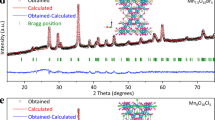
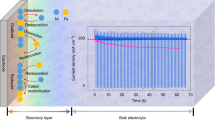
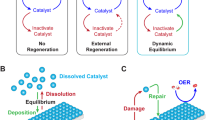
Abstract
Electrochemical water splitting in acidic conditions offers important advantages over that in alkaline systems, but the technological progress is limited by the lack of inexpensive and efficient anode catalysts that can stably operate at a low pH and elevated temperature. Here we demonstrate oxygen evolution catalysts that are based on non-noble metals, are formed in situ during electrooxidation of acidic water and exhibit a high stability in operation due to a self-healing mechanism. The highly disordered mixed metal oxides generated from dissolved cobalt, lead and iron precursors sustain high water oxidation rates at reasonable overpotentials. Moreover, utilizing a sufficiently robust electrode substrate allows for a continuous water oxidation at temperatures up to 80 °C and rates up to 500 mA cm−2 at overpotentials below 0.7 V with an essentially flat support and with no loss in activity. This robust operation of the catalysts is provided by the thermodynamically stable lead oxide matrix that accommodates homogeneously distributed catalytic dopants.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support all findings of this study are available from the corresponding author upon request.
References
Lewis, N. S. & Nocera, D. G. Powering the planet: chemical challenges in solar energy utilization. Proc. Natl Acad. Sci. USA 103, 15729–15735 (2006).
Lewis, N. S. Research opportunities to advance solar energy utilization. Science 351, 353 (2016).
Nielander, A. C., Shaner, M. R., Papadantonakis, K. M., Francis, S. A. & Lewis, N. S. A taxonomy for solar fuels generators. Energy Environ. Sci. 8, 16–25 (2015).
González, E. L., Llerena, F. I., Pérez, M. S., Iglesias, F. R. & Macho, J. G. Energy evaluation of a solar hydrogen storage facility: Comparison with other electrical energy storage technologies. Int. J. Hydrog. Energy 40, 5518–5525 (2015).
Moreno-Hernandez, I. A. et al. Crystalline nickel manganese antimonate as a stable water-oxidation catalyst in aqueous 1.0 M H2SO4. Energy Environ. Sci. 10, 2103–2108 (2017).
Jiao, F. & Frei, H. Nanostructured cobalt and manganese oxide clusters as efficient water oxidation catalysts. Energy Environ. Sci. 3, 1018–1027 (2010).
Gong, M. & Dai, H. A mini review of NiFe-based materials as highly active oxygen evolution reaction electrocatalysts. Nano Res. 8, 23–39 (2014).
Chatti, M. et al. Sustainable energy & fuels highly dispersed and disordered nickel–iron layered hydroxides and sulphides: robust and high-activity water oxidation catalysts. Sustain. Energ. Fuels 2, 1561–1573 (2018).
Roger, I., Shipman, M. A. & Symes, M. D. Earth-abundant catalysts for electrochemical and photoelectrochemical water splitting. Nat. Rev. Chem. 1, 0003 (2017).
Kanan, M. W. & Nocera, D. G. In situ formation of an oxygen-evolving catalyst in neutral water containing phosphate and Co2+. Science 321, 1072–1075 (2008).
McKone, J. R., Sadtler, B. F., Werlang, C. A., Lewis, N. S. & Gray, H. B. Ni–Mo nanopowders for efficient electrochemical hydrogen evolution. ACS Catal. 3, 166–169 (2013).
Carmo, M., Fritz, D. L., Mergel, J. & Stolten, D. A comprehensive review on PEM water electrolysis. Int. J. Hydrog. Energy 38, 4901–4934 (2013).
Huynh, M., Ozel, T., Liu, C., Lau, E. C. & Nocera, D. G. Design of template-stabilized active and Earth-abundant oxygen evolution catalysts in acid. Chem. Sci. 8, 4779–4794 (2017).
Park, S., Shao, Y., Liu, J. & Wang, Y. Oxygen electrocatalysts for water electrolyzers and reversible fuel cells : status and perspective. Energy Environ. Sci. 5, 9331–9344 (2012).
Leng, Y. et al. Solid-state water electrolysis with an alkaline membrane. J. Am. Chem. Soc. 134, 9054–9057 (2012).
Reier, T., Nong, H. N., Teschner, D., Schlögl, R. & Strasser, P. Electrocatalytic oxygen evolution reaction in acidic environments—reaction mechanisms and catalysts. Adv. Energy Mater. 7, 1601275 (2017).
Schalenbach, M. et al. Acidic or alkaline? Towards a new perspective on the efficiency of water electrolysis. J. Electrochem. Soc. 163, F3197–F3208 (2016).
Hinnemann, B. et al. Biomimetic hydrogen evolution : MoS2 nanoparticles as catalyst for hydrogen evolution. J. Am. Chem. Soc. 127, 5308–5309 (2005).
McCrory, C. C. L. et al. Benchmarking hydrogen evolving reaction and oxygen evolving reaction electrocatalysts for solar water splitting devices. J. Am. Chem. Soc. 137, 4347–4357 (2015).
Mondschein, J. S. et al. Crystalline cobalt oxide films for sustained electrocatalytic oxygen evolution under strongly acidic conditions. Chem. Mater. 29, 950–957 (2017).
Mondschein, J. S. et al. Intermetallic Ni2Ta electrocatalyst for the oxygen evolution reaction in highly acidic electrolytes. Inorg. Chem. 57, 6010–6015 (2018).
Li, X., Pletcher, D. & Walsh, F. C. Electrodeposited lead dioxide coatings. Chem. Soc. Rev. 40, 3879–3894 (2011).
Musiani, M. Anodic deposition of PbO2/Co3O4 composites and their use as electrodes for oxygen evolution reaction. Chem. Commun. 2, 2403–2404 (1996).
Velichenko, A. B. et al. Oxygen evolution on lead dioxide modified with fluorine and iron. Russ. J. Electrochem. 36, 1216–1220 (2000).
Yu, P. & Keefe, T. J. O. Evaluation of lead anode reactions in acid sulfate electrolytes. J. Electrochem. Soc. 149, 558–569 (2002).
Velichenko, A. B., Amadelli, R., Baranova, E. A., Girenko, D. V. & Danilov, F. I. Electrodeposition of Co-doped lead dioxide and its physicochemical properties. J. Electroanal. Chem. 527, 56–64 (2002).
Nikoloski, A. N. & Nicol, M. J. Addition of cobalt to lead anodes used for oxygen evolution: a literature review. Miner. Process. Extr. Metall. Rev. 31, 30–57 (2010).
Yu, P. & OKeefe, T. J. Evaluation of lead anode reactions in acid sulfate electrolytes. I. Lead alloys with cobalt additives. J. Electrochem. Soc. 146, 1361–1369 (1999).
Nikoloski, A. N. & Barmi, M. J. Novel lead–cobalt composite anodes for copper electrowinning. Hydrometallurgy 137, 45–52 (2013).
Schmachtel, S. et al. Simulation of electrochemical processes during oxygen evolution on Pb–MnO2 composite electrodes. Electrochim. Acta 245, 512–525 (2017).
Lutterman, D. A., Surendranath, Y. & Nocera, D. G. A self-healing oxygen-evolving catalyst. J. Am. Chem. Soc. 131, 3838–3839 (2009).
Bloor, L. G., Molina, P. I., Symes, M. D. & Cronin, L. Low pH electrolytic water splitting using Earth-abundant metastable catalysts that self-assemble in situ. J. Am. Chem. Soc. 136, 3304–3311 (2014).
Bonke, S. A. et al. Electrolysis of natural waters contaminated with transition-metal ions: identification of a metastable FePb-based oxygen-evolution catalyst operating in weakly acidic solutions. ChemPlusChem 83, 704–710 (2018).
Cattarin, S. & Musiani, M. Electrosynthesis of nanocomposite materials for electrocatalysis. Electrochim. Acta 52, 2796–2805 (2007).
Huang, L. F., Hutchison, M. J., Santucci, R. J., Scully, J. R. & Rondinelli, J. M. Improved electrochemical phase diagrams from theory and experiment: the Ni–water system and its complex compounds. J. Phys. Chem. C 121, 9782–9789 (2017).
Tekerlekopoulou, A. G., Pavlou, S. & Vayenas, D. V. Removal of ammonium, iron and manganese from potable water in biofiltration units: a review. J. Chem. Technol. Biotechnol. 88, 751–773 (2013).
Velichenko, A. B., Amadelli, R., Zucchini, G. L., Girenko, D. V. & Danilov, F. I. Electrosynthesis and physicochemical properties of Fe-doped lead dioxide electrocatalysts. Electrochim. Acta 45, 4341–4350 (2000).
Musiani, M., Furlanetto, F. & Bertoncello, R. Electrodeposited PbO2 + RuO2: a composite anode for oxygen evolution from sulphuric acid solution. J. Electroanal. Chem. 465, 160–167 (1999).
Han, S., He, S., Xu, R., Wang, J. & Chen, B. An RDE research on the preparation process of β-PbO2–CoOx composite coatings. Int. J. Electrochem. Sci. 11, 8391–8404 (2016).
Cattarin, S., Guerriero, P. & Musiani, M. Preparation of anodes for oxygen evolution by electrodeposition of composite Pb and Co oxides. Electrochim. Acta 46, 4229–4234 (2001).
Bonke, S. A., Bond, A. M., Spiccia, L. & Simonov, A. N. Parameterization of water electrooxidation catalyzed by metal oxides using Fourier transformed alternating current voltammetry. J. Am. Chem. Soc. 138, 16095–16104 (2016).
Costentin, C., Porter, T. R. & Savéant, J. M. Conduction and reactivity in heterogeneous-molecular catalysis: new insights in water oxidation catalysis by phosphate cobalt oxide films. J. Am. Chem. Soc. 138, 5615–5622 (2016).
Fang, Y. & Liu, Z. Mechanism and Tafel lines of electro-oxidation of water to oxygen on RuO2(110). J. Am. Chem. Soc. 132, 18214–18222 (2010).
Halck, N. B., Petrykin, V., Krtil, P. & Rossmeisl, J. Beyond the volcano limitations in electrocatalysis—oxygen evolution reaction. Phys. Chem. Chem. Phys. 16, 13682–13688 (2014).
Mahalingam, T. et al. Electrosynthesis and characterization of lead oxide thin films. Mater. Charact. 58, 817–822 (2007).
Figueiredo, M. O., Silva, T. P. & Veiga, J. P. A XANES study of the structural role of lead in glazes from decorated tiles, XVI to XVIII century manufacture. Appl. Phys. A 83, 209–211 (2006).
Liang, Y. et al. Co3O4 nanocrystals on graphene as a synergistic catalyst for oxygen reduction reaction. Nat. Mater. 10, 780–786 (2011).
Kanan, M. W. et al. Structure and valency of a cobalt–phosphate water oxidation catalyst determined by in situ X-ray spectroscopy. J. Am. Chem. Soc. 132, 13692–13701 (2010).
King, H. J. et al. Engineering disorder into heterogenite-like cobalt oxides by phosphate doping: implications for the design of water-oxidation catalysts. ChemCatChem 9, 511–521 (2017).
Westre, T. E. et al. A multiplet analysis of Fe K-edge 1s → 3d pre-edge features of iron complexes. J. Am. Chem. Soc. 119, 6297–6314 (1997).
Benck, J. D., Pinaud, B. A., Gorlin, Y. & Jaramillo, T. F. Substrate selection for fundamental studies of electrocatalysts and photoelectrodes: inert potential windows in acidic, neutral, and basic electrolyte. PLoS ONE 9, e107942 (2014).
Staszak-jirkovský, J. et al. A highly active and stable IrOx/SrIrO3 catalyst for the oxygen evolution reaction. Science 353, 1–8 (2015).
Geiger, S. et al. The stability number as a metric for electrocatalyst stability benchmarking. Nat. Catal. 1, 508–515 (2018).
Hu, F. et al. Amorphous metallic NiFeP: a conductive bulk material achieving high activity for oxygen evolution reaction in both alkaline and acidic media. Adv. Mater. 29, 1–9 (2017).
Lei, C. et al. Fe-N4 sites embedded into carbon nanofiber integrated with electrochemically exfoliated graphene for oxygen evolution in acidic medium. Adv. Energy Mater. 8, 1801912 (2018).
Blasco-Ahicart, M., Soriano-Lopez, J., Carbo, J. J., Poblet, J. M. & Galan-Mascaros, J. R. Polyoxometalate electrocatalysts based on Earth-abundant metals for efficient water oxidation in acidic media. Nat. Chem 10, 24–30 (2018).
Yang, H. T. et al. Electrochemical behavior of rolled Pb–0.8%Ag anodes. Hydrometallurgy 140, 144–150 (2013).
Ma, R. et al. Oxygen evolution and corrosion behavior of low-MnO2-content Pb–MnO2 composite anodes for metal electrowinning. Hydrometallurgy 159, 6–11 (2016).
Hoogeveen, D. A. et al. Photo-electrocatalytic hydrogen generation at dye-sensitised electrodes functionalised with a heterogeneous metal catalyst. Electrochim. Acta 219, 773–780 (2016).
Hoogeveen, D. A. et al. Origin of photoelectrochemical generation of dihydrogen by a dye-sensitized photocathode without an intentionally introduced catalyst. J. Phys. Chem. C 121, 25836–25846 (2017).
Glover, C. et al. Status of the X-ray absorption spectroscopy (XAS) beamline at the Australian synchrotron. AIP Conf. Proc. 882, 884–886 (2007).
Cramer, S. P. & Hodgson, K. O. X-ray absorption spectroscopy: a new structural method and its applications to bioinorganic chemistry. Prog. Inorg. Chem 25, 1–39 (1979).
Ravel, B. & Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 12, 537–541 (2005).
Acknowledgements
The authors thank B.H.R. Suryanto (Monash University) for the instrumental support throughout the study, and P. Kappen and C. Glover (Australian Synchrotron) for support in the XAS experiments. The authors acknowledge the use of facilities within the Monash Centre for Electron Microscopy (funded by the Australian Research Council grant LE110100223) and Monash X-ray Platform (funded by Australian Research Council grant LE130100072). Part of this research was undertaken on the XAS beamline at the Australian Synchrotron, part of ANSTO. Funding of this work by the Australian Research Council through the ARC Centre of Excellence for Electromaterials Science (CE140100012) and by the Australian Renewable Energy Agency (ARENA contract no. 2018/RND008) is appreciated.
Author information
Authors and Affiliations
Contributions
M.C. designed and undertook electrochemical experiments, performed the SEM/EDX and XRD analysis, analysed and interpreted data, and co-wrote the paper. J.G. undertook electrochemical experiments, analysed and interpreted data, and co-wrote the paper. M.F. undertook O2 detection and ICP-OES analysis, and contributed to data analysis. B.J. undertook XAS experiments. T.W. collected and analysed TEM data. T.R.G. collected and analysed XPS data. N.P. undertook cross-sectional SEM and ICP-OES analyses. C.N. undertook SEM analysis and assisted with data analysis. D.R.M. interpreted data and contributed to the manuscript preparation. R.K.H. collected and analysed XAS data, interpreted data and contributed to the manuscript preparation. A.N.S. conceived and directed the project, designed experiments, analysed and interpreted data, and co-wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Information
Supplementary Figures 1–29, Supplementary Tables 1–6 and Supplementary References.
Supplementary Video
In situ generation and operation of the CoFePbOx water oxidation catalyst in 0.1 M H2SO4 at 23 °C.
Rights and permissions
About this article
Cite this article
Chatti, M., Gardiner, J.L., Fournier, M. et al. Intrinsically stable in situ generated electrocatalyst for long-term oxidation of acidic water at up to 80 °C. Nat Catal 2, 457–465 (2019). https://doi.org/10.1038/s41929-019-0277-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-019-0277-8
This article is cited by
-
Acid-stable manganese oxides for proton exchange membrane water electrolysis
Nature Catalysis (2024)
-
Distance effect of single atoms on stability of cobalt oxide catalysts for acidic oxygen evolution
Nature Communications (2024)
-
Surface oxidation/spin state determines oxygen evolution reaction activity of cobalt-based catalysts in acidic environment
Nature Communications (2024)
-
Elemental partitioning and corrosion resistance of Ni–Cr alloys revealed by accurate ab-initio thermodynamic and electrochemical calculations
npj Materials Degradation (2023)
-
Non-covalent ligand-oxide interaction promotes oxygen evolution
Nature Communications (2023)