and normalized relative luciferase units (%) for each labeled condition. All bar graphs (c, g) show individual data points, with error bars representing 95% confidence interval for the medians, based on three biological replicates. P-values are for the two-sided unpaired Student’s t-test.
The A3B3 isoform is generated by skipping of exon 5 in A3B and encodes truncated and likely unstable protein without the catalytic domain, in which a 132 aa fragment in the C terminus of A3B is replaced by 62 aa of an aberrant frame-shifted sequence (Fig. 2b). The stop codon in the penultimate exon also makes the A3B3 transcript a potential target for NMD. Indeed, A3B3 mRNA expression was significantly increased after treatment of HT-1376 cells with digoxin, a known NMD inhibitor21 (Fig. 2c). Due to NMD, expression studies, including by RNA-seq, would detect only residual levels of A3B3, underestimating its expression.
Evaluation of the deaminating (mutagenic) potential of recombinant APOBEC3 proteins (Fig. 2d) showed activities for the canonical A3A1 and A3B1 isoforms, but not for the alternative A3A2, A3B2, and A3B3 proteins (Fig. 2e, f). The alternative protein isoforms did not show dominant-negative effects on the activities of A3A1 and A3B1 evaluated either by deamination assays (Supplementary Fig. 2) or by HIV-1 infectivity inhibition assays22,23,24 (Fig. 2g and Supplementary Fig. 3). Based on these results, we conclude that AS of A3A and A3B results in the production of catalytically inactive, non-mutagenic protein isoforms of these enzymes.
Isoform-specific analysis refines the correlations between A3A and A3B expression and APOBEC-mediated mutagenesis
Previously, multiple reports showed correlations between the total expression of A3A and A3B genes and the burden of APOBEC-signature mutations in several tumor types8,25. Now, we revisited these conclusions based on correlations between the expression of the mutagenic A3A1 and A3B1 isoforms quantified by RNA-seq read counts for specific exon–exon junctions and “APOBEC mutation pattern” proposed as the most stringent estimate of APOBEC-signature mutation burden7. The analysis was performed in six cancer types, with ≥10% of tumors carrying these mutations (Supplementary Fig. 4).
We observed significant positive correlations between the expression of both mutagenic isoforms—A3A1 and A3B1 and APOBEC mutation burden in several cancer types. A3A1-specific correlations were observed in BRCA (P = 5.49E−08; rho = 0.17), CESC (P = 7.84E−08; rho = 0.37), and HNSC (P = 3.684E−08; rho = 0.24), while A3B1-specific correlations in BLCA (P = 2.76E−07; rho = 0.25). These results corroborate previous findings based on germline variants, which implicated A3A as a more prominent mutagen in BRCA and A3B—in BLCA1,11.
In LUAD, the correlations were comparable for both A3A1 (P = 3.12E−12; rho = 0.31) and A3B1 (P = 7.73E−12; rho = 0.30) and in LUSC—the correlation was moderately significant only for A3A1 (P = 1.86E−02; rho = 0.17) (Fig. 3a, b). Notably, these tumor-specific correlations were not reported by studies based on total gene expression8,25.
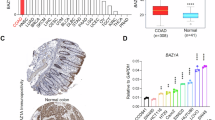
Correlation analysis between mRNA expression of a A3A1 and b A3B1 and APOBEC-mediated mutagenesis, measured as “APOBEC mutation load minimum estimate” in six cancer types in TCGA. Spearman correlation coefficients with P-values for two-sided tests are shown. The line in scatterplots represents the linear trend and the shading represents the 95% confidence interval around the line of best fit. c Significant correlation between APOBEC-signature mutation score (S3) and A3B1 but not A3A1 expression in non-muscle-invasive bladder cancer (NMIBC) in the UROMOL study. d Kaplan–Meier plots for progression-free survival (PFS) of NMIBC based on A3A1 and A3B1 isoform expression in the UROMOL study. P-values are for multivariable Cox-regression models adjusted for sex, age, and tumor stage. Grouping into “No”, “Low”, and “High” groups was done based on A3B1 RNA-seq read counts, separating samples with no expression (zero reads) and then below and above the median for the remaining samples.
A3B as a driver of APOBEC-mediated mutagenesis and a predictor of progression-free survival in bladder cancer
It was reported that episodes of APOBEC-mediated mutagenesis could be observed in cell lines propagated in culture26. Those included bladder cancer cell lines, in which we detected the expression of A3B1 but not A3A1 (Supplementary Note 1), thus nominating A3B1 rather than A3A1 as the primary mutagenic APOBEC3 enzyme responsible for these episodes of mutagenic activity.
Our analysis of non-muscle-invasive bladder tumors from the UROMOL study27 further supported the role of A3B1 in bladder cancer. Specifically, using the same RNA-seq exon junction isoform-specific quantification of expression as in TCGA samples, we observed that higher expression of A3B1 was significantly associated with increased APOBEC-mediated mutation burden (P = 5.51E−06) and with shorter progression-free survival (P = 5.69E−05) in patients with non-muscle-invasive bladder cancer (Fig. 3c, d). Similar quantification of the A3A1 expression in the UROMOL tumors showed no association with APOBEC-mediated mutagenesis or progression-free survival (Fig. 3d).
Non-mutagenic isoform A3B3 is more common in normal tissues compared to tumors
Alternative A3B isoforms (A3B2 and A3B3) were detected in ≥50% of 11,058 TCGA samples (Fig. 1), albeit at very low levels compared to canonical A3B1 isoform. Although these isoforms may not produce any functional proteins (Fig. 1), they could still be important. For example, splicing towards alternative, non-mutagenic isoforms might decrease the production of the canonical, mutagenic isoforms. Because mutagenesis is considered tumorigenic, we hypothesized that the proportion of splicing towards non-mutagenic isoforms might be higher in normal tissues compared to tumors. To test this hypothesis, we calculated the percent spliced-in index (PSI)28 based on RNA-seq read counts of specific exon–exon junctions in TCGA paired tumor and adjacent normal tissues. A3A was excluded from this analysis because the expression of the alternative A3A2 isoform was detected only in 2.57% (18 of 698) of normal samples (Supplementary Fig. 5a). Thus, we calculated PSI for exons 5 and 6 of A3B in 17 TCGA cancer types with ≥5 tumor/normal pairs. We observed that the proportion of A3B canonical splicing was higher, while the proportion of alternative splicing was lower in tumors compared to corresponding adjacent normal tissues.
Specifically, the proportion of A3B2 splicing was lower in tumors of KICH (P = 1.0E−03) and LUSC (P = 3.30E−02) (Supplementary Fig. 5b) and the proportion of A3B3 splicing was lower in tumors of BLCA (P = 6.65E−03), HNSC (P = 7.09E−03), LIHC (P = 6.01E−03), LUAD (P = 6.69E−04), and LUSC (P = 5.55E−07) (Supplementary Fig. 5c). All other cancer types showed no significant differences in this analysis (Supplementary Fig. 5d, e). Most cancer types with a low proportion of non-mutagenic isoforms, specifically of A3B3, also had a higher rate of APOBEC-mediated mutation burden7,8, with exceptions for tumors of KICH and LIHC, in which loads of APOBEC-signature mutations were negligible (Supplementary Fig. 4). In these cancers, A3B might play a mitogenic rather than a mutagenic role as has been shown for hepatocellular carcinoma29 and suggested for breast cancer30.
We also performed RT-PCR analysis and sequencing of splicing junctions between exons 4 and 6 of A3B in a panel of 33 muscle-invasive bladder tumors and 30 adjacent normal tissues (Fig. 4a). Exon 5 skipping, generating A3B3, was the most frequently observed AS event, similar to the pattern observed in TCGA, present in 30% of normal tissues compared to 13% of tumors (Fig. 4a). Western blot analysis in the same tissue samples showed that A3B1 protein expression was common in tumors but rare in normal tissues, although the frame-shifted and truncated A3B3 protein could not be detected (Fig. 4b and Supplementary Note 2). We then used isoform-specific TaqMan assays to quantify the expression of A3B isoforms in the same set of bladder tissues. A3B1 was expressed significantly higher in tumors, while A3B3 was higher in normal tissues, and A3B2 was not quantifiable (Fig. 4c). The fact that AS of A3B was more common in normal tissues suggests that increased generation of alternative, non-mutagenic A3B isoforms might be anti-tumorigenic or/and inhibited in tumors.
a RT-PCR analysis in 63 bladder tissue samples. Sanger sequencing of RT-PCR products generated with primers for A3B exons 4 and 6 shows the canonical (A3B1) and alternative (A3B2 and A3B3) splicing events. A splicing event (labeled as “Novel”) that involves both exon 5 skipping and the use of a cryptic splice site in exon 6, was detected in three samples. An AS event (mainly A3B3) was observed in 19% of all samples (13 of 63 samples), including 13% of tumors (4 of 30) and 27% of adjacent normal tissues (9 of 33). b Western blot analysis for the A3B1 protein. c qRT–PCR analysis for A3B1 and A3B3 isoforms. mRNA expression levels of A3B1 were significantly higher in tumors and of A3B3—in adjacent normal tissues. A3B2 expression was not detected in >95% samples (Supplementary Data 1). Plots show all individual data points with error bars representing 95% confidence intervals for the medians. P-values are for the non-parametric Mann–Whitney U test.
A3B exon 5 skipping is sensitive to expression levels of splicing factors
Considering that A3B1 is clinically relevant, at least in bladder cancer (Fig. 3c, d), decreasing its expression might be of therapeutic importance. We hypothesized this could be achieved by shifting A3B pre-mRNA splicing from the mutagenic A3B1 to non-mutagenic A3B2 or A3B3 isoforms. To this end, we first sought to explore the regulation of these splicing events. AS outcomes are regulated by the interaction of trans-acting spliceosomal and splicing factors (SFs) with cis-acting intronic and exonic pre-mRNA motifs31. To explore AS of A3B, we created mini-genes by cloning the corresponding alternative exons with 80 bp of flanking intronic sequences into an Exontrap vector (Supplementary Fig. 6a). These mini-genes were transiently transfected into HEK293T cells, and their splicing patterns were evaluated.
The mini-gene experimental system was not informative for evaluation of splicing occurring via a cryptic splicing site in exon 6 of A3B, as the observed splicing pattern represented only the canonical (A3B1) but not the alternative (A3B2) isoform (Supplementary Fig. 6b). However, we could capture the AS pattern of A3B exon 5, with its inclusion representing a proxy of the A3B1 isoform and its skipping representing a proxy of the A3B3 isoform. We used this mini-gene for exon 5 (E5) to further explore the regulation of A3B1 versus A3B3 splicing patterns.
We hypothesized that splicing of A3B exon 5 might be affected by SFs and bioinformatically predicted several candidate SFs that bind within this exon (Supplementary Table 3). To experimentally test these predictions, we co-transfected HEK293T cells with expression constructs for 10 of these SFs (Supplementary Table 4 and Fig. 5a) together with the E5 mini-gene. Of these, four SFs—SRSF2, SRSF3, CELF1-T4, and ELAVL2—significantly affected exon 5 splicing (Fig. 5b), with SRSF2 showing the strongest effect. The effect of SRSF2 on exon 5 splicing was further confirmed in three additional bladder cancer cell lines—HT-1376, SW-780, and HTB-9 (Supplementary Fig. 7). Screening of exon 5 splicing in 10 cell lines of different tissue origin also showed variable exon 5 skipping (Fig. 5c), presumably due to differences in levels of expression/activity of endogenous SFs in these cell lines.
a Exontrap mini-gene E5 as an experimental model for the analysis of AS of A3B exon 5. b An agarose gel showing variable ratios of RT-PCR for splicing products of the E5 mini-gene at baseline (No SF control) and after co-transfection with 10 candidate SFs in HEK293T cells; negative control—untransfected cells; M—100-bp size marker; positive control—vector-specific amplicon of 84 bp. Bands from separate gels representing three biological replicates were quantified by densitometry, and the A3B1/A3B3 splicing shifts were calculated in relation to no SF control and plotted as a bar graph. Overexpression of SRSF2 significantly shifted splicing towards increased A3B3 expression (about 10%). c Splicing patterns of the E5 mini-gene analyzed by RT-PCR and agarose gel electrophoresis in 10 human cell lines. Bands from separate gels representing three biological replicates were quantified by densitometry to assess the skipping of A3B exon 5, as shown in the bar graph. d Branch points site (BPS) prediction for all 7 introns of A3B. Shown are the BPS with the highest predicted scores for each intron, with indicated positions upstream of the corresponding exons and the numbers of predicted BPS in a 100 bp window. The two predicted BPS within intron 4 have the lowest scores of all A3B introns. e Exon splicing enhancer (ESE) and exon splicing silencer (ESS) prediction within A3B exons. Exon 5 has the strongest ESS motif of all A3B exons. f Comparison of splicing patterns for A3B exons 5 and 6 using mini-genes with (E5BPS+ and E6BPS+) and without (E5BPS− and E6BPS−) predicted BPS. “E” refers to exon and “VE” to vector exon. Splicing between vector exons—VE1-VE2 is a proxy for A3B3 isoform created by exon 5 skipping. All bar graphs (b, c) show individual data points with error bars representing 95% confidence intervals for the medians, based on three biological replicates. P-values are for the two-sided unpaired Student’s t-test.
To test this, we performed a correlation analysis between the percentage of A3B exon 5 skipping in the mini-gene experiment (Fig. 5c) and total mRNA expression of the eight SFs evaluated in this experiment (Fig. 5c), using RNA-seq data available in the Cancer Cell Line Encyclopedia (CCLE). HEK293T cell line was excluded from this analysis because RNA-seq data for this cell line was not available in CCLE. A3B exon 5 skipping showed significant positive correlations with the expression of SRSF2, KHDRBS1, and MBNL1, and negative correlations with the expression of ELAVL2 (Supplementary Note 5). The correlation with SFRS2 expression was the strongest and supported the results of the overexpression model (Fig. 5b). Thus, our results suggested that A3B exon 5 skipping is sensitive to expression levels of multiple SFs, which can bind to cis-regulatory motifs within this exon.
Skipping of A3B exon 5 is facilitated by weak intronic branch point sites within intron 4
Bioinformatics analysis of A3B showed differences in the distribution of predicted cis-regulatory motifs - intronic branch point sites (BPS) (Fig. 5d and Supplementary Table 5) and exonic splicing silencers/enhancers (Fig. 5e). The most striking differences were found within intron 4 that harbored the fewest and the weakest BPS of those scored in all A3B introns. The highest scored intronic BPS located 38 and 50 bp upstream of exon 5 were included in the 80 bp of flanking intronic sequences in the E5 mini-gene (referred to as E5BPS+) and supported the observed partial inclusion of exon 5 that generates the A3B1-type canonical splicing event (Fig. 5e).
We then created an E5 mini-gene version with only 20 bp of flanking intronic sequences to exclude the putative BPS and observed no canonical A3B1-type splicing in this model (E5BPS−, Fig. 5e), confirming the importance of the putative BPS in intron 4 for exon 5 inclusion/skipping. In agreement with the stronger predicted BPS in intron 5 compared to those in intron 4, only canonical splicing with complete inclusion of exon 6 was observed in a corresponding mini-gene for exon 6 of A3B with 80 bp of flanking intronic sequences (E6BPS+). Consistently, no canonical splicing was observed in the E6BPS− mini-gene, which lacks the BPS in intron 5 (Fig. 5e). Based on these results, we conclude that the skipping of A3B exon 5 is facilitated by weak BPS in intron 4. The magnitude of exon 5 skipping is likely to be determined by expression levels of some SFs that might differ between tissue types and disease conditions, including normal tissues vs. tumors.
SF3B1 inhibitor pladienolide B promotes skipping of A3B exon 5 and reduces A3B1 production
Efficient canonical splicing requires robust interaction of the spliceosomal machinery with intronic BPS; thus, exon 5 skipping could result from a weak engagement of the spliceosome to BPS in intron 4 (Fig. 6a). Interaction between spliceosome and BPS is facilitated by the SF3b complex, with SF3B1 splicing factor as a core protein32. We hypothesized that interference with SF3B1 function, such as by treatment with a spliceosome inhibitor pladienolide B32,33, would destabilize interaction of the SF3b complex at weak BPS within intron 4, resulting in enhanced exon 5 skipping, but may not affect exon 6 splicing that is regulated by strong BPS in intron 5 (Fig. 6b). We tested this hypothesis by evaluating splicing patterns of E5BPS+ and E6BPS+ mini-genes in cells treated with pladienolide B.
a Schematic representation of the A3B splicing module (exon 4–exon 6) featuring interactions between trans-factors of the spliceosomal machinery with splicing cis-elements: splice sites (SS), branch point sites (BPS), polypyrimidine tract (Py), and exonic splicing enhancers/silencers (ESE)/(ESS); possible outcomes of A3B exon 5 splicing resulting from interactions between SF3B1 and other spliceosomal proteins with weak versus strong BPS. b Schematic representation of the hypothesis depicting that inhibition of SF3B1 is expected to result in skipping of A3B exon 5 and downregulation of A3B1 protein. c PCR analysis of splicing patterns of A3B exons 5 and 6 using Exontrap mini-genes E5BPS+ and E6BPS+ in HT-1376 cells treated for 36 h with increasing concentrations of pladienolide B. Exon 5 skipping is increased in the presence of the SF3B1 inhibitor, while splicing of exon 6 is not affected. d Results of qRT–PCR in HT-1376 cells treated with increasing concentrations of pladienolide B for indicated time points show splicing shift from A3B1 to A3B3. Shown are means ± SD of biological triplicates. e Western blot protein analysis of HT-1376 cells treated with increasing concentrations of pladienolide B for 36 h showed downregulation of A3B1 protein, while expression of endogenous A3B3 protein was not detected. f RT-PCR analysis of exon 5 splicing representing A3B1 and A3B3 transcripts in HT-1376 cells treated with DMSO (vehicle), digoxin (an NMD inhibitor), pladienolide B alone, and pladienolide B with digoxin. A3B3 mRNA levels are increased in digoxin-treated cells due to its inhibition of NMD. g Deamination assays using whole-cell extracts from HT-1376 cells treated with bleomycin and pladienolide B. Equal amounts of total protein were used for each reaction based on densitometry pre-assessment of GAPDH protein levels in each sample. Deamination was observed in reactions with protein lysate of HT-1376 cells treated with bleomycin alone or bleomycin plus 1 nM of pladienolide B. Higher concentrations of pladienolide B (10, 20, 40, and 100 nM) reduced A3B1 levels and prevented deamination of the ssDNA probe in bleomycin-treated conditions. A3B1 was barely detectable in DMSO and pladienolide B only conditions because of low total protein input in reactions (2 μg of total protein lysate). h Separate experiment showing that pladienolide B (10, 20, 40, and 100 nM) suppresses induction of A3B by bleomycin. Western blot (WB) analysis with an anti-A3B1 antibody shows the amounts of A3B1 in corresponding reactions. Each lane was loaded with equal amounts of the whole-cell lysate (15 μg of protein/lane). Shown are representative results of one of the three independent experiments.
This treatment not only enhanced but also caused complete exon 5 skipping in a concentration-dependent manner while not affecting exon 6 splicing in the mini-gene models (Fig. 6c). Skipping of exon 5 in endogenous A3B was also increased by pladienolide B treatment, resulting in increased expression of A3B3 mRNA and decreased expression of A3B1 mRNA and A3B1 protein (Fig. 6d, e and Supplementary Fig. 8a). Importantly, the reduction of A3B1 protein caused by pladienolide B treatment was more prominent and consistent than the induction of A3B3 expression. A3B3 expression was much higher in cells treated with pladienolide B in the presence of digoxin, an NMD blocker21 (Fig. 6f), supporting the NMD-based degradation of A3B3 transcript (Fig. 1c) as a mechanism contributing to its poor detection on mRNA level and no detection on the protein level.
These experiments confirmed that exon 5 skipping is facilitated by weak BPS in intron 4 but is also dependent on the expression levels of SF3B1. The role of SF3B1 in this process was further confirmed by the effect of its siRNA-mediated knockdown in HT-1376 cells, which resulted in increased skipping of A3B exon 5 (Supplementary Fig. 8b). Thus, interfering with SF3B1 activity by its inhibitors should increase A3B exon 5 skipping, leading to reduced levels of mutagenic A3B1.
Reducing the A3B1 levels increased by various endogenous and exogenous factors, including treatments, might restrict APOBEC mutagenesis. This restriction could be important for preventing tumor progression, recurrence, clonal evolution, and chemotherapy resistance34. To explore this, we simultaneously treated cells with bleomycin, a chemotherapy drug, which induces A3B expression1 and pladienolide B. Endogenous bleomycin-induced A3B caused deaminating (mutagenic) activity detectable in lysates of these cells, but this activity was completely blocked by pladienolide B (Fig. 6g). In the samples co-treated with bleomycin and pladienolide B, A3B1 protein levels remained similar to untreated samples (Fig. 6h). These results suggest that increased A3B1 production can be prevented, such as by treatments that switch A3B1 mRNA to A3B3, such as with pladienolide B. The APOBEC-mediated mutagenic activity was also inhibited by pladienolide B in a cell-based cytosine deamination assay (Supplementary Fig. 9).
Identification of splice-switching oligonucleotides to facilitate A3B exon 5 skipping
Splice-switching oligonucleotides (SSOs) can specifically and effectively modulate splicing events as therapeutic targets in various conditions, including cancer35. To this end, we designed SSOs to target A3B splicing through two BPS in intron 4 (BPS-50 and BPS-38) and the 3′-splice site of exon 5 (3′SS), and tested their ability to affect A3B exon 5 skipping in vitro (Fig. 7a). The SSO-BPS-50 had no effect and SSO-BPS-38 had a modest effect on exon 5 skipping (10%) (Fig. 7b), which could be due to RNA folding at these sites creating steric hindrance for binding, a common problem in identifying efficient SSOs36. However, the SSO-3′SS caused ~80% of A3B exon 5 skipping (Fig. 7b), providing first insights into the utility of using SSOs for modulating splicing of A3B through exon 5 skipping for controlling APOBEC-mediated mutagenesis (Fig. 7c).
a Schematic representation of the A3B splicing module that includes exons 4–6. The splice sites (SS), branch point sites (BPS), and SSOs for intron 4 BPS (positions −50 and −38 bp) and 3′ SS of exon 5 are depicted. b A representative agarose gel showing RT-PCR products of exon 5 inclusion and skipping in two bladder cancer cell lines (HT-1376 and HTB-9) that were nucleofected with 2 nM of SSOs. c Bar graphs based on image analysis of b, showing the % of A3B exon 5 skipping. Error bars represent 95% confidence intervals for the medians, based on three biological replicates. SSO-3′SS resulted in exon 5 skipping in ~80% of A3B mRNA.
Discussion
High activity of the A3A and A3B enzymes is mutagenic and genotoxic, as suggested by in vitro overexpression studies37,38,39,40 and positive correlations between mRNA expression of genes encoding these enzymes and burden of APOBEC-signature mutations in tumors1,8,25,41. This potent mutagenic activity must be tightly regulated by multiple mechanisms to restrict the cell damage it may cause. Here, we showed that AS of APOBEC3s, and particularly of A3B, is one of several possible regulatory mechanisms that control the expression of the mutagenic A3B1 enzyme. We present proof-of-principle data suggesting that AS can be modulated to shift the A3B balance from producing mutagenic to non-mutagenic isoforms.
Initially, we explored the functional consequences of splicing events within exon 2 in A3A and exon 6 in A3B, both occurring via cryptic splicing sites, and another event, occurring through skipping of the entire exon 5 in A3B. In all these cases, AS resulted in a shift from the production of the canonical isoforms (A3A1 and A3B1) that encode mutagenic enzymes to corresponding alternative isoforms (A3A1, A3B2, and A3B3) that encode non-mutagenic enzymes. The expression of the canonical isoforms positively correlated with a load of APOBEC-signature mutations in TCGA tumors of different types. The isoform-level analysis also suggested that A3A1 and A3B1 might contribute to APOBEC mutagenesis in a cancer-type specific manner. Our previous finding that a common genetic variant identified by a GWAS for bladder cancer risk was also associated with A3B expression and APOBEC mutagenesis1 nominated A3B as the primary mutagenic APOBEC in bladder tumors. This was further supported by the association of increased A3B1 expression with higher APOBEC mutation score and shorter progression-free survival in patients with non-muscle-invasive bladder cancer.
AS is regulated by a complex interplay between the core spliceosomal and alternative SFs31 that bind cis-acting exonic and intronic elements. These interactions can depend on various tissue- and disease-specific environments42. Analysis of splicing patterns using our in vitro mini-gene Exontrap system identified splicing plasticity of A3B exon 5, which we attributed to the weak BPS in intron 4 of this gene and variable levels of SF3B1 expression. Splicing of endogenous exon 5 also varied in cell lines of different tissue origin and was sensitive to changes in expression levels of some other SFs predicted to bind within this exon, such as SRSF2.
We observed that A3B splicing events were more common in adjacent normal tissues compared to tumors of several types. This suggests that AS of A3B is an intrinsic, tissue-specific regulatory mechanism rather than a result of general dysregulation of splicing machinery in tumors43,44, manifested in the inactivation of tumor suppressor genes and generation of tumor-specific isoforms45. On the other hand, mutations in splicing or other regulatory factors that would affect splicing globally could also result in decreased splicing towards alternative A3B2 and A3B3 isoforms, failing to prevent shunting of A3B1 mRNA and consequently lead to increased APOBEC mutagenesis. Thus, AS of A3B might be a natural mechanism restricting the expression of mutagenic isoforms in some conditions, such as in normal tissues.
The observed splicing plasticity of A3B mRNA might represent an adaptive biological mechanism of tuning down the excessive effects of mutagenic APOBEC3 proteins to safeguard the cells from the genotoxic activity of these enzymes. A similar role has been proposed for several SFs, including SRSF2, which affected A3B exon 5 splicing in our experiments. These SFs regulate the expression of DNA repair proteins to protect the genome from DNA damage and the toxic effects of mutagens46,47,48. Splicing re-routing, such as by exclusion of exon 5 in A3B, followed by elimination of the alternative frame-shifted A3B3 transcript by NMD might be a mechanism to tweak APOBEC mutagenesis in specific conditions. The entire functional role of A3B3 could be just to use up some pre-mRNA that otherwise would be used to produce the mutagenic A3B1 enzyme and then get degraded by NMD. Thus, the NMD-targeted A3B3 transcript not producing a functional protein might still play an important role in the regulation of APOBEC mutagenesis, regardless of its very low residual expression levels observed in tumors and tissues.
A recent study has analyzed mutational signatures in a large set of cell lines and suggested that the initiators of APOBEC mutagenesis in vitro are cell-intrinsic factors with continuous but intermittent activity26. As the authors did not observe correlations between APOBEC mutagenesis and expression of APOBEC3 genes, they suggested that these initiators may include modulators such as the availability of single-stranded DNA (ssDNA) substrate, etc. Based on our observations that A3B1 and not A3A1 is expressed in bladder cancer cell lines, which have a high load of APOBEC-signature mutations, it is likely that at least in these cell lines, episodic APOBEC-mediated mutagenesis is caused by the A3B1 activity.
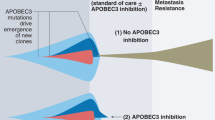
Therapeutic targeting of AS to treat cancer is a rapidly developing field49 with several types of SF3B1 inhibitors, such as E710750,51, an analog of pladienolide B, and H3B-880052 being evaluated for acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS)33,49,52. Because tumor cells depend on wild-type SFs for survival, these drugs are particularly effective in killing cancer cells with already impaired splicing machinery due to mutations in SFs, such as SRSF2 and SF3B152. Mutations in these and other SFs have been reported in many solid tumors, including bladder cancer53 and these tumors tend to have more APOBEC-signature mutations (Supplementary Fig. 10). Our results suggest that SF3B1 inhibitors and other tools affecting alternative splicing might be tested to eliminate cancer cells with mutations in SFs and also to control APOBEC mutagenesis in clinically relevant conditions. It is interesting to note that FGFR3-S249C, the most common somatic mutation found in the highly recurrent non-muscle-invasive bladder cancer, is likely caused by APOBEC mutagenic activity54. As we found A3B1 the primary mutagenic APOBEC enzyme in bladder tumors, enhancing A3B exon 5 skipping might help to restrict APOBEC-mediated mutagenesis, including FGFR3-S249C mutation in bladder cancer, as well as prevent tumor progression and recurrence, clonal evolution, and resistance to chemotherapy (Fig. 8). In addition to using SF3B1 inhibitors, we also demonstrate the utility of SSOs for inducing exon 5 skipping and decreasing the expression of mutagenic A3B isoform, possibly opening up avenues for targeting APOBEC-mediated mutagenesis by oligo-based drugs55. Using SSOs to increase the skipping of exon 5 of A3B would result in natural NMD-based elimination of the non-functional A3B3 isoform produced in this process. Targeting any other gene region of the highly homologous APOBEC3 gene family (Supplementary Note 1) might be more challenging and cause off-target effects.
Canonical splicing of A3B pre-mRNA generates the mutagenic A3B1 enzyme that causes APOBEC mutagenesis and fuels tumor progression, recurrence, clonal evolution, and chemotherapy resistance. Alternative splicing of A3B, and, specifically, skipping of exon 5, generates A3B3 isoform with a premature termination codon (PTC). Some of this aberrant transcript is degraded by nonsense-mediated decay (NMD), and the residual transcript encodes catalytically inactive protein isoform. Therapeutic enhancing of exon 5 skipping by SF3B1 inhibitors or other tools affecting alternative splicing, such as splicing-switching oligonucleotides (SSOs), may restrict and even prevent A3B-mediated mutagenesis in clinically relevant conditions.
In conclusion, our study showed that AS of A3B, which creates non-mutagenic isoforms, represents an intrinsic regulatory mechanism that keeps in check the expression of the mutagenic A3B1 enzyme, modulates APOBEC-mediated mutagenesis in different disease-, tissue- and environment-specific conditions, and can be therapeutically targeted. Additional functional studies will be needed to better understand the consequences of this modulation for clinical outcomes.
Methods
Data acquisition and processing of TCGA samples
The mutation dataset for all TCGA cancer types was downloaded from the Broad GDAC Firehose in October 2016. For analysis of APOBEC-signature mutations, we used the variable “APOBEC mutation load minimum estimate”, which represents APOBEC mutation pattern8. mRNA expression of A3A and A3B genes was analyzed in 11,058 TCGA samples (10,328 tumors and 730 adjacent normal tissues) based on RNA-seq BAM slices generated using workflow https://docs.gdc.cancer.gov/API/Users_Guide/BAM_Slicing/ through the NCI Genomics Data Commons (GDC) portal accessed in August 2019. RNA-seq BAM files for cell lines from the Cancer Cell Line Encyclopedia (CCLE) were acquired from the GDC legacy archive (https://portal.gdc.cancer.gov/legacy-archive/search/f) and Stringtie-2.1. 4 was used for whole-transcriptome level gene expression quantification using hg19 as the reference.
Estimation of A3A and A3B RNA-seq read counts for exons and exon–exon junctions
For estimation of sequencing reads corresponding to all exons as well as canonical and alternatively spliced exon–exon junctions, RNA-seq BAM slices were processed through R package ASpli version 1.5.1. We used strict ASpli pipeline settings, requiring a minimum of 10 nucleotides of perfect match covering exon–exon junctions.
Correlation analysis of mRNA expression and APOBEC mutagenesis
Expression levels of the mutagenic isoforms A3A1 and A3B1 were calculated based on their RNA-seq exon–exon junction read counts. The counts of sequencing reads for E1–E2 junctions (for A3A1) and E5–E6 junctions (for A3B1) and APOBEC mutation load minimum estimate counts were Log10-transformed (after adding 1 to all raw values). The Spearman correlation analysis and data plotting were performed using R packages for six cancer types with ≥10% of samples with APOBEC-signature mutations. SKCM was excluded from the analysis because mutations induced by APOBEC in melanoma likely overlap with mutations induced by UV56.
Analysis of AS levels in paired tumor and adjacent normal tissue samples
The analysis was performed in 17 cancer types with ≥5 tumor-normal pairs. Splicing ratios were calculated as PSI based on RNA-seq reads using ASpli pipeline, as was previously described28 and statistical significance between PSI in tumors vs. normal tissues was evaluated by non-parametric Wilcoxon matched-pairs signed-rank test.
RNA-sequencing of SeV-infected T47D cells
Total RNA extracted from the T47D breast cancer cells infected or not infected (control) with Sendai Virus (SeV)1 was used for paired-end RNA-seq on HiSeq 2500 (Illumina) in biological duplicates (SRA accession number: PRJNA512015). The library was prepared with KAPA Stranded RNA-seq Kit with RiboErase (Roche). RNA-seq reads (120 bp) were filtered and aligned with STAR alignment tool57 using the GRChg37/hg19 genome assembly and visualized using the Integrative Genomics Viewer (IGV). The RNA-seq reads aligned to A3A were BAM-sliced with SAMtools and then re-aligned to the reference genome by STAR to identify cross-alignment with A3B.
Bioinformatics analysis of the A3A and A3B protein isoforms
Protein sequences of the APOBEC3 isoforms A3A1 (UCSC ID uc003awn.2), A3A2 (UCSC ID uc011aob.1), A3B1 (UCSC ID uc003awo.1), A3B2 (UCSC ID uc003awp.1), and A3B3 (UCSC ID uc003awq.1) were downloaded from the UCSC genome browser. Multi-sequence alignments were generated using the web-based tool Clustal Omega58.
Cell lines
Cell lines—embryonic kidney HEK293T, bladder cancer cell lines - HT-1376, RT-4, HTB-9, and SW-780, breast cancer cell lines—MDA-MB-231 (HTB-26), and T47D (HTB-133), hepatocellular carcinoma HepG2, cervical adenocarcinoma HeLa and lung carcinoma H460—were purchased from the American Type Culture Collection (ATCC) and maintained per ATCC recommendations. A pancreatic cancer cell line PA-TU-8998T was purchased from Leibniz Institute DSMZ-German Collection of Microorganisms and Cell Cultures (DSMZ Scientific). No commonly misidentified cell lines were used in this project. If used longer than for a year after initial purchase, cell lines were authenticated by the Cancer Genomics Research Laboratory of DCEG/NCI by genotyping a panel of microsatellite markers (Identifiler kit, ThermoFisher Scientific). All cell lines were tested bi-monthly for mycoplasma contamination using the MycoAlert Mycoplasma Detection kit (Lonza).
Analysis in non-muscle-invasive bladder tumors from the UROMOL study
A set of low-stage (Ta and T1) bladder tumors representing non-muscle-invasive bladder cancer (NMIBC) has been described27. Mutations were scored based on RNA-seq data and used for deconvolution into mutational signatures S1–S6, with S3 corresponding to the APOBEC-signature mutations27. The FASTQ files for RNA-seq data were aligned with STAR and BAM-sliced to include A3A and A3B genes, followed by estimation of all exon and exon–exon junction reads using ASpli R package, similar to the analysis in TCGA samples. We performed a Spearman correlation analysis of log10-transformed read counts for A3A1 and A3B1 with APOBEC-signature mutation score (S3). Progression-free survival analysis was performed based on the expression of A3A1 and A3B1 mutagenic isoforms with samples divided into three groups: “No”—samples with undetectable expression (0 value); the remaining samples were split into two groups based on the expression below and above the median as “Low” and “High” groups, respectively. The Cox-regression models were adjusted for sex, age, and tumor stage.
Analysis of additional bladder tumor and adjacent normal tissue samples
The panel of muscle-invasive bladder tumors (n = 42) and adjacent normal (n = 32) tissue samples has been described59. For each of these samples, cDNA was synthesized from 250 ng of total RNA per 20 µl reactions using the RT2 First-Strand cDNA kit and random hexamers (Qiagen). For detection of splicing events between A3B exons 4 and 6, we performed PCR with primers: F_ex4: 5′-GCCTTGGTACAAATTCGATGA-3′ and R_ex6: 5′-TGTGTTCTCCTGAAGGAACG-3′, with cDNA input corresponding to 30 ng of total RNA per 25 μl reactions using AmpliTaq Gold™ 360 Master Mix (ThermoFisher Scientific). PCR-amplified products were resolved on 2% agarose gel, and each distinct PCR product was cut, purified, and Sanger-sequenced. For quantification of A3B1 and A3B3 splicing products, the cDNA input corresponding to 10 ng of total RNA per reaction was used, as previously described1, and subjected to qRT–PCR using isoform-specific TaqMan assays (Supplementary Note 4). Expression of A3B1 and A3B3 was normalized by the expression of endogenous controls GAPDH (assay 4326317E) and PPIA (assay 4326316E). Western blotting for A3B1 protein and GAPDH (loading control) was performed as described in Supplementary Note 2.
Generation and partial purification of the recombinant A3A and A3B protein isoforms
Expression constructs for the C-terminally Myc-DDK tagged canonical isoforms A3A1 (NM_145699) and A3B1 (NM_004900) cloned in the pCMV6 vector were purchased from OriGene (Rockville, MD). Open reading frames for the C-terminally Myc-DDK tagged alternative isoforms A3A2, A3B2, and A3B3 (Supplementary Table 1) were synthesized (ThermoFisher Scientific) and cloned into BamHI/XbaI restriction sites of the pcDNA3.1(+) vector. The HEK293T cells (4 ×106 cells/20 ml) were seeded in 175 cm2 flasks (Corning) and transiently transfected with plasmids after 24 h at 75% confluency using Lipofectamine 3000 (ThermoFisher Scientific). Cells were harvested 24 h post-transfection and proteins were purified with c-Myc tagged Protein Mild Purification Kit (MBL, Japan) and eluted with 20 μl of 1 mg/ml Myc peptide provided with the kit. The concentration of the total eluted protein, which included both purified protein and Myc peptide, was estimated using a BCA protein assay (ThermoFisher Scientific). For evaluating protein purity and enrichment, ~25 μg of total protein was resolved on 4–12% Tris-glycine SDS polyacrylamide gel (Life Technologies) and used for Western blot analysis. Densitometry analysis of Western blots showed at least 10-fold enrichment of all eluted isoforms compared to whole-cell lysates (Supplementary Note 3). All candidate antibodies were tested for the detection of A3A and A3B protein isoforms after overexpression of corresponding expression constructs in cell lines (Supplementary Note 2). Detection was done using a HyGLO chemiluminescent HRP antibody detection reagent (Denville Scientific Inc).
Cytosine deamination assays with recombinant proteins
Deamination activity of the recombinant A3A and A3B protein isoforms was tested as previously described60. Specifically, reactions were carried out in 10 µl of deamination buffer containing 10 mM Tris/HCl, pH 7.5, 50 mM NaCl, 1 mM DTT, 0.25–1 µg of partially purified A3A and A3B proteins, and 1–5 pM of single-stranded DNA substrate—the target probe 5′-5Alexa488N/(ATA)8TCC (ATA)7-3′, or a positive control probe 5′-5Alexa488N/(ATA)8TUU (ATA)7-3′ (Invitrogen). Reactions were incubated in a water bath at 37 °C for 2 h, treated with the Uracil DNA Glycosylase (UDG) for 40 min at 37 °C, and then with 0.6 N NaOH for 20 min at 37 °C. Final products were mixed with 2x RNA loading dye (ThermoFisher Scientific) and heated at 95 °C for 2–3 min. Of the final 40 μl reaction volume, one set of 14 µl aliquots was resolved on 15% TBE-urea polyacrylamide gel (Life Technologies) at 150 V for 1 h and 20 min at room temperature in 1× TBE buffer. Gels were imaged with Gel Doc (Bio-Rad) using Alexa 488 fluorescence settings. Another set of 14 µl aliquots from the same reactions was used for the detection of APOBEC3 proteins by Western blotting. Concentrations of eluted proteins were estimated based on densitometry of Western blots. For competition assays, the amounts of mutagenic isoforms and total reaction volumes were kept constant while the amounts of non-mutagenic isoforms were increased. Proteins extracted from the lysates of untransfected cells were used as a negative control to account for inhibition caused by non-specific endogenous proteins. In the 1:1 control competition reaction, the non-mutagenic isoform was replaced by an equal amount of the negative control protein.
Deamination activity of endogenous A3B1 in the presence of pladienolide B was evaluated using a previously described protocol14 with some modifications. Briefly, HT-1376 cells, treated with bleomycin (25 µg/ml) alone, pladienolide B alone (1, 10, 20, 40, 100 nM) and with combined bleomycin and pladienolide B, were harvested after 48 h in HED buffer (25 mM HEPES, 5 mM EDTA, 10% glycerol, 1 mM DTT and protease inhibitor). The protein concentrations were determined based on a densitometric assessment of GAPDH by Western blot from 7 μl of total lysate for each condition. Each 20 μl deamination reaction contained equal amounts of total protein in 15 μl (adjusted with H2O), 1 μl (10 pM) of single-stranded DNA substrate–probe 5′-5Alexa488N/(ATA)8TCC (ATA)7-3′, 2 μl 10× UDG buffer (ThermoFisher Scientific), 1 μl (1 U/μl) UDG and 1 μl RNaseA (100 mg/ml, Qiagen) and reactions were incubated at 37 °C for 3 h. Subsequently, 100 mM NaOH was added to each reaction, and the samples were then incubated at 37 °C for 30 min to cleave the abasic sites. Final products were mixed with 2× RNA loading dye (ThermoFisher Scientific) and heated at 95 °C for 2–3 min, and reactions were resolved on a 15% urea-TBE gel and imaged as described above.
HIV-1 infectivity inhibition assays
The activity of the A3B protein isoforms was evaluated with single-cycle infection assays for HIV-1 restriction as has been described for APOBEC3G (A3G)22,23,24. Briefly, a G/B-A3B1 plasmid was constructed starting from the previously engineered A3B plasmid22 by replacing 63 aa at the N-terminus with a similar region of A3G. This replacement increases the packaging of A3B protein into HIV-1 viral particles, which is necessary for the ability of A3B to inhibit HIV-1 infectivity22,24. This system is suitable for analysis of the activity of A3B, which like A3G, has two cytidine deaminase domains. However, this approach is not appropriate for evaluating the activity of A3A as it contains only one cytidine deaminase domain, and A3G/A3A swap would not be compatible. Fusion constructs G/B-A3B-V1, G/B-A3B-V2, G/B-A3B-V3, were generated to represent three A3B expression constructs, all with C-terminal hemagglutinin epitope tags (3X-HA). All plasmids were verified by Sanger sequencing. Because the A3B plasmids were from three sources, we found two protein-changing single point variations that might be functionally relevant (Supplementary Fig. 3). To generate the virus for infection, HEK293T cells (4 × 105 cells/6-well dish) were transfected using LT1 reagent (Mirus Bio) with HDV-EGFP (1 ug), pHCMV-G (0.25 ug), and variable concentrations of plasmids (0, 680, and 1200 ng), individually or in combinations. The virus was harvested 48 h post-infection, filtered with 0.45-um-pore filters, and stored at −80 °C. Capsid p24 measurements were determined using the HIV-1 p24 capsid (CA) ELISA Kit (XpressBio). Normalized p24 CA amounts were used to infect TZM-bl cells containing HIV-1 Tat-inducible luciferase reporter gene in a 96-well plate (4000 cells/well). Luciferase activity was measured 48 h after infection, using a 96-well luminometer (LUMIstar Galaxy, BMG LABTECH). For some experiments, portions of the viral supernatant were spun through a 20% sucrose cushion (15,000 rpm, 2 h, 4 °C, in a Sorvall WX80 + ultracentrifuge), concentrated 10-fold, and used in experiments to determine virion encapsidation of APOBEC3 proteins by western blotting analysis as previously described23.
Evaluation of A3B3 mRNA degradation by nonsense-mediated decay (NMD)
The HT-1376 cells were treated with DMSO (vehicle) or with 2, 5, and 10 µM of digoxin (Sigma), an NMD inhibitor21 dissolved in DMSO. The cells were harvested after 16 and 24 h of treatment; total RNA was isolated with an RNeasy kit with on-column DNase I treatment (Qiagen), and RNA quantity and quality were analyzed with NanoDrop 8000 (Thermo Scientific). After an additional DNA removal step, cDNA for each sample was prepared from equal amounts of total RNA, using the RT2 First-Strand cDNA kit and random hexamers (Qiagen). Expression was measured in the same cDNA with TaqMan expression assays (all from ThermoFisher Scientific) for endogenous controls GAPDH (assay 4326317E) and PPIA (assay 4326316E), and positive control ATF4 (assay Hs00909569_g1), which is induced by NMD inhibition61; custom assays were used for detection of A3B1, A3B3, and A3B1/A3B2 combined (Supplementary Note 4). Experiments were performed in biological triplicates per condition and expression was measured in four technical replicates on QuantStudio 7 (Life Technologies) using TaqMan Gene Expression buffer (Life Technologies). Water and genomic DNA were used as negative controls for all assays. Expression was measured as Ct values (PCR cycle at detection threshold) and calculated as fold change using the 2−(ΔΔCt) method in relation to control (untreated) groups of samples.
Bioinformatics analysis of A3B splicing cis-elements and SF binding sites
Exons and 100 bp of flanking intronic sequences were used for the prediction of exonic splicing enhancer (ESE)/silencer (ESS) motifs and branch point sites (BPS) using the Human Splicing Finder (HSF, www.umd.be/HSF3/)62. Per HSF guidelines, BPS with scores above 67 were considered high-confidence; the strength of ESE and ESS was evaluated based on the relative ESE/ESS ratio. SF binding sites were predicted using SFmap63 and SpliceAid264 (Supplementary Table 3).
Exontrap analysis of alternatively spliced exons of A3A and A3B genes
Exon 2 of A3A and exons 5 and 6 of A3B with either 20 or 80 bp of flanking intronic sequences were custom-synthesized (ThermoFisher Scientific) and cloned in sense orientation using XhoI and NotI restriction sites of Exontrap vector pET01 (MoBiTec) to generate mini-genes that were validated by Sanger sequencing. The HEK293T cells were seeded in a 96-well plate at a cell density of 1.5 × 104 and transfected the next day with 100 ng of mini-genes using Lipofectamine 3000 transfection reagent (Invitrogen), in 4 biological replicates. Cells were harvested 48 h post-transfection, and total RNA was extracted with QIACube using RNeasy kit with on-column DNase I treatment (Qiagen). For each sample, 0.5–1 μg of total RNA was converted into cDNA with SuperScript III reverse transcriptase (Invitrogen) and a vector-specific primer: 5′-AGGGGTGGACAGGGTAGTG-3′. Samples were diluted with water, and cDNA corresponding to 1.5 ng of RNA input was used for each qRT–PCR reaction. Splicing products of each mini-gene were amplified using a common primer pair F: 5′-CACCTTTGTGGTTCTCACTTGG-3′ and R: 5′-AGCACTGATCCACGATGCC-3′, corresponding to vector exons V1 and V2 (Figs. 4, 5 and Supplementary Fig. 6). An assay with primers F: 5′-CCGTGACCTTCAGACCTTGG-3′ and R: 5′-AGAGAGCAGATGCTGGTGCA-3′ targeting Exontrap vector exon V2 was used as a control. All PCR-amplified splicing products were resolved by agarose gel electrophoresis. Specific bands were cut out from the gel, purified, and validated by Sanger sequencing. Co-transfection of E5 mini-gene with 10 SFs was performed in HEK293T cells for 48 h, followed by RNA extraction and analysis of splicing patterns. Similar analyses were performed for four select SFs (SRSF2, SRSF3, CELF1, and ELAVL2) in SW-780, HT-1376, and HTB-9 cells. The E5 construct was also transfected into a panel of 10 cell lines for 24 h, and splicing analysis was performed as described above.
Modulation of A3B exon 5 splicing
Cell lines (HT-1376, HTB-9, and HeLa) were grown in 12-well plates at a density of 1.5 × 105 cells/well and treated with DMSO or 1, 10, 20, 40, and 100 nM pladienolide B (Santa Cruz, sc-391691) reconstituted in DMSO, in biological triplicates per condition. Cells were harvested after 8, 16, 24, and 36 h of treatment separately for RNA and protein analysis. cDNA was synthesized from 300 ng of RNA in a 20 µl reaction using the RT2 First-Strand cDNA kit and random hexamers (Qiagen). cDNA corresponding to 30 ng of total RNA per reaction was used for PCR performed with a primer pair to amplify different splicing products of A3B: Forward primer on exon 4: 5′-GCCTTGGTACAAATTCGATGA-3′, Reverse primer on exon 6: 5′-TGTGTTCTCCTGAAGGAACG-3′. PCR-amplified splicing products, A3B1 (397 bp), A3B2 (322 bp), and A3B3 (243 bp), were resolved by agarose gel electrophoresis, and specific bands were cut out from the gel, purified, and validated by Sanger sequencing. Quantification of A3B1 and A3B3 splicing products was performed with qRT–PCR using isoform-specific TaqMan assays (Supplementary Note 4) and cDNA corresponding to 10 ng of total RNA per reaction, as previously described1. Expression of A3B1 and A3B3 was normalized by the expression of endogenous controls GAPDH (assay 4326317E) and PPIA (assay 4326316E). Water and genomic DNA were used as negative controls for all assays. Expression was measured as Ct values (PCR cycle at detection threshold) and calculated as ΔΔCt in relation to control (DMSO) groups of samples. For protein analysis, whole-cell extracts were harvested in RIPA buffer with proteinase inhibitor (ThermoFisher Scientific) and used for Western blotting as described in Supplementary Note 2. Shown are representative results of one of three independent experiments.
Cell-based cytosine deamination assay
To determine base substitution rates, we used an ssDNA oligo (Supplementary Fig. 9a), modified based on a previously reported oligo65. HT-1376 cells were nucleofected with 100 pmol of oligo alone or together with A3B1 plasmid (program CM-130) with SF Cell Line 4D X Kit L (V4XC-2024) on Lonza 4D-Nucleofector. Nucleofected cells were then plated in a six-well plate and after 8 h, cells transfected only with oligo were treated with DMSO or pladienolide B for 64 h. Cells were then lysed with QuickExtract DNA Extraction Solution (Lucigen) and PCR-amplified using primers: Forward, 5′-TGATGATGTGAGTGGTGGATGA-3′; Reverse, 5′- TCATCAACACCTACCACACAC-3′. PCR products were gel-purified with a DNA gel extraction kit (QIAquick Gel Extraction Kit, Qiagen) and used for library preparation with TruSeq/ChIP-Seq reagents (Illumina), to generate 75 bp paired-end sequencing reads. Samples were barcoded, multiplexed, and subjected to deep sequencing on the Illumina MiSeq instrument. The FASTQ files were aligned with custom reference (sequence of ssDNA oligo) using the BWA-MEM algorithm and then indexed by SAMtools.
siRNA knockdown of SF3B1
The HT-1376 cells were transfected with scrambled siRNA (#1022076), SF3B1 siRNA-1 (SI00715932), or siRNA-2 (SI04154647), all from Qiagen, using Lipofectamine RNAiMAX Reagent (ThermoFisher). Cells were harvested after 36 h for RNA and protein and analyzed for A3B exon 5 skipping by PCR as described above. SF3B1 knockdown was confirmed by Western blot with an anti-SF3B1 antibody (Abcam, ab172634, 1:1000 dilution) and GAPDH control, as described above.
Modulation of A3B exon 5 skipping by splice-switching oligonucleotides (SSOs)
We designed SSOs targeting two predicted BPS within intron 4 of A3B (at positions -38 and -50 bp) and the 3′ splice site (3′SS) of exon 5 (Fig. 7a). The SSOs were 2-OMe-substituted phosphorothioate RNA (Integrated DNA Technologies): BPS-50 (5-GACUCCUCCUGGGACUACCUCUC-3), BPS-38 (5-GAAACCAAAAGCCCUAAGACUCCUC-3), 3′SS (5-CAUCAGGUAUCUGUGGACAAAGAC-3), and negative control (5-AGUAGGAAAAGGGAUAUAUGAUGGAAAU-3). HT-1376 and HTB-9 cells were nucleofected with 2 nM of SSOs (program CM-130) using SF Cell Line 4D X Kit L (V4XC-2024) on Lonza 4D-Nucleofector. Nucleofected cells were then plated in a 12-well plate and harvested after 48 h for RNA extraction and analysis of A3B exon 5 skipping by PCR as described above.
Computational analysis
All data processing and analyses were performed using R package versions (3.2.4-3.4.0), SPSS version 25, and NIH High-Performance Computing Biowulf cluster.
URLs
Firehose Broad GDAC: https://gdac.broadinstitute.org/; Firebrowse: http://firebrowse.org/#; The Cancer Genome Atlas (TCGA): http://cancergenome.nih.gov; cBioPortal: http://www.cbioportal.org/index.do; Broad Institute Cancer Cell Line Encyclopedia (CCLE) https://portals.broadinstitute.org/ccle; Protein Data Bank (PDB): http://www.rcsb.org/pdb/home/home.do; Clustal Omega: (http://www.ebi.ac.uk/Tools/msa/clustalo/); SpliceAid2 (http://193.206.120.249/splicing_tissue.html); SFmap (http://sfmap.technion.ac.il/). Human Splicing Finder (HSF), www.umd.be/HSF3/; The R project for statistical computing: http://www.r-project.org/; Integrative Genomics Viewer (IGV): http://www.broadinstitute.org/igv; ASpli R package: https://bioconductor.org/packages/release/bioc/vignettes/ASpli/inst/doc/ASpli.pdf
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
The authors declare that data supporting the findings of this study are available from TCGA or within the paper and its supplementary information files. Source data for the main and supplementary figures are available in Supplementary Data 1–8. Unprocessed blot and gel images are provided in Supplementary Fig. 11. The RNA-seq dataset for T47D cells infected with SeV and uninfected cells has been deposited to NCBI Short Read Archive (SRA), accession number PRJNA512015. Additional information, protocols, and reagents can be provided on request to the corresponding author (L.P.-O.).
References
Middlebrooks, C. D. et al. Association of germline variants in the APOBEC3 region with cancer risk and enrichment with APOBEC-signature mutations in tumors. Nat. Genet. 48, 1330–1338 (2016).
Faltas, B. M. et al. Clonal evolution of chemotherapy-resistant urothelial carcinoma. Nat. Genet. 48, 1490–1499 (2016).
Law, E. K. et al. The DNA cytosine deaminase APOBEC3B promotes tamoxifen resistance in ER-positive breast cancer. Sci. Adv. 2, e1601737 (2016).
Roper, N. et al. APOBEC mutagenesis and copy-number alterations are drivers of proteogenomic tumor evolution and heterogeneity in metastatic thoracic tumors. Cell Rep. 26, 2651–2666 e6 (2019).
Shi, J. et al. Somatic genomics and clinical features of lung adenocarcinoma: a retrospective study. PLoS Med. 13, e1002162 (2016).
Miao, D. et al. Genomic correlates of response to immune checkpoint blockade in microsatellite-stable solid tumors. Nat. Genet. 50, 1271–1281 (2018).
Chan, K. et al. An APOBEC3A hypermutation signature is distinguishable from the signature of background mutagenesis by APOBEC3B in human cancers. Nat. Genet. 47, 1067–106 (2015).
Roberts, S. A. et al. An APOBEC cytidine deaminase mutagenesis pattern is widespread in human cancers. Nat. Genet. 45, 970–976 (2013).
Burns, M. B. et al. APOBEC3B is an enzymatic source of mutation in breast cancer (vol 494, 366, 2013). Nature 502, 580–580 (2013).
Starrett, G. J. et al. The DNA cytosine deaminase APOBEC3H haplotype I likely contributes to breast and lung cancer mutagenesis. Nat. Commun. 7, 12918 (2016).
Nik-Zainal, S. et al. Association of a germline copy number polymorphism of APOBEC3A and APOBEC3B with burden of putative APOBEC-dependent mutations in breast cancer. Nat. Genet. 46, 487–491 (2014).
Caval, V., Suspene, R., Shapira, M., Vartanian, J. P. & Wain-Hobson, S. A prevalent cancer susceptibility APOBEC3A hybrid allele bearing APOBEC3B 3’UTR enhances chromosomal DNA damage. Nat. Commun. 5, 5129 (2014).
Henderson, S., Chakravarthy, A., Su, X., Boshoff, C. & Fenton, T. R. APOBEC-mediated cytosine deamination links PIK3CA helical domain mutations to human papillomavirus-driven tumor development. Cell Rep. 7, 1833–1841 (2014).
Kanu, N. et al. DNA replication stress mediates APOBEC3 family mutagenesis in breast cancer. Genome Biol. 17, 185 (2016).
Mockenhaupt, S. & Makeyev, E. V. Non-coding functions of alternative pre-mRNA splicing in development. Semin. Cell Dev. Biol. 47-48, 32–39 (2015).
Harari, A., Ooms, M., Mulder, L. C. & Simon, V. Polymorphisms and splice variants influence the antiretroviral activity of human APOBEC3H. J. Virol. 83, 295–303 (2009).
Ebrahimi, D. et al. Genetic and mechanistic basis for APOBEC3H alternative splicing, retrovirus restriction, and counteraction by HIV-1 protease. Nat. Commun. 9, 4137 (2018).
Lassen, K. G., Wissing, S., Lobritz, M. A., Santiago, M. & Greene, W. C. Identification of two APOBEC3F splice variants displaying HIV-1 antiviral activity and contrasting sensitivity to Vif. J. Biol. Chem. 285, 29326–29335 (2010).
Bohn, M. F. et al. The ssDNA Mutator APOBEC3A is regulated by cooperative dimerization. Structure 23, 903–911 (2015).
Shi, K., Carpenter, M. A., Kurahashi, K., Harris, R. S. & Aihara, H. Crystal structure of the DNA deaminase APOBEC3B catalytic domain. J. Biol. Chem. 290, 28120–28130 (2015).
Nickless, A. et al. Intracellular calcium regulates nonsense-mediated mRNA decay. Nat. Med. 20, 961–966 (2014).
Pak, V., Heidecker, G., Pathak, V. K. & Derse, D. The role of amino-terminal sequences in cellular localization and antiviral activity of APOBEC3B. J. Virol. 85, 8538–8547 (2011).
Maiti, A. et al. Crystal structure of the catalytic domain of HIV-1 restriction factor APOBEC3G in complex with ssDNA. Nat. Commun. 9, 2460 (2018).
Stenglein, M. D., Matsuo, H. & Harris, R. S. Two regions within the amino-terminal half of APOBEC3G cooperate to determine cytoplasmic localization. J. Virol. 82, 9591–9599 (2008).
Burns, M. B., Temiz, N. A. & Harris, R. S. Evidence for APOBEC3B mutagenesis in multiple human cancers. Nat. Genet 45, 977–983 (2013).
Petljak, M. et al. Characterizing mutational signatures in human cancer cell lines reveals episodic APOBEC mutagenesis. Cell 176, 1282–1294 e20 (2019).
Hedegaard, J. et al. Comprehensive transcriptional analysis of early-stage urothelial carcinoma. Cancer Cell 30, 27–42 (2016).
Schafer, S. et al. Alternative splicing signatures in RNA-seq data: percent spliced in (PSI). Curr. Protoc. Hum. Genet. 87, 11 16 1–11 16 14 (2015).
Wang, D. W. et al. APOBEC3B interaction with PRC2 modulates microenvironment to promote HCC progression. Gut 68, 1846–1857 (2019).
Udquim, K. I., Zettelmeyer, C., Banday, A. R., Lin, S. H. & Prokunina-Olsson, L. APOBEC3B expression in breast cancer cell lines and tumors depends on the estrogen receptor status. Carcinogenesis 41, 1030–1037 (2020).
Lee, Y. & Rio, D. C. Mechanisms and regulation of alternative Pre-mRNA splicing. Annu. Rev. Biochem. 84, 291–323 (2015).
Teng, T. et al. Splicing modulators act at the branch point adenosine binding pocket defined by the PHF5A-SF3b complex. Nat. Commun. 8, 15522 (2017).
Webb, T. R., Joyner, A. S. & Potter, P. M. The development and application of small molecule modulators of SF3b as therapeutic agents for cancer. Drug Discov. Today 18, 43–49 (2013).
Vlachostergios, P. J. & Faltas, B. M. Treatment resistance in urothelial carcinoma: an evolutionary perspective. Nat. Rev. Clin. Oncol. 15, 495–509 (2018).
Havens, M. A. & Hastings, M. L. Splice-switching antisense oligonucleotides as therapeutic drugs. Nucleic Acids Res. 44, 6549–6563 (2016).
Kralovicova, J. et al. Optimal antisense target reducing INS intron 1 retention is adjacent to a parallel G quadruplex. Nucleic Acids Res. 42, 8161–8173 (2014).
Landry, S., Narvaiza, I., Linfesty, D. C. & Weitzman, M. D. APOBEC3A can activate the DNA damage response and cause cell-cycle arrest. Embo Rep. 12, 444–450 (2011).
Suspene, R. et al. Somatic hypermutation of human mitochondrial and nuclear DNA by APOBEC3 cytidine deaminases, a pathway for DNA catabolism. Proc. Natl Acad. Sci. USA 108, 4858–4863 (2011).
Aynaud, M. M. et al. Human tribbles 3 protects nuclear DNA from cytidine deamination by APOBEC3A. J. Biol. Chem. 287, 39182–39192 (2012).
Shinohara, M. et al. APOBEC3B can impair genomic stability by inducing base substitutions in genomic DNA in human cells. Sci. Rep. 2, 806 (2012).
Burns, M. B. et al. APOBEC3B is an enzymatic source of mutation in breast cancer. Nature 494, 366–370 (2013).
Cieply, B. & Carstens, R. P. Functional roles of alternative splicing factors in human disease. Wiley Interdiscip. Rev. RNA 6, 311–326 (2015).
Anczukow, O. & Krainer, A. R. Splicing-factor alterations in cancers. RNA 22, 1285–1301 (2016).
Li, Y. S. et al. Revealing the determinants of widespread alternative splicing perturbation in cancer. Cell Rep. 21, 798–812 (2017).
Martinez-Montiel, N., Anaya-Ruiz, M., Perez-Santos, M. & Martinez-Contreras, R. D. Alternative splicing in breast cancer and the potential development of therapeutic tools. Genes 8, 217 (2017).
Naro, C., Bielli, P., Pagliarini, V. & Sette, C. The interplay between DNA damage response and RNA processing: the unexpected role of splicing factors as gatekeepers of genome stability. Front. Genet. 6, 142 (2015).
Dutertre, M., Sanchez, G., Barbier, J., Corcos, L. & Auboeuf, D. The emerging role of pre-messenger RNA splicing in stress responses: sending alternative messages and silent messengers. RNA Biol. 8, 740–747 (2011).
Li, X. L. & Manley, J. L. Inactivation of the SR protein splicing factor ASF/SF2 results in genomic instability. Cell 122, 365–378 (2005).
Lee, S. C. & Abdel-Wahab, O. Therapeutic targeting of splicing in cancer. Nat. Med. 22, 976–986 (2016).
Lee, S. C. et al. Modulation of splicing catalysis for therapeutic targeting of leukemia with mutations in genes encoding spliceosomal proteins. Nat. Med. 22, 672–678 (2016).
Aird, D. et al. Sensitivity to splicing modulation of BCL2 family genes defines cancer therapeutic strategies for splicing modulators. Nat. Commun. 10, 137 (2019).
Seiler, M. et al. H3B-8800, an orally available small-molecule splicing modulator, induces lethality in spliceosome-mutant cancers. Nat. Med. 24, 497–49 (2018).
Seiler, M. et al. Somatic mutational landscape of splicing factor genes and their functional consequences across 33 cancer types. Cell Rep. 23, 282–296 e4 (2018).
Shi, M. J. et al. APOBEC-mediated mutagenesis as a likely cause of FGFR3 S249C mutation over-representation in bladder cancer. Eur. Urol. 76, 9–13 (2019).
Quemener, A. M. et al. The powerful world of antisense oligonucleotides: from bench to bedside. Wiley Interdiscip. Rev. RNA 11, e1594 (2020).
Roberts, S. A. & Gordenin, D. A. Hypermutation in human cancer genomes: footprints and mechanisms. Nat. Rev. Cancer 14, 786–800 (2014).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Sievers, F. et al. Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol. Syst. Biol. 7, 539 (2011).
Fu, Y. P. et al. Common genetic variants in the PSCA gene influence gene expression and bladder cancer risk. Proc. Natl Acad. Sci. USA 109, 4974–4979 (2012).
Nair, S. & Rein, A. In vitro assay for cytidine deaminase activity of APOBEC3 protein. Bio Protoc. 4, 820 (2014).
Mendell, J. T., Sharifi, N. A., Meyers, J. L., Martinez-Murillo, F. & Dietz, H. C. Nonsense surveillance regulates expression of diverse classes of mammalian transcripts and mutes genomic noise. Nat. Genet. 36, 1073–1078 (2004).
Desmet, F. O. et al. Human Splicing Finder: an online bioinformatics tool to predict splicing signals. Nucleic Acids Res. 37, e67 (2009).
Paz, I., Akerman, M., Dror, I., Kosti, I. & Mandel-Gutfreund, Y. SFmap: a web server for motif analysis and prediction of splicing factor binding sites. Nucleic Acids Res. 38, W281–W285 (2010).
Piva, F., Giulietti, M., Burini, A. B. & Principato, G. SpliceAid 2: a database of human splicing factors expression data and RNA target motifs. Hum. Mutat. 33, 81–85 (2012).
Lei, L. et al. APOBEC3 induces mutations during repair of CRISPR-Cas9-generated DNA breaks. Nat. Struct. Mol. Biol. 25, 45–52 (2018).
Acknowledgements
The results presented here are in part based upon data generated by the TCGA Research Network. The study was supported by the Intramural Research Program of the National Cancer Institute - Division of Cancer Epidemiology and Genetics (L.P.-O.), AIDS Targeted Antiviral Program, Center for Cancer Research (V.K.P.), and the NCI Innovation Award (R.B.). We thank the Cancer Genomics Research Laboratory of NCI for RNA-sequencing, Nathan Cole for help with retrieval of sliced RNA-seq BAM files from the NCI Data Commons portal, and Dr. Marc-Henri Stern, Institute Curie, Paris, for critical comments and suggestions.
Funding
Open Access funding provided by the National Institutes of Health (NIH).
Author information
Authors and Affiliations
Contributions
A.R.B. and L.P.-O. conceived and designed the study. L.P.-O. supervised the study. A.R.B., O.O.O., S.H.-Y.L., A.O., J.M.V., K.A.D.-F, A.B., and C.Z. performed the experiments and analyzed the data. A.R.B., P.L., and O.F.-V. analyzed the RNA-seq data and performed statistical analyses. A.R.B., O.O.O., V.K.P., L.D., and L.P.-O. contributed reagents/materials/analysis tools. A.R.B. and L.P.-O. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rouf Banday, A., Onabajo, O.O., Lin, S.HY. et al. Targeting natural splicing plasticity of APOBEC3B restricts its expression and mutagenic activity. Commun Biol 4, 386 (2021). https://doi.org/10.1038/s42003-021-01844-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-021-01844-5
This article is cited by
-
APOBEC3-mediated mutagenesis in cancer: causes, clinical significance and therapeutic potential
Journal of Hematology & Oncology (2023)
-
ILF2 enhances the DNA cytosine deaminase activity of tumor mutator APOBEC3B in multiple myeloma cells
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.