Abstract
C4 photosynthesis is characterised by a CO2 concentrating mechanism that operates between mesophyll and bundle sheath cells increasing CO2 partial pressure at the site of Rubisco and photosynthetic efficiency. Electron transport chains in both cell types supply ATP and NADPH for C4 photosynthesis. Cytochrome b6f is a key control point of electron transport in C3 plants. To study whether C4 photosynthesis is limited by electron transport we constitutively overexpressed the Rieske FeS subunit in Setaria viridis. This resulted in a higher Cytochrome b6f content in mesophyll and bundle sheath cells without marked changes in the abundances of other photosynthetic proteins. Rieske overexpression plants showed better light conversion efficiency in both Photosystems and could generate higher proton-motive force across the thylakoid membrane underpinning an increase in CO2 assimilation rate at ambient and saturating CO2 and high light. Our results demonstrate that removing electron transport limitations can increase C4 photosynthesis.
Similar content being viewed by others
Introduction
Crop yield gains achieved during the Green Revolution by conventional plant breeding were not based on photosynthetic traits1 and the theoretical maximum yield of the light conversion in crops is yet to be achieved2. Increasing photosynthetic efficiency by targeted genetic manipulation could potentially double the yield of crop plants3. Electron transfer reactions of photosynthesis (also called light reactions) supply ATP and NADPH essential for CO2 assimilation and therefore are a target for improvement4. It has been demonstrated in C3 plants that facilitating electron transport by overexpressing the components of electron transfer chain can result in higher assimilation rates5,6. As C4 plants play a key role in world agriculture with maize and sorghum being major contributors to world food production and sugarcane, miscanthus and switchgrass being major plant sources of bioenergy7, improvement of electron transport reactions could further increase the rates of C4 photosynthesis and yield8,9.
In C3 plants the electron transport chain is localised to the thylakoid membranes of mesophyll cells and consists of four major protein complexes: Photosystem II (PSII), Cytochrome b6f (cytb6f), Photosystem I (PSI) and ATP synthase. The first three complexes sustain linear electron flow from the water-oxidising complex of PSII to NADP+, the terminal acceptor of PSI, which is accompanied by acidification of the internal compartments of thylakoids (lumen). Transmembrane difference in electrochemical potentials of protons, or proton-motive force (pmf), serves as the driving force for ATP synthesis.
Cytb6f oxidises plastoquinol reduced by PSII and reduces plastocyanin, which then diffuses to PSI; plastoquinol oxidation is a rate-limiting step in the intersystem chain10. As a result of Q-cycle operating between the two binding sites of cytb6f, two protons are translocated from the stroma to the lumen per one electron11. Once pools of inorganic phosphate are exhausted, ATP synthesis slows down and causes a build-up of the pmf across the membrane12. Both components of pmf, the pH gradient (ΔpH) and the electrochemical gradient (ΔΨ), are equally capable of driving ATP synthesis13. However, the pH component has a major regulatory effect on the electron transport chain and at pH < 6 causes slowing down of the plastoquinol oxidation14 and down-regulation of PSII activity via non-photochemical quenching (NPQ). NPQ is a mechanism that triggers the attenuation of PSII activity by the dissipation of excess light energy in the form of heat in the light-harvesting antenna of PSII (LHCII)15. Fast, pH-dependent NPQ component in higher plants, qE, is regulated by the PHOTOSYSTEM II SUBUNIT S (PsbS) protein16 and xanthophyll cycle17.
Cytb6f forms a homodimer where each monomer consists of eight subunits: major subunits, Rieske FeS protein (PetC), Cytochrome b6 (PetB), Cytochrome f (PetA) and subunit IV (PetD), and minor subunits, PetG, PetL, PetM and PetN18. There is an increasing amount of evidence that the amount of Rieske FeS protein, one of the two nuclear-encoded subunits along with PetM, regulates the abundance of cytb6f6,19,20,21,22,23,24. Transgenic Arabidopsis thaliana plants overexpressing Rieske FeS showed an increase in the amounts of other cytb6f subunits, positive effects on PSII electron transport rate and CO2 assimilation rate and decreased NPQ6. Studies on transgenic tobacco plants indicate that cytb6f determines the rate of electron transport through the electron transport chain and concomitantly the CO2 assimilation rate19,20,21,22,23.
C4 photosynthesis is a biochemical CO2 concentrating pathway operating between mesophyll (M) and bundle sheath (BS) cells and there are three biochemical C4 subtypes25. PEP carboxylase (PEPC) catalyses primary carbon fixation in the cytoplasm of mesophyll cells into C4 acids. In C4 plants like maize, sorghum and setaria malate diffuses to the BS cells where it is decarboxylated inside chloroplasts by NADP-malic enzyme (NADP-ME) to provide CO2 for ribulose bisphosphate carboxylase oxygenase (Rubisco). Pyruvate resulting from malate decarboxylation diffuses back to mesophyll cells where it is regenerated into PEP by pyruvate ortophosphate dikinase. C4 species with NADP-ME biochemistry require a minimum of 1 NADPH and 2 ATP in mesophyll cells and 1 NADPH and 3 ATP in BS cells per one CO2 fixed8. The components of mesophyll electron transport chain are very similar to those described above for C3 plants but BS cells of NADP-ME C4 species are effectively supplied with NADPH via malate coming from the mesophyll cells and therefore are more specialised for ATP production. BS cells of NADP-ME species usually have little or no PSII activity and operate active cyclic electron flow (CEF) leading to the formation of pmf but not to NADP+ reduction26. There are two pathways for CEF: one via PROTON GRADIENT REGULATION 5 protein (PGR5), cytb6f and PSI, and another one via chloroplastic NAD(P)H:Quinone oxidoreductase 1-like complex (NDH complex), cytb6f and PSI27.
Since cytb6f is a component of electron transport chain in both mesophyll and BS cells, we used Setaria viridis, a model NADP-ME-type C4 plant, to study effects of constitutive Rieske FeS overexpression on C4 photosynthesis. We demonstrate that in both cell types increased abundance of Rieske FeS results in higher content of cytb6f and allows higher photosynthesis rates without notable changes of Rubisco and chlorophyll content. Our results indicate that in C4 plants electron transport is one of the limitations for CO2 assimilation, particularly at high light and non-limiting CO2 concentrations, and it is under cytb6f control.
Results
Generation of transgenic plants with Rieske overexpression
For Rieske FeS overexpression, the coding sequence of PetC gene from Brachypodium distachyon (BdPetC) was codon-optimised for the Golden Gate cloning system and assembled into two constructs (230 and 231) under the control of the maize ubiquitin promoter (Supplementary Fig. 1). The constructs were transformed into S. viridis using stable agrobacterium-mediated transformation. Eleven T0 transgenic plants were selected on the basis of hygromicin resistance and a subset of nine lines was analysed for Rieske FeS protein abundance, the presence of the BdPetC transcript and insertion numbers (Fig. 1a). Plants that went through the transformation process but tested negative for the insertion (“escapes”) were used as control for T0 plants and T1 progeny. Rieske FeS and Rubisco large subunit protein abundances per leaf area were highly variable between transgenic and control plants in the T0 generation (Fig. 1a), because T0 plants have been regenerated from the tissue culture at different times and thus varied substantially in age. Therefore three lines, 230(4), 231(3) and 231(6), were selected for further analysis of the T1 progeny based on the presence of the BdPetC transcript (Fig. 1a).
Selection of the transgenic S. viridis plants with Rieske FeS overexpression by immunodetection of Rieske FeS and Rubisco large subunit (RbcL) protein abundances on leaf area basis. a T0 lines selected based on hygromycin resistance after transformation with constructs 230 and 231; insertion numbers indicate copy numbers of the hygromycin phosphotransferase gene; (+), transcript of the PetC from B. distachyon (BdPetC) was detected; (−) BdPetC transcript was not detected. b T1 plants of three independent lines and their insertion numbers. c. Quantitative estimate of Rieske FeS abundance in T1 plants from the immunoblots shown in (b) relative to control plants (=1). The plants that went through the transformation process and tested negative for the hygromycin phosphotransferase gene (“escapes”) were used as control plants in the T0 and T1 generations. Positions of the molecular weight markers (kDa) are indicated on the left. Asterisks indicate plants selected for further analysis in the T2 generation. Uncropped images of the membranes used for immunodetection are available in Supplementary Fig. 5. Data points for the item c are available in Supplementary data 1
In the T1 generation, a quantitative estimate of the changes in Rieske FeS levels from the immunoblots showed that a number of transgenic plants contained increased amounts of Rieske FeS protein per leaf area compared to control plants with a maximum increase of 10–15% in 230(4)-7, 231(3)-3 and 231(6)-1 plants (Fig. 1b, c). Seeds from those plants were collected and used for the analysis of their T2 progeny. Interestingly, analysis of the BdPetC expression in the T1 generation of the line 230(4) (Supplementary Fig. 2) demonstrated that, although homozygous plants had about twofold higher transcript abundance compared to heterozygous plants, only heterozygous plants showed an increase of Rieske FeS protein level (Fig. 1b). Similar effect was observed in T1 plants of the line 231(3) and consequently also in the T2 progenies of the lines 230(4)-7 and 231(3)-3: only some heterozygous plants had increased Rieske FeS protein abundance (Supplementary Fig. 3). Moreover, Rieske FeS protein overexpression could only be detected when plants were grown at a low irradiance of 200 µmol m−2 s−1.
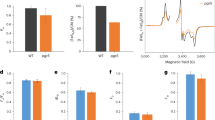
Transgenic plants with higher Rieske FeS protein abundance (FeS-OE hereafter) could be identified using pulse modulated chlorophyll fluorescence measurements of photochemical and non-photochemical processes at the incident light intensity with the MultispeQ (Table 1). In T2 progeny, FeS-OE plants, when compared to Zero(Null)-segregates control plants that are genetically identical to wild type, demonstrated significantly higher quantum yield of PSII (φPSII), a parameter estimating a proportion of the light absorbed by chlorophyll associated with PSII that is used in photochemistry, and a lower quantum yield of NPQ (φNPQ), i.e. a proportion of absorbed light actively dissipated in the PSII antennae. Electron transport parameters measured in transgenic plants harbouring the transgene but showing a control plant-level of Rieske FeS (“Control-level FeS” in Table 1) did not differ from control plants. Other parameters such as relative chlorophyll content, measured by Soil Plant Analysis Development (SPAD) chlorophyll metre, and leaf thickness did not differ between the three groups (Table 1).
Rieske FeS overexpression increases Cytochrome b 6 f content
Protein abundance of Rieske FeS and Cytochrome f subunits of cytb6f was analysed on leaf area basis in the T2 progeny of the line 230(4)-7 using specific antibodies (Fig. 2a). Both cytb6f subunits were significantly more abundant in FeS-OE plants relative to control plants (Fig. 2b). Immunoblotting with specific antibodies against representative subunits of other thylakoid complexes suggested that there were no significant differences in abundances of PSI, PSII, ATP synthase, NDH complex and light-harvesting complexes of PSI and PSII between FeS-OE and control plants on the leaf level (Fig. 2a, b). In addition, no changes in PsbS abundance were detected. Abundances of Rubisco large subunit (RbcL) and PEPC in FeS-OE plants were also similar to the levels detected in control plants (Fig. 2a, b). In line with the immunoblotting results, amounts of Rubisco active centers in FeS-OE and control plants, estimated by [14C] carboxyarabinitol bisphosphate binding assay, did not differ and neither did leaf chlorophyll (a + b) content and chlorophyll a/b ratio (Table 1).
Relative abundance of photosynthetic proteins and Cytochrome b6f complex in plants with Rieske FeS overexpression (FeS-OE) and control plants. a Western blots with antibodies against Rieske FeS, Cyt f (Cytochrome f), RbcL (Rubisco large subunit), PsaB (Photosystem I), D1 (Photosystem II), NdhH (NDH complex), AtpB (ATP synthase), Lhca1 (light-harvesting complex of Photosystem I), Lhcb2 (light-harvesting complex of Photosystem II), PsbS (Photosystem II subunit S) and PEPC (PEP carboxylase). Positions of the molecular weight markers (kDa) are indicated on the left. b Quantification of protein abundances in FeS-OE plants on leaf area basis relative to control plants (=1). Mean ± SE, n = 3 biological replicates. Asterisks indicate statistically significant differences between transgenic and control plants (P < 0.05). c Blue Native gel electrophoresis of the thylakoid protein complexes isolated from mesophyll (M) and bundle sheath (BS) cells; 10 µg of chlorophyll (a+b) loaded in each lane. d Immunodetection of Cytochrome b6f complex from the Blue-Native gel with Rieske FeS antibodies; Cytb6f and Cytb6f*—two distinct forms of the complex detected. Zero(Null)-segregates were used as control plants. Uncropped images of the membranes used for immunodetection are available in Supplementary Fig. 6. Data points for the item b are available in Supplementary data 1
To see if the higher level of Rieske FeS protein resulted in increased abundance of the whole cytb6f complex, thylakoid protein complexes from the BS and mesophyll cells of FeS-OE and control plants were separated using Blue Native gel electrophoresis. No major changes were found between transgenic and control plants in composition of complexes within the thylakoid membranes of each cell type (Fig. 2c). Thylakoid protein complexes were probed with Rieske FeS antibodies to obtain the relative abundance of cytb6f. Whilst one band matching the cytb6f band reported previously28 was detected in the mesophyll thylakoids, in the BS thylakoids two bands were recognised by the Rieske FeS antibodies: a minor one with a similar molecular weight to the mesophyll cytb6f and a major one with a slightly higher molecular weight (cytb6f*) (Fig. 2d). Relative abundance of cytb6f was higher in the mesophyll thylakoids and cytb6f* was more abundant in the BS thylakoids of FeS-OE plants compared to control plants (Fig. 2d).
Rieske FeS overexpression increases CO2 assimilation rate
CO2 assimilation rate was measured in the T1 plants of the lines 230(4), 231(3) and 231(6) and the T2 progenies of the lines 230(4)-7, 231(3)-3 and 231(6)-1 at 1500 µmol m−2 s−1 and CO2 of 400 ppm and plotted against the relative abundance of Rieske FeS protein on leaf area basis estimated from the immunoblots shown on Fig. 1b, Fig. 2a and Supplementary Fig. 3. All three lines showed a positive correlation between Rieske FeS abundance and CO2 assimilation rate, suggesting that higher abundance of cytb6f supported higher rates of C4 photosynthesis (Fig. 3).
Relationships of Rieske FeS abundance and CO2 assimilation rate in T1 (filled symbols) and T2 (open symbols) plants of three independent lines relative to control plants. Rieske FeS abundance was quantified from the immunoblots shown on the Fig. 1b, Fig. 2a and Supplementary Fig. 3. Steady-state CO2 assimilation rates were measured at 1500 µmol m−2 s−1 and CO2 partial pressure of 400 ppm. The plants that went through the transformation process and tested negative for the hygromycin phosphotransferase gene were used as control for T1 plants and Zero(Null)-segregates were used as control for T2 plants. Average rates of CO2 assimilation measured from control plants were 26.47 µmol m−2 s−1 for T1 plants and 32.65 µmol m−2 s−1 for T2 plants. Uncropped images of the membranes used for immunodetection are available in Supplementary Fig. 5, Supplementary Fig. 6 and Supplementary Fig. 7. Data points for this item are available in Supplementary data 2
To study the effect of Rieske FeS overexpression on CO2 assimilation rate in more detail, the CO2 response of assimilation was examined at a constant irradiance of 1500 µmol m−2 s−1 in the T2 progenies of the lines 230(4)-7 and 231(3)-3. At lower intercellular CO2 partial pressure no differences in assimilation rates were found between two FeS-OE lines and control plants (Fig. 4a, Supplementary Fig. 4). However, at Ci above 150 µbar FeS-OE plants had higher CO2 assimilation rates with a significant increase at the Ci above 250 µbar. Maximum CO2 assimilation rates reached by FeS-OE plants of the line 230(4)-7 and control plants at high Ci were 41.30 ± 0.89 and 38.14 ± 0.81 µmol m−2 s−1 (mean ± SE, n = 3, P = 0.049), respectively, indicating about 8% increase of photosynthesis in FeS-OE plants (Fig. 4a). FeS-OE plants of the line 231(3)-3 also demonstrated about 10% increase of CO2 assimilation rates at ambient and high CO2 (Supplementary Fig. 4). CO2 response of φPSII measured in FeS-OE and control plants showed similar trends: no difference at low Ci but significantly higher φPSII in FeS-OE plants at Ci above 250 µbar (Fig. 4b). Stomatal conductance in FeS-OE plants estimated from the gas-exchange measurements was higher at Ci above 250 µbar but not significantly (0.05 < P < 0.15) (Fig. 4c).
Gas-exchange and fluorescence analysis of plants with Rieske FeS overexpression (FeS-OE) and control plants. a–c CO2 response of assimilation rate (A), quantum yield of Photosystem II (φPSII) and stomatal conductance (gsw) at 1500 µmol m−2 s−1. d–f Light response of A, φPSII and gsw at CO2 partial pressure of 400 ppm. Mean ± SE, n = 3 biological replicates. Asterisks indicate statistically significant differences between FeS-OE and control plants (P < 0.05). Results are for the T2 progeny of the line 230(4)-7, results for the T2 progeny of the line 231(3)-3 are shown on Supplementary Fig. 4. Zero(Null)-segregates were used as control plants. Data points are available in Supplementary data 3
The light response of CO2 assimilation was examined at a constant CO2 of 400 ppm. Assimilation rates did not differ between FeS-OE plants of the line 230(4)-7 and control plants at the range of irradiances below 1500 µmol m−2 s−1 (Fig. 4d). However at light levels above 1500 µmol m−2 s−1, FeS-OE plants had significantly higher assimilation rates compared to control plants with a maximum increase of about 8%. φPSII measured concurrently was significantly higher in FeS-OE plants compared to control plants over the range of irradiances above 200 µmol m−2 s−1 (Fig. 4e) whilst stomatal conductance did not differ in transgenic plants independently of irradiance (Fig. 4f).
Rieske FeS overexpression plants have lower NPQ
NPQ as well as photochemical and non-photochemical yields of PSI at different irradiances were analysed in FeS-OE plants of the line 230(4)-7 and control plants with the Dual-PAM-100. FeS-OE plants had significantly lower NPQ at the irradiances above 220 µmol m−2 s−1 (Fig. 5a). The effective quantum yield of PSI (φPSI) in FeS-OE plants was significantly higher compared to control plants at the irradiances above 57 µmol m−2 s−1 (Fig. 5b). φND, a non-photochemical loss due to the oxidised primary donor of PSI, was significantly lower in FeS-OE plants compared to control plants at the range of irradiances between 57 and 220 µmol m−2 s−1 (Fig. 5c). φNA, a non-photochemical loss due to the reduced PSI acceptor, did not differ significantly between FeS-OE and control plants but FeS-OE showed a tendency to lower φNA at high irradiances (0.1 > P > 0.05) (Fig. 5d).
Light response of photosynthetic parameters in plants with Rieske FeS overexpression (FeS-OE) and control plants. a NPQ, non-photochemical quenching. b φPSI, quantum yield of PSI. c φND, non-photochemical loss due to the oxidised primary donor of PSI. d φNA, non-photochemical loss due to the reduced PSI acceptor. e pmf, proton-motive force, estimated from the electrochromic shift signal. f ∆ψ, electrochemical gradient component of pmf. g ∆pH, proton gradient component of pmf. h gH+, proton conductivity of the thylakoid membrane. Results are for the T2 progeny of the line 230(4)-7. Mean ± SE, n = 3 biological replicates. Asterisks indicate statistically significant differences between FeS-OE and control plants (P < 0.05). Zero(Null)-segregates were used as control plants. Data points are available in Supplementary data 4
Dark-interval relaxation of the electrochromic shift signal has been recorded to analyse the generation of pmf in leaves at various irradiances. Total pmf was significantly higher in FeS-OE plants at 1287 µmol m−2 s−1 (Fig. 5e). Decomposition of pmf into electrochemical gradient (ΔΨ) and proton gradient (ΔpH) components revealed that the higher pmf at 1287 µmol m−2 s−1 was due to the higher ΔpH (Fig. 5f, g). Fitting of the first-order relaxation kinetics of electrochromic shift signal showed that there was no difference in thylakoid proton conductivity (gH+) between FeS-OE and control plants irrespective of irradiance (Fig. 5h).
Light-induced changes of photosynthetic parameters were studied on dark-adapted leaves over the course of illumination with red actinic light of 220 µmol m−2 s−1 (Fig. 6). During the first three minutes of illumination, FeS-OE plants had significantly lower NPQ indicating a slower induction of qE compared to control plants whilst no difference in NPQ relaxation kinetics was observed in darkness. Lower NPQ in FeS-OE plants was accompanied by the higher φPSII during the first minutes of irradiance whilst φPSI was higher in FeS-OE plants over the whole course of illumination compared to control plants (Fig. 6).
Photosynthetic parameters of plants with Rieske FeS overexpression (FeS-OE) and control plants during dark/light transitions measured on dark-adapted leaves. a Induction and relaxation of NPQ during dark-light-dark shift: black bars, darkness; white bar, red actinic light of 220 µmol m−2 s−1. b, c Dynamic changes of φPSII and φPSI, quantum yields of Photosystem II and Photosystem I, during first minutes of illumination with red actinic light of 220 µmol m−2 s−1. Results are for the T2 plants of the line 230(4)-7. Mean ± SE, n = 3 biological replicates. Asterisks indicate statistically significant differences between FeS-OE and control plants (P < 0.05). Zero(Null)-segregates were used as control plants. Data points are available in Supplementary data 5
Discussion
C4 plants already have high photosynthetic rates when compared to C3 plants which is achieved by operating a biochemical carbon concentrating mechanism, thereby reducing photorespiratory energy losses and allowing Rubisco to operate close to its maximum capacity. However, further enhancing C4 photosynthesis could have the potential for major agricultural impact. Maize, sorghum and sugarcane are among the most productive crops and all three belong to the NADP-ME biochemical subtype. Here, we used the closely related model NADP-ME species S. viridis to study the effect of increased electron transport capacity on C4 photosynthesis. Since cytb6f controls electron transport in C3 plants and its abundance is likely determined by the abundance of Rieske FeS subunit6,19, we attempted to accelerate electron transport rate in S. viridis by overexpressing Rieske FeS. As shown by immunodetection of the thylakoid protein complexes isolated from mesophyll and BS cells, constitutive overexpression of Rieske FeS protein in S. viridis resulted in higher abundance of the complete cytb6f complex in both cell types (Fig. 2d).
The role of cytb6f in the regulation of electron transport is defined by its position between the two photosystems. PSII activity can be controlled by cytb6f levels either by limiting the rate of plastoquinol oxidation and hence oxidation of the primary electron acceptor QA or by down-regulation of photochemical efficiency via NPQ. Plants generated here with more cytb6f had higher effective quantum yields of PSII at irradiances above 200 µmol m−2 s−1 which indicated that a higher portion of absorbed light reached PSII reaction centres (Fig. 4e). Lower NPQ measured in plants with higher cytb6f abundance at those irradiances suggested that the increase in φPSII was attributed to the lower loss of energy via thermal dissipation (Fig. 5a). Both observations suggested that in FeS-OE plants QA was more oxidised and linear electron flow was less limited by the rate of plastoquinol oxidation. Since in NADP-ME monocots mesophyll cells contain a much higher portion of PSII than BS cells (95–98%29), leaf level changes in PSII light-use efficiency can be attributed predominantly to mesophyll cells suggesting a higher capacity for whole chain electron transport in mesophyll cells. Steady-state electron flow measurements in leaves under the growth irradiance also confirmed the increased capacity for the linear electron flow in FeS-OE plants (Table 1).
PSI does not have an effective repair mechanism, such as the D1 protein turnover process of PSII30, and when the transfer of electrons to PSI exceeds the capacity of the stromal acceptors, PSI photoinhibition causes long-term inhibitory effects on electron transport and carbon fixation31,32. Cytb6f determines the amount of electrons reaching PSI from the intersystem chain, especially at low irradiance when CEF is mostly inactive33. In line with that, at irradiances below 340 µmol m−2 s−1 higher abundance of cytb6f increased electron supply to the donor side of PSI (Fig. 5c) which resulted in the higher effective quantum yield of PSI (Fig. 5b).
Calculations of electron transport rates through photosystems are complicated in C4 plants because of more complex distribution of light energy between PSI and PSII in the two-cell system. However, we attribute more efficient use of absorbed light for photochemical reactions in both photosystems to a “release” of cytb6f control in both mesophyll and BS cells. In C3 A. thaliana plants overexpressing Rieske FeS, increased φPSI and φPSII were also accompanied by increased abundances of all electron transport complexes6. In FeS-OE plants generated here, due to unaltered abundances of other photosynthetic complexes (Fig. 2b), higher φPSI and φPSII clearly demonstrate the effect of the cytb6f control on whole leaf electron transport.
In C3 plants, the rate of CEF around PSI increases at high light and generates a higher ∆pH which consequently decreases light harvesting efficiency via generation of a higher NPQ34. In line with CEF increasing electron supply to the donor side of PSI35, no difference in electron flux on the donor side of PSI was detected in plants with higher cytb6f abundance at higher irradiance (Fig. 5c). Instead, a higher yield of PSI detected at higher irradiance corresponded to the increased availability of PSI electron acceptors (Fig. 5d). It has been suggested that heme ci of cytb6f exposed to the stroma may be involved in the PGR5-mediated CEF route36 and cytb6f, PSI and PGR5 can form a super-complex together with ferredoxin, ferredoxin:NADPH oxidoreductase and PROTON GRADIENT REGULATION5-like137. If this is the case, higher abundance of cytb6f could directly increase the rate of PGR5-mediated CEF and contribute to alleviating PSI acceptor side limitation. During the C3–C4 evolutionary transition increased PGR5 abundance appears to have been favoured in both cell types38 and therefore stimulatory effect of Rieske FeS overexpression on PGR5-mediated CEF could be valid for both mesophyll and BS cells.
C4 plants typically grow in high light environments which allow them to accommodate the higher ATP requirements of the pathway39. Higher pmf generated by the plants with increased cytb6f abundance at high irradiance (Fig. 5e,g), conceivably, indicated a higher capacity for ATP production and could underpin higher assimilation rates (Fig. 4d). Since the effect of cytb6f abundance on pmf was most prominent above 825 µmol m−2 s−1, this irradiance might be a prerequisite for a realisation of increased photosynthesis in C4 plants.
Since the electron transport chain in BS cells is specifically optimised for active CEF27, higher proton-motive force (pmf) and ∆pH detected in plants with higher cytb6f abundance at high light (Fig. 5e, Fig. 5g) could be present in BS cells. The NDH-mediated route of CEF is considered to be prevalent in bundle sheath cells of NADP-ME plants40. Similar protein abundance of the NdhH subunit between FeS-OE and control plants (Fig. 2b) suggested that CEF activity in BS cells could be regulated also at the level of cytb6f abundance. Lateral heterogeneity of the thylakoid membranes where PSII is localised to the stacked grana and PSI to the stromal lamellae41 provides the basis for functional specialisation of cytb6f. Since about 55% of cytb6f is found in appressed grana and 45% is distributed over the stromal lamellae42, these two fractions of cytb6f might be more specific for either linear or CEF. In line with this we detected cytb6f*complex (Fig. 2c) in BS cells of S. viridis that might represent a CEF-specific cytb6f potentially forming a complex with its electron transport-partners.
Our results suggest that lower NPQ detected in plants with increased cytb6f levels underpinned more efficient use of light energy that could result in higher assimilation rates. Remarkably, higher abundance of Rieske FeS resulted in slower NPQ induction in both C36 and C4 plants (Fig. 6) showing an apparent effect on pH-dependent qE component of NPQ. Moreover, we demonstrate that FeS-OE plants generated the same or higher levels of pmf (and ∆pH) compared to control plants despite the decreased NPQ (Fig. 5e). Plants with higher cytb6f levels retained linear relationships between pmf and NPQ, but showed an offset in the NPQ response (Fig. 7).
Regulation of ATP synthase is thought to play a key role in modulating pmf and NPQ43. However the effect of Rieske FeS overexpression presented on Fig. 7 was not due to changes in thylakoid proton conductivity (Fig. 5h). In addition, no decrease in violaxanthin content could be detected in A.thaliana plants overexpressing Rieske FeS6 and no difference in PsbS abundance was found between FeS-OE and cotrol plants in this work (Fig. 2b). Although detailed characterisation of the cell-specific changes in FeS-OE plants is required to clarify whether there are alterations in ratios of protein abundance and/or activity of the regulatory mechanisms between mesophyll and BS cells, our observations suggest that there might be other mechanisms regulating NPQ in FeS-OE plants. PGR5-mediated CEF might serve also for redox poising of the electron transport chain44 to prevent the reduction of QA and the closure of PSII centres. If higher cytb6f abundance stimulated this pathway, it could result in a sustained change in the redox state of the QA(i.e. constantly 10% more oxidised QA) and hence a corresponding offset of the NPQ response in FeS-OE plants (Fig. 7). While lower NPQ might be detrimental for C3 plants in high light conditions, due to higher availability of CO2 as terminal electron acceptor, C4 photosynthesis is more resistant to photo-damage by high irradiance8 and the “release” of cytb6f control could have positive effects on photosynthesis without photoinhibitory effects on photosystems.
At low intercellular CO2 (Ci), the assimilation rate is largely limited by the rate of primary CO2 fixation in mesophyll cells45, whilst at high Ci, according to the C4 photosynthesis model46,47, assimilation rate is co-limited by the amount and activity of Rubisco, the capacity for regeneration of either PEP or ribulose bisphosphate and the rate of whole leaf electron transport (the sum of electron transport rates of the two cell types). In line with the model, saturating rates of CO2 assimilation were about 8% higher in plants with about 10% higher Rieske FeS abundance (and hence with higher electron transport rate as discussed above). When measured in the steady-state, there was a strong positive correlation between Rieske FeS abundance and CO2 assimilation rate at ambient Ci in three independent transgenic lines (Fig. 3).
While very little work has been done on improving C4 photosynthesis by genetic modification, maize plants overexpressing both subunits of Rubisco together with the chaperonin RUBISCO ASSEMBLY FACTOR 1 demonstrated higher assimilation rates at high Ci and high light48. Importantly, since plants with higher cytb6f abundance examined here did not have higher Rubisco content, both abundance of Rubisco and cytb6f are likely co-limiting factors of C4 photosynthesis under high light and non-limiting CO2, in accordance with the C4 model predictions.
Although increasing electron transport by means of Rieske FeS overexpression is a viable approach for improving C4 photosynthesis, our results also demonstrate that Rieske FeS abundance is tightly regulated on a post-transcriptional level since no increase of protein abundance was detected in homozygous plants with high transcript levels (Fig. 1b, Supplementary Fig. 2). Moreover, overexpression of Rieske FeS in S. viridis could only be detected when plants were grown at low irradiance of 200 µmol m−2 s−1. Due to the long cytb6f lifetime of at least one week and fast light-induced degradation of its unassembled components49, cytb6f biogenesis is practically restricted to a short period during the growth of young leaves. Using leaf-specific promoters and overexpressing all cytb6f subunits simultaneously might allow these problems to be overcome by ensuring the presence of all parts required for cytb6f assembly at the right time.
In conclusion, we provide evidence that cytb6f exerts a high level of control over the rate of electron transport in both mesophyll and BS cells in the C4 model plant S. viridis. We have demonstrated that increasing cytb6f content can help improve light conversion efficiency in both photosystems and support generation of higher proton-motive force providing the basis for higher CO2 assimilation rates. Our results support the idea that C4 photosynthesis at ambient and high CO2 and high irradiance is co-limited by the electron transport rate. Engineering C4 crops with higher cytb6f abundance may result in higher CO2 assimilation rates and higher yield.
Methods
Construct generation
Two constructs, 230 and 231, were created using the Golden Gate MoClo Plant Parts Kit50 (Supplementary Fig. 1). The Golden Gate cloning system allows assembly of multiple independent transcription modules in a plant binary vector pAGM4723. In both constructs the first expression module was occupied by the hygromycin phosphotransferase gene (hpt) driven by the rice actin promoter (pOsAct). The second module in both constructs was taken by the coding sequence of PetC gene encoding for the Rieske FeS subunit of the Cytochrome b6f complex from Brachypodium distachyon domesticated for the Golden Gate (Supplementary Fig. 1) and driven by the maize ubiquitin promoter (pZmUbi). Construct 231 contained a third expression module with the coding sequence of the cyan fluorescent protein mTurquoise driven by the 2x35S promoter. The bacterial terminator tNos was used in all transcription modules. Both constructs were verified by sequencing and transformed into Agrobacterium tumefaciens strain AGL1 to proceed with stable plant transformation.
Generation and selection of transgenic plants
Stable agrobacterium-mediated transformation of S. viridis (accession A10.1) tissue culture was performed as described in Osborn et al.51. Hygromycin-resistant plants were transferred to soil and grown in control environmental chambers with 2% CO2, the irradiance of 200 µmol m−2 s−1, 16 h photoperiod, 28 °C day, 20 °C night and 60% humidity. T0 plants were analysed for the hpt gene copy number by digital droplet PCR (iDNA genetics, UK) as described in Osborn et al51. The plants that went through the transformation process and tested negative for the hpt gene were used as control for T0 and T1 generations. Zero(Null)-segregates were used as control for T2 plants. T1 and T2 progenies were analysed with digital droplet PCR to confirm the presence of insertions. MultispeQ v1.0 leaf photosynthesis V1.0 protocol was used for a fast screening of transgenic plants for altered photosynthetic properties52. MultispeQ data were analysed with the PhotosynQ web application (https://photosynq.org).
Plant growth conditions
The T1 progenies of T0 plants were analysed to select the lines with Rieske FeS overexpression. T1 seeds were sterilised and germinated on medium containing 2.15 g L−1 Murashige and Skoog salts, 10 mL L−1 100× Murashige and Skoog vitamins stock, 30 g L−1 sucrose, 7 g L−1 Phytoblend, 20 mg L−1 hygromycin (pH 5.7). Seedlings that were able to develop secondary roots were transferred to 1 L pots with garden soil mix fertilised with 1 g L−1 Osmocote (Scotts, Bella Vista, Australia). Plants were grown in controlled environmental chambers under the 16 h photoperiod, 28 °C day, 20 °C night, 60% humidity and ambient CO2 concentrations. Light at the intensity of 200 µmol m−2 s−1 was supplied by 1000 W red sunrise 3200 K lamps (Sunmaster Growlamps, Solon, OH). Seeds of the T1 plants with confirmed Rieske FeS overexpression were collected to be analysed in the T2 generation. T2 seeds were incubated in 5% liquid smoke (Wrights, B&G foods, Parsippany, NJ) overnight and germinated in 1 L pots with garden soil mix layered on top with 2 cm seed raising mix (Debco, Tyabb, Australia) both containing 1 g L−1 Osmocote. Youngest fully expanded leaves of the 3–4 weeks plants were used for all analyses. Plants of different genotypes were placed randomly in growth chambers to reduce any position effects.
Gas exchange measurements
Rates of CO2 assimilation were measured over a range of intercellular CO2 partial pressure and irradiances simultaneously with chlorophyll fluorescence using a portable gas-exchange system LI-6800 (LI-COR Biosciences, Lincoln, NE) and a Fluorometer head 6800-01 A (LI-COR Biosciences). Leaves were first equilibrated at 400 ppm CO2 in the reference side, an irradiance of 1500 µmol m−2 s−1, leaf temperature 28 °C, 60% humidity and flow rate 300 µmol s−1. CO2 response curves were conducted under the constant irradiance of 1500 µmol m−2 s−1 by imposing a stepwise increase of CO2 concentrations from 0 to 1600 ppm at 3 min intervals. Light response curves were measured at constant CO2 partial pressure of 400 ppm in the reference cell under a stepwise increase of irradiance from 0 to 3000 µmol m−2 s−1 at 2 min intervals. Red-blue actinic light (90%/10%) was used in all measurements.
Protein isolation and Western blotting
To isolate proteins from leaves, leaf discs of 0.71 cm2 were collected and frozen immediately in liquid N2. One disc was ground in ice-cold glass homogeniser in 0.5 mL of protein extraction buffer: 100 mM trisaminomethane-HCl, pH 7.8, supplemented with 25 mM NaCl, 20 mM ethylenediaminetetraacetic acid, 2% sodium dodecyl sulfate (w/v), 10 mM dithiothreitol and 2% (v/v) protease inhibitor cocktail (Sigma, St Louis, MO). Protein extracts were incubated at 65 °C for 10 min and then centrifuged at 13,000 g for 1 min at 4 °C to obtain clear supernatant. Protein extracts were supplemented with 4× XT Sample buffer (BioRad, Hercules, CA), loaded on leaf area basis and separated by polyacrylamide gel electrophoresis [Nu-PAGE 4–12% Bis-(2-hydroxy-ethyl)-amino-tris(hydroxymethyl)-methane (Bis-Tris) gel, Invitrogen, Life Technologies Corporation, Carlsbad, CA] in running buffer (pH 7.3) containing 50 mM 2-(N-morpholino)ethanesulfonic acid, 50 mM trisaminomethane, 0.1% sodium dodecyl sulfate (w/v), 20 mM ethylenediaminetetraacetic acid. Proteins were transferred to a nitrocellulose membrane and probed with antibodies against various photosynthetic proteins purchased from Agrisera (Vännäs, Sweden) in dilution recommended by the producer: PetC (Agrisera, AS08 330), PsaB (Agrisera, AS10 695), D1 (Agrisera, AS10 704), NdhH (Agrisera, AS16 4065), AtpB (Agrisera, AS05 085), Lhca1 (Agrisera, AS01 005), Lhcb2 (Agrisera, AS01 003), PsbS (Agrisera, AS09 533), PEPC (Agrisera, AS09 458), RbcL (Agrisera, AS03 037), Cyt f (Agrisera, AS08 306). Quantification of immunoblots was performed with Image Lab software (Biorad, Hercules, CA).
Thylakoid isolation and Blue Native gel electrophoresis
These procedures were done in dim light and at 4 °C (or on ice) to reduce light-induced damage of isolated thylakoid complexes. Ten youngest fully expanded leaves were collected from one plant and midribs were removed. Leaves were cut into 3 mm pieces and ground in 100 mL of ice-cold grinding buffer (50 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (Hepes)-NaOH, pH 7.5, 330 mM sorbitol, 5 mM MgCl2) in Omni Mixer (Thermo Fisher Scientific, Tewksbury, MA) at the intensity #10 for 2 s. This fast treatment was used to break only mesophyll cells that don’t have suberised cell walls. The homogenate was passed through the 80-µm nylon filter and the filtrate containing mesophyll suspension was collected. All tissues collected on the filter were again homogenised in 100 mL of grinding buffer in Omni Mixer during three 10-s cycles at the intensity #7. The homogenate was passed through a tea strainer and then BS strands from the filtrate were collected on the 80-µm nylon filter. BS strands were further ground in 10 mL of grinding buffer in an ice-cold glass homogeniser.
Mesophyll and BS suspensions were filtered through a layer of Miracloth (Merck Millipore, Burlingtone, MA) and centrifuged at 6000 rpm, 4 °C for 5 min. Pellets were first resuspended in ice-cold shock buffer (50 mM Hepes-NaOH, pH 7.5, 5 mM MgCl2) and centrifuged again. Second time pellets were resuspended in ice-cold storage buffer (50 mM Hepes-NaOH, pH 7.5, 100 mM sorbitol, 10 mM MgCl2) and centrifuged again. Finally pellets were resuspended in an equal aliquot of the storage buffer, snap-frozen in liquid N2 and stored at −80 °C.
For Blue Native gel electrophoresis aliquots of thylakoid samples containing 10 µg chlorophyll (a+b) were taken for solubilisation. Aliquots were centrifuged at 6000 rpm, 4 °C for 5 min and then thylakoids were resuspended in ice-cold sample buffer (25 mM BisTris-HCl, pH 7.0, 20% (w/v) glycerol, 2.5% (v/v) protease inhibitor cocktail) to obtain chlorophyll (a+b) concentration of 1 µg µL−1. An equal volume of 2% (w/v) of n-dodecyl β-D-maltoside in sample buffer was added to thylakoids and the mixture was incubated for 5 min in darkness at 4 °C. Traces of unsolubilised material were removed by centrifugation at 14,000 × g at 4 °C for 30 min. Supernatants were supplemented with 10% (v/v) of Serve Blue G buffer (100 mM BisTris-HCl, pH 7.0, 0.5 M e-amino-n-caproic acid, 30% (w/v) sucrose, 50 mg mL−1 Serva Blue G) and applied to the precast native 3–12% Bis-Tris polyacrylamide gel (Invitrogen, Life Technologies Corporation, Carlsbad, CA). Blue Native gel electrophoresis was run using cathode and anode buffers and voltage regime according to Rantala et al.53 The gel was scanned, then incubated for 30 min in transfer buffer (25 mM trisaminomethane, 25 mM glycine, 20% methanol, 0.1% sodium dodecyl sulphate) and blotted to a nitrocellulose membrane. Western blotting was then performed as usually.
Chlorophyll and Rubisco assays
For chlorophyll analysis, frozen leaf discs were ground using the Qiagen TissueLyser II (Qiagen, Venlo, The Netherlands) and total chlorophyll was extracted in 80% acetone buffered with 25 mM Hepes-KOH. Chlorophyll a and b contents were measured at 750.0 nm, 663.3 nm and 646.6 nm, and calculated according to Porra et al.54. Amount of Rubisco active sites was estimated by [14C] carboxyarabinitol bisphosphate binding assay as described in Ruuska et al.22
Electron transport and electrochromic shift
Effective quantum yield of PSII (φPSII)55 was probed simultaneously with the gas-exchange measurements under red-blue actinic light (90%/10%) using multiphase saturating flash of 8000 µmol m−2 s−1. NPQ as well as effective yields of photochemical and non-photochemical reactions in PSI were measured with Dual-PAM-100 (Heinz Walz, Effeltrich, Germany) under red actinic light with 300-ms saturating pulses of 10000 µmol m−2 s−1. The effective quantum yield of PSI (φPSI), the non-photochemical yield of PSI caused by donor side limitation (φND) and the non-photochemical yield of PSI caused by acceptor side limitation of PSI (φNA) were calculated as described earlier56. To monitor light-induced response of photosynthetic parameters, leaves were dark-adapted for 30 min to record F0 and FM, the minimal and maximal levels of fluorescence in the dark, respectively. Afterwards saturating pulse was given after pre-illumination with far-red light to record PM, the maximal level of P700 oxidation, and P0, the minimal P700 signal, after the pulse. Next, photosynthetic parameters were monitored by the saturating pulse application every 45 s, first for 8 min under actinic light of 220 µmol m−2 s−1 and afterwards for 5 min in darkness to record NPQ relaxation. After that photosynthetic parameters were assessed in the same leaf over a range of irradiances from 0 to 1287 µmol m−2 s−1 at 1 min intervals.
The electrochromic shift signal was monitored as the absorbance change at 515 nm using Dual-PAM-100 equipped with the P515/535 emitter-detector module (Heinz Walz, Effeltrich, Germany). Leaves were first dark adapted for 10 min and the absorbance change induced by a single turnover flash (ECSST) was measured. Dark-interval relaxation of electrochromic shift signal was recorded during 3 min in darkness after 3-min intervals of illumination with actinic light at a stepwise increasing irradiance from 0 to 1287 µmol m−2 s−1. Total proton-motive force (pmf) was estimated from the amplitude of the rapid decay of the electrochromic shift signal and slow relaxation of the signal showed the contribution of proton gradient (ΔpH) and electrochemical gradient (ΔΨ) across the thylakoid membrane57,58. Pmf levels and the magnitudes of ΔpH and ΔΨ were normalised against the ECSST. Proton conductivity (gH+) of the thylakoid membrane through the ATP synthase was calculated as an inverse of the time constant obtained by fitting the first-order electrochromic shift relaxation59.
RNA isolation and quantitative real-time PCR (qPCR)
Frozen leaf discs (0.71 cm2) were ground using the Qiagen TissueLyser II and RNA was extracted using the RNeasy Plant Mini Kit (Qiagen, Venlo, The Netherlands). DNA from the samples was removed using the Ambion TURBO DNA free kit (Thermo Fisher Scientific, Tewksbury, MA), and RNA quantity and quality were determined using a NanoDrop (Thermo Fisher Scientific, Tewksbury, MA). One microgram of RNA was reverse transcribed into cDNA using SuperScript™ III Reverse Transcriptase (Thermo Fisher Scientific, Tewksbury, MA). qPCR and melt curve analysis were performed on a Viia7 Real-time PCR system (Thermo Fisher Scientific, Tewksbury, MA) using the Power SYBR green PCR Master Mix (Thermo Fisher Scientific, Tewksbury, MA) according to the manufacturer’s instructions. Primer pairs to distinguish between the PetC gene transcript from S. viridis and B. distachyon were designed using Primer3 in Geneious R9.1.1 (https://www.geneious.com): GCTGGGCAACGACATCAAG and CAAAGGAACTTGTTCTCGGC for SvPetC, GGCTCCGGGAGCAACAC and CAAAGGAACTTGTTCTCGGC for BdPetC. Relative fold change was calculated by the ΔΔCt method, using the geometric mean of the Ct values for three reference genes described in Osborn et al.51.
Statistics and reproducibility
One-way ANOVA was performed for electron transport parameters measured with MultispeQ using the PhotosynQ web application (https://photosynq.org). Measurements collected with MultispeQ at significantly different light intensities were excluded, otherwise no data were excluded from the analysis. For other measurements the relationship between mean values for transgenic and control plants were tested using a two-tailed, heteroscedastic Student’s t-test (Excel 2016). Eight plants were grown and analysed for each of three transgenic lines in T1 generation. Up to 30 plants were grown for each of three transgenic lines in T2 generation and plants with Rieske FeS overexpression confirmed by immunoblotting comprised 10–20% of plants depending on the line. Three T2 plants with the highest overexpression levels from each line were used for analyses. Experiments were partially replicated in T1 and T2 generations for three independent lines.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
The datasets analysed in this paper are included in this published article and supplementary information files. MultipeQ data are available at PhotosynQ web application (https://photosynq.org), project ID 3400. Plasmids used for generation of plants with Rieske FeS overexpression can be obtained from Addgene (deposit 77017). Further datasets generated during the current study as well as seeds of the WT and FeS-OE S.viridis are available from the corresponding author on request.
References
Andralojc, P. J. et al. Raising yield potential of wheat. II. Increasing photosynthetic capacity and efficiency. J. Exp. Bot. 62, 453–467 (2010).
Long, Stephen, P., Marshall-Colon, A. & Zhu, X.-G. Meeting the global food demand of the future by engineering crop photosynthesis and yield potential. Cell 161, 56–66 (2015).
Furbank, R. T., Quick, W. P. & Sirault, X. R. R. Improving photosynthesis and yield potential in cereal crops by targeted genetic manipulation: Prospects, progress and challenges. Field Crops Res. 182, 19–29 (2015).
Andrew J. et al. Feeding the world: improving photosynthetic efficience for sustainable crop production. J. Exp. Bot., https://doi.org/10.1093/jxb/ery445 (2019).
Chida, H. et al. Expression of the algal Cytochrome c6 gene in Arabidopsis enhances photosynthesis and growth. Plant Cell Physiol. 48, 948–957 (2007).
Simkin, A. J., McAusland, L., Lawson, T. & Raines, C. A. Overexpression of the RieskeFeS protein increases electron transport rates and biomass yield. Plant Physiol. 175, 134–145 (2017).
Byrt, C. S., Grof, C. P. L. & Furbank, R. T. C4 plants as biofuel feedstocks: optimising biomass production and feedstock quality from a lignocellulosic perspective free access. J. Integr. Plant Biol. 53, 120–135 (2011).
von Caemmerer, S. & Furbank, R. T. Strategies for improving C4 photosynthesis. Curr. Opin. Plant Biol. 31, 125–134 (2016).
Wu, A., Doherty, A., Farquhar, G. D. & Hammer, G. L. Simulating daily field crop canopy photosynthesis: an integrated software package. Funct. Plant Biol. 45, 362–377 (2018).
Tikhonov, A. N. The cytochrome b6f complex at the crossroad of photosynthetic electron transport pathways. Plant Physiol. Biochem. 81, 163–183 (2014).
Mitchell, P. Possible molecular mechanisms of the protonmotive function of cytochrome systems. J. Theor. Biol. 62, 327–367 (1976).
Takizawa, K., Kanazawa, A. & Kramer, D. M. Depletion of stromal Pi induces high ‘energy-dependent’ antenna exciton quenching (qE) by decreasing proton conductivity at CFO-CF1 ATP synthase. Plant, Cell Environ. 31, 235–243 (2008).
Kramer, D. M., Cruz, J. A. & Kanazawa, A. Balancing the central roles of the thylakoid proton gradient. Trends Plant Sci. 8, 27–32 (2003).
Finazzi, G. Redox-coupled proton pumping activity in Cytochrome b6f, as evidenced by the pH dependence of electron transfer in whole cells of Chlamydomonas reinhardtii. Biochemistry 41, 7475–7482 (2002).
Müller, P., Li, X.-P. & Niyogi, K. K. Non-photochemical quenching. A response to excess light energy. Plant Physiol. 125, 1558–1566 (2001).
Li, X.-P. et al. A pigment-binding protein essential for regulation of photosynthetic light harvesting. Nature 403, 391 (2000).
Demmig-Adams, B. Carotenoids and photoprotection in plants: a role for the xanthophyll zeaxanthin. Biochim. et. Biophys. Acta (BBA) - Bioenerg. 1020, 1–24 (1990).
Schöttler, M. A., Tóth, S. Z., Boulouis, A. & Kahlau, S. Photosynthetic complex stoichiometry dynamics in higher plants: biogenesis, function, and turnover of ATP synthase and the cytochrome b6f complex. J. Exp. Bot. 66, 2373–2400 (2015).
Price, G. et al. Chloroplast cytochrome b6/f and ATP synthase complexes in tobacco: transformation with antisense RNA against nuclear-encoded transcripts for the Rieske FeS and ATPδ polypeptides. Funct. Plant Biol. 22, 285–297 (1995).
Price, G. D. et al. Photosynthesis is strongly reduced by antisense suppression of chloroplastic cytochrome bf complex in transgenic tobacco. Funct. Plant Biol. 25, 445–452 (1998).
Anderson, J. M., Dean Price, G., Soon Chow, W., Hope, A. B. & Badger, M. R. Reduced levels of cytochrome bf complex in transgenic tobacco leads to marked photochemical reduction of the plastoquinone pool, without significant change in acclimation to irradiance. Photosynth. Res. 53, 215–227 (1997).
Ruuska, S. A., Andrews, T. J., Badger, M. R., Price, G. D. & von Caemmerer, S. The role of chloroplast electron transport and metabolites in modulating Rubisco activity in tobacco. Insights from transgenic plants with reduced amounts of cytochrome b/f complex or glyceraldehyde 3-phosphate dehydrogenase. Plant Physiol. 122, 491–504 (2000).
Yamori, W. et al. The roles of ATP synthase and the cytochrome b6f complexes in limiting chloroplast electron transport and determining photosynthetic capacity. Plant Physiol. 155, 956–962 (2011).
Barkan, A., Miles, D. & Taylor, W. C. Chloroplast gene expression in nuclear, photosynthetic mutants of maize. EMBO J. 5, 1421–1427 (1986).
Hatch, M. D. C4 photosynthesis - a unique blend of modified biochemistry, anatomy and ultrastructure. Biochim Biophys. Acta 895, 81–106 (1987).
Chapman, K. S. R., Berry, J. A. & Hatch, M. D. Photosynthetic metabolism in bundle sheath cells of the C4 species Zea mays: sources of ATP and NADPH and the contribution of photosystem II. Arch. Biochem. Biophys. 202, 330–341 (1980).
Munekage, Y. N. Light harvesting and chloroplast electron transport in NADP-malic enzyme type C4 plants. Curr. Opin. Plant Biol. 31, 9–15 (2016).
Romanowska, E. et al. Structural organization of photosynthetic apparatus in agranal chloroplasts of maize. J. Biol. Chem. 283, 26037–26046 (2008).
Ghannoum, O. et al. Faster Rubisco is the key to superior nitrogen-use efficiency in NADP-malic enzyme relative to NAD-malic enzyme C4 Grasses. Plant Physiol. 137, 638–650 (2005).
Aro, E.-M., Zhang, L., Suorsa, M., Paakkarinen, V. & Battchikova, N. Maintenance of functional photosystem II by D1 protein turnover. Sci. Access 3, https://doi.org/10.1071/SA0403002 (2001).
Sonoike, K. Photoinhibition of photosystem I. Physiol. Plant. 142, 56–64 (2011).
Lima-Melo, Y., Gollan, P. J., Tikkanen, M., Silveira, J. A. G. & Aro, E.-M. Consequences of photosystem-I damage and repair on photosynthesis and carbon use in Arabidopsis thaliana. Plant J. 0, https://doi.org/10.1111/tpj.14177.
Laisk, A., Eichelmann, H., Oja, V. & Peterson, R. B. Control of cytochrome b6f at low and high light intensity and cyclic electron transport in leaves. Biochim. et. Biophys. Acta (BBA) - Bioenerg. 1708, 79–90 (2005).
Miyake, C. et al. Effects of light intensity on cyclic electron flow around PSI and its relationship to non-photochemical quenching of Chl fluorescence in tobaccoleaves. Plant Cell Physiol. 46, 1819–1830 (2005).
Joliot, P. & Joliot, A. Quantification of cyclic and linear flows in plants. Proc. Natl Acad. Sci. USA 102, 4913–4918 (2005).
Cramer, W. A., Zhang, H., Yan, J., Kurisu, G. & Smith, J. L. Transmembrane traffic in the Cytochrome b 6 f complex. Annu. Rev. Biochem. 75, 769–790 (2006).
DalCorso, G. et al. A complex containing PGRL1 and PGR5 is involved in the switch between linear and cyclic electron flow in Arabidopsis. Cell 132, 273–285 (2008).
Nakamura, N., Iwano, M., Havaux, M., Yokota, A. & Munekage, Y. N. Promotion of cyclic electron transport around photosystem I during the evolution of NADP–malic enzyme-type C4 photosynthesis in the genus Flaveria. New Phytol. 199, 832–842 (2013).
Edwards, E. J., Osborne, C. P., Strömberg, C. A. E. & Smith, S. A., Consortium, C. G. The origins of C4 grasslands: integrating evolutionary and ecosystem science. Science 328, 587–591 (2010).
Takabayashi, A., Kishine, M., Asada, K., Endo, T. & Sato, F. Differential use of two cyclic electron flows around photosystem I for driving CO2-concentration mechanism in C4 photosynthesis. Proc. Natl Acad. Sci. USA 102, 16898–16903 (2005).
Andersson, B. & Anderson, J. M. Lateral heterogeneity in the distribution of chlorophyll-protein complexes of the thylakoid membranes of spinach chloroplasts. Biochim. et. Biophys. Acta (BBA) - Bioenerg. 593, 427–440 (1980).
Albertsson, P.-Å. A quantitative model of the domain structure of the photosynthetic membrane. Trends Plant Sci. 6, 349–354 (2001).
Kanazawa, A. & Kramer, D. M. In vivo modulation of nonphotochemical exciton quenching (NPQ) by regulation of the chloroplast ATP synthase. Proc. Natl Acad. Sci. USA 99, 12789–12794 (2002).
Nandha, B., Finazzi, G., Joliot, P., Hald, S. & Johnson, G. N. The role of PGR5 in the redox poising of photosynthetic electron transport. Biochim. et. Biophys. Acta (BBA) - Bioenerg. 1767, 1252–1259 (2007).
Alonso-Cantabrana, H. et al. Diffusion of CO2 across the mesophyll-bundle sheath cell interface in a C4 Plant with genetically reduced PEP carboxylase activity. Plant Physiol. 178, 72–81 (2018).
von Caemmerer, S. Biochemical Models of Leaf Photosynthesis. (CSIRO Publishing, Clayton, Australia, 2000).
von Caemmerer, S. & Furbank, R. T. in The Biology of C4 Photosynthesis (eds Rowan F. S. & Russell K. M.) 173–211 (Academic Press, Cambridge, MA, 1999).
Salesse-Smith, C. E. et al. Overexpression of Rubisco subunits with RAF1 increases Rubisco content in maize. Nat. Plants 4, 802–810 (2018).
Ostersetzer, O. & Adam, Z. Light-stimulated degradation of an unassembled Rieske FeS protein by a thylakoid-bound protease: the possible role of the FtsH protease. Plant Cell 9, 957–965 (1997).
Engler, C. et al. A golden gate modular cloning toolbox for plants. ACS Synth. Biol. 3, 839–843 (2014).
Osborn, H. L. et al. Effects of reduced carbonic anhydrase activity on CO2 assimilation rates in Setaria viridis: a transgenic analysis. J. Exp. Bot. 68, 299–310 (2016).
Kuhlgert, S. et al. MultispeQ Beta: a tool for large-scale plant phenotyping connected to the open PhotosynQ network. Royal Soc. Open Sci. 3, 160592. https://doi.org/10.1098/rsos.160592.
Rantala, M., Paakkarinen, V. & Aro, E.-M. Separation of thylakoid protein complexes with two-dimensional native-PAGE. Bio-Protoc. 8, e2899 (2018).
Porra, R. J., Thompson, W. A. & Kriedemann, P. E. Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim. et. Biophys. Acta (BBA) - Bioenerg. 975, 384–394 (1989).
Genty, B., Briantais, J. M. & Baker, N. R. The relationship between the quantum yield of photosynthetic electron-transport and quenching of chlorophyll fluorescence. Biochim Biophys. Acta 990, 87–92 (1989).
Klughammer, C. & Schreiber, U. Saturation Pulse method for assessment of energy conversion in PS I. PAM Appl. Notes 1, 3 (2008).
Sacksteder, C. A. & Kramer, D. M. Dark-interval relaxation kinetics (DIRK) of absorbance changes as a quantitative probe of steady-state electron transfer. Photosynth. Res. 66, 145 (2000).
Cruz, J. A., Sacksteder, C. A., Kanazawa, A. & Kramer, D. M. Contribution of electric field (Δψ) to steady-state transthylakoid proton motive force (pmf) in vitro and in vivo. Control of pmf parsing into Δψ and ΔpH by ionicstrength. Biochemistry 40, 1226–1237 (2001).
Kanazawa, A. et al. Chloroplast ATP synthase modulation of the thylakoid proton motive force: implications for photosystem I and photosystem II photoprotection. Fron. Plant Sci. 8, https://doi.org/10.3389/fpls.2017.00719 (2017).
Acknowledgements
We thank Soumi Bala for the help with [14C] carboxyarabinitol bisphosphate binding assays and Tegan Norley for the help with electron transport measurements. This research was supported by the Australian Research Council Centre of Excellence for Translational Photosynthesis (CE140100015).
Author information
Authors and Affiliations
Contributions
S.vC. and R.F. conceived the project, S.vC., R.F., C.R. and M.E. planned experiments, P.L.-C. assembled constructs, M.E. performed experiments, M.E. analysed data and wrote the manuscript with contribution of all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ermakova, M., Lopez-Calcagno, P.E., Raines, C.A. et al. Overexpression of the Rieske FeS protein of the Cytochrome b6f complex increases C4 photosynthesis in Setaria viridis. Commun Biol 2, 314 (2019). https://doi.org/10.1038/s42003-019-0561-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s42003-019-0561-9
This article is cited by
-
Application of Acinetobacter indicus to promote cigarette smoke particulate matter phytoremediation: removal efficiency and plant–microbe interactions
Environmental Science and Pollution Research (2024)
-
Genetic manipulation of photosynthesis to enhance crop productivity under changing environmental conditions
Photosynthesis Research (2023)
-
Integrated biomarker responses of rice associated with grain yield in copper-contaminated soil
Environmental Science and Pollution Research (2022)
-
Overexpression of differentially expressed AhCytb6 gene during plant-microbe interaction improves tolerance to N2 deficit and salt stress in transgenic tobacco
Scientific Reports (2021)
-
The role of Cytochrome b6f in the control of steady-state photosynthesis: a conceptual and quantitative model
Photosynthesis Research (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.