Abstract
Currently, a sample for small-angle scattering (SAS) is usually highly purified and looks monodispersed: The Guinier plot of its SAS intensity shows a fine straight line. However, it could include the slight aggregates which make the experimental SAS profile different from the monodispersed one. A concerted method with analytical-ultracentrifugation (AUC) and SAS, named as AUC-SAS, offers the precise scattering intensity of a concerned biomacromolecule in solution even with aggregates as well that of a complex under an association-dissociation equilibrium. AUC-SAS overcomes an aggregation problem which has been an obstacle for SAS analysis and, furthermore, has a potential to lead to a structural analysis for a general multi-component system.
Similar content being viewed by others
Introduction
Structural investigations of biomacromolecules and their complexes in solution are essential to understanding physiological phenomena in biological systems. Several analytical methods have been developed and/or improved to address the investigations of such systems. Small-angle X-ray and neutron scatterings (SAXS or SANS) are classical techniques that give size information of particles in solution1. In addition, improved modern SAS gives further structural information: three-dimensional structure and structural fluctuation by combining scattering data with computational analyses, such as ab initio modeling1,2 and molecular dynamics3,4,5, respectively. These analyses require high-quality scattering data, i.e. the data purely from the target biomacromolecule.
There is an intrinsic obstacle to satisfy this requirement. Because SAS offers an ensemble-averaged scattering intensity of all particles in solution, unspecified aggregates in a solution (Supplementary Fig. 1a) will pollute the scattering intensity—especially the aggregates make abnormally upturn on the scattering intensity in the lower scattering angles (Supplementary Fig. 1b, c). In addition, a hidden problem has appeared6,7. We usually prepare for a purified sample in a structural analysis of a single molecule. However, even though the measured scattering intensity does not show such abnormal upturn and holds Guinier approximation6, it could include a small amount of aggregates and they should be removed to obtain correct structure parameters7. In other words, it is difficult to judge that there still remains a small amount of aggregates in a solution only with SAS: A highly purified sample (Supplementary Fig. 2) looks nicely holds Guinier approximation but it included a small amount of aggregates. Practically, “revealing and removal of unspecified aggregates” has been one of the most significant challenges for SAS in many years.
The breakthrough for the “removal” is the development of size exclusion chromatography SAXS (SEC-SAXS)7,8,9 which directly observes scattering of a size-separated particle solution eluting from a SEC-column. However, even with SEC-SAXS, we still have problems: demand of a relatively large amount of sample (>2 mg for a typical case), quick re-aggregation, and destruction of a weakly bound complex.
Analytical ultracentrifugation (AUC)10 is an interesting technique for the “revealing” because it provides the concentration distribution of particles in solution with a small amount of sample (~0.05 mg) and is less destructive to complexes. Therefore, several previous studies used AUC to check the quality of SAS samples11,12,13. On the other hand, we have conceived in the advance to utilize AUC for the “removal”: The SAS intensity of multi-component solution can be decomposed into those of components by utilizing the information of their concentration distribution obtained with AUC. In this way, the SAS intensity of a certain component, such as a monomer in solution, can be extracted from that of the whole solution even though the solution includes the unspecified aggregates.
By integral use of AUC and SAS, we have succeeded in extracting the SAS intensity of a specific monomer in a solution with also contained its aggregates. Furthermore, we have applied the method to obtain the SAS intensity of a weakly bound complex under an association–dissociation equilibrium. In this paper, we report on this newly developed “AUC-SAS” method.
Results
Extraction of monomer scattering (removing aggregation)
A scattering intensity Imul(q) (q: magnitude of scattering vector) of a multi-component system is represented by
where c, imul(q), and n are total mass concentration, scattering intensity per c, and number of components, and cj and ij(q) are mass concentration and scattering intensity per cj for the jth component, respectively. Here, because imul(q) is provided with SAS experiment as \(i_{{\mathrm{exp}}}\left( q \right)( = I_{{\mathrm{exp}}}\left( q \right)/c)\) to solve a scattering intensity of a concerned component k, ik(q), the followings are required; “number of components, n”, “concentrations of all components, {cj}”, and “scattering intensity(s) except for the concerned component, ij(q) (j ≠ k)”. AUC gives n and {cj}. Therefore, the remains are ij(q) (j ≠ k).
Firstly, our purpose is confined to extract the scattering intensity of the monomer (k = 1) from the scattering of the whole solution including unspecified aggregates (j ≥ 2). Accordingly, the task is to figure out the scattering intensities of unspecified aggregates ij(q) (j ≥ 2). In general, it is difficult to know these scattering intensities, individually. Here, we notice that samples for a general SAS are highly purified, for which the following three conditions hold in most cases. Condition (i): Highly denatured contaminates have already been removed by purification. The remaining aggregates are simple oligomers with the low aggregation degree (2 ≤ j ≤ 4 at most) and their total weight fraction is less than ca 10%. Condition (ii): The inner structures of aggregates are identical with that of the monomer because the aggregates are assumed to be simple homo-oligomers, such as assembly of neat monomers. Condition (iii): Guinier approximation is established for experimental scattering intensity ciexp(q). This is the dangerous point because this could mislead us that the solution is monodispersed as described in “Introduction”.
Under these conditions, we have developed a method, which extracts i1(q) from iexp(q) including the unspecified aggregates. Here, we explain the developed five-steps protocol shown in Fig. 1, taking a purified bovine serum albumin (BSA) solution as an example (see “Methods” and Supplementary Fig. 2).
For aggregation system, in Steps 3 and 4, the contribution of monomer in the scattering intensity is estimated because that of aggregation is not measurable directly. On the other hand, if all ij(q) (j ≠ k) are obtained by individual measurements, such as an association–dissociation equilibrium system, Steps 3 and 4 are skipped.
Step 1: SAS measurement
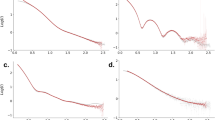
Scattering intensity of multi-component system cimul(q) is measured as ciexp(q) (open black circles in Fig. 2a). The mean gyration radius Rge and forward scattering intensity ciexp(0) are calculated with Guinier analysis (black circles and line in Fig. 2b and non-treated SAXS in Supplementary Table 1). If the scattering intensity deviates from the Guinier approximation (Supplementary Fig. 1b, c) or the obtained Rge is abnormally large, further purification should be applied.
a SAXS intensities and b Guinier plots for the BSA1 solution. Black, blue, and red circles indicate \(ci_{{\mathrm{exp}}}\left( q \right)\) (non-treated SAXS), \(c_1i_1\left( q \right)\) and \(c_{\mathrm{a}}i_{\mathrm{a}}\left( q \right)\) obtained with AUC-SAS as the final result, respectively. Solid lines represent the least-square fitting with Guinier formula. Inset shows an ab initio model based on i1(q).
Step 2: AUC measurement
Sedimentation velocity-AUC (SV-AUC)14 is conducted for the same solution subjected to the SAS measurement. The measured sedimentation coefficient distribution c(s20,w) offers n and {cj} (Fig. 3, Supplementary Table 2).
Step 3: Forward scattering intensity of monomer, i1(0)
The forward scattering intensity ratio t1 of monomer \(c_1i_1(0)\) to the whole forward scattering cimul(0) is calculated with AUC-measured {cj} as follows (Supplementary Note 1):
The i1(0) is calculated as \(t_1ci_{{\mathrm{mul}}}(0)/c_1 = t_1ci_{{\mathrm{exp}}}(0)/c_1\): cimul(0) is provided as ciexp(0) with a SAS experiment for a multi-component system as described before. Here, ciexp(0) and \(c_1i_1(0)\) are black and blue squares in Fig. 4, respectively, clearly indicating that a few % of aggregates (5.9% in the example, BSA1) generates the excess scattering \(c_{\mathrm{a}}i_{\mathrm{a}}(0)\) (\(= \mathop {\sum}\nolimits_{j = 2}^4 {c_j} i_j\left( 0 \right)\) in the example) (red bar in Fig. 4) in the observed whole ciexp(0).
Closed black and blue squares represent \(ci_{{\mathrm{exp}}}(0)\) and \(c_1i_1\left( 0 \right)\), respectively. The bar graph expresses the contributions of monomer \(c_1i_1\left( 0 \right)\) (blue part) and aggregates \(c_ai_a\left( 0 \right)\) (red part) in \(ci_{{\mathrm{exp}}}\left( 0 \right)\).
Step 4: Scattering of monomer in the high q, ilh(qh)
It is assumed that the aggregates are simple associated homo-oligomers (condition (ii)). In this case, an inner structure of the aggregates is same as a monomer. Therefore, the scattering intensity of monomer in the high q-range, \(i_{1{\mathrm{h}}}\left( {q_{\mathrm{h}}} \right)\), is almost identical to that of oligomer \(i_{j{\mathrm{h}}}\left( {q_{\mathrm{h}}} \right)(j \ge 2)\).
It should be considered that the difference between ilh(qh) and imul(qh) arises toward lower q-range. Here, the consideration is briefly described as follows (Supplementary Figs. 3 and 4, and Supplementary Note 2 in detail). To estimate the lowest q*, where Eq. (3) holds, and to calculate the maximum difference at q*, the intensity ratio r(q) of whole scattering intensity to that of monomer is introduced as follows:
Here, two simple models for oligomers are introduced: linearly aligned and closed packing oligomerization models (j ≤ 4; Supplementary Fig. 3). As the first assumption, the orientation of monomers in the oligomers is averaged and then their scattering intensities \(i_j\left( q \right)(j \ge 2)\) (Eqs. (S7)–(S12) in Supplementary Note 2-1) are calculated based on Debye function with monomer scattering intensity i1(q) (Eqs. (S6) in Supplementary Note 2-1).
As shown in Supplementary Fig. 4, r(q) with ij(q) and {cj} for the present example (BSA1) showed rapidly asymptotical approach to unity, and the deviation from unity is less than 1.8% in \(q^ \ast R_{{\mathrm{g}}1} \ge 1.0\), where Rg1 is the gyration radius of the monomer. Therefore, in the case with a few % of aggregates (5.9% in the example, BSA1), it is approximated to be \(i_{1{\mathrm{h}}}\left( {q_{\mathrm{h}}} \right) \approx i_{{\mathrm{mul}}}\left( {q_{\mathrm{h}}} \right) = i_{{\mathrm{exp}}}\left( {q_{\mathrm{h}}} \right)\) within the maximum difference of 1.8% in \(q_{\mathrm{h}} \, > \,q^ \ast = 1.0/R_{{\mathrm{g}}1}\) (Fig. 5a). Accordingly, in the present AUC-SAS protocol, the initial (tentative) scattering intensity of monomer in the high q-range ilh(qh)* is set to be iexp (qh) (qh > q*): The closed blue circles in Fig. 5b represents \(c_1i_{1{\mathrm{h}}}\left( {q_{\mathrm{h}}} \right)^ \ast\) and open blue circles do extrapolation of \(c_1i_{1{\mathrm{h}}}\left( q \right)^ \ast\) in the lower q-range (q < q*).
a Scattering intensity ratios r(q) at Rg1 = 27.2 Å with the {cj} for the present example (BSA1) (green: linearly aligned model and red: closed packing model: Supplementary Note 2). b Scattering intensities \(ci_{{\mathrm{exp}}}\left( q \right)\) (open black circles), c1i1h(qh)* (closed blue circles), and extrapolation of \(c_1i_{1{\mathrm{h}}}\left( q \right)\ast\) in the lower q-range (open blue circles).
Step 5A: Extraction of scattering intensity of monomer
To obtain the scattering intensity of monomer in whole q-range, it is necessary to find the scattering intensity in low q-range i1l(ql) filling the gap between i1(0) calculated in Step 3 and \(i_{1{\mathrm{h}}}\left( {q_{\mathrm{h}}} \right) ^\ast ( = i_{{\mathrm{exp}}}(q_{\mathrm{h}}))\) set in Step 4.
5A-1. Setting the initial scattering intensity, i1(q)*:
In general, i1l(q) should satisfy Guinier approximation, holding the y-intercept of i1(0) (\(= t_1ci_{{\mathrm{mul}}}(0)/c_1\)). Therefore, the initial scattering intensity in low q-range \(i_{1{\mathrm{l}}}\left( {q_{\mathrm{l}}} \right) ^\ast\) is semi-empirically set as follows:
Here, the initial gyration radius of monomer \(R_{{\mathrm{g}}1}^ \ast\) is chosen for making a smooth connection of \(i_{1{\mathrm{l}}}\left( {q_{\mathrm{l}}} \right) ^\ast\) to \(i_{1{\mathrm{h}}}( {q_{\mathrm{h}}} ) ^\ast\) at the connection point qc: \({\mathrm{d}}{\mathrm{ln}}( {i_{1{\mathrm{l}}}( {q_{\mathrm{c}}} ) ^\ast } )/{\mathrm{d}}q^2 = {\mathrm{d}}{\mathrm{ln}}( {i_{1{\mathrm{h}}}( {q_{\mathrm{c}}} ) ^\ast } )/{\mathrm{d}}q^2\) (Supplementary Note 3-1). Then, the initial whole scattering intensity of monomer \(i_1\left( q \right)^ \ast\) is provided with \(i_{1{\mathrm{l}}}\left( {q_{\mathrm{l}}} \right) ^\ast (q_{\mathrm{l}} \, < \, q_{\mathrm{c}})\) and \(i_{1{\mathrm{h}}}\left( {q_{\mathrm{h}}} \right) ^\ast (q_{\mathrm{h}} \ge q_{\mathrm{c}})\) (Supplementary Fig. 5).
5A-2. Refinement of the whole scattering intensity, \({\cal{R}}(i_1(q)^ \ast )\)):
There is a possibility that the initial whole scattering intensity \(i_1\left( q \right) ^\ast\) involves errors caused from the semi-empirically obtained \(i_{1{\mathrm{h}}}\left( {q_{\mathrm{h}}} \right) ^\ast\) and i1l(q1)*. In order to refine \(i_1\left( q \right) ^\ast\), we utilized the expanded Guinier formula which holds to a relatively higher q-range: The expanded Guinier formula is prepared with the polynomial expansion of Debye formula15 (Supplementary Figs. 6 and 7, Supplementary Notes 3-2 and 3-3). The final result i1(q), the full scattering profile of monomer extracted by AUC-SAS, is shown with closed blue circles in Fig. 2a and Supplementary Fig. 8. The structural parameters extracted by the above procedure completely agree with those from SEC-SAXS as listed in Supplementary Table 1, and the derived three-dimension ab initio model2 well reproduced the crystal structure (Fig. 2a). Furthermore, AUC-SAS also succeeded to extract the scatting intensities of target protein in the other samples (ovalbumin and apoferritin: Supplementary Figs. 9 and 10, Supplementary Tables 1 and 2).
The AUC-SAS has advantages over the SEC-SAXS to obtain the monomer scattering intensity from solution including aggregates. A required sample amount for AUC-SAS is 0.1–0.25 mg of proteins (1–3 mg/mL in 30 μL for SAXS and 50 μL for SV-AUC) whereas that even for the recent high performance SEC-SAXS (https://www-ssrl.slac.stanford.edu/smb-saxs/content/documentation/sec-saxs/introduction) is 0.2–0.5 mg of proteins (4–10 mg/mL in 50 μL). Furthermore, AUC-SAS has better resolution for molecular separation than SEC and does not make it problem that the monomers quickly re-assemble after the separation with SEC. However, there is also limitation to provide correct result with the present AUC-SAS protocol about the upper concentration of aggregates around 12%. This is mentioned in Supplementary Note 4 (Supplementary Figs. 11–13 and Supplementary Table 3).
Extraction of complex scattering
AUC-SAS can extract the scattering intensity of a complex under an association–dissociation equilibrium. It should be noted that SEC-SAXS is unavailable to observe a weakly bound complex due to destruction of it by the SEC process (Supplementary Fig. 14). To the contrary, AUC has an ability to reveal concentration distribution of all components under an association–dissociation equilibrium even though the complex is weakly bounded.
Considering the equilibrium system of A + B ↔ AB, Eq. (1) is explicitly rewritten as
AUC-SAS protocol is slightly modified to extract iAB(q) from Iexp(q). Here, iA(q) and iB(q) are known with their individual SAXS measurements (Step 1) and then Steps 3 and 4 are skipped. Therefore, the key subject is to know the accurate concentrations for all components with AUC (Step 2) and combined analysis (Step 5B). Here, the modified protocol is demonstrated with an association–dissociation equilibrium system of hHR23b-UBL (component A) and PNGase-PUB (component B), which makes a weakly bounded complex16.
Step 1: SAS measurement
iexp(q), iA(q), and iB(q) are individually measured with SAXS (Fig. 6a, Supplementary Fig. 15, and Supplementary Table 4).
a SAXS intensities for the hHR23b-UBL and PNGase-PUB system. Black, pink, green, and blue circles represent \(ci_{{\mathrm{exp}}}\left( q \right)\) of the experimental SAXS intensity for the mixture solution, \(c_{\mathrm{A}}i_{\mathrm{A}}\left( q \right)\) for hHR23b-UBL, \(c_{\mathrm{B}}i_{\mathrm{B}}\left( q \right)\) for PNGase-PUB by the individual experiments, and \(c_{{\mathrm{AB}}}i_{{\mathrm{AB}}}\left( q \right)\) for the complex obtained with AUC-SAS as the final result, respectively. Inset shows the refined structure with normal mode analysis (https://files.inria.fr/NanoDFiles/Website/Software/Pepsi-SAXS/MacOS/Pepsi-SAXS-NMA)22 (Supplementary Note 7). b Representative sedimentation equilibrium curves by SE-AUC at [hHR23b-UBL] = [PNGase-PUB] = 100 μM. Closed black circles, open black squares, and open gray circles represent the result at 20,000, 30,000, and 35,000 r.p.m., respectively. Red lines show the fitting curves (see “Methods”).
Step 2: AUC measurement
SV-AUC cannot provide the concentrations of all components (cA, cB, and cAB) for the system under the fast association–dissociation process (Supplementary Fig. 16, Supplementary Note 5). Therefore, the concentrations of all components should be calculated with the dissociation constant KD by measured with the sedimentation equilibrium-AUC method (SE-AUC). Here, KD for the demonstration system were measured at three concentrations and with three rotation speeds by SE-AUC and cA, cB, and cAB were obtained (Fig. 6b, Supplementary Fig. 17, Supplementary Table 5, and Supplementary Note 6). Here, it is important that the absence of aggregates should be confirmed with SV-AUC (Supplementary Note 5) prior to SE-AUC measurements.
Step 5B: Extraction of scattering intensity of complex
The scattering intensity of complex iAB(q) (blue closed circles in Fig. 6a) is obtained from Eq. (6) with the intensities and concentrations obtained in Steps 1 and 2. The extracted scattering intensity was subjected to three-dimension modeling as well as the size analysis (Supplementary Fig. 18, Supplementary Table 4, Supplementary Note 7). This is the first report of the detailed structural information of this complex in solution.
Discussion
Recently, SAS is required to study biomacromolecular structures in more complicated multi-component solution, for example, an association–dissociation equilibrium system involving aggregates. As an advanced example for demonstrating this technique, the nucleosome was measured with AUC-SAS (Supplementary Note 8). The sample was highly purified but still included small amounts of aggregates, liberated histone complex, and DNA (Supplementary Fig. 19a, Supplementary Table 6). Therefore, both techniques, removing aggregation and complex scattering extraction, were required to find the precise scattering intensity of the nucleosome in solution. As shown in Supplementary Fig. 19b, c and Supplementary Table 7, AUC-SAS succeeded to provide the scattering intensity of nucleosome and, using it, the 3D-structure was reconstructed (Supplementary Fig. 20).
For many years, aggregation has been a real difficult obstacle in bio-SAS. AUC-SAS overcomes this aggregation problem and offers a precise scattering intensity of a concerned biomacromolecule. In these days, the structural analysis of oligomers17 in a multi-component solution attracts attention as well as that of monomer. For such cases, AUC-SAS is applicable if all scattering intensities except for the concerned component are individually available: the demonstration of AUC-SAS for nucleosome could be one of the examples (Supplementary Note 8). Furthermore, AUC-SAS also embraces structural analysis of a weakly bounded complex. In conclusion, AUC-SAS has a potential to become one of the standard methods to analyze structures of biomacromolecules in solution as shown in Supplementary Fig. 20.
Finally, we would like to remak the following. AUC-SAS does not require the very high-intensity beam for a sample-flow experiment such as SEC-SAXS. Therefore, AUC-SAS has a potential to be a complementary method for a laboratory-based SAXS and a standard SANS to synchrotron-based SEC-SAXS for structural analysis of biomacromolecules in solution.
Methods
Samples
BSA (product# A2153), ovalbumin (OVA; product# A5503), and apoferritin (AF; product# A3641) purchased from Sigma Aldrich Co. were dissolved in 100 mM Tris/HCl (pH 7.5) buffer containing 100 mM NaCl. The solutions were purified by the anion-exchange chromatography with Resource Q 6 mL column (GE Healthcare) followed by the size exclusion chromatography with Superdex 200 increase 10/300GL column (GE Healthcare). The mass concentrations subjected to SAXS and AUC measurements were 2.29 mg/mL for BSA1 (used for demonstration of AUC-SAS protocol in the main text), 3.15 mg/mL for BSA2-4 (used for concentration boundary check in Supplementary Note 4), 2.00 mg/mL for OVA, and 1.81 mg/mL for AF, respectively. A quality of BSA1 was checked with SDS-PAGE. As shown in Supplementary Fig. 2a, no clear aggregation and contamination were observed in the sample.
The mixture solution of ubiquitin-like domain of the proteasome shuttle factor hHR23b (hHR23b-UBL; PDB code, 1P1A) and PUB domain of peptide:N-glycanase (PNGase-PUB; PDB code, 2D5U) were utilized as a demonstrated system which forms a weakly bounded complex under an association–disassociation equilibrium. The molecular weights of hHR23b-UBL and PNGase-PUB are 9.5 and 12.5 kDa, respectively. The expression and purification of the proteins have described previously16. The proteins were dissolved and dialyzed in 10 mM sodium phosphate buffer (pH7.0) (for all experiments) and PBS (only for SV-AUC). SE-AUC was carried out at three concentrations ([hHR23b-UBL] = [PNGase-PUB] = 100, 75, and 50 μM) and three rotation speeds (20,000, 30,000, and 35,000 r.p.m.). SAXS measurements are carried out at [hHR23b-UBL] = 200 μM, [PNGase-PUB] = 200 μM, and [hHR23b-UBL] = [PNGase-PUB] = 100 μM for mixture, where the intermolecular interference effect can be negligible on the scattering profile.
The nucleosome was prepared as the same manner in the previous report18. The sample was dialyzed against 20 mM Tris/HCl (pH7.5) buffer containing 50 mM NaCl and 1 mM DTT. The mass concentrations subjected to SAXS and AUC measurements were 1.29 mg/mL.
Small-angle X-ray scattering
All SAXS measurements were carried out with a laboratory-based instrument NANOPIX (Rigaku) equipped with high-brilliance point-focused generator of a Cu-Kα source (MicroMAX-007 HFMR, wavelength (λ) = 1.54 Å). The scattered X-rays were detected using a two-dimensional semiconductor detector (HyPix-6000) with the spatial resolution of 100 μm. The sample-to-detector-distances were set to be 1280 mm (covered q-range: 0.010–0.20 Å−1) for BSA, OVA, AF, and nucleosome experiments, and 355 mm (covered q-range: 0.030–0.70 Å−1) for PNGase-PUB + hHR23b-UBL and nucleosome experiments, respectively. Two-dimensional scattering pattern was converted to a one-dimensional scattering intensity with SAngler software19. After the correction by the transmittance and subtraction by the buffer scattering, the absolute scaled scattering intensity was obtained by referring to a standard scattering intensity of water (1.632 × 10−2 cm−1)20. All measurements were conducted at 25 °C.
Analytical ultracentrifugation
AUC measurements were conducted with a ProteomeLab XL-I analytical-ultracentrifuge (Beckman Coulter). The cell with a small volume (optical path: 1.5 mm, volume: 50 μL, Nanolytics) was used for the measurements. Two measuring methods, sedimentation velocity-AUC (SV-AUC) and sedimentation equilibrium-AUC (SE-AUC), were conducted depending upon the sample situations. The former measures sedimentation speed of particles and gives sedimentation coefficient distribution c(s20,w). The later observes a concentration gradient under sedimentation equilibrium, which provides a dissociation constant KD for an association–dissociation system. The sample solutions were loaded at 50 μL for SV-AUC and 20 μL for SE-AUC. SV-AUC was performed using Rayleigh interference optics at 40,000 r.p.m. of rotor speed for BSA, OVA, AF, and nucleosome, and at 60,000 r.p.m. of rotor speed for hHR23b-UBL, PNGase-PUB, and their mixture, respectively. SE-AUC was carried out using absorbance optics at 20,000, 30,000, and 35,000 r.p.m. for PNGase-PUB and hHR23b-UBL. All measurements were conducted at 25 °C. The SV-AUC data were analyzed with SEDFIT (http://www.analyticalultracentrifugation.com/sedfit.htm) software which executed the fitting with Lamm formula14. The sedimentation coefficient was converted to the value at 20 °C in pure water (s20,w). The molecular weight for each component was calculated using the peak s20,w and friction ratio fr. The weight fraction cj for each component was obtained from the peak area. The SE-AUC data were analyzed with SEDPHAT (http://www.analyticalultracentrifugation.com/sedphat/default.htm) software, which conducts the fitting to the SE-AUC results with the association–dissociation equilibrium model as follows21:
where a(r, KD) is an absorbance at radius r for the equilibrium system with the dissociation constant KD, ε is the extinction coefficient, c(r0) is the concentration at the reference radius r0, ω is the angular velocity, M is the molecular weight, \(\bar v\) is the partial specific volumes, ρ is the solvent density, and RT is the multiplication of the gas constant and absolute temperature. For accurate determination of the free parameters, we carried out the global fitting analysis for the three different concentrations and three different rotation speeds with same KD. For the analysis, the partial specific volumes (\(\bar v\)) of each protein were calculated from their amino acid sequences with SEDNTERP (http://www.jphilo.mailway.com/download.htm) software. The density and viscosity of solvents were measured with the density meter DMA4500M (Anton Paar) and the viscometer Lovis 2000 M/ME (Anton Paar), respectively.
Statistics and reproducibility
The fittings for “Guinier formula” to derive i(0) and Rg were performed with the linear least-square method by Igor Pro (7.04) and the fitting for SE-AUC to derive KD was done with the non-linear least-square method (Levenberg–Marquardt algorithm) also by Igor Pro (7.04). The errors were calculated considering the error propagation theory with the χ2, which are listed in Supplementary Tables 1, 3, 4 and 7.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Code availability
All software used in this study is publicly available at the URLs below.
Igor Pro 7.04: https://www.wavemetrics.net/previous7.html
SAngler 2.0.0: http://pfwww.kek.jp/saxs/SAngler.html
SEDFIT 15.01c: http://www.analyticalultracentrifugation.com/sedfit.htm.
SEDPHAT 14.0: http://www.analyticalultracentrifugation.com/sedphat/default.htm.
SEDNTERP 1.10: http://www.jphilo.mailway.com/download.htm.
Pepsi-SAXS-NMA: https://files.inria.fr/NanoDFiles/Website/Software/Pepsi-SAXS/MacOS/Pepsi-SAXS-NMA
References
Walther, D., Cohen, F. E. & Doniach, S. Reconstruction of low-resolution three-dimensional density maps from one-dimensional small-angle X-ray solution scattering data for biomolecules. J. Appl. Crystallogr. 33, 350–363 (2000).
Svergun, D. I. Restoring low resolution structure of biological macromolecules from solution scattering using simulated annealing. Biophys. J. 76, 2879–2886 (1999).
Oroguchi, T., Hashimoto, H., Shimizu, T., Sato, M. & Ikeguchi, M. Intrinsic dynamics of restriction endonuclease EcoO109I studied by molecular dynamics simulations and X-ray scattering data analysis. Biophys. J. 96, 2808–2822 (2009).
Saikusa, K. et al. Characterisation of an intrinsically disordered protein complex of Swi5–Sfr1 by ion mobility mass spectrometry and small-angle X-ray scattering. Analyst 138, 1441–1449 (2013).
Anami, Y. et al. Apo-and antagonist-binding structures of vitamin D receptor ligand-binding domain revealed by hybrid approach combining small-angle X-ray scattering and molecular dynamics. J. Med. Chem. 59, 7888–7900 (2016).
Mylonas, E. & Svergun, D. I. Accuracy of molecular mass determination of proteins in solution by small-angle X-ray scattering. Appl. Crystallogr. 40, 245–249 (2007).
Ryan, T. M. et al. An optimized SEC-SAXS system enabling high X-ray dose for rapid SAXS assessment with correlated UV measurements for biomolecular structure analysis. J. Appl. Crystallogr. 51, 97–111 (2018).
David, G. & Pérez, J. Combined sampler robot and high-performance liquid chromatography: a fully automated system for biological small-angle X-ray scattering experiments at the Synchrotron SOLEIL SWING beamline. J. Appl. Crystallogr. 42, 892–900 (2009).
Inoue, R. et al. Newly developed laboratory-based size exclusion chromatography small-angle x-ray scattering system (La-SSS). Sci. Rep. 9, 12610 (2019).
Lebowitz, J., Lewis, M. S. & Schuck, P. Modern analytical ultracentrifugation in protein science: a tutorial review. Protein Sci. 11, 2067–2079 (2002).
Vivan, A. L. et al. Structural studies of prephenate dehydratase from Mycobacterium tuberculosis H37Rv by SAXS, ultracentrifugation, and computational analysis. Proteins: Struct., Funct., Bioinforma. 72, 1352–1362 (2008).
Goertz, V., Dingenouts, N. & Nirschl, H. Comparison of nanometric particle size distributions as determined by SAXS, TEM and analytical ultracentrifuge. Part. Part. Syst. Charact. 26, 17–24 (2009).
Patel, T. R., Chojnowski, G., Koul, A., McKenna, S. A. & Bujnicki, J. M. Structural studies of RNA-protein complexes: a hybrid approach involving hydrodynamics, scattering, and computational methods. Methods 118, 146–162 (2017).
Schuck, P. Size-distribution analysis of macromolecules by sedimentation velocity ultracentrifugation and lamm equation modeling. Biophys. J. 78, 1606–1619 (2000).
Feigin, L. & Svergun, D. Structure Analysis by Small-Angle X-Ray and Neutron Scattering (Plenum Press, New York, 1987).
Kamiya, Y. et al. NMR characterization of the interaction between the PUB domain of peptide: N-glycanase and ubiquitin-like domain of HR23. FEBS Lett. 586, 1141–1146 (2012).
Marchenkova, M. A. et al. Dodecamers derived from the crystal structure were found in the pre-crystallization solution of the transaminase from the thermophilic bacterium Thermobaculum terrenum by small-angle X-ray scattering. J. Biomol. Struct. Dyn. 37, 3058–3064 (2019).
Arimura, Y. et al. Structural basis of a nucleosome containing histone H2A. B/H2A. Bbd that transiently associates with reorganized chromatin. Sci. Rep. 3, 3510 (2013).
Shimizu, N. et al. Software development for analysis of small-angle X-ray scattering data. AIP Conf. Proc. 1741, 050017 (AIP Publishing LLC, 2016).
Orthaber, D., Bergmann, A. & Glatter, O. SAXS experiments on absolute scale with Kratky systems using water as a secondary standard. J. Appl. Crystallogr. 33, 218–225 (2000).
Schuck, P. Analytical ultracentrifugation as a tool for studying protein interactions. Biophys. Rev. 5, 159–171 (2013).
Grudinin, S., Garkavenko, M. & Kazennov, A. Pepsi-SAXS: an adaptive method for rapid and accurate computation of small-angle X-ray scattering profiles. Acta Crystallogr. Sect. D 73, 449–464 (2017).
Acknowledgements
We especially wish to thank to Prof. Susumu Uchiyama (Osaka University) for suggestive and fruitful comments on the analysis of AUC and also would like to thank to Dr. Soji Murayama (Beckman Coulter) for kind help for instruction of AUC. We would like to expree our appliciation to Prof. Martin E. Vigild (Thechical University of Denmark) for his comments on SAXS data analysis. We also thank Ms. Yukiko Isono (IMS/ExCELLS) for protein purification. Preliminary SAXS experiments at Photon Factory were performed under Proposal No. 2017G100 and 2018G680. This work was supported by MEXT/JSPS KAKENHI Grant Numbers (JP19K16088 to K.M., JP17K07361, JP19KK0071, and JP20K06579 to R.I., JP17K07816 to N.S., JP18H05229, JP18H05534, and JP18H03681 to M.S.) and by the Future Development Funding Program ofKyoto University Research Coordination Alliance, Research Fund for Young Scientists in Kyoto University and Fund for Project Research in Institute for Integrated Radiation and Nuclear Science, Kyoto University (KURNS). This work was also partially supported by the project for Construction of the basis for the advanced materials science and analytical study by the innovative use of quantum beam and nuclear sciences in KURNS and by Joint Research by IMS (IMS program No. 205) and by the Platform Project for Supporting Drug Discovery and Life Science Research (Basis for Supporting Innovative Drug Discovery and Life Science Research (BINDS)) from AMED under Grant Number (JP20am0101076 to H.K.).
Author information
Authors and Affiliations
Contributions
K.M., A.O., Y.M., R.I., N.S., R.U., and M.S. performed SAXS measurements and analysis. K.M., Y.M., and M.S. performed AUC measurements and analysis. M.Y.-U. and K.K. prepared hHR23b-UBL and PNGase-PUB. R.H, T.K., and H.K. prepared H2A nucleosome. K.M. and M.S. designed research and all the authors wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Morishima, K., Okuda, A., Inoue, R. et al. Integral approach to biomacromolecular structure by analytical-ultracentrifugation and small-angle scattering. Commun Biol 3, 294 (2020). https://doi.org/10.1038/s42003-020-1011-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-020-1011-4
This article is cited by
-
Genome-wide mapping and cryo-EM structural analyses of the overlapping tri-nucleosome composed of hexasome-hexasome-octasome moieties
Communications Biology (2024)
-
Overall structure of fully assembled cyanobacterial KaiABC circadian clock complex by an integrated experimental-computational approach
Communications Biology (2022)
-
Solution structure of multi-domain protein ER-60 studied by aggregation-free SAXS and coarse-grained-MD simulation
Scientific Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.