Abstract
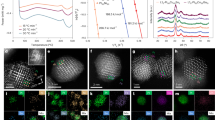
The emerging oxide–zeolite bifunctional catalysis for direct syngas conversion has drawn extensive interest, both academically and industrially, with further exploration urging a clear mechanistic understanding of this complex catalytic network. Herein, using a specially designed quasi-in situ, solid-state nuclear magnetic resonance-gas chromatography/gas chromatography-mass spectrometry analysis strategy, this reaction is fully monitored from the very early induction period to steady-state conversion under high-pressure flow-reaction conditions, using ZnAlOx/H-ZSM-5 composites as model catalysts. We identify abundant critical and/or transient intermediates in dynamic evolution, including carboxylates, alkoxyls, acid-bounded methyl-cyclopentenones and methyl-cyclopentenyl carbocations, providing direct evidence of vigorous regulation by unique, oxygenate-based pathways of the reaction network. This proposed mechanism overturns the general cognition of oxide–zeolite reactions as simple tandem catalysis, and highlights the many roles (both positive and negative) of CO and H2 molecules via oxygenate-based routes, thus dictating the final product. The current characterization technology and its mechanistic understanding would benefit further exploration in bifunctional catalysis.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
Data supporting the findings of this study are available within this paper and its Supplementary Information or from the corresponding author upon reasonable request. The datasets for catalytic performance tests of syngas conversion are available in the figshare repository (https://doi.org/10.6084/m9.figshare.19722955.v1).
References
Dry, M. E. The Fischer-Tropsch process: 1950–2000. Catal. Today 71, 227–241 (2002).
Sartipi, S., Makkee, M., Kapteijn, F. & Gascon, J. Catalysis engineering of bifunctional solids for the one-step synthesis of liquid fuels from syngas: a review. Catal. Sci. Technol. 4, 893–907 (2014).
Torres Galvis, H. M. & de Jong, K. P. Catalysts for production of lower olefins from synthesis gas: a review. ACS Catal. 3, 2130–2149 (2013).
Zhao, B. et al. Direct transformation of syngas to aromatics over Na-Zn-Fe5C2 and hierarchical HZSM-5 tandem catalysts. Chem 3, 323–333 (2017).
Li, J. et al. Integrated tuneable synthesis of liquid fuels via Fischer–Tropsch technology. Nat. Catal. 1, 787–793 (2018).
Zhou, W. et al. New horizon in C1 chemistry: breaking the selectivity limitation in transformation of syngas and hydrogenation of CO2 into hydrocarbon chemicals and fuels. Chem. Soc. Rev. 48, 3193–3228 (2019).
Zhang, Q., Yu, J. & Corma, A. Applications of zeolites to C1 chemistry: recent advances, challenges, and opportunities. Adv. Mater. 32, 2002927 (2020).
Pan, X., Jiao, F., Miao, D. & Bao, X. Oxide-zeolite-based composite catalyst concept that enables syngas chemistry beyond Fischer-Tropsch synthesis. Chem. Rev. 121, 6588–6609 (2021).
Jiao, F. et al. Selective conversion of syngas to light olefins. Science 351, 1065–1068 (2016).
Cheng, K. et al. Direct and highly selective conversion of synthesis gas into lower olefins: design of a bifunctional catalyst combining methanol synthesis and carbon-carbon coupling. Angew. Chem. Int. Ed. Engl. 55, 4725–4728 (2016).
Jiao, F. et al. Shape-selective zeolites promote ethylene formation from syngas via a ketene intermediate. Angew. Chem. Int. Ed. Engl. 57, 4692–4696 (2018).
Zhou, W. et al. Direct conversion of syngas into methyl acetate, ethanol, and ethylene by relay catalysis via the intermediate dimethyl ether. Angew. Chem. Int. Ed. Engl. 57, 12012–12016 (2018).
Li, N. et al. High-quality gasoline directly from syngas by dual metal oxide–zeolite (OX-ZEO) catalysis. Angew. Chem. Int. Ed. Engl. 58, 7400–7404 (2019).
Chang, C. D., Lang, W. H. & Silvestri, A. J. Synthesis gas conversion to aromatic hydrocarbons. J. Catal. 56, 268–273 (1979).
Cheng, K. et al. Bifunctional catalysts for one-step conversion of syngas into aromatics with excellent selectivity and stability. Chem 3, 334–347 (2017).
Yang, J., Pan, X., Jiao, F., Li, J. & Bao, X. Direct conversion of syngas to aromatics. Chem. Commun. 53, 11146–11149 (2017).
Ni, Y. et al. Realizing and recognizing syngas-to-olefins reaction via a dual-bed catalyst. ACS Catal. 9, 1026–1032 (2018).
Wang, C.-M., Wang, Y.-D. & Xie, Z.-K. Methylation of olefins with ketene in zeotypes and its implications for the direct conversion of syngas to light olefins: a periodic DFT study. Catal. Sci. Technol. 6, 6644–6649 (2016).
Chowdhury, A. D. & Gascon, J. The curious case of ketene in zeolite chemistry and catalysis. Angew. Chem. Int. Ed. Engl. 57, 14982–14985 (2018).
Haw, J. F., Song, W., Marcus, D. M. & Nicholas, J. B. The mechanism of methanol to hydrocarbon catalysis. Acc. Chem. Res. 36, 317–326 (2003).
Svelle, S. et al. Conversion of methanol into hydrocarbons over zeolite H-ZSM-5: ethene formation is mechanistically separated from the formation of higher alkenes. J. Am. Chem. Soc. 128, 14770–14771 (2006).
Olsbye, U. et al. Conversion of methanol to hydrocarbons: how zeolite cavity and pore size controls product selectivity. Angew. Chem. Int. Ed. Engl. 51, 5810–5831 (2012).
Ilias, S. & Bhan, A. Mechanism of the catalytic conversion of methanol to hydrocarbons. ACS Catal. 3, 18–31 (2013).
Tian, P., Wei, Y., Ye, M. & Liu, Z. Methanol to olefins (MTO): from fundamentals to commercialization. ACS Catal. 5, 1922–1938 (2015).
Yarulina, I., Chowdhury, A. D., Meirer, F., Weckhuysen, B. M. & Gascon, J. Recent trends and fundamental insights in the methanol-to-hydrocarbons process. Nat. Catal. 1, 398–411 (2018).
Cheng, Y.-T. & Huber, G. W. Chemistry of furan conversion into aromatics and olefins over HZSM-5: a model biomass conversion reaction. ACS Catal. 1, 611–628 (2011).
Rezaei, P. S., Shafaghat, H. & Daud, W. M. A. W. Production of green aromatics and olefins by catalytic cracking of oxygenate compounds derived from biomass pyrolysis: a review. Appl. Catal. A 469, 490–511 (2014).
Liu, Y. et al. Formation mechanism of the first carbon-carbon bond and the first olefin in the methanol conversion into hydrocarbons. Angew. Chem. Int. Ed. Engl. 55, 5723–5726 (2016).
Chowdhury, A. D. et al. Initial carbon-carbon bond formation during the early stages of the methanol-to-olefin process proven by zeolite-trapped acetate and methyl acetate. Angew. Chem. Int. Ed. Engl. 55, 15840–15845 (2016).
Chowdhury, A. D. et al. Bridging the gap between the direct and hydrocarbon pool mechanisms of the methanol-to-hydrocarbons process. Angew. Chem. Int. Ed. Engl. 57, 8095–8099 (2018).
Plessow, P. N., Smith, A., Tischer, S. & Studt, F. Identification of the reaction sequence of the MTO initiation mechanism using ab initio-based kinetics. J. Am. Chem. Soc. 141, 5908–5915 (2019).
Yang, L. et al. Role of acetaldehyde in the roadmap from initial carbon-carbon bonds to hydrocarbons during methanol conversion. ACS Catal. 9, 6491–6501 (2019).
Arora, S. S., Nieskens, D. L. S., Malek, A. & Bhan, A. Lifetime improvement in methanol-to-olefins catalysis over chabazite materials by high-pressure H2 co-feeds. Nat. Catal. 1, 666–672 (2018).
Zhao, X. et al. Achieving a superlong lifetime in the zeolite-catalyzed MTO reaction under high pressure: synergistic effect of hydrogen and water. ACS Catal. 9, 3017–3025 (2019).
Chen, Z. et al. Coupling of methanol and carbon monoxide over H-ZSM-5 to form aromatics. Angew. Chem. Int. Ed. Engl. 57, 12549–12553 (2018).
Gilani, S. Z. A. et al. Two-way desorption coupling to enhance the conversion of syngas into aromatics by MnO/H-ZSM-5. Catal. Sci. Technol. 10, 3366–3375 (2020).
Arslan, M. T. et al. Selective conversion of syngas into tetramethylbenzene via an aldol-aromatic mechanism. ACS Catal. 10, 2477–2488 (2020).
Lazo, N. D., Murray, D. K., Kieke, M. L. & Haw, J. F. In situ carbon-13 solid-state NMR study of the Cu/ZnO/Al2O3 methanol synthesis catalyst. J. Am. Chem. Soc. 114, 8552–8559 (1992).
Chen, Y. et al. C−C bond formation in syngas conversion over zinc sites grafted on ZSM-5 Zeolite. Angew. Chem. Int. Ed. Engl. 59, 6529–6534 (2020).
Wang, W., Buchholz, A., Seiler, M. & Hunger, M. Evidence for an Initiation of the methanol-to-olefin process by reactive surface methoxy groups on acidic zeolite catalysts. J. Am. Chem. Soc. 125, 15260–15267 (2003).
Wang, W. & Hunger, M. Reactivity of surface alkoxy species on acidic zeolite catalysts. Acc. Chem. Res. 41, 895–904 (2008).
Wang, C. et al. Extra-framework aluminum-assisted initial C−C bond formation in methanol-to-olefins conversion on zeolite H-ZSM-5. Angew. Chem. Int. Ed. Engl. 57, 10197–10201 (2018).
Stepanov, A. G., Luzgin, M. V., Romannikov, V. N. & Zamaraev, K. I. NMR observation of the Koch reaction in zeolite H-ZSM-5 under mild conditions. J. Am. Chem. Soc. 117, 3615–3616 (1995).
Luzgin, M. V., Stepanov, A. G., Sassi, A. & Sommer, J. Formation of carboxylic acids from small alkanes in zeolite H-ZSM-5. Chem. Eur. J. 6, 2368–2376 (2000).
Qi, L., Wei, Y., Xu, L. & Liu, Z. Reaction behaviors and kinetics during induction period of methanol conversion on HZSM-5 zeolite. ACS Catal. 5, 3973–3982 (2015).
Liu, Z., Dong, X., Liu, X. & Han, Y. Oxygen-containing coke species in zeolite-catalyzed conversion of methanol to hydrocarbons. Catal. Sci. Technol. 6, 8157–8165 (2016).
Gabrienko, A. A., Arzumanov, S. S., Toktarev, A. V. & Stepanov, A. G. Solid-state NMR characterization of the structure of intermediates formed from olefins on metal oxides (Al2O3 and Ga2O3). J. Phys. Chem. C Nanomater. Interfaces 116, 21430–21438 (2012).
Xu, T., Munson, E. J. & Haw, J. F. Toward a systematic chemistry of organic reactions in zeolites: in situ NMR studies of ketones. J. Am. Chem. Soc. 116, 1962–1972 (1994).
Ramirez, A. et al. Effect of zeolite topology and reactor configuration on the direct conversion of CO2 to light olefins and aromatics. ACS Catal. 9, 6320–6334 (2019).
Chang, C. D. & Silvestri, A. J. The conversion of methanol and other o-compounds to hydrocarbons over zeolite catalysts. J. Catal. 47, 249–259 (1977).
Khare, R., Arora, S. S. & Bhan, A. Implications of cofeeding acetaldehyde on ethene selectivity in methanol-to-hydrocarbons conversion on MFI and Its mechanistic interpretation. ACS Catal. 6, 2314–2331 (2016).
Yan, T. et al. On the deactivation mechanism of zeolite catalyst in ethanol to butadiene conversion. J. Catal. 367, 7–15 (2018).
Ni, Y., Zhu, W. & Liu, Z. H-ZSM-5-catalyzed hydroacylation involved in the coupling of methanol and formaldehyde to aromatics. ACS Catal. 9, 11398–11403 (2019).
Wang, C. et al. New insight into the hydrocarbon-pool chemistry of the methanol-to-olefins conversion over zeolite H-ZSM-5 from GC-MS, solid-state NMR spectroscopy, and DFT calculations. Chem. Eur. J. 20, 12432–12443 (2014).
Olsbye, U. et al. The formation and degradation of active species during methanol conversion over protonated zeotype catalysts. Chem. Soc. Rev. 44, 7155–7176 (2015).
Wang, C. et al. Experimental evidence on the formation of ethene through carbocations in methanol conversion over H-ZSM-5 zeolite. Chem. Eur. J. 21, 12061–12068 (2015).
Dokania, A. et al. Acidity modification of ZSM-5 for enhanced production of light olefins from CO2. J. Catal. 381, 347–354 (2020).
Ramirez, A. et al. Selectivity descriptors for the direct hydrogenation of CO2 to hydrocarbons during zeolite-mediated bifunctional catalysis. Nat. Commun. 12, 5914 (2021).
Chen, H. et al. A mechanistic study of syngas to light olefins over OXZEO bifunctional catalysts: insights into the initial carbon-carbon bond formation on the oxide. Catal. Sci. Technol. 12, 1289–1295 (2022).
Gao, P. et al. DNP NMR reveals the hidden surface C–C bond growth mechanism over ZnAlOx during syngas conversion. J. Energy Chem. 67, 640–644 (2022).
Haw, J. F. et al. Roles for cyclopentenyl cations in the synthesis of hydrocarbons from methanol on zeolite catalyst HZSM-5. J. Am. Chem. Soc. 122, 4763–4775 (2000).
Gao, P. et al. A mechanistic study of methanol-to-aromatics reaction over Ga-modified ZSM-5 zeolites: understanding the dehydrogenation process. ACS Catal. 8, 9809–9820 (2018).
Hu, M. et al. Establishing a link between the dual cycles in methanol-to-olefins conversion on H-ZSM-5: aromatization of cycloalkenes. ACS Catal. 10, 4299–4305 (2020).
Luzgin, M. V., Romannikov, V. N., Stepanov, A. G. & Zamaraev, K. I. Interaction of olefins with carbon monoxide on zeolite H-ZSM-5. NMR observation of the Friedel−Crafts acylation of alkenes at ambient temperature. J. Am. Chem. Soc. 118, 10890–10891 (1996).
Frontier, A. J. & Hernandez, J. J. New twists in Nazarov cyclization chemistry. Acc. Chem. Res. 53, 1822–1832 (2020).
Liu, X. et al. Tandem catalysis for hydrogenation of CO and CO2 to lower olefins with bifunctional catalysts composed of spinel oxide and SAPO-34. ACS Catal. 10, 8303–8314 (2020).
Massiot, D. et al. Modelling one- and two-dimensional solid-state NMR spectra. Magn. Reson. Chem. 40, 70–76 (2002).
Hou, G., Yan, S., Trébosc, J., Amoureux, J.-P. & Polenova, T. Broadband homonuclear correlation spectroscopy driven by combined R2nv sequences under fast magic angle spinning for NMR structural analysis of organic and biological solids. J. Magn. Reson. 232, 18–30 (2013).
Shu, J., Li, P., Chen, Q. & Zhang, S. Quantitative measurement of polymer compositions by NMR spectroscopy:targeting polymers with marked difference in phase mobility. Macromolecules 43, 8993–8996 (2010).
Zhang, R., Mroue, K. H. & Ramamoorthy, A. Hybridizing cross-polarization with NOE or refocused-INEPT enhances the sensitivity of MAS NMR spectroscopy. J. Magn. Reson. 266, 59–66 (2016).
Kulminskaya, N. et al. Access to side-chain carbon information in deuterated solids under fast MAS through non-rotor-synchronized mixing. Chem. Commun. 52, 268–271 (2016).
Acknowledgements
This work was financially supported by the National Key R&D Programme of China (no. 2021YFA1502803), National Natural Science Foundation of China (grant nos. 21773230, 91945302, 21902158 and 22002165), LiaoNing Revitalization Talents Programme (no. XLYC1807207), China National Postdoctoral Programme for Innovative Talents (no. BX20190321) and the China Postdoctoral Science Foundation (nos. 2019M651161 and 2019M651154). P.G. thanks the Special Assistant Funding of the Chinese Academy of Science. The authors thank Y. Ding for NH3-TPD measurements and S. Li for help in the operation of SEM.
Author information
Authors and Affiliations
Contributions
G.H. conceived the project. G.H., P.G. and Y.J. designed the experiments, analysed the data and wrote the paper. Y.J. and P.G. prepared the catalysts, conducted catalytic tests and carried out NMR measurements. Y.J. carried out structural characterization of catalysts. Z.Z. and K.C. participated in experiment design. D.X. carried out partial NMR measurements. Q.H. and H.C. prepared partial catalysts. Q.H. participated in TEM measurement. All authors participated in the analysis of experimental data and discussion of the results.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–27, Notes 1–5 and Tables 1–6.
Rights and permissions
About this article
Cite this article
Ji, Y., Gao, P., Zhao, Z. et al. Oxygenate-based routes regulate syngas conversion over oxide–zeolite bifunctional catalysts. Nat Catal 5, 594–604 (2022). https://doi.org/10.1038/s41929-022-00806-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-022-00806-2
This article is cited by
-
Upgrading CO2 to sustainable aromatics via perovskite-mediated tandem catalysis
Nature Communications (2024)
-
Giving oxygenates a new spin
Nature Catalysis (2022)