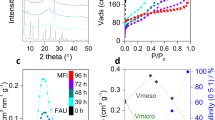
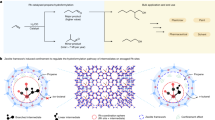
Abstract
The catalytic shuttling of functional moieties has emerged as a promising strategy to substitute and diversify traditional hydrofunctionalization technologies. However, these reactions are reversible due to their isodesmic nature, which limits their applicability to a select array of donor and acceptor molecules, and poses substantial challenges with regard to atom economy and practicality. Here we show an approach that harnesses the shape-selective and catalytic properties of zeolites to drive the shuttling equilibrium of transfer hydrocyanation and transfer hydroformylation reactions to near-completion. The zeolites irreversibly convert the transfer reaction co-products in an exergonic tandem reaction while excluding the substrates via pore size restrictions. Through fine-tuning of the zeolite’s properties, yield increases of up to 80% can be achieved, enabling diversification of nitrile donors to propionitrile and aldehyde acceptors to unactivated olefins. Mechanistic and spectroscopic studies highlight the unique synergy between the zeolites and the homogeneous transfer catalysts.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The findings of this study are available within the article and its supplementary information. All data are available from the authors upon reasonable request.
References
Beller, M., Seayad, J., Tillack, A. & Jiao, H. Catalytic Markovnikov and anti-Markovnikov functionalization of alkenes and alkynes: recent developments and trends. Angew. Chem. Int. Ed. 43, 3368–3398 (2004).
Bhawal, B. N. & Morandi, B. Catalytic transfer functionalization through shuttle catalysis. ACS Catal. 6, 7528–7535 (2016).
Bhawal, B. N. & Morandi, B. Catalytic isofunctional reactions—expanding the repertoire of shuttle and metathesis reactions. Angew. Chem. Int. Ed. 58, 10074–10103 (2019).
Bhawal, B. N. & Morandi, B. Shuttle catalysis—new strategies in organic synthesis. Chem. A Eur. J. 23, 12004–12013 (2017).
Fang, X., Yu, P. & Morandi, B. Catalytic reversible alkene-nitrile interconversion through controllable transfer hydrocyanation. Science 351, 832–836 (2016).
Fang, X., Cacherat, B. & Morandi, B. CO- and HCl-free synthesis of acid chlorides from unsaturated hydrocarbons via shuttle catalysis. Nat. Chem. 9, 1105–1109 (2017).
Murphy, S. K., Park, J. W., Cruz, F. A. & Dong, V. M. Rh-catalyzed C-C bond cleavage by transfer hydroformylation. Science 347, 56–60 (2015).
Simonneau, A. & Oestreich, M. 3-silylated cyclohexa-1,4-dienes as precursors for gaseous hydrosilanes: the B(C6F5)3-catalyzed transfer hydrosilylation of alkenes. Angew. Chem. Int. Ed. 52, 11905–11907 (2013).
Lian, Z., Bhawal, B. N., Yu, P. & Morandi, B. Palladium-catalyzed carbon-sulfur or carbon-phosphorus bond metathesis by reversible arylation. Science 356, 1059–1063 (2017).
Mulryan, D., Rodwell, J., Phillips, N. A. & Crimmin, M. R. Au(I) Catalyzed HF transfer: tandem alkyne hydrofluorination and perfluoroarene functionalisation. ACS Catal 12, 3411–3419 (2022).
De La Higuera Macias, M. & Arndtsen, B. A. Functional group transposition: a palladium-catalyzed metathesis of Ar-X σ-bonds and acid chloride synthesis. J. Am. Chem. Soc. 140, 10140–10144 (2018).
Rong, Z. Q., Lim, H. N. & Dong, G. Intramolecular acetyl transfer to olefins by catalytic C-C bond activation of unstrained ketones. Angew. Chem. Int. Ed. 57, 475–479 (2018).
Tan, G., Wu, Y., Shi, Y. & You, J. Syngas-free highly regioselective rhodium-catalyzed transfer hydroformylation of alkynes to α,β-unsaturated aldehydes. Angew. Chem. Int. Ed. 58, 7440–7444 (2019).
Yu, P., Bismuto, A. & Morandi, B. Iridium-catalyzed hydrochlorination and hydrobromination of alkynes by shuttle catalysis. Angew. Chem. Int. Ed. 59, 2904–2910 (2020).
Li, Y., Bao, G. & Wu, X. F. Palladium-catalyzed intermolecular transthioetherification of aryl halides with thioethers and thioesters. Chem. Sci. 11, 2187–2192 (2020).
Delcaillau, T., Boehm, P. & Morandi, B. Nickel-catalyzed reversible functional group metathesis between aryl nitriles and aryl thioethers. J. Am. Chem. Soc. 143, 3723–3728 (2021).
Dong, X., Roeckl, J. L., Waldvogel, S. R. & Morandi, B. Merging shuttle reactions and paired electrolysis for reversible vicinal dihalogenations. Science 371, 507–514 (2021).
Veth, L., Grab, H. A., Martínez, S., Antheaume, C. & Dydio, P. Transfer C-H borylation of alkenes under Rh(I) catalysis: insight into the synthetic capacity, mechanism and selectivity control. Chem. Catal. 2, 762–778 (2022).
Bhunia, A., Bergander, K. & Studer, A. Cooperative palladium/Lewis acid-catalyzed transfer hydrocyanation of alkenes and alkynes using 1-methylcyclohexa-2,5-diene-1-carbonitrile. J. Am. Chem. Soc. 140, 16353–16359 (2018).
Bhawal, B. N., Reisenbauer, J. C., Ehinger, C. & Morandi, B. Overcoming selectivity issues in reversible catalysis: a transfer hydrocyanation exhibiting high kinetic control. J. Am. Chem. Soc. 142, 10914–10920 (2020).
Luo, X. et al. Mechanism of rhodium-catalyzed formyl activation: a computational study. J. Org. Chem. 81, 2320–2326 (2016).
Fürstner, A. Olefin metathesis and beyond. Angew. Chem. Int. Ed. 39, 3012–3043 (2000).
Allian, A. D. et al. Process safety in the pharmaceutical industry—Part I: thermal and reaction hazard evaluation processes and techniques. Org. Process Res. Dev. 24, 2529–2548 (2020).
Orecchia, P., Yuan, W. & Oestreich, M. Transfer hydrocyanation of α- and α,β-substituted styrenes catalyzed by boron Lewis acids. Angew. Chem. Int. Ed. 58, 3579–3583 (2019).
Wang, C. S., Yu, Y., Sunada, Y., Wang, C. & Yoshikai, N. Cobalt-catalyzed carbo- and hydrocyanation of alkynes via C-CN bond activation. ACS Catal 12, 4054–4066 (2022).
Landis, C. R. Construction and deconstruction of aldehydes by transfer hydroformylation. Science 347, 29–30 (2015).
Wasilke, J. C., Obrey, S. J., Baker, R. T. & Bazan, G. C. Concurrent tandem catalysis. Chem. Rev. 105, 1001–1020 (2005).
Frye, N. L., Bhunia, A. & Studer, A. Nickel-catalyzed Markovnikov transfer hydrocyanation in the absence of Lewis acid. Org. Lett. 22, 4456–4460 (2020).
Fan, C. & Zhou, Q. L. Nickel-catalyzed group transfer of radicals enables hydrocyanation of alkenes and alkynes. Chem. Catal. 1, 117–128 (2021).
Reisenbauer, J. C., Bhawal, B. N., Jelmini, N. & Morandi, B. Development of an operationally simple, scalable and HCN-free transfer hydrocyanation protocol using an air-stable Nickel precatalyst. Org. Process Res. Dev. 26, 1165–1173 (2022).
Morandi, B., Fang, Y. & Yu, P. Process for the catalytic reversible alkene-nitrile interconversion. US patent 10,597,356 B2 (2020).
Ni, S. F., Yang, T. L. & Dang, L. Transfer hydrocyanation by Nickel(0)/Lewis acid cooperative catalysis, mechanism investigation and computational prediction of shuttle catalysts. Organometallics 36, 2746–2754 (2017).
Lenges, C. P. & Brookhart, M. Isomerization of aldehydes catalyzed by rhodium(I) olefin complexes. Angew. Chem. Int. Ed. 38, 3533–3537 (1999).
Corma, A. & Iborra, S. Oligomerization of alkenes. Catal. Fine Chem. Synth. 4, 125 (2006).
Sanati, M., Hörnell, C. & Järäs, S. G. The oligomerization of alkenes by heterogeneous catalysts. Catalysis 14, 236–288 (2007).
Skupińska, J. Oligomerization of α-olefins to higher oligomers. Chem. Rev. 91, 613–648 (1991).
Al-Jarallah, A. M., Anabtawi, J. A., Siddiqui, M. A. B., Aitani, A. M. & Al-Sa’doun, A. W. Ethylene dimerization and oligomerization to butene-1 and linear α-olefins. A review of catalytic systems and processes. Catal. Today 14, 1–121 (1992).
Ghosh, A. K. & Kevan, L. Catalytic and ESR studies of ethylene dimerization on palladium-exchanged Na-X and Ca-X zeolites. J. Phys. Chem. 92, 4439–4446 (1988).
Lallemand, M., Finiels, A., Fajula, F. & Hulea, V. Catalytic oligomerization of ethylene over Ni-containing dealuminated Y zeolites. Appl. Catal. A Gen. 301, 196–201 (2006).
O’connor, C. T. & Kojima, M. Alkene oligomerization. Catal. Today 6, 329–349 (1990).
Finiels, A., Fajula, F. & Hulea, V. Nickel-based solid catalysts for ethylene oligomerization—a review. Catal. Sci. Technol. 4, 2412–2426 (2014).
Wang, S. & Iglesia, E. Mechanism of isobutanal-isobutene Prins condensation reactions on solid Brønsted acids. ACS Catal. 6, 7664–7684 (2016).
Arundale, E. & Mikeska, L. A. The olefin-aldehyde condensation. The Prins reaction. Chem. Rev. 51, 505–555 (1952).
Vasiliadou, E. S., Li, S., Caratzoulas, S. & Lobo, R. F. Formaldehyde-isobutene Prins condensation over MFI-type zeolites. Catal. Sci. Technol. 8, 5794–5806 (2018).
Wilshier, K. G. Propene oligomerization over H-ZSM-5 zeolite. Stud. Surf. Sci. Catal. 36, 621–625 (1988).
Sarazen, M. L., Doskocil, E. & Iglesia, E. Effects of void environment and acid strength on alkene oligomerization selectivity. ACS Catal. 6, 7059–7070 (2016).
Kuo, T. W. & Tan, C. S. Alkylation of toluene with propylene in supercritical carbon dioxide over chemical liquid deposition HZSM-5 pellets. Ind. Eng. Chem. Res. 40, 4724–4730 (2001).
Doyle, M., Us, I. L., Anderson, S. L. & Plaines, D. Alkylation of aromatic hydrocarbons. US patent 4,469,908 (1984).
Grigor’eva, N. G., Serebrennikov, D. V., Bubennov, S. V. & Kutepov, B. I. Isoamylene oligomerization over zeolite catalysts. Pet. Chem. 61, 183–189 (2021).
Hauge, K., Bergene, E., Chen, D., Fredriksen, G. R. & Holmen, A. Oligomerization of isobutene over solid acid catalysts. Catal. Today 100, 463–466 (2005).
Chang, C. D., Chu, C. T. W., Miale, J. N., Bridger, R. F. & Calvert, R. B. Aluminum insertion into high silica zeolite frameworks. 1. Reaction with aluminum halides. J. Am. Chem. Soc. 106, 8143–8146 (1984).
Batool, S. R., Sushkevich, V. L. & van Bokhoven, J. A. Correlating Lewis acid activity to extra-framework aluminum species in zeolite Y introduced by ion-exchange. J. Catal. 408, 24–35 (2022).
Lang, S. et al. Mechanisms of the AlCl3 modification of siliceous microporous and mesoporous catalysts investigated by multi-nuclear solid-state NMR. Top. Catal. 60, 1537–1553 (2017).
Marcilly, C. R. Where and how shape selectivity of molecular sieves operates in refining and petrochemistry catalytic processes. Top. Catal. 13, 357–366 (2000).
Csicsery, S. M. Catalysis by shape selective zeolites-science and technology. Pure Appl. Chem. 58, 841–856 (1986).
Teketel, S. et al. Shape selectivity in the conversion of methanol to hydrocarbons: the catalytic performance of one-dimensional 10-ring zeolites: ZSM-22, ZSM-23, ZSM-48 and EU-1. ACS Catal. 2, 26–37 (2011).
Xue, Y. et al. Enhancing propene selectivity in methanol and/or butene conversion by regulating channel systems over ZSM-5/ZSM-48 composite zeolites. Microporous Mesoporous Mater. 312, 110803 (2021).
Bugaev, A. L. et al. The role of palladium carbides in the catalytic hydrogenation of ethylene over supported palladium nanoparticles. Catal. Today 336, 40–44 (2019).
Rodriguez-González, L. et al. The acid properties of H-ZSM-5 as studied by NH3-TPD and 27Al-MAS-NMR spectroscopy. Appl. Catal. A Gen. 328, 174–182 (2007).
Wagschal, S. et al. Formation of XPhos-ligated palladium(0) complexes and reactivity in oxidative additions. Chem. Eur. J. 25, 6980–6987 (2019).
Fang, X., Yu, P., Prina Cerai, G. & Morandi, B. Unlocking Mizoroki-Heck-type reactions of aryl cyanides using transfer hydrocyanation as a turnover-enabling step. Chem. Eur. J. 22, 15629–15633 (2016).
Kleemann, A., Engel, J., Kutscher, B. & Reichert, D. Pharmaceutical Substances (Thieme, 2011).
Acknowledgements
We thank J. Vercammen for insights on zeolite synthesis and manipulation, K. Lomachenko for help with XAS measurements, G. O’Rourke for assistance with TGA experiments, C. Marquez for help with ICP measurements, and C. Cheung for providing the scanning electron microscopy images. The XAS experiments were performed at beamline BM23 of the European Synchrotron Radiation Facility (ESRF, Grenoble, France). D.D.V. and J.D. are grateful to KULeuven (C1 project) and FWO (G0781118N with ARSS, Slovenia; G0F2320N) for funding. A.K. and G.M. acknowledge financial support from the Slovenian Research Agency (research core funding no. P1-0021 and project no. N1-0079).
Author information
Authors and Affiliations
Contributions
D.D.V. and J.D. were responsible for the conception, design and interpretation of the experiments. J.D. and J.W. performed the experiments and analysed the data. J.D., N.V.V. and A.L.B. conceived the design of the XAS experiments, and A.L.B. and O.A.U. were responsible for data processing. A.K. and G.M. conceived and performed the NMR experiments. The paper was written by J.D. and D.D.V., with contributions from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Discussion, Figs. 1–47, Tables 1–28 and References.
Supplementary Data 1
Computational geometry optimization data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dallenes, J., Wuyts, J., Van Velthoven, N. et al. Zeolites as equilibrium-shifting agents in shuttle catalysis. Nat Catal 6, 495–505 (2023). https://doi.org/10.1038/s41929-023-00967-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-023-00967-8