Abstract
Appendicular lean mass (ALM) is a heritable trait associated with loss of lean muscle mass and strength, or sarcopenia, but its genetic determinants are largely unknown. Here we conducted a genome-wide association study (GWAS) with 450,243 UK Biobank participants to uncover its genetic architecture. A total of 1059 conditionally independent variants from 799 loci were identified at the genome-wide significance level (p < 5 × 10−9), all of which were also significant at p < 5 × 10–5 in both sexes. These variants explained ~15.5% of the phenotypic variance, accounting for more than one quarter of the total ~50% GWAS-attributable heritability. There was no difference in genetic effect between sexes or among different age strata. Heritability was enriched in certain functional categories, such as conserved and coding regions, and in tissues related to the musculoskeletal system. Polygenic risk score prediction well distinguished participants with high and low ALM. The findings are important not only for lean mass but also for other complex diseases, such as type 2 diabetes, as ALM is shown to be a protective factor for type 2 diabetes.
Similar content being viewed by others
Introduction
Lean body mass is an important physiological index. Low lean body mass, together with low muscle strength and low physical performance, represents a key component for the definition of sarcopenia, which is a critical condition with functional impairment and physical disability and a major modifiable cause of frailty in the elderly1,2. Lean body mass is associated with bone mineral density and hence may also influence the risk for osteoporosis3. Other lean body mass-related conditions include dysmotility syndrome4, sarcopenic obesity5 and cachexia6.
Lean body mass has a significant genetic component, as evidenced by a high heritability of 50–80% observed in twin studies7,8. However, findings of specific genes for human lean mass variation remain limited, even with the powerful genome-wide association study (GWAS) approach. A key reason for the limited findings, as in other human complex traits, is the modest sample size used in most GWASs performed for lean body mass9,10,11,12,13, resulting in few single nucleotide polymorphisms (SNPs) identified with genome-wide significance.
As a notable example, one previous large meta-analysis of GWASs amassed 20 cohorts of European ancestry with a total sample size of >38,000 for whole body lean mass and of >28,000 for appendicular lean mass (ALM)14. Of the 20 studied cohorts, 10 (n = 21,074, 55%) were characterized with the dual-energy X-ray absorptiometry (DEXA)-derived measure of lean mass, while the other 10 (n = 17,218, 45%) were characterized with the bioelectrical impedance analysis (BIA)-derived measure. Despite the large sample used, the percentage of phenotypic variance explained by the identified SNPs was still only 0.23% and 0.16% for whole body lean mass and ALM, respectively, suggesting that most of the heritability of lean body mass was still undetected. Therefore, even with such a large GWAS meta-analysis, it is still necessary to boost the sample size further to enhance the statistical power for detecting more causal SNPs underlying lean body mass.
In a more recent study, a GWAS of ALM was conducted in a population of 85,750 middle-aged (aged 38–49 years) individuals from the UK Biobank (UKB)15. A total of 182 loci were identified, 78% of which were replicated in a population of 181,862 elderly (aged 60–74 years) individuals from the same UKB cohort.
Unlike ALM, which is mainly affected by skeletal muscle, whole body lean mass is determined by skeletal muscle, smooth muscle and cardiac muscle. Therefore, ALM has a higher predictive power for sarcopenia-related health outcomes because sarcopenia is mainly due to a low skeletal muscle amount. ALM is also slightly more heritable than whole body lean mass16 and as such a more suitable trait for sarcopenia-related genetic analyses.
Here, in this study, with a sample containing approximately half-million participants of European origin, we performed a GWAS of ALM in the full UKB cohort. At a stringent genome-wide significance level (p < 5 × 10–9), we identified >1000 independent variants that were significant, all of which were significant in both sexes at p < 5 × 10–5. Our findings revealed a large number of genetic variants for lean body mass and contributed to the characterization of the genetic architecture of this complex trait. Through this GWAS, we demonstrated the power for mapping the genetic landscape of common human complex traits/diseases using extraordinarily large samples.
Results
A flow chart of this study is displayed in Fig. 1. The study sample came from the UKB cohort, which is a large prospective cohort of ~500,000 participants from across the United Kingdom, aged between 48 and 73 at recruitment. The basic characteristics of the sample are listed in Supplementary Data 1. In this study, we quantified ALM by appendicular fat-free mass measured by BIA. This measurement of lean mass is reliable based on its strong correlation with ALM measured by DEXA in 4294 UKB participants (Pearson’s correlation coefficient 0.96, p < 2.2 × 10–16). The Pearson correlation coefficient between ALM and AFM was 0.68 in males and 0.79 in females. Neither ALM nor AFM followed a normal distribution. Instead, both distributions were right skewed (Supplemental Fig. 1).
The study sample came from the UKB cohort. After QC, 450,243 self-reported white participants were eligible for analysis. The two sex groups were analyzed separately and then were combined for a meta-analysis. A series of in-depth analyses, including heritability enrichment, gene and gene sets enrichment, genetic correlation, Mendelian randomization, and polygenic risk score profiling were performed to uncover the genetic architecture of the trait.
ALM for all eligible participants was adjusted by AFM and other covariates, and the residuals were transformed into a standard normal distribution so that no outliers were observed in the transformed phenotype ALMadj.
Main association results
Following QC of both ALMadj and genome-wide genotypes, data from 19.4 million variants with a minor allele frequency (MAF) > 0.1% and an imputation quality score >0.3 were available in 244,730 female and 205,513 male participants.
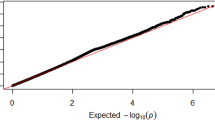
In each sex group, the additive effect of each variant was tested on ALMadj with BOLT-LMM17 (Fig. 2). The genomic inflation factor showed notable inflation in both sex groups (λfemale = 2.09, λmale = 1.84). The LDSC mean Chi-square and intercept were 3.05 and 1.17 for females and 2.63 and 1.13 for males, corresponding to attenuation ratios of 0.083 and 0.077, respectively.
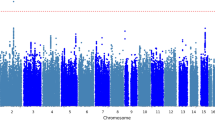
a Manhattan plot of the meta-analyses of both sexes (males: top, females: bottom). The horizontal red line indicates the genome-wide significance level (α = 5 × 10−9) on the −log10 scale. All significant SNPs are marked in green. b Per allele effect size versus minor allele frequency (MAF). The X-axis is the MAF of the 1059 identified variants, and the Y-axis is the per allele effect size (regression coefficient). The black solid line is the power function fitted to the data.
LDSC estimated a genetic correlation coefficient as high as 0.93 (s.e. 0.01) between the two sexes, implying that most GWAS-attributable heritability was shared between sexes.
Given the shared heritability between sexes, between-sex meta-analysis was performed with an inverse-variance-weighted fixed-effects model to combine the sex-specific GWAS results. The meta-analysis signals had an attenuation ratio of 0.078 (mean Chi-square = 4.50, intercept = 1.27), which was equal to that estimated by analysing the subset of genetically determined “Caucasian” participants (N = 400,879, attenuation ratio = 0.078), suggesting a limited effect of population structure. However, some residual stratification may remain uncorrected because ancestry was based upon self-reporting.
A total of 121,109 variants were significant at the genome-wide significance (GWS, α = 5.0 × 10–9) level in the combined meta-analysis and were significant at p < 5 × 10–5 in both sexes. Based on their physical coordinates, these variants were divided into 828 loci that were at least 500 kb apart. It was found that 57 lead variants were not in complete linkage equilibrium (LD r2 < 0.1) with each other due to the long-range LD pattern. After removing 33 lead variants, the remaining 795 lead variants were all in linkage equilibrium. Therefore, they were treated as independent loci.
Approximate conditional association analysis followed by between-sex meta-analysis was recursively performed, which further identified 4 additional loci and 264 conditionally significant variants at the GWS level that were also significant in both sexes at p < 5 × 10–5. These additional variants were also in linkage equilibrium (LD r2 < 0.1) with the 795 primary lead variants.
In total, 1059 (i.e. 795 + 264) independent variants from 799 distinct loci were associated with ALMadj (Supplementary Data 2). Among them, 353 achieved the strongest significance level (p < 5 × 10–9) in both sexes (categorized here as Tier 1 variants). Additionally, 208 variants achieved p values <5 × 10–9 in females and p values <5 × 10–5 in males; 94 variants achieved p values <5 × 10–9 in males and p values <5 × 10–5 in females (categorized here as Tier 2 variants). Finally, 404 variants achieved p values <5 × 10–5 in both sexes and p values <5 × 10–9 in the between-sex meta-analysis (categorized here as Tier 3 variants).
Replication in UKB South Asian participants
The associations of the 1059 lead SNPs in the 7,452 UKB South Asian (Indian, Pakistani and Bangladeshi) participants are listed in Supplementary Data 3. At the stringent significance level of 4.72 × 10–5 (0.05/1,059), only one SNP, rs4338565, was significant (p = 2.30 × 10–5), which is not unexpected given the limited sample size. At the nominal level of 0.05, 124 SNPs were significant. Overall, the effect direction at 767 SNPs was consistent between the two analyses (binomial p < 2.2 × 10–16), and the correlation coefficient of the regression between them was 0.27 (95% CI [0.22, 0.33], p < 2.2 × 10–16), partially demonstrating the replicability in the smaller South Asian sample.
Replication of previously reported loci
Associations of previously reported SNPs in the present study are listed in Supplementary Data 4. Among them, all five lead SNPs reported by Zillikens et al.14 were successfully replicated at the nominal level of p < 0.05, and up to four of them were replicated at the 5.0 × 10–8 level. For another bivariate study in which eight loci were identified18, two were significant at the 5.0 × 10–8 level, five additional were significant at the nominal level, but the last SNP rs7672749 was not significant (p = 0.26). For the recent study of ALM in middle-aged and older UKB participants, all 182 lead SNPs were nominally significant in the present study, and up to 168 (92.3%) were significant at the GWS level. In sum, the present study well replicates the findings from previous smaller studies.
Of the 799 identified loci, 102 were reported in the abovementioned previous studies, while the remaining 697 were not.
Overlap with loci for obesity-related traits
We also evaluated the overlap of the identified loci with those identified for three obesity traits obtained through the GIANT consortium. Flanking variants surrounding 478 lead SNPs (defined as the lead SNP + 500 kb flanking region to either side) were associated with one or more obesity traits at the 5.0 × 10–8 level, while no flanking variants surrounding the remaining 581 lead SNPs showed associations at the 5.0 × 10–8 level, demonstrating their novelty and possible specificity to lean but not fat mass.
Sex specificity
Our analysis identified an additional 107 loci that were significant at the GWS level in the between-sex meta-analysis but not significant at the suggestive level (p < 5 × 10–5) in each sex group (Supplementary Data 5). These loci may represent sex-specific signals pending further replication.
Sex heterogeneity
Of the 1059 identified variants, 181 (17.1%) exhibited high between-sex meta-analysis heterogeneity (I2 > 50%), most (175) of which were Tier 1 or 2 variants. A statistical test of sex differences in genetic effects revealed no difference in any SNP after accounting for multiple testing (α = 0.05/1,059 = 4.72 × 10–5), suggesting that all of the identified variants had similar effect sizes between sexes.
Age-dependent effect
The 1059 lead variants were evaluated for their age-dependent effects. At the Bonferroni-corrected significance level (p < 0.05/1,059 = 4.72 × 10–5), no age-dependent effects were identified at the 1059 lead SNPs (Supplementary Data 6). The most significant hit was rs2310876 (p = 2.01 × 10–3), where the per allele effect became larger as age increased. The second strongest hit was rs550793660 (p = 3.48 × 10–3). In contrast, the allele effect at this SNP tended to become zero as age increased.
Heritability distribution
The 1059 identified variants included 938 common (MAF > 5%), 97 less common (5%≥MAF > 1%) and 24 rare (MAF ≤ 1%) variants. Variants with a smaller MAF generally had a larger per allele effect size (Fig. 2). For example, the average per allele effect size of rare variants (mean 0.14, s.d 0.08) was 7-fold larger than that of common variants (mean 0.02, s.d 0.009).
The total phenotypic variance explained by the 1059 lead variants was 17.8% before correction for the winner’s curse effect and 15.5% after the correction. Using BOLT-REML19, GWAS-attributable total heritability was estimated to be 0.486 (s.e 3.14 × 10–3) and 0.476 (s.e 3.63 × 10–3) in females and males, respectively. After removing all variants in the flanking 500 kb regions surrounding each of the 1059 lead variants from raw genotypes, the remaining heritability was estimated to be 0.207 (s.e 2.94 × 10–3) and 0.188 (s.e 3.34 × 10–3) in females and males, respectively, implying that the identified loci collectively explained heritabilities of 0.279 and 0.288 in females and males, respectively. Therefore, ~10% of the heritability at the identified loci remains undefined.
Applying the stratified LDSC analysis, the GWAS-attributable heritability was partitioned into 24 functional categories20. Statistically significant enrichment was observed for 19 functional categories (p < 0.05/24 = 2.08 × 10–3, Fig. 3). In line with the observations by Finucane et al.20, regions conserved in mammals showed the strongest enrichment of any category, with 2.6% of SNPs explaining an estimated 31.3% of SNP heritability (enrichment ratio = 12.0, P = 1.48 × 10–18). Other categories with significant enrichment included coding regions (enrichment ratio = 8.0, P = 4.10 × 10–10), 3′ UTRs (enrichment ratio = 7.6, P = 9.29 × 10–5), transcription start sites (enrichment ratio = 6.5, P = 6.07 × 10–5), and H3K9ac histone marks (enrichment ratio = 4.9, P = 1.02 × 10–13). Neither the promoter nor the 5′-UTR region showed significant enrichment, although the 5′-UTR region had a high estimate of the enrichment ratio (13.2, p = 0.04).
Enrichment of genome-wide association signals in 24 main annotations using LDSC regression. The Y-axis represents the ratio of phenotypic variance explained by variants in a particular annotation category against that explained in the remaining regions. Error bars represent jackknife standard errors around the estimates of enrichment. A single asterisk indicates significance at p < 0.05 after Bonferroni correction for the 24 hypotheses tested, and two asterisks indicate significance at p < 0.01.
Using a new function of the stratified LDSC method21, three tissues were enriched at a Bonferroni-corrected significance level (P < 0.05/205 = 2.44 × 10–4), including cartilage (p = 1.01 × 10–7), chondrocytes (p = 9.48 × 10–5) and uterus (2.02 × 10–4).
Candidate gene prioritization
To prioritize candidate genes at the associated loci, we used multiple analytical strategies. A set of credible risk variants (CRVs) at each locus were defined as variants with high LD with the lead variant (r2 > 0.8). A total of 27,988 CRVs were identified (Supplementary Data 7). Based on these CRVs, six sources of supporting evidence were used to prioritize 1755 candidate genes (Supplementary Data 8–12).
A number of genes had multiple lines of supporting evidence (Supplemental Fig. 2). The IGFBP3 (IGF-Binding Protein 3) gene at 7p12.3, for example, had four lines of supporting evidence. This gene was closest to the lead SNP chr7:46262082 at the locus. One CRV, rs11977526, was associated with its mRNA expression level in skeletal muscle (p = 3.21 × 10–5) and associated with its protein level (p = 3.80 × 10–5) in whole blood. Finally, it was prioritized by DEPICT (p = 5.66 × 10–4). Another example is CXXC5 (CXXC Finger Protein 5) at 5q31.2. The lead SNP rs3822742 is an intron variant in CXXC5. Multiple CRVs around rs3822742, such as rs356452, were associated with CXXC5 mRNA expression in whole blood samples. This gene was also prioritized by both DEPICT and SMR.
Comparison between imputation- and sequencing-based association signals
Of the 1059 identified variants, 51 were missense coding variants. They are also available in the recently released UKB exome-sequencing data that contain a subset of ~50,000 participants from the whole UKB cohort. Using a set of 45,554 unrelated European participants who were both genotyped/imputed and sequenced, we compared the imputation-based association results with exome-sequencing-based results. Raw genotypes were highly concordant between the imputed and sequenced data (r2 = 0.84–1.00). The imputation-based and sequencing-based p values were highly concordant (Supplementary Data 13). For example, the imputation-based p values were within a 2-fold difference of the sequencing-based p values for up to 48 variants. Overall, these observations support that imputation-based association signals are close to the real sequencing-based association signals in a large sample. Therefore, imputation-based GWAS may be able to identify true associations, even those of rare variants.
Missense variants and the associated genes
As mentioned above, of the 1059 identified variants, 51 were missense coding variants. The majority (28) of these 51 missense mutations were predicted to be deleterious by one or more bioinformatics tools, including PolyPhen-222, SIFT23, PROVEAN24 and FATHMM25 (Supplementary Data 14), supporting their potential functional relevance.
Missense mutations were enriched among rare variants. Eight of the 24 rare variants were missense mutations, in clear contrast to the 43 missense mutations among the remaining 1035 variants (odds ratio = 11.53, Fisher’s exact test p = 6.77 × 10–6). Evidence of the enrichment was strengthened by comparing 24 missense mutations from 121 rare or less common variants with 27 missense mutations from 938 common variants (odds ratio = 6.89, Fisher’s exact test p = 1.10 × 10–9), suggesting that low-frequency mutations are more likely to play a direct role in changing protein function.
The top five loci containing missense lead variants are described below, while all 51 loci are listed in Table 1.
2p23.3 (GCKR). The common lead SNP rs1260326 (MAF = 39.6%, beta = −0.03, pmale = 5.30 × 10–29, pfemale = 4.20 × 10–37, pmeta = 6.16 × 10–64) is located in the exon of the GCKR (glucokinase regulator) gene, resulting in an amino acid change from leucine to proline. This SNP was previously reported to be associated with multiple metabolic traits26. GCKR encodes a regulatory protein that inhibits glucokinase in liver and pancreatic islet cells by binding noncovalently to form an inactive complex with the enzyme.
14q32.12 (RIN3). The lead SNP rs117068593 (MAF 19.0%, beta = 0.04, pmale = 5.60 × 10–34, pfemale = 1.50 × 10–33, pmeta = 8.83 × 10–62) results in a change from arginine to cysteine at the 204th amino acid of the protein encoded by the RIN3 (ras and rab interactor 3) gene. This change is predicted to be harmful by PROVEAN, Polyphen-2 and SIFT. This locus was previously reported to be bivariately associated with both bone mass and lean mass at the lead SNP rs75438818. rs754388 is in nearly perfect LD with rs117068593 (r2 = 0.95), implying the same association signal for both SNPs. The gene product of RIN3 is a member of the RIN family of Ras interaction-interference proteins. It functions as a guanine nucleotide exchange for RAB5B and RAB31.
5q35.2 (STC2). The lead SNP rs148833559 (MAF 0.14%, beta = 0.38, pmale = 6.70 × 10–25, pfemale = 1.10 × 10–30, pmeta = 9.21 × 10–52) is a rare mutation whose per allele effect is 10-fold larger than those of the above two common ones. The substitution of arginine to leucine at the 44th amino acid of the STC2 (Stanniocalcin 2) protein is predicted to be harmful by PROVEAN, Polyphen-2 and SIFT. This rare variant was previously reported to be associated with human height27. In a recent study of ALM in middle-aged and older participants of the UKB cohort, the same SNP was identified. The authors also verified that knockdown of STC2 had a significant effect on myotube length in C2C12 cells15.
8q24.22 (ZFAT). Two missense SNPs in the ZFAT (zinc finger and AT-hook domain containing) gene are associated with ALMadj. The primary SNP is a common SNP rs12541381 (MAF 25.8%, beta = −0.03, pmale = 2.50 × 10–19, pfemale = 3.30 × 10–32, pmeta = 2.81 × 10–49), resulting in a change from proline to serine at the 102nd amino acid of the protein encoded by ZFAT, where the change is predicted to be benign by all four prediction tools. Conditional analysis identified a secondary rare SNP, rs112892337 (MAF 0.4%, beta = 0.14, pmale = 4.53 × 10–14, pfemale =2.32 × 10–11, pmeta = 1.23 × 10–23 after conditioning), which results in a change from serine to cysteine at the 470th amino acid. The change is predicted to be harmful by three prediction tools. ZFAT encodes a protein that likely binds DNA and functions as a transcriptional regulator involved in apoptosis and cell survival.
8q24.12 (ENPP2). The lead SNP rs10283100 (MAF 5.6%, beta = −0.06, pmale = 4.00 × 10–20, pfemale = 7.40 × 10–29, pmeta = 4.11 × 10–44) results in a change from serine to proline at the 545th amino acid of the protein encoded by the ENPP2 (Ectonucleotide pyrophosphatase/phosphodiesterase 2) gene. This change is predicted to be benign by all prediction tools. ENPP2 encodes a protein functioning as both a phosphodiesterase and a phospholipase.
Gene-based and geneset enrichment analyses
A total of 2885 genes were significant at the gene-based GWS level (α = 0.05/17,788 = 2.81 × 10–6, Supplementary Data 15). The most significant gene was DLEU1 (p = 5.83 × 10–170), followed by ZBTB38 (p = 1.94 × 10–77), FNDC3B (p = 9.68 × 10–73), EDEM2 (p = 1.76 × 10–72) and CRADD (p = 2.36 × 10–71). Using gene level association statistics as input, a total of 123 gene sets were significant at the geneset significance level (α = 0.05/15,481 = 3.23 × 10–6, Supplementary Data 16). The top gene sets included genes with known functions related to the musculoskeletal and connective systems, such as GO:0001501 ‘skeletal system development’ (p = 3.31 × 10–18, Ngene = 483), GO:0002062 ‘chondrocyte differentiation’ (p = 3.22 × 10–14, Ngene = 115) and GO:0051216 ‘cartilage development’ (p = 3.55 × 10–14, Ngene = 197).
Polygenic risk score profiling
To assess the ability of the GWAS findings to predict ALM, a polygenic risk score (PRS) analysis was performed on the subset of unrelated white participants from the UKB cohort. Three-quarters of the participants (n = 277,762, including 149,329 females) were randomly selected as the training sample, with the remaining participants (n = 92,206, including 49,660 females) as the validation sample.
The training sample revealed 134,277 variants with a p value <1 × 10–5 for association with ALMadj. Using these variants as predictors, the predicted genome-wide PRS and the real phenotype residual in the validation sample were significantly correlated (Pearson’s correlation coefficient 0.32, 95% CI (0.32, 0.33), p < 2.2 × 10–16). Mean phenotype residuals in the top tail were significantly higher than those in the bottom tail of the PRS distribution (Fig. 4). For example, the predicted top 1% of participants had an increased average residual of 1.16 compared to that of the predicted bottom 1% of participants (0.57 (s.d 0.96) vs. −0.59 (s.d 0.94)), corresponding to a 1.69 kilogram (kg) increase in raw ALM (24.61 kg (s.d 5.89 kg) vs. 22.92 kg (s.d 5.27 kg)). In the female group, the predicted top 1% of participants had an average 1.39 kg increase in raw ALM compared with that of the predicted bottom 1% of participants (20.26 kg (s.d 2.75 kg) vs. 18.87 kg (s.d 2.45 kg)). In males, the increase was 2.29 kg (29.82 kg (s.d 4.18 kg) vs. 27.53 kg (s.d 3.56 kg)). These results demonstrate that PRS prediction based on the current GWAS finding is capable of identifying participants with high or low levels of ALM.
A total of 277,504 participants were randomly selected as the training sample, and another independent 92,108 participants were selected as the validation sample. The variants achieving a p value of <1 × 10−5 in the training sample were selected and used for prediction in the validation sample via the LDpred approach. Participants in the two extreme tails of the predicted genome-wide polygenic risk score (PRS) distribution were compared in terms of raw phenotypic mean (after correction). The X-axis represents the fraction of participants drawn from both extreme tails of the predicted PRS distribution. The Y-axis represents the mean ALMadj (with the standard error of the mean difference between the two tails).
Using the same approach, we evaluated the capability of the GWAS finding to predict DEXA-derived ALM. The correlation coefficient between PRS and DEXA-derived ALM was 0.18 (95% CI [0.15, 0.21] p < 2.2 × 10–16), again demonstrating the capacity of the present findings to predict DEXA-measured lean mass.
Genetic correlations with other traits
To test whether lean mass has a shared genetic aetiology with other diseases and relevant traits, a genetic correlation analysis was performed with LDSC28. ALMadj is strongly genetically correlated with whole body lean mass and ALM studied by a previous GWAS meta-analysis14 (rg = 0.70 and 0.56, p < 2.2 × 10–16) (Fig. 5). Strikingly, ALM was highly correlated with height (rg = 0.71, p < p < 2.2 × 10–16), implying that the two traits share a large number of developmental pathways. Furthermore, ALMadj was modestly correlated with BMI (rg = 0.12, p = 1.59 × 10–7). However, the correlation with heel bone mineral density was low (rg = −0.03, p = 0.13). ALMadj was most negatively correlated with BMI-adjusted 2-h glucose (rg = −0.26, p = 7.79 × 10–5) and BMI-adjusted leptin (rg = −0.23, p = 2.63 × 10–7). It was also negatively correlated with body fat (rg = −0.21, p = 1.36 × 10–10). However, this correlation should be interpreted with caution given the following two confounding factors. The first is collider bias, as the trait that we analysed was fat mass-adjusted lean mass. The second is the phenotypic constraint between lean mass and fat mass, as the sum of the two measures defines body weight and body size.
Genetic correlations (rg) between ALMadj and 51 traits and diseases were estimated. LD score regression tested for genome-wide SNP associations in these participants against similar data for various other traits and diseases, including musculoskeletal system, anthropometric, obesity, cognition, metabolism, psychiatry, reproduction and neuropsychiatric outcome traits. Error bars represent the standard errors of these estimates. Blue bars represent significant positive correlations at the nominal level of p < 0.05; pink bars represent significant negative correlations (p < 0.05); grey bars represent nonsignificant correlations.
Mendelian randomization analysis
To investigate whether ALMadj is causally linked with other complex diseases and traits, a Mendelian randomization analysis was performed with GSMR29. Ten diseases and eight continuous traits from a variety of categories were chosen for evaluation. The scatter plot for all 18 traits is displayed in Supplemental Fig. 3. At the Bonferroni-corrected significance level of 2.78 × 10–3 (0.05/18), ALMadj was causally associated with five diseases (coronary artery disease, p = 2.09 × 10–56; fracture, p = 3.45 × 10–10; type 2 diabetes, p = 1.20 × 10–8; insomnia, p = 2.77 × 10–5; and inflammatory bowel disease p = 8.09 × 10–4) (Supplementary Data 17). The causal association between ALMadj and type 2 diabetes was negative, indicating that ALMadj is a protective factor for the latter. A one-standard deviation increase in the ALMadj residual corresponded to a decrease in the odds ratio of 0.92 (95% CI [0.89, 0.95]).
At the same significance level, ALMadj was also causally associated with 6 metabolic traits, including four serum lipid traits and two blood pressure traits.
Discussion
This study of lean mass with approximately half a million participants, the largest sample used for a GWAS of lean mass so far, was successful. More than 1000 variants were identified at the genome-wide significance scale (p < 5 × 10–9). In particular, more than half of these variants achieved genome-wide significance (p < 5 × 10–9) in one sex and were replicated in the other sex (p < 5 × 10–5). Overall, these >1000 variants accounted for ~15% of ALM variation, again, the largest explainable fraction of variation in lean mass reported so far in a GWAS. Our finding of >1000 variants is expected for a complex trait with high heritability, particularly considering another trait with comparable heritability, height, for which ~700 variants were detected30. Interestingly, the majority of the loci detected in a previous smaller GWAS12 and meta-analysis14 of lean mass were also significant in the present study, providing solid evidence of replication.
The inability of GWAS to detect and replicate specific genetic variants for human complex traits, contradicting a trait’s established high heritability, e.g. height, was formally recognized as the missing heritability problem a decade ago31,32. An explanation is the so-called polygenic model, where hundreds or even thousands of common SNP variants act additively, with each contributing only a tiny fraction of the trait variation. The genetic findings from the present study support this explanation for lean mass. The total 1059 conditionally independent variants explained 15.5% of the phenotypic variance, corresponding to an average per variant variance as low as 0.015%. It is worth noting that the present study had nearly 100% power to detect variants with an effect size larger than 0.015% and indeed did so. On the other hand, the power to detect variants with effect sizes as low as 0.001% was nearly zero. Therefore, there might exist more variants with effect sizes smaller than those identified in the present study, further supporting the polygenic model.
The functional relevance of our identified variants was supported by geneset enrichment analysis, where GO terms, including GO:0001501 ‘skeletal system development’, GO:0061448 ‘connective tissue development’ and GO:0051216 ‘cartilage development’, were among the significant gene sets. Specifically, the common genes involved in these terms were tightly connected into a network that contained TGF pathway genes, BMP pathway genes and SMAD family genes, which are all important musculoskeletal development genes/pathways. This finding is concordant with knowledge of developmental biology since cells from bone, cartilage, muscle and fat share the same progenitor, mesenchymal stem cells, and pleiotropy of muscle and bone is well recognized in both humans33 and animal models34.
To declare an association as significant, we required that the signal not only be significant at the GWS level in the combined analysis but also be significant at the 5 × 10–5 level for each sex group. This significance level was essentially equivalent to that in a two-stage design, where the first stage involved a GWAS in one group (e.g. the male group) and the second stage involved replicating top hits in the other group (e.g. the female group). As a maximal number of 1000 independent loci was assumed, we could have selected the top 1000 hits from the first stage for replication at the second stage. As a result, a significance level of 5 × 10–5 (0.05/1000) was sufficiently conservative to declare successful replication. In our actual analysis, the numbers of independent loci with p < 5 × 10–5 were 1988 and 1713 in the female and male groups, respectively, which were almost twice the presumed number (n = 1000) of independent loci. This may have inflated the type I error rate for the variants whose p values fell within the range from 5 × 10–5 to 5 × 10–9 (i.e. the Tier 3 variants).
The present study had the following strengths. First, the large sample size of over 400,000 participants is the largest used for a lean mass GWAS to date, offering a unique opportunity to discover loci that were undetected by previous smaller GWASs. Second, instead of analysing the sample as a whole, the two sexes were analysed separately, and then meta-analysis was performed. This may have reduced the statistical power for identifying new loci but allowed us to replicate significant findings between the sexes. Third, via a series of comprehensive downstream analyses annotating the identified SNPs, a deep understanding was achieved for the genetic mechanism underlying ALM and its interplay with other complex traits and diseases.
Certain limitations existed in the present study. First, lean mass was measured by the BIA approach, which is not as reliable as the gold standards for quantifying lean mass, such as magnetic resonance imaging and computed tomography, because these latter methods are direct measures. Instead of measuring lean mass directly, BIA derives an estimate of lean mass based on electrical conductivity. Therefore, it can be influenced by the hydration status of the subject. Moreover, the derivation equation of BIA relies on a calibrated reference population, which may not be well validated across populations. Second, the genetic findings for lean mass alone are inadequate for characterizing the full genetic basis of sarcopenia. This is because there is a consensus that sarcopenia is defined not only by low lean mass but also, more importantly, by low muscle strength and poor physical performance35. Therefore, the present study only discovered the genetic mechanism of sarcopenia from the lean mass perspective, far more than enough to begin understanding the genetic basis of sarcopenia as a whole. Third, physical activity is known to influence lean mass36, the confounding effect of which was not controlled for in the present study.
In summary, we performed a GWAS using approximately a half-million participants for lean mass. The variation (~15%) in lean mass explained by the identified variants represents a significant leap in explaining the hidden heritability of this complex trait using the GWAS approach. The translational value of these findings lies in the importance of lean mass for other complex diseases, such as type 2 diabetes, as our Mendelian randomization analysis showed that ALM is a protective factor for the latter. Overall, our study provides another example in which a GWAS with a very large sample size ultimately and thoroughly delineates the genetic architecture of a complex human trait. This epitomizes the value of big data in human genetic research.
Methods
Study participants
The study sample came from the UKB cohort, which is a large prospective cohort of ~500,000 participants from across the United Kingdom, aged between 48 and 73 at recruitment. Ethics approval for the UKB study was obtained from the North West Centre for Research Ethics Committee (11/NW/0382), and informed consent was provided by all participants. This study (project number 41542) was covered by general ethical approval for the UKB study.
As careful data quality control is critical to avoid false positives, we only analysed self-reported white individuals (data field 21000). Participants who had a self-reported sex inconsistent with the genetic sex, whose sex chromosome was aneuploid, who had unusually high heterozygosity and/or missing rates or who withdrew their consent were removed. Overall, 487,378 participants were identified to have both phenotypic and genotypic data, 37,135 of whom were excluded. The final sample consisted of 450,243 participants, including 244,730 females and 205,513 males.
Phenotype and modelling
Body composition was measured by the BIA approach. ALM was quantified by the sum of fat-free mass at the arms (data fields 23121 and 23125) and legs (data fields 23113 and 23117). Appendicular fat mass (AFM) was quantified by the sum of fat mass at the arms (data fields 23120 and 23124) and legs (data fields 23112 and 23116). In each sex, covariates, including AFM, age, age squared, the top 10 principal components, assessment centre (23 levels) and genotyping array (2 levels), were used to adjust raw ALM values. To avoid collider bias due to height, height was not included as a covariate. The adjusted residuals were normalized into inverse quantiles of a standard normal distribution, which were used for subsequent association analysis.
A small subset of 4294 participants also received a DEXA body composition scan, and hence, their DEXA-derived ALM was also available. Therefore, raw ALM values derived from DEXA and from BIA were compared in these participants to evaluate phenotypic consistency from these two measurements.
Genotype quality control
Genome-wide genotypes were available for all participants at 784,256 genotyped autosome markers and were imputed into UK10K haplotype, 1000 Genomes project phase 3 and Haplotype Reference Consortium reference panels. Genotype imputation was performed by the UKB. A total of ~92 million variants were generated by imputation. We excluded variants with a MAF < 0.1% and with an imputation r2 < 0.3. As a result, ~19.4 million well-imputed variants were retained for subsequent analysis.
Genetic association analysis
In each sex group, we used BOLT-LMM to perform linear mixed model analysis17. Upon the completion of sex-specific association analyses, we meta-analysed the summary statistics of the two sexes by an inverse-variance-weighted fixed-effects model with METAL37. The GWS level was set at α = 5 × 10–9 to account for both common and rare variants38. The variants that passed this threshold in the between-sex meta-analysis were then checked for significance in both sexes. Previous studies on human height and body mass index (BMI), two of the most polygenic traits, identified 712 and 536 loci in up to ~700,000 European participants39. By referring to these previous findings, we selected an arbitrary but reasonable number, i.e. 1000, for the maximal number of independent loci for ALMadj and set the suggestive significance level to be 5 × 10–5 (0.05/1000) to account for multiple testing. An association was declared only if the signal was significant at the GWS level (p < 5 × 10–9) in the meta-analysis and was significant at the suggestive level (p < 5 × 10–5) in both sexes.
Differences in genetic effects between females and males were examined by a two-sided z-score test with the following equation:
where βfemale and βmale are regression coefficients for females and males and var(·) are their variances, respectively.
The identified variants were annotated by Variant Effect Predictor (VEP)40, which invokes dbNSFP41 to annotate nonsynonymous SNPs.
Conditional association analysis
To identify additional signals in regions of association, approximate joint and conditional association analysis was performed using the GCTA tool42. From the UKB sample, a reference sample of 100,000 unrelated participants was generated for estimating LD patterns. Specifically, KING software43 (with a kinship coefficient cut-off at 0.0884) was used to infer 369,968 unrelated participants, from whom the 100,000 participants of the reference sample were randomly drawn.
A recursive conditional association analysis was performed. In each iteration, an approximate conditional analysis conditioned on the current list of lead variants was performed in each sex, followed by a between-sex meta-analysis. Again, an association was defined as significant if it achieved both a conditional meta-analysis GWS signal and a conditional suggestive signal (p < 5 × 10–5) in both sexes. In addition, each such identified variant was required to be independent of all variants in the lead SNP list (LD r2 < 0.1). The variant with the lowest p value among such identified variants was added to the list of lead variants. Iterations of the conditional analysis were run until no significant signal could be detected.
Overlap with loci related to obesity traits
GWAS summary statistics for three obesity traits, including BMI39, waist circumference and waist-hip ratio44, were downloaded from the GIANT consortium website. For each trait, SNPs located within all the identified loci (lead SNP +500 kb flanking region to either side) were extracted from the GWAS summary statistics. The significance level for the obesity traits was set at the conventional level of 5.0 × 10–8.
Replication in the South Asian population
The UKB participants from the South Asian population were analysed to replicate the findings identified in the white population. Specifically, self-reported South Asian participants (data field 21000, including Indian, Pakistani and Bangladeshi) were collected. Quality control criteria were the same as those for the main analysis. The final sample consisted of 7452 participants. Phenotype modelling was the same as that for the main analysis, with the only exception that both sexes were analysed together, and sex was used as a covariate. Association analysis was performed again with BOLT-LMM.
Comparing imputation accuracy with exome-sequencing data
During the preparation of this manuscript, the UKB released exome-sequencing data on a selected subset of ~50,000 participants. While we are aware of the systematic assembly issue announced by the UKB, we used the current released SPB dataset to assess the imputation accuracy of the present GWAS data by comparing association signals from imputed genotypes to those from direct sequencing. BOLT-LMM involves sophisticated two-step implementation, where the first step fits the model to a set of directly typed variants and the second step examines the association in another set of imputed and/or typed variants. When using BOLT-LMM for comparison, the first-step model parameters fitted to imputed versus sequenced data may incur differences, which is nonrelevant to imputation accuracy. To make the comparison as fair as possible, we used a simple linear model instead. Specifically, we generated an unrelated sample consisting of participants who were both exome-sequenced and genotype-imputed.
As the QC procedure, we removed participants who were not self-reported as white, whose self-reported sex was inconsistent with their genetic sex, and who withdrew their consent. KING software was again used to select unrelated participants43 according to genotyped SNPs. The final sample consisted of 45,554 participants, including 24,740 females and 20,814 males.
Sequence variant coordinates, which were annotated to the GRCH38 assembly, were converted back to the GRCH37 assembly with Liftover (http://genome.ucsc.edu/cgi-bin/hgLiftOver). For each participant, variants that were missing in the sequenced data were set to missing in the imputed data as well. In both datasets, genetic association with normalized phenotype residuals was analysed in R.
Genetic architecture
The LDSC method was used to estimate the amount of genomic inflation due to confounding factors such as population stratification and cryptic relatedness28. Precomputed LD scores from the 1000 Genomes Project for European participants were used for estimation. The relative contribution of confounding factors was measured by the attenuation ratio, which is defined as (intercept-1)/(mean chi^2−1), where intercept and mean chi^2 are estimates of confounding and the overall association inflation, respectively28.
BOLT-REML was used to estimate heritability tagged by all the analysed variants19. It was applied to raw genotypic data with or without removing all variants among the total identified loci (each defined as the lead SNP +500 kb flanking region to either side) to estimate respective values of heritability. The difference between the two measures provides an estimate of the heritability explained by the identified loci.
The variance explained by all lead variants was calculated as the sum of all individual variant effect sizes, which is defined as the percentage of phenotypic variance explained by the variant and estimated with the formula 2 f(1 − f)β2, where f is allele frequency and β is the regression coefficient associated with the variant. To account for the winner’s curse effect, the effect sizes were shrunk with a false discovery rate (FDR)-based method45.
Age-dependent effect
The identified lead variants were evaluated for their age-dependent effects on ALMadj. Specifically, the sample was divided into the following six age strata defined by bins of 5 years: 45 or less (N = 54,608), 46–50 (N = 58,865), 51–55 (N = 70,253), 56–60 (N = 89,479), 61–65 (N = 109,696) and 66 or more (N = 67,342). Participants of both sexes within each age stratum were pooled together for analysis. Phenotype modelling within each age stratum was similar to that in the main association analysis, with the only exception that sex was added to the covariates. The association within each age stratum was examined by BOLT-LMM. The generated regression coefficients from all age strata were meta-regressed against the mean age of each stratum to examine the effect of age on the genetic effect. Meta-regression was implemented by a linear regression analysis weighted by the inverse variance of each regression coefficient. Evidence of significance was determined by the Bonferroni-corrected significance level.
Enrichment analysis
Stratified LDSC was used to partition heritability from GWAS summary statistics into different functional categories20. The analysis was based on the ‘full baseline model’ created by Finucane et al.20 from 24 publicly available main annotations that are not specific to any cell type. The significance level of enrichment was set at p < 2.08 × 10–3 (0.05/24).
Stratified LDSC was also used to assess the enrichment of heritability in specific tissues and cell types21. This method analyses gene expression data together with GWAS summary statistics, for which the two precompiled gene expression datasets in LDSC were used. The first is the GTEx project dataset46, and the second is the Franke Laboratory dataset47. The GTEx dataset contains 53 tissues with an average of 161 samples per tissue. The Franke Laboratory dataset is an aggregation of publicly available microarray gene expression datasets comprising 37,427 human samples from 152 tissues. A total of 205 (=53 + 152) tissues were classified into nine categories for visualization. Again, evidence of significance was determined by a Bonferroni-corrected significance level of p < 2.44 × 10–4 (0.05/205).
Candidate gene prioritization
At each associated locus, CRVs were defined as variants in strong LD with the lead variant (r2 > 0.8, including the lead variant itself). The LD r2 measure was estimated based on the above 100,000 unrelated reference samples with PLINK48. Six sources of information were used to evaluate a gene’s causality: (1) being nearest to the lead CRV; (2) containing a missense coding CRV; (3) being a target gene for a cis-eQTL CRV; (4) being a target gene for a cis-protein QTL (cis-pQTL) CRV; (5) being prioritized by DEPICT analysis49 and (6) being prioritized by SMR analysis50.
Cis-eQTLs revealed by the GTEx (v7) project were accessed from the GTEx web portal (www.gtexportal.org/)46. Cis-eQTL information is available for over 50 tissues. We selected skeletal muscle and whole blood for our analysis. Cis-eQTL was searched within a 500 kb distance of the target gene. Significant cis-eQTLs were declared at p < 5 × 10–5.
Cis-pQTL information was accessed from Sun et al.51. GWAS summary statistics for 3284 proteins were downloaded from the study’s website. Cis-pQTL was searched within a 500 kb distance of the target gene. Significant cis-eQTLs were declared at p < 5 × 10–5.
DEPICT is an integrative tool that takes advantage of predicted gene functions to systematically prioritize the most likely causal genes at loci of interest49. The input of DEPICT includes a list of variant identifiers, and the output contains all genes located in the loci and their p values as candidate genes. All lead variants were submitted to DEPICT for analysis. Significant genes were declared at a false discovery rate <5%.
The SMR (Summary data–based Mendelian Randomization) method50 is another gene prioritization program that integrates summary-level data from GWASs with data from eQTL studies to identify genes whose expression levels are associated with traits due to causal or pleiotropic effects. Here, the pleiotropy effect means that a SNP is causally associated with both gene expression and phenotypic variation. SMR uses SNPs as an instrumental variable and tests the causal relation of gene expression to phenotype variation. The results are interpreted as the effect of gene expression on the phenotype free of confounding from nongenetic factors. We used a precompiled eQTL dataset of whole blood tissue for estimation52. Evidence of a pleiotropic instead of causal relationship between an eQTL and ALMadj was examined by the HEIDI test29. We set a loose significance level of 0.05 for the HEIDI test to exclude potential pleiotropy.
The intersections of candidate genes prioritized from different sources were plotted using the R package UpSetR53.
Gene-based and geneset enrichment analyses
Gene-based association analysis was performed with MAGMA v1.654, as implemented on the FUMA website (http://fuma.ctglab.nl/). GWAS meta-analysis summary statistics were mapped to 19,427 protein-coding genes, resulting in 17,788 genes that were covered by at least one SNP. A gene-based association test was performed, taking into account the LD between variants. The significance level was set at a stringent Bonferroni-corrected threshold of 2.81 × 10–6, i.e. 0.05/17,788.
The generated gene-based summary statistics were further used to test for enrichment of associations with specific biological pathways or gene sets. A geneset’s association signal was evaluated by integrating all signals from the genes in the set with MAGMA. A competitive geneset analysis model was used to test whether the genes in a geneset were more strongly associated with the phenotype than other genes.
Gene sets were obtained through the MSigDB website (http://software.broadinstitute.org/gsea/msigdb/index.jsp)55. Each gene was assigned to a geneset as annotated by Gene Ontology (GO), Kyoto Encyclopedia of Genes and Genomes (KEGG), Reactome and BioCarta geneset databases and other gene sets curated by domain experts or from the biomedical literature55. A total of 15,481 gene sets were used in this analysis. Significance was set at a Bonferroni-corrected level of 0.05/15,481 = 3.23 × 10–6.
Polygenic risk score profiling
To assess the capability of the GWAS findings to predict ALM, a PRS analysis was conducted with a training sample and a validation sample. It was required that the two samples be independent from each other. To accomplish this, both samples were drawn from unrelated participants extracted from the main UKB sample. Specifically, three-quarters of the participants (277,504, including 149,172 females) were randomly selected as the training sample, and the remaining one quarter of the participants (92,108, including 49,604 females) were selected as the validation sample. Participants of both sexes were pooled together for analysis.
Raw phenotypes were adjusted by age, age squared, sex, assessment centre, genotyping array, AFM and the top 10 PCs. The residuals were converted to the standard normal distribution quantiles for downstream analysis. Genetic association analysis was performed with PLINK 256 because of its computational efficiency.
PRS calculation was conducted with LDpred57. LDpred infers the posterior mean effect size of each marker by using a prior of effect sizes and LD information from an external reference panel. To save computer memory usage, only the variants achieving a p value of <1 × 10–5 in the training sample were selected and used for prediction in the validation sample. Specifically, the validation sample with original genotypes was used as a reference panel for LD estimation. The number of SNPs used to adjust LD from each side of the target SNP was set to 1000. Other software parameters were set to the default.
Using the same approach, we also evaluated the capability of the GWAS finding to predict DEXA-derived ALM. To accomplish this, we divided the total 450,243 eligible participants into one training sample including participants who did not receive the DEXA scan and one validation sample including participants who received the DEXA scan. The correlation between the risk score and DEXA-derived ALM was examined in the validation sample.
Genetic correlations with other traits
To test whether lean mass has a shared genetic aetiology with other diseases and relevant traits, a genetic correlation analysis was performed with the LDSC method28. An online web tool, LDHub (http://ldsc.broadinstitute.org/ldhub/), was used to estimate the genetic correlations between ALMadj and 49 complex traits and diseases. The standalone version of the software was used to estimate the correlations between ALMadj and two additional traits, ALM and whole body lean mass, which are not available in the LDHub GWAS summary statistics collections and were downloaded from the GEFOS consortium website (http://www.gefos.org).
Both the LDHub and standalone analyses adopted the same QC criteria. Specifically, only HapMap3 autosomal SNPs were included to minimize poor imputation quality28. SNPs were further removed given the following conditions: MAF < 0.01, palindromic strand (A/T or C/G), duplicated ID, or reported sample size less than 60% of the total sample size. LD scores precomputed with the European participants in 1000 Genomes Project were used for calculation.
Mendelian randomization analysis
To investigate whether ALMadj (as exposure) is causally associated with complex traits and diseases (as outcomes), a Mendelian randomization analysis was performed with GSMR29 on 10 diseases and 8 metabolic traits. The 10 diseases included fracture, type 2 diabetes, asthma, insomnia, inflammatory bowel disease, smoking addiction, coronary artery disease, amyotrophic lateral sclerosis, bipolar disorder and autistic spectrum disorder. Although some of the selected diseases were distantly related to lean mass, they could serve as negative controls for the MR analysis. The metabolic traits included high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, total cholesterol, triglycerides, insulin, glucose, diastolic blood pressure and systolic blood pressure.
GWAS summary statistics for each trait were downloaded from the corresponding study’s website. From the list of SNPs associated with ALMadj at the 5 × 10–9 level, qualified SNPs were included based on the following criteria: concordant alleles between exposure and outcome GWAS summary statistics, nonpalindromic SNPs with certain strands, a MAF > 1%, and allele frequency difference between exposure and outcome GWAS summary statistics <0.2.
Independent SNPs were further clumped with PLINK 256 by using an independence LD threshold r2 < 0.05 and a 1 MB window size. For each pair of studied traits, the clumped independent SNPs were examined for their pleiotropic effects on both exposure and outcome by the HEIDI test29. The significance level for the HEIDI test was set to α = 0.05. After removing pleiotropic SNPs on an outcome-by-outcome basis, the remaining independent SNPs were taken as instrumental variables to test for a causal effect of exposure on outcomes. The estimated causal effect coefficients are approximately equal to the natural log odds ratio for a case–control trait. The MR analysis significance level was set to 2.78 × 10–3 (0.05/18).
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
Genome-wide summary statistics are available through the NHGRI-EBI GWAS Catalog (https://www.ebi.ac.uk/gwas/downloads/summary-statistics), with accession IDs GCST90000025-GCST90000027. Additional data are available in the Supplementary Data file in Supplementary Data 1–17
References
Giles, J. T., Bartlett, S. J., Andersen, R. E., Fontaine, K. R. & Bathon, J. M. Association of body composition with disability in rheumatoid arthritis: impact of appendicular fat and lean tissue mass. Arthritis Rheum. 59, 1407–1415 (2008).
Janssen, I., Heymsfield, S. B. & Ross, R. Low relative skeletal muscle mass (sarcopenia) in older persons is associated with functional impairment and physical disability. J. Am. Geriatr. Soc. 50, 889–896 (2002).
Miyakoshi, N., Hongo, M., Mizutani, Y. & Shimada, Y. Prevalence of sarcopenia in Japanese women with osteopenia and osteoporosis. J. Bone Min. Metab. 31, 556–561 (2013).
Binkley, N., Krueger, D. & Buehring, B. What’s in a name revisited: should osteoporosis and sarcopenia be considered components of “dysmobility syndrome?”. Osteoporos. Int. 24, 2955–2959 (2013).
Wannamethee, S. G. & Atkins, J. L. Muscle loss and obesity: the health implications of sarcopenia and sarcopenic obesity. Proc. Nutr. Soc. 74, 405–412 (2015).
Evans, W. J. et al. Cachexia: a new definition. Clin. Nutr. 27, 793–799 (2008).
Arden, N. K. & Spector, T. D. Genetic influences on muscle strength, lean body mass, and bone mineral density: a twin study. J. Bone Min. Res. 12, 2076–2081 (1997).
Livshits, G. et al. Contribution of heritability and epigenetic factors to skeletal muscle mass variation in United Kingdom twins. J. Clin. Endocrinol. Metab. 101, 2450–2459 (2016).
Liu, X. G. et al. Genome-wide association and replication studies identified TRHR as an important gene for lean body mass. Am. J. Hum. Genet. 84, 418–423 (2009).
Ran, S. et al. Gene-based genome-wide association study identified 19p13.3 for lean body mass. Sci. Rep. 7, 45025 (2017).
Hai, R. et al. Bivariate genome-wide association study suggests that the DARC gene influences lean body mass and age at menarche. Sci. China Life Sci. 55, 516–520 (2012).
Urano, T., Shiraki, M., Sasaki, N., Ouchi, Y. & Inoue, S. Large-scale analysis reveals a functional single-nucleotide polymorphism in the 5′-flanking region of PRDM16 gene associated with lean body mass. Aging Cell 13, 739–743 (2014).
Klimentidis, Y. C. et al. Genetic variant in ACVR2B is associated with lean mass. Med. Sci. Sports Exerc. 48, 1270–1275 (2016).
Zillikens, M. C. et al. Large meta-analysis of genome-wide association studies identifies five loci for lean body mass. Nat. Commun. 8, 80 (2017).
Hernandez Cordero, A. I. et al. Genome-wide associations reveal human-mouse genetic convergence and modifiers of myogenesis, CPNE1 and STC2. Am. J. Hum. Genet. 105, 1222–1236 (2019).
Hsu, F. C. et al. Heritability of body composition measured by DXA in the diabetes heart study. Obes. Res. 13, 312–319 (2005).
Loh, P. R. et al. Efficient Bayesian mixed-model analysis increases association power in large cohorts. Nat. Genet. 47, 284–290 (2015).
Medina-Gomez, C. et al. Bivariate genome-wide association meta-analysis of pediatric musculoskeletal traits reveals pleiotropic effects at the SREBF1/TOM1L2 locus. Nat. Commun. 8, 121 (2017).
Loh, P. R. et al. Contrasting genetic architectures of schizophrenia and other complex diseases using fast variance-components analysis. Nat. Genet. 47, 1385–1392 (2015).
Finucane, H. K. et al. Partitioning heritability by functional annotation using genome-wide association summary statistics. Nat. Genet. 47, 1228–1235 (2015).
Finucane, H. K. et al. Heritability enrichment of specifically expressed genes identifies disease-relevant tissues and cell types. Nat. Genet. 50, 621–629 (2018).
Adzhubei, I. A. et al. A method and server for predicting damaging missense mutations. Nat. Methods 7, 248–249 (2010).
Kumar, P., Henikoff, S. & Ng, P. C. Predicting the effects of coding non-synonymous variants on protein function using the SIFT algorithm. Nat. Protoc. 4, 1073–1081 (2009).
Choi, Y., Sims, G. E., Murphy, S., Miller, J. R. & Chan, A. P. Predicting the functional effect of amino acid substitutions and indels. PLoS ONE 7, e46688 (2012).
Shihab, H. A. et al. Predicting the functional, molecular, and phenotypic consequences of amino acid substitutions using hidden Markov models. Hum. Mutat. 34, 57–65 (2013).
Sabatti, C. et al. Genome-wide association analysis of metabolic traits in a birth cohort from a founder population. Nat. Genet. 41, 35–46 (2009).
Marouli, E. et al. Rare and low-frequency coding variants alter human adult height. Nature 542, 186–190 (2017).
Bulik-Sullivan, B. K. et al. LD Score regression distinguishes confounding from polygenicity in genome-wide association studies. Nat. Genet. 47, 291–295 (2015).
Zhu, Z. et al. Causal associations between risk factors and common diseases inferred from GWAS summary data. Nat. Commun. 9, 224 (2018).
Wood, A. R. et al. Defining the role of common variation in the genomic and biological architecture of adult human height. Nat. Genet. 46, 1173–1186 (2014).
Manolio, T. A. et al. Finding the missing heritability of complex diseases. Nature 461, 747–753 (2009).
Zuk, O., Hechter, E., Sunyaev, S. R. & Lander, E. S. The mystery of missing heritability: genetic interactions create phantom heritability. Proc. Natl Acad. Sci. USA 109, 1193–1198 (2012).
Karasik, D. & Kiel, D. P. Genetics of the musculoskeletal system: a pleiotropic approach. J. Bone Min. Res. 23, 788–802 (2008).
Blank, R. D. Bone and muscle pleiotropy: the genetics of associated traits. Clin. Rev. Bone Min. Metab. 12, 61–65 (2014).
Cruz-Jentoft, A. J. et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 48, 16–31 (2019).
Distefano, G. & Goodpaster, B. H. Effects of exercise and aging on skeletal muscle. Cold Spring Harb. Perspect. Med. 8, a029785 (2018).
Willer, C. J., Li, Y. & Abecasis, G. R. METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics 26, 2190–2191 (2010).
Jones, S. E. et al. Genome-wide association analyses of chronotype in 697,828 individuals provides insights into circadian rhythms. Nat. Commun. 10, 343 (2019).
Yengo, L. et al. Meta-analysis of genome-wide association studies for height and body mass index in approximately 700000 individuals of European ancestry. Hum. Mol. Genet. 27, 3641–3649 (2018).
McLaren, W. et al. The ensembl variant effect predictor. Genome Biol. 17, 122 (2016).
Liu, X., Jian, X. & Boerwinkle, E. dbNSFP v2.0: a database of human non-synonymous SNVs and their functional predictions and annotations. Hum. Mutat. 34, E2393–2402 (2013).
Yang, J., Lee, S. H., Goddard, M. E. & Visscher, P. M. GCTA: a tool for genome-wide complex trait analysis. Am. J. Hum. Genet. 88, 76–82 (2011).
Manichaikul, A. et al. Robust relationship inference in genome-wide association studies. Bioinformatics 26, 2867–2873 (2010).
Shungin, D. et al. New genetic loci link adipose and insulin biology to body fat distribution. Nature 518, 187–196 (2015).
Bigdeli, T. B. et al. A simple yet accurate correction for winner’s curse can predict signals discovered in much larger genome scans. Bioinformatics 32, 2598–2603 (2016).
Consortium, T. G. Human genomics. The Genotype-Tissue Expression (GTEx) pilot analysis: multitissue gene regulation in humans. Science 348, 648–660 (2015).
Fehrmann, R. S. et al. Gene expression analysis identifies global gene dosage sensitivity in cancer. Nat. Genet. 47, 115–125 (2015).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Pers, T. H. et al. Biological interpretation of genome-wide association studies using predicted gene functions. Nat. Commun. 6, 5890 (2015).
Zhu, Z. et al. Integration of summary data from GWAS and eQTL studies predicts complex trait gene targets. Nat. Genet. 48, 481–487 (2016).
Sun, B. B. et al. Genomic atlas of the human plasma proteome. Nature 558, 73–79 (2018).
Westra, H. J. et al. Systematic identification of trans eQTLs as putative drivers of known disease associations. Nat. Genet. 45, 1238–1243 (2013).
Conway, J. R., Lex, A. & Gehlenborg, N. UpSetR: an R package for the visualization of intersecting sets and their properties. Bioinformatics 33, 2938–2940 (2017).
de Leeuw, C. A., Mooij, J. M., Heskes, T. & Posthuma, D. MAGMA: generalized gene-set analysis of GWAS data. PLoS Comput. Biol. 11, e1004219 (2015).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl Acad. Sci. USA 102, 15545–15550 (2005).
Chang, C. C. et al. Second-generation PLINK: rising to the challenge of larger and richer datasets. Gigascience 4, 7 (2015).
Vilhjalmsson, B. J. et al. Modeling linkage disequilibrium increases accuracy of polygenic risk scores. Am. J. Hum. Genet. 97, 576–592 (2015).
Acknowledgements
This research was conducted using the UK Biobank resource under application number 41542. Y.F.P. and L.Z. were partially supported by funding from the National Natural Science Foundation of China (31771417 and 31571291), and by a project funded by the Priority Academic Program Development (PAPD) of Jiangsu Higher Education Institutions. Y.Z.L. was supported by GoMRI (Gulf of Mexico Research Initiative) grant G-23818 and NIH grants NIH/Fogarty 1U2RTW010673-01, R01 MH116844-01 and R01 AI147372-01. The numerical calculations in this paper were performed on the supercomputing system of the National Supercomputing Center in Changsha.
Author information
Authors and Affiliations
Contributions
Y.F.P. and L.Z. designed and supervised the study. L.Z. collected the data. Y.F.P., L.Z., X.L.Y. and H.Z. analysed the data. G.J.F. and X.T.W. performed the literature review and provided technical assistance. Y.F.P. and Y.Z.L. interpreted the data and drafted the early version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pei, YF., Liu, YZ., Yang, XL. et al. The genetic architecture of appendicular lean mass characterized by association analysis in the UK Biobank study. Commun Biol 3, 608 (2020). https://doi.org/10.1038/s42003-020-01334-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-020-01334-0
This article is cited by
-
Sarcopenia as a predictor of nutritional status and comorbidities: a cross-sectional and mendelian randomization study
BMC Geriatrics (2024)
-
Causal relationship of interleukin-6 and its receptor on sarcopenia traits using mendelian randomization
Nutrition Journal (2024)
-
Lipid metabolites and sarcopenia-related traits: a Mendelian randomization study
Diabetology & Metabolic Syndrome (2024)
-
The causal relationship of human blood metabolites with the components of Sarcopenia: a two-sample Mendelian randomization analysis
BMC Geriatrics (2024)
-
Appendicular lean mass and the risk of stroke and Alzheimer’s disease: a mendelian randomization study
BMC Geriatrics (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.