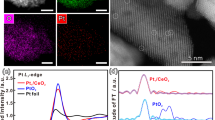
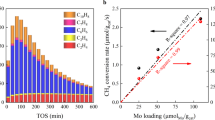
Abstract
Efficient and direct conversion of methane to value-added products has been a long-term challenge in shale gas applications. Here, we show that atomically thin nanolayers of Pt with a single or double atomic layer thickness, supported on a two-dimensional molybdenum titanium carbide (MXene), catalyse non-oxidative coupling of methane to ethane/ethylene (C2). Kinetic and theoretical studies, combined with in-situ spectroscopic and microscopic characterizations, demonstrate that Pt nanolayers anchored at the hexagonal close-packed sites of the MXene support can activate the first C–H bond of methane to form methyl radicals that favour desorption over further dehydrogenation and thus suppress coke deposition. At 750 °C and 7% methane conversion, the catalyst runs for 72 hours of continuous operation without deactivation and exhibits >98% selectivity towards C2 products, with a turnover frequency of 0.2–0.6 s−1. Our findings provide insights into the design of highly active and stable catalysts for methane activation and create a platform for developing atomically thin supported metal catalysts.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are deposited at https://iastate.box.com/s/sf9go743qngg8ta0ufni0q102nu7hvjv. All the data and access are available from the corresponding authors on reasonable request.
References
Qiao, B. et al. Single-atom catalysis of CO oxidation using Pt1/FeOx. Nat. Chem. 3, 634–641 (2011).
Coperet, C. et al. Surface organometallic and coordination chemistry toward single-site heterogeneous catalysts: strategies, methods, structures, and activities. Chem. Rev. 116, 323–421 (2016).
Jones, J. et al. Thermally stable single-atom platinum-on-ceria catalysts via atom trapping. Science 353, 150–154 (2016).
Kuznetsov, D. A. et al. Single site cobalt substitution in 2D molybdenum carbide (MXene) enhances catalytic activity in the hydrogen evolution reaction. J. Am. Chem. Soc. 141, 17809–17816 (2019).
Liu, P. et al. Photochemical route for synthesizing atomically dispersed palladium catalysts. Science 352, 797–800 (2016).
Xiong, H. et al. Thermally stable and regenerable platinum-tin clusters for propane dehydrogenation prepared by atom trapping on ceria. Angew. Chem. Int. Ed. Engl. 56, 8986–8991 (2017).
Zhu, Y. et al. Lattice-confined Sn (IV/II) stabilizing raft-like Pt clusters: high selectivity and durability in propane dehydrogenation. ACS Catal. 7, 6973–6978 (2017).
Kwak, J. H., Kovarik, L. & Szanyi, J. Heterogeneous catalysis on atomically dispersed supported metals: CO2 reduction on multifunctional Pd catalysts. ACS Catal. 3, 2094–2100 (2013).
Zhu, Y. et al. Covalent-bonding to irreducible SiO2 leads to high-loading and atomically dispersed metal catalysts. J. Catal. 353, 315–324 (2017).
Hou, Y., Nagamatsu, S., Asakura, K., Fukuoka, A. & Kobayashi, H. Trace mono-atomically dispersed rhodium on zeolite-supported cobalt catalyst for the efficient methane oxidation. Commun. Chem. 1, 41 (2018).
DeRita, L. et al. Catalyst architecture for stable single atom dispersion enables site-specific spectroscopic and reactivity measurements of CO adsorbed to Pt atoms, oxidized Pt clusters, and metallic Pt clusters on TiO2. J. Am. Chem. Soc. 139, 14150–14165 (2017).
Liu, J. Catalysis by supported single metal atoms. ACS Catal. 7, 34–59 (2017).
Kwak, J. H. et al. Coordinatively unsaturated Al3+ centers as binding sites for active catalyst phases of platinum on γ-Al2O3. Science 325, 1670–1673 (2009).
Yao, S. et al. Atomic-layered Au clusters on α-MoC as catalysts for the low-temperature water-gas shift reaction. Science 357, 389–393 (2017).
Wang, S. et al. In situ atomic-scale studies of the formation of epitaxial Pt nanocrystals on monolayer molybdenum disulfide. ACS Nano 11, 9057–9067 (2017).
Göhl, D. et al. Engineering stable electrocatalysts by synergistic stabilization between carbide cores and Pt shells. Nat. Mater. 19, 287–291 (2020).
Wang, L. et al. Tunable intrinsic strain in two-dimensional transition metal electrocatalysts. Science 363, 870–874 (2019).
Esposito, D. V., Hunt, S. T., Kimmel, Y. C. & Chen, J. G. A new class of electrocatalysts for hydrogen production from water electrolysis: metal monolayers supported on low-cost transition metal carbides. J. Am. Chem. Soc. 134, 3025–3033 (2012).
Schweitzer, N. M. et al. High activity carbide supported catalysts for water gas shift. J. Am. Chem.Soc. 133, 2378–2381 (2011).
Hunt, S. T. et al. Self-assembly of noble metal monolayers on transition metal carbide nanoparticle catalysts. Science 352, 974–978 (2016).
Guo, X. et al. Direct, nonoxidative conversion of methane to ethylene, aromatics, and hydrogen. Science 344, 616–619 (2014).
Marcinkowski, M. D. et al. Pt/Cu single-atom alloys as coke-resistant catalysts for efficient C-H activation. Nat. Chem. 10, 325–332 (2018).
Spivey, J. J. & Hutchings, G. Catalytic aromatization of methane. Chem. Soc. Rev. 43, 792–803 (2014).
Belgued, M., Pareja, P., Amariglio, A. & Amariglio, H. Conversion of methane into higher hydrocarbons on platinum. Nature 352, 789–790 (1991).
Naguib, M. et al. Two-dimensional nanocrystals produced by exfoliation of Ti3AlC2. Adv. Mater. 23, 4248–4253 (2011).
Thakur, R. et al. Insights into the genesis of a selective and coke-resistant MXene-based catalyst for the dry reforming of methane. ACS Catal. 10, 5124–5134 (2020).
Anasori, B. et al. Two-dimensional, ordered, double transition metals carbides (MXenes). ACS Nano 9, 9507–9516 (2015).
Zhang, J. et al. Single platinum atoms immobilized on an MXene as an efficient catalyst for the hydrogen evolution reaction. Nat. Catal. 1, 985–992 (2018).
Li, Z. et al. Reactive metal-support interactions at moderate temperature in two-dimensional niobium-carbide-supported platinum catalysts. Nat. Catal. 1, 349–355 (2018).
Li, Z. et al. Two-dimensional transition metal carbides as supports for tuning the chemistry of catalytic nanoparticles. Nat. Commun. 9, 5258 (2018).
Li, Z. et al. In situ formed Pt3Ti nanoparticles on a two-dimensional transition metal carbide (MXene) used as efficient catalysts for hydrogen evolution reactions. Nano Lett. 19, 5102–5108 (2019).
Liu, J. Advanced electron microscopy of metal-support interactions in supported metal catalysts. ChemCatChem 3, 934–948 (2011).
Schultz, T. et al. Surface termination dependent work function and electronic properties of Ti3C2Tx MXene. Chem. Mater. 31, 6590–6597 (2019).
Gerceker, D. et al. Methane conversion to ethylene and aromatics on PtSn catalysts. ACS Catal. 7, 2088–2100 (2017).
Xiao, Y. & Varma, A. Highly selective nonoxidative coupling of methane over Pt-Bi bimetallic catalysts. ACS Catal. 8, 2735–2740 (2018).
Xie, P. et al. Nanoceria-supported single-atom platinum catalysts for direct methane conversion. ACS Catal. 8, 4044–4048 (2018).
Ghose, R., Hwang, H. T. & Varma, A. Oxidative coupling of methane using catalysts synthesized by solution combustion method: catalyst optimization and kinetic studies. Appl. Catal. A 472, 39–46 (2014).
Dutta, K., Shahryari, M. & Kopyscinski, J. Direct non-oxidative methane coupling to ethylene over gallium nitride. A catalyst regeneration study. Ind. Eng. Chem. Res. 59, 4245–4256 (2020).
Deeva, E. B. et al. In situ XANES/XRD study of the structural stability of two-dimensional molybdenum carbide Mo2CTx: implications for the catalytic activity in the water-gas shift reaction. Chem. Mater. 31, 4505–4513 (2019).
Wegener, E. C. et al. Intermetallic compounds as an alternative to single-atom alloy catalysts: geometric and electronic structures from advanced X-ray spectroscopies and computational studies. ChemCatChem 12, 1325–1333 (2020).
Nørskov, J. K., Abild-Pedersen, F., Studt, F. & Bligaard, T. Density functional theory in surface chemistry and catalysis. Proc. Natl Acad. Sci. USA 108, 937–943 (2011).
Xi, Y. & Heyden, A. Direct oxidation of methane to methanol enabled by electronic atomic monolayer–metal support interaction. ACS Catal. 9, 6073–6079 (2019).
Wang, W., Jiang, Y. & Hunger, M. Mechanistic investigations of the methanol-to-olefin (MTO) process on acidic zeolite catalysts by in situ solid-state NMR spectroscopy. Catal. Today 113, 102–114 (2006).
Kosinov, N. et al. Structure and evolution of confined carbon species during methane dehydroaromatization over Mo/ZSM-5. ACS Catal. 8, 8459–8467 (2018).
Ohnishi, R., Liu, S., Dong, Q., Wang, L. & Ichikawa, M. Catalytic dehydrocondensation of methane with CO and CO2 toward benzene and naphthalene on Mo/HZSM-5 and Fe/Co-modified Mo/HZSM-5. J. Catal. 182, 92–103 (1999).
Kosinov, N. et al. Reversible nature of coke formation on Mo/ZSM-5 methane dehydroaromatization catalysts. Angew. Chem. Int. Ed. Engl. 58, 7068–7072 (2019).
Finiels, A., Fajula, F. & Hulea, V. Nickel-based solid catalysts for ethylene oligomerization-a review. Catal. Sci. Technol. 4, 2412–2426 (2014).
Ravel, B. & Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 12, 537–541 (2005).
Weisz, P. & Prater, C. Interpretation of measurements in experimental catalysis. Adv. Catal. 6, 60390–60399 (1954).
Mears, D. E. Diagnostic criteria for heat transport limitations in fixed bed reactors. J. Catal. 20, 127–131 (1971).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169 (1996).
Gunasooriya, G. K. K. & Nørskov, J. K. Analysis of acid-stable and active oxides for the oxygen evolution reaction. ACS Energy Lett. 5, 3778–3787 (2020).
Campbell, C. T., Árnadóttir, L. & Sellers, J. R. Kinetic prefactors of reactions on solid surfaces. Z. Phys. Chem. 227, 1435–1454 (2013).
Acknowledgements
Y.W. appreciates the support from the Herbert L. Stiles Professorship and Iowa State University College of Engineering exploratory research projects. J.C.Z., Z.W. and J.T.M. were supported in part by the National Science Foundation under Cooperative Agreement no. EEC-1647722. Y.X. and A.V. thank the R. Games Slayter Fund and the Varma Reaction Engineering Research Fund of Purdue University. Z.Z. and J.P.G. acknowledge financial support from NSF-CBET Award 1804712. T.M. acknowledges the financial support of the University of Michigan College of Engineering and technical support from the Michigan Center for Materials Characterization. Use of the Advanced Photon Source was supported by the US Department of Energy, Office of Basic Energy Sciences under contract no. DE-AC02-06CH11357. MRCAT operations, beamline 10-BM, are supported by the Department of Energy and the MRCAT member institutions. Use of the Stanford Synchrotron Radiation Lightsource, SLAC National Accelerator Laboratory, is supported by the US Department of Energy, Office of Science, Office of Basic Energy Sciences under contract no. DE-AC02-76SF00515. All TEM-related work was performed using instruments in the Sensitive Instrument Facility in Ames Laboratory. Ames Laboratory is operated for the US Department of Energy by Iowa State University under contract no. DE-AC02-07CH11358.
Author information
Authors and Affiliations
Contributions
Z.L. and Y.X. conceived the idea and designed the present work. Y.X., P.R.C. and Z.Z. conducted DFT calculations. Z.L., Y.X., P.J.P. and P.H. synthesized the catalysts and performed the catalytic evaluation. J.Z.C., Z.W., G.W., D.J. and J.T.M. carried out the spectroscopic characterizations. L.Z., T.M., T.-H.K. and Z.L. performed the detailed microscopic experiments. Y.W., J.T.M., X.R., J.P.G. and A.V. supervised the research.
Corresponding authors
Ethics declarations
Competing interests
Z.L., Y.W. and X.Y. are inventors on US. Provisional Patent Application 62/937,055, submitted by Iowa State University. The remaining authors declare no competing interests.
Additional information
Peer review information Nature Catalysis thanks Chen Chen and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Notes 1–3, Figs. 1–36, Tables 1–8,and refs. 1–5,
Supplementary Data 1
Atomic coordinates of optimized structures for DFT calculations conducted in this study
Rights and permissions
About this article
Cite this article
Li, Z., Xiao, Y., Chowdhury, P.R. et al. Direct methane activation by atomically thin platinum nanolayers on two-dimensional metal carbides. Nat Catal 4, 882–891 (2021). https://doi.org/10.1038/s41929-021-00686-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-021-00686-y
This article is cited by
-
Accurate atomic scanning transmission electron microscopy analysis enabled by deep learning
Nano Research (2024)
-
Synergistically improved tracking resistance of silicone rubber with ZrP nanosheet and urethane-containing silane
Journal of Thermal Analysis and Calorimetry (2024)
-
Two-dimensional atomically thin Pt layers on MXenes: The role of electronic effects during catalytic dehydrogenation of ethane and propane
Nano Research (2024)
-
2D MXenes polar catalysts for multi-renewable energy harvesting applications
Nature Communications (2023)
-
Conjugated dual size effect of core-shell particles synergizes bimetallic catalysis
Nature Communications (2023)