Abstract
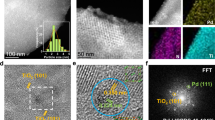
The selective activation of inert C–H bonds has emerged as a promising tool for avoiding the use of wasteful traditional coupling reactions. Oxidative coupling of simple aromatics allows for a cost-effective synthesis of biaryls. However, utilization of this technology is severely hampered by poor regioselectivity and by the limited stability of state-of-the-art homogeneous Pd catalysts. Here, we show that confinement of cationic Pd in the pores of a zeolite allows for the shape-selective C–H activation of simple aromatics without a functional handle or electronic bias. For instance, out of six possible isomers, 4,4′-bitolyl is produced with high shape selectivity (80%) in oxidative toluene coupling on Pd-Beta. Not only is a robust, heterogeneous catalytic system obtained, but this concept is also set to control the selectivity in transition-metal-catalysed arene C–H activation through spatial confinement in zeolite pores.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The findings of this study are available in the main text or the supplementary materials. Atomic coordinates of optimized computational models and initial and final configurations of molecular dynamics trajectories are supplied in a Supplementary Data file. All data are available from the authors upon reasonable request.
References
Hassan, J., Sévignon, M., Gozzi, C., Schulz, E. & Lemaire, M. Aryl−aryl bond formation one century after the discovery of the Ullmann reaction. Chem. Rev. 102, 1359–1469 (2002).
Mondschein, R. J. et al. Synthesis and characterization of amorphous bibenzoate (co)polyesters: permeability and rheological performance. Macromolecules 50, 7603–7610 (2017).
De Smit, E. et al., Hydroalkylation catalyst and process for use thereof. US patent 2014/0378697 (2014).
Dakka, J. M. et al. Biphenyl esters, their production and their use in the manufacture of plasticizers. US patent 9,556,103 (2017).
Chen, X., Engle, K. M., Wang, D. ‐H. & Yu, J. ‐Q. Palladium(II)-catalyzed C–H activation/C–C cross-coupling reactions: versatility and practicality. Angew. Chem. Int. Ed. 48, 5094–5115 (2009).
Xu, B.-Q., Sood, D., Iretskii, A. V. & White, M. G. Direct synthesis of dimethylbiphenyls by toluene coupling in the presence of palladium triflate and triflic acid. J. Catal. 187, 358–366 (1999).
Izawa, Y. & Stahl, S. S. Aerobic oxidative coupling of o‐xylene: discovery of 2‐fluoropyridine as a ligand to support selective Pd‐catalyzed C–H functionalization. Adv. Synth. Catal. 352, 3223–3229 (2010).
Wang, D., Izawa, Y. & Stahl, S. S. Pd-catalyzed aerobic oxidative coupling of arenes: evidence for transmetalation between two Pd(ii)–aryl intermediates. J. Am. Chem. Soc. 136, 9914–9917 (2014).
Wang, D. & Stahl, S. S. Pd-catalyzed aerobic oxidative biaryl coupling: non-redox cocatalysis by Cu(OTf)2 and discovery of Fe(OTf)3 as a highly effective cocatalyst. J. Am. Chem. Soc. 139, 5704–5707 (2017).
Álvarez‐Casao, Y. et al. Palladium‐catalyzed cross‐dehydrogenative coupling of o‐xylene: evidence of a new rate‐limiting step in the search for industrially relevant conditions. ChemCatChem 10, 2620–2626 (2018).
Yang, Y., Lan, J. & You, J. Oxidative C–H/C–H coupling reactions between two (hetero)arenes. Chem. Rev. 117, 8787–8863 (2017).
Kuhl, N., Hopkinson, M. N., Wencel-Delord, J. & Glorius, F. Beyond directing groups: transition-metal-catalyzed C–H activation of simple arenes. Angew. Chem. Int. Ed. 51, 10236–10254 (2012).
Sambiagio, C. et al. A comprehensive overview of directing groups applied in metal-catalysed C–H functionalisation chemistry. Chem. Soc. Rev. 47, 6603–6743 (2018).
Leow, D., Li, G., Mei, T.-S. & Yu, J.-Q. Activation of remote meta-C–H bonds assisted by an end-on template. Nature 486, 518–522 (2012).
Boursalian, G. B., Ham, W. S., Mazzotti, A. R. & Ritter, T. Charge-transfer-directed radical substitution enables para-selective C–H functionalization. Nat. Chem. 8, 810–815 (2016).
Berger, F. et al. Site-selective and versatile aromatic C–H functionalization by thianthrenation. Nature 567, 223–228 (2019).
Dey, A., Maity, S. & Maiti, D. Reaching the south: metal-catalyzed transformation of the aromatic para-position. Chem. Commun. 52, 12398–12414 (2016).
Van Speybroeck, V. et al. Advances in theory and their application within the field of zeolite chemistry. Chem. Soc. Rev. 44, 7044–7111 (2015).
Kosinov, N., Liu, C., Hensen, E. J. M. & Pidko, E. A. Engineering of transition metal catalysts confined in zeolites. Chem. Mater. 30, 3177–3198 (2018).
Lawton, S. L., Leonowicz, M. E., Partridge, R. D., Chu, P. & Rubin, M. K. Twelve-ring pockets on the external surface of MCM-22 crystals. Micropor. Mesopor. Mat. 23, 109–117 (1998).
Van Velthoven, N. et al. Single-site metal–organic framework catalysts for the oxidative coupling of arenes via C–H/C–H activation. Chem. Sci. 10, 3616–3622 (2019).
Kaplan, G. Preparation of biphenols by oxidative coupling of alkylphenols using a recyclable copper catalyst. US patent 2003/0050515 (2003)
Eckardt, M., Greb, A. & Simat, T. J. Polyphenylsulfone (PPSU) for baby bottles: a comprehensive assessment on polymer-related non-intentionally added substances (NIAS). Food Addit. Contam. A 35, 1421–1437 (2018).
Davies, D. L., Macgregor, S. A. & McMullin, C. L. Computational studies of carboxylate-assisted C–H activation and functionalization at group 8–10 transition metal centers. Chem. Rev. 117, 8649–8709 (2017).
Chen, B., Hou, X., Li, Y. & Wu, Y. Mechanistic understanding of the unexpected meta selectivity in copper-catalyzed anilide C–H bond arylation. J. Am. Chem. Soc. 133, 7668–7671 (2011).
Deng, C., Zhang, J. & Lin, Z. Theoretical studies on Pd(ii)-catalyzed meta-selective C–H bond arylation of arenes. ACS Catal. 8, 2498–2507 (2018).
Lane, B. S., Brown, M. A. & Sames, D. Direct palladium-catalyzed C-2 and C-3 arylation of indoles: a mechanistic rationale for regioselectivity. J. Am. Chem. Soc. 127, 8050–8057 (2005).
Popp, B. V. & Stahl, S. S. Insertion of molecular oxygen into a palladium−hydride bond: computational evidence for two nearly isoenergetic pathways. J. Am. Chem. Soc. 129, 4410–4422 (2007).
Konnick, M. & Stahl, S. S. Reaction of molecular oxygen with a PdII-hydride to produce a PdII-hydroperoxide: experimental evidence for an HX-reductive-elimination pathway. J. Am. Chem. Soc. 130, 5753–5762 (2008).
Acknowledgements
We thank N. Van Velthoven for discussion. The XAS experiments were performed on beamline BM26A at the European Synchrotron Radiation Facility (ESRF), Grenoble, France. This work was funded by grants from FWO (1S17620N for J.V.; project G0D0518N, G0F2320N, G078118N; EoS BioFACT), the Flemish government (CASAS Methusalem programme for D.D.V.). V.V.S., J.H. and M.B. acknowledge the Research Board of Ghent University (BOF) and funding from the European Union’s Horizon 2020 research and innovation programme (consolidator ERC grant agreement No. 647755 – DYNPOR (2015-2020)). The computational resources and services used were provided by Ghent University (Stevin Supercomputer Infrastructure) and the VSC (Flemish Supercomputer Center), funded by the Research Foundation - Flanders (FWO). A.S. and A.B. acknowledge the funding from Russian Science Foundation (joint RSF-FWO grant No. 20-43-01015). A.K. and G.M. acknowledge the financial support from the Slovenian Research Agency (research core funding No. P1-0021 and project No. N1-0079).
Author information
Authors and Affiliations
Contributions
Under the supervision of D.D.V., J.V. was responsible for the conception, design and interpretation of the experiments. S.V.M. performed additional experiments. Under the supervision of V.V.S., J.H., S.N. and M.B. performed the DFT calculations. A.B. and A.S. conceived and performed the XAS experiments. A.K. and G.M. conceived and performed the NMR experiments. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
J.V., P.T. and D.D.V. filed a patent application GB1804905.6 prior to an international patent application PCT/EP2019/057746.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Discussion, Figs. 1–53, Tables 1–16.
Supplementary Data 1
Atomic coordinates of optimized computational models and initial and final configurations of molecular dynamics trajectories.
Rights and permissions
About this article
Cite this article
Vercammen, J., Bocus, M., Neale, S. et al. Shape-selective C–H activation of aromatics to biarylic compounds using molecular palladium in zeolites. Nat Catal 3, 1002–1009 (2020). https://doi.org/10.1038/s41929-020-00533-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-020-00533-6
This article is cited by
-
Investigation of the Suzuki–Miyaura cross-coupling reaction on a palladium H-beta zeolite with DFT calculations
Scientific Reports (2024)
-
Ensemble effect for single-atom, small cluster and nanoparticle catalysts
Nature Catalysis (2022)