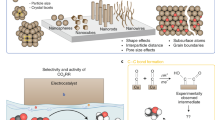
Abstract
Renewable energy conversion and storage play an important role in our global efforts to limit the drastic effects of climate change. In particular, the electrocatalytic reduction of carbon dioxide to chemicals and fuels can bring us closer towards a closed-loop anthropogenic carbon cycle. Significant breakthroughs are often the result of deeper understandings of reaction mechanisms, material structures and surface sites. To this end, operando techniques have been invaluable in combining advanced characterization of a catalyst with simultaneous measurements of its activity and selectivity under real working conditions. This Review aims to highlight significant progress in the use of operando characterization techniques that enhance our understanding of heterogeneous electrocatalytic CO2 reduction. We provide a summary of the most recent mechanistic understanding using operando optical, X-ray and electron-based techniques, along with key questions that need to be addressed. We conclude by offering some insight on emerging directions and prospects in the field.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Electron microscopy and tomography micrographs are adapted from ref. 116, Springer Nature.
Similar content being viewed by others
References
UNFCCC Report of the Conference of the Parties on its Twenty-First Session, Held in Paris from 30 November to 13 December 2015 Decision 1/CP.21 (United Nations, 2015).
van Vuuren, D. P. et al. Alternative pathways to the 1.5 °C target reduce the need for negative emission technologies. Nat. Clim. Change 8, 391–397 (2018).
Graves, C., Ebbesen, S. D., Mogensen, M. & Lackner, K. S. Sustainable hydrocarbon fuels by recycling CO2 and H2O with renewable or nuclear energy. Renew. Sustain. Energy Rev. 15, 1–23 (2011).
Seh, Z. W. et al. Combining theory and experiment in electrocatalysis: Insights into materials design. Science 355, eaad4998 (2017).
Kuhl, K. P., Cave, E. R., Abram, D. N. & Jaramillo, T. F. New insights into the electrochemical reduction of carbon dioxide on metallic copper surfaces. Energy Environ. Sci. 5, 7050–7059 (2012).
Kortlever, R., Shen, J., Schouten, K. J. P., Calle-Vallejo, F. & Koper, M. T. M. Catalysts and reaction pathways for the electrochemical reduction of carbon dioxide. J. Phys. Chem. Lett. 6, 4073–4082 (2015).
Goldsmith, B. R., Esterhuizen, J., Liu, J.-X., Bartel, C. J. & Sutton, C. Machine learning for heterogeneous catalyst design and discovery. AlChE J. 64, 2311–2323 (2018).
Hansen, H. A., Varley, J. B., Peterson, A. A. & Nørskov, J. K. Understanding trends in the electrocatalytic activity of metals and enzymes for CO2 reduction to CO. J. Phys. Chem. Lett. 4, 388–392 (2013).
Peterson, A. A., Abild-Pedersen, F., Studt, F., Rossmeisl, J. & Norskov, J. K. How copper catalyzes the electroreduction of carbon dioxide into hydrocarbon fuels. Energy Environ. Sci. 3, 1311–1315 (2010).
Kirk, C. et al. Theoretical investigations of the electrochemical reduction of CO on single metal atoms embedded in graphene. ACS Cent. Sci. 3, 1286–1293 (2017).
Wain, A. J. & O’Connell, M. A. Advances in surface-enhanced vibrational spectroscopy at electrochemical interfaces. Adv. Phy. X 2, 188–209 (2017).
Yang, C. & Wöll, C. IR spectroscopy applied to metal oxide surfaces: adsorbate vibrations and beyond. Adv. Phy. X 2, 373–408 (2017).
Wuttig, A., Yaguchi, M., Motobayashi, K., Osawa, M. & Surendranath, Y. Inhibited proton transfer enhances Au-catalyzed CO2-to-fuels selectivity. Proc. Natl Acad. Sci. USA 113, E4585–E4593 (2016).
Hori, Y., Koga, O., Yamazaki, H. & Matsuo, T. Infrared spectroscopy of adsorbed CO and intermediate species in electrochemical reduction of CO2 to hydrocarbons on a Cu electrode. Electrochim. Acta 40, 2617–2622 (1995).
Gunathunge, C. M., Ovalle, V. J., Li, Y., Janik, M. J. & Waegele, M. M. Existence of an electrochemically inert CO population on Cu electrodes in alkaline pH.ACS Catal. 8, 7507–7516 (2018). Observation of two different *CO ads geometries, and demonstration that *CO bridge is inactive for further reduction using operando IR spectroscopy. Relates to the deactivation and surface reconstruction of Cu during electrocatalysis.
Dunwell, M., Yan, Y. & Xu, B. In situ infrared spectroscopic investigations of pyridine-mediated CO2 reduction on Pt electrocatalysts. ACS Catal. 7, 5410–5419 (2017).
Sheng, W. et al. Electrochemical reduction of CO2 to synthesis gas with controlled CO/H2 ratios. Energy Environ. Sci. 10, 1180–1185 (2017).
Firet, N. J. & Smith, W. A. Probing the reaction mechanism of CO2 electroreduction over Ag films via operando infrared spectroscopy. ACS Catal. 7, 606–612 (2017).
Baruch, M. F., Pander, J. E., White, J. L. & Bocarsly, A. B. Mechanistic insights into the reduction of CO2 on tin electrodes using in situ ATR-IR spectroscopy. ACS Catal. 5, 3148–3156 (2015).
Dutta, A., Kuzume, A., Rahaman, M., Vesztergom, S. & Broekmann, P. Monitoring the chemical state of catalysts for CO2 electroreduction: an in operando study. ACS Catal. 5, 7498–7502 (2015).
Murata, A. & Hori, Y. Product selectivity affected by cationic species in electrochemical reduction of CO2 and CO at a Cu electrode. Bull. Chem. Soc. Jpn 64, 123–127 (1991).
Dunwell, M. et al. The central role of bicarbonate in the electrochemical reduction of carbon dioxide on gold. J. Am. Chem. Soc. 139, 3774–3783 (2017). Demonstrates that bicarbonate is the primary source of carbon in the CO formed at the Au electrode through equilibrium exchange with dissolved CO 2 using operando ATR-SEIRAS.
Zhu, S., Jiang, B., Cai, W.-B. & Shao, M. Direct observation on reaction intermediates and the role of bicarbonate anions in CO2 electrochemical reduction reaction on Cu surfaces. J. Am. Chem. Soc. 139, 15664–15667 (2017).
Resasco, J. et al. Promoter effects of alkali metal cations on the electrochemical reduction of carbon dioxide. J. Am. Chem. Soc. 139, 11277–11287 (2017).
Resasco, J., Lum, Y., Clark, E., Zeledon, J. Z. & Bell, A. T. Effects of anion identity and concentration on electrochemical reduction of CO2. ChemElectroChem 5, 1064–1072 (2018).
Pérez-Gallent, E., Marcandalli, G., Figueiredo, M. C., Calle-Vallejo, F. & Koper, M. T. M. Structure- and potential-dependent cation effects on CO reduction at copper single-crystal electrodes. J. Am. Chem. Soc. 139, 16412–16419 (2017).
Lum, Y., Cheng, T., Goddard, W. A. & Ager, J. W. Electrochemical CO reduction builds solvent water into oxygenate products. J. Am. Chem. Soc. 140, 9337–9340 (2018). Demonstration of the involvement of O atoms from water in producing oxygenate products from CO 2 . This calls into question previous models of oxygenate formation as O atoms in oxygenates have always been presumed to originate from CO 2 (or CO) intermediate.
Pérez-Gallent, E., Figueiredo, M. C., Calle-Vallejo, F. & Koper, M. T. M. Spectroscopic observation of a hydrogenated CO dimer intermediate during CO reduction on Cu(100) electrodes. Angew. Chem. Int. Ed. 129, 3675–3678 (2017). The initial observation of hydrogenated CO dimer (*OCCOH) on (100) cu using operando IR spectroscopy.
Schouten, K. J. P., Qin, Z., Gallent, E. P. & Koper, M. T. M. Two pathways for the formation of ethylene in CO reduction on single-crystal copper electrodes. J. Am. Chem. Soc. 134, 9864–9867 (2012).
Deng, Y. & Yeo, B. S. Characterization of electrocatalytic water splitting and CO2 reduction reactions using in situ/operando raman spectroscopy. ACS Catal. 7, 7873–7889 (2017).
Albrecht, M. G. & Creighton, J. A. Anomalously intense Raman spectra of pyridine at a silver electrode. J. Am. Chem. Soc. 99, 5215–5217 (1977).
Zeng, Z.-C. et al. Novel electrochemical Raman spectroscopy enabled by water immersion objective. Anal. Chem. 88, 9381–9385 (2016).
Yeo, B. S., Klaus, S. L., Ross, P. N., Mathies, R. A. & Bell, A. T. Identification of hydroperoxy species as reaction intermediates in the electrochemical evolution of oxygen on gold. ChemPhysChem 11, 1854–1857 (2010).
Ren, D., Ang, B. S.-H. & Yeo, B. S. Tuning the selectivity of carbon dioxide electroreduction toward ethanol on oxide-derived CuxZn catalysts. ACS Catal. 6, 8239–8247 (2016).
Ren, D. et al. Selective electrochemical reduction of carbon dioxide to ethylene and ethanol on copper(I) oxide catalysts. ACS Catal. 5, 2814–2821 (2015).
Oda, I., Ogasawara, H. & Ito, M. Carbon monoxide adsorption on copper and silver electrodes during carbon dioxide electroreduction studied by infrared reflection absorption spectroscopy and surface-enhanced Raman spectroscopy. Langmuir 12, 1094–1097 (1996).
Ichinohe, Y., Wadayama, T. & Hatta, A. Electrochemical reduction of CO2 on silver as probed by surface‐enhanced Raman scattering. J. Raman Spectrosc. 26, 335–340 (1995).
Mahoney, M. R., Howard, M. W. & Cooney, R. P. Carbon dioxide conversion to hydrocarbons at silver electrode surfaces: Raman spectroscpic evidence for surface carbon intermediates. Chem. Phys. Lett. 71, 59–63 (1980).
Smith, B. D., Irish, D. E., Kedzierzawski, P. & Augustynski, J. A surface enhanced Raman scattering study of the intermediate and poisoning species formed during the electrochemical reduction of CO2 on Copper. J. Electrochem. Soc. 144, 4288–4296 (1997).
Schmitt, K. G. & Gewirth, A. A. In situ surface-enhanced Raman spectroscopy of the electrochemical reduction of carbon dioxide on silver with 3,5-diamino-1,2,4-triazole. J. Phys. Chem. C 118, 17567–17576 (2014).
Deng, Y. et al. On the role of sulfur for the selective electrochemical reduction of CO2 to formate on CuSx catalysts. ACS Appl. Mater. Interfaces 10, 28572–28581 (2018).
Zhang, Z., Sheng, S., Wang, R. & Sun, M. Tip-enhanced Raman spectroscopy. Anal. Chem. 88, 9328–9346 (2016).
Zhang, R. et al. Chemical mapping of a single molecule by plasmon-enhanced Raman scattering. Nature 498, 82–86 (2013).
Schmid, T., Yeo, B. S., Leong, G., Stadler, J. & Zenobi, R. Performing tip‐enhanced Raman spectroscopy in liquids. J. Raman Spectrosc. 40, 1392–1399 (2009).
Mondal, B., Rana, A., Sen, P. & Dey, A. Intermediates Involved in the 2e–/2H+ Reduction of CO2 to CO by iron(0) porphyrin. J. Am. Chem. Soc. 137, 11214–11217 (2015).
Mistry, H. et al. Highly selective plasma-activated copper catalysts for carbon dioxide reduction to ethylene. Nat. Commun. 7, 12123 (2016).
Clausen, B. S. et al. A new procedure for particle size determination by EXAFS based on molecular dynamics simulations. J. Catal. 141, 368–379 (1993).
Eilert, A., Roberts, F. S., Friebel, D. & Nilsson, A. Formation of copper catalysts for CO2 reduction with high ethylene/methane product ratio investigated with in situ X-ray absorption spectroscopy. J. Phys. Chem. Lett. 7, 1466–1470 (2016).
Jiang, K. et al. Isolated Ni single atoms in graphene nanosheets for high-performance CO2 reduction. Energy Environ. Sci. 11, 893–903 (2018).
Lin, S. et al. Covalent organic frameworks comprising cobalt porphyrins for catalytic CO2 reduction in water. Science 349, 1208–1213 (2015).
Weng, Z. et al. Active sites of copper-complex catalytic materials for electrochemical carbon dioxide reduction. Nat. Commun. 9, 415 (2018).
Matsui, H. et al. Operando 3d visualization of migration and degradation of a platinum cathode catalyst in a polymer electrolyte fuel cell. Angew. Chem. Int. Ed. 56, 9371–9375 (2017).
Mistry, H. et al. Enhanced carbon dioxide electroreduction to carbon monoxide over defect-rich plasma-activated silver catalysts. Angew. Chem. Int. Ed. 56, 11394–11398 (2017).
Gao, D. et al. Plasma-activated copper nanocube catalysts for efficient carbon dioxide electroreduction to hydrocarbons and alcohols. ACS Nano 11, 4825–4831 (2017).
Lum, Y. & Ager, J. W. Stability of residual oxides in oxide-derived copper catalysts for electrochemical CO2 reduction investigated with 18O labeling. Angew. Chem. Int. Ed. 57, 551–554 (2018).
Velasco-Velez, J. J. et al. Photoelectron spectroscopy at the graphene–liquid interface reveals the electronic structure of an electrodeposited cobalt/graphene electrocatalyst. Angew. Chem. Int. Ed. 54, 14554–14558 (2015).
Nemšák, S. et al. Interfacial electrochemistry in liquids probed with photoemission electron microscopy. J. Am. Chem. Soc. 139, 18138–18141 (2017).
Siegbahn, H. & Siegbahn, K. ESCA applied to liquids. J. Electron. Spectrosc. Relat. Phenom. 2, 319–325 (1973).
Roy, K., Artiglia, L. & Bokhoven, J. A. V. Ambient pressure photoelectron spectroscopy: opportunities in catalysis from solids to liquids and introducing time resolution. ChemCatChem 10, 666–682 (2018).
Liu, Z. & Bluhm, H. in Hard X-ray Photoelectron Spectroscopy (HAXPES) (ed Joseph Woicik) 447–466 (Springer International Publishing, 2016).
Axnanda, S. et al. Using “tender” X-ray ambient pressure X-ray photoelectron spectroscopy as a direct probe of solid-liquid interface. Sci. Rep. 5, 9788 (2015).
Eilert, A. et al. Subsurface oxygen in oxide-derived copper electrocatalysts for carbon dioxide reduction. J. Phys. Chem. Lett. 8, 285–290 (2017).
Gao, S. et al. Partially oxidized atomic cobalt layers for carbon dioxide electroreduction to liquid fuel. Nature 529, 68–71 (2016).
Li, C. W., Ciston, J. & Kanan, M. W. Electroreduction of carbon monoxide to liquid fuel on oxide-derived nanocrystalline copper. Nature 508, 504–507 (2014).
De Luna, P. et al. Catalyst electro-redeposition controls morphology and oxidation state for selective carbon dioxide reduction. Nat. Catal. 1, 103–110 (2018). Experimental demonstration of higher Cu oxidation state using in situ soft XAS. Time-dependant spectra of Cu + at CO 2 RR relevant potentials were tracked for up to one hour.
Zakaria, S. N. A. et al. Insight into nature of iron sulfide surfaces during the electrochemical hydrogen evolution and CO2 reduction reactions. ACS Appl. Mater. Interfaces 10, 32078–32085 (2018).
Favaro, M. et al. Subsurface oxide plays a critical role in CO2 activation by Cu(111) surfaces to form chemisorbed CO2, the first step in reduction of CO2. Proc. Natl Acad. Sci. USA 114, 6706–6711 (2017).
Xiao, H., Goddard, W. A., Cheng, T. & Liu, Y. Cu metal embedded in oxidized matrix catalyst to promote CO2 activation and CO dimerization for electrochemical reduction of CO2. Proc. Natl Acad. Sci. USA 114, 6685–6688 (2017).
Zhang, Y.-J. & Peterson, A. A. Oxygen-induced changes to selectivity-determining steps in electrocatalytic CO2 reduction. Phys. Chem. Chem. Phys. 17, 4505–4515 (2015).
Garza, A. J., Bell, A. T. & Head-Gordon, M. Is subsurface oxygen necessary for the electrochemical reduction of CO2 on copper? J. Phys. Chem. Lett. 9, 601–606 (2018).
Fields, M., Hong, X., Nørskov, J. K. & Chan, K. Role of subsurface oxygen on Cu surfaces for CO2 electrochemical reduction. J. Phys. Chem. C 122, 16209–16215 (2018).
Liu, C. et al. Stability and effects of subsurface oxygen in oxide-derived Cu catalyst for CO2 reduction. J. Phys. Chem. C 121, 25010–25017 (2017).
Penner-Hahn, J. E. in eLS Ch. 2.13 (John Wiley and Sons, London, 2005).
Glatzel, P., Singh, J., Kvashnina, K. O. & van Bokhoven, J. A. In situ characterization of the 5d density of states of Pt nanoparticles upon adsorption of CO. J. Am. Chem. Soc. 132, 2555–2557 (2010).
van Schooneveld, M. M. et al. A multispectroscopic study of 3d orbitals in cobalt carboxylates: the high sensitivity of 2p3d resonant X-ray emission spectroscopy to the ligand field. Angew. Chem. Int. Ed. 52, 1170–1174 (2012).
Ishihara, T., Tokushima, T., Horikawa, Y., Kato, M. & Yagi, I. Development of a spectro-electrochemical cell for soft X-ray photon-in photon-out spectroscopy. Rev. Sci. Instrum. 88, 104101 (2017).
Chang, K. C. et al. in In-situ Spectroscopic Studies of Adsorption at the Electrode and Electrocatalysis (eds Sun, S.-G., Christensen, P. A. & Wieckowski, A.) 383–407 (Elsevier Science, New York, 2007).
Gul, S. et al. Simultaneous detection of electronic structure changes from two elements of a bifunctional catalyst using wavelength-dispersive X-ray emission spectroscopy and in situ electrochemistry. Phys. Chem. Chem. Phys. 17, 8901–8912 (2015). Demonstration of the feasibility of operando XES in tracking the chemical state of multi-component electrocatalysts during the oxygen evolution reaction.
Liu, Y., Barbour, A., Komanicky, V. & You, H. X-ray crystal truncation rod studies of surface oxidation and reduction on Pt(111). J. Phys. Chem. C 120, 16174–16178 (2016).
Bouldin, C. E. et al. Diffraction anomalous fine structure: XAFS with virtual photoelectrons. Jpn J. Appl. Phys. 32, 198 (1993).
Kawaguchi, T. et al. Roles of transition metals interchanging with lithium in electrode materials. Phys. Chem. Chem. Phys. 17, 14064–14070 (2015).
He, J., Johnson, N. J. J., Huang, A. & Berlinguette, C. P. Electrocatalytic alloys for CO2 reduction. ChemSusChem 11, 48–57 (2018).
Mehdi, B. L. et al. Observation and quantification of nanoscale processes in lithium batteries by operando electrochemical (S)TEM. Nano Lett. 15, 2168–2173 (2015).
Hodnik, N., Dehm, G. & Mayrhofer, K. J. J. Importance and challenges of electrochemical in situ liquid cell electron microscopy for energy conversion research. Acc. Chem. Res. 49, 2015–2022 (2016).
Mierwaldt, D. et al. Environmental TEM investigation of electrochemical stability of perovskite and ruddlesden–popper type manganite oxygen evolution catalysts. Adv. Sustain. Syst. 1, 1700109 (2017).
Mildner, S. et al. Environmental TEM study of electron beam induced electrochemistry of Pr0.64Ca0.36MnO3 catalysts for oxygen evolution. J. Phys. Chem. C 119, 5301–5310 (2015).
Yu, Y. et al. Three-dimensional tracking and visualization of hundreds of Pt−Co fuel cell nanocatalysts during electrochemical aging. Nano Lett. 12, 4417–4423 (2012).
Zhang, D. et al. An in situ TEM study of the surface oxidation of palladium nanocrystals assisted by electron irradiation. Nanoscale 9, 6327–6333 (2017).
Mancera, L. A., Behm, R. J. & Groβ, A. Structure and local reactivity of PdAg/Pd(111) surface alloys. Phys. Chem. Chem. Phys. 15, 1497–1508 (2013).
Ustarroz, J. et al. Electrodeposition of highly porous Pt nanoparticles studied by quantitative 3D electron tomography: influence of growth mechanisms and potential cycling on the active surface area. ACS Appl. Mater. Interfaces 9, 16168–16177 (2017).
Binnig, G., Rohrer, H., Gerber, C. & Weibel, E. Surface studies by scanning tunneling microscopy. Phys. Rev. Lett. 49, 57–61 (1982).
Itaya, K. & Tomita, E. Scanning tunneling microscope for electrochemistry - a new concept for the in situ scanning tunneling microscope in electrolyte solutions. Surf. Sci. 201, L507–L512 (1988).
Kim, Y.-G., Baricuatro, J. H., Javier, A., Gregoire, J. M. & Soriaga, M. P. The evolution of the polycrystalline copper surface, first to Cu(111) and then to Cu(100), at a fixed CO2RR potential: a study by operando EC-STM. Langmuir 30, 15053–15056 (2014).
Binnig, G., Quate, C. F. & Gerber, C. Atomic force microscope. Phys. Rev. Lett. 56, 930–933 (1986).
Grosse, P. et al. Dynamic changes in the structure, chemical state and catalytic selectivity of Cu nanocubes during CO2 electroreduction. Angew. Chem. Int. Ed. 57, 6192–6197 (2018). Dynamic morphological and chemical monitoring of Cu cubes during CO 2 RR using operando EC-AFM and EXAFS. Roughening, loss of (100) facets and loss of Cu atoms from edge and corner sites, and the reduction of CuO x species were observed, concommitant with reduced CO 2 RR activity.
Nellist, M. R. et al. Potential-sensing electrochemical atomic force microscopy for in operando analysis of water-splitting catalysts and interfaces. Nat. Energy 3, 46–52 (2018).
Jung, C., Sánchez-Sánchez, C. M., Lin, C.-L., Rodríguez-López, J. & Bard, A. J. Electrocatalytic activity of Pd−Co bimetallic mixtures for formic acid oxidation studied by scanning electrochemical microscopy. Anal. Chem. 81, 7003–7008 (2009).
Bard, A. J., Fan, F. R. F., Kwak, J. & Lev, O. Scanning electrochemical microscopy. Introduction and principles.Anal. Chem. 61, 132–138 (1989).
Lucas, M. & Boily, J.-F. Mapping electrochemical heterogeneity at iron oxide surfaces: a local electrochemical impedance study. Langmuir 31, 13618–13624 (2015).
Sreekanth, N. & Phani, K. L. Selective reduction of CO2 to formate through bicarbonate reduction on metal electrodes: new insights gained from SG/TC mode of SECM. Chem. Commun. 50, 11143–11146 (2014).
Mariano, R. G., McKelvey, K., White, H. S. & Kanan, M. W. Selective increase in CO2 electroreduction activity at grain-boundary surface terminations. Science 358, 1187–1192 (2017).The initial experimental demonstration of heightened CO 2 RR activity at the grain boundary using SECM, as corroborated by electron backscattering diffraction (EBSD) grain mapping.
Kai, T., Zhou, M., Duan, Z., Henkelman, G. A. & Bard, A. J. Detection of CO2 •– in the electrochemical reduction of carbon dioxide in N,N-dimethylformamide by scanning electrochemical microscopy. J. Am. Chem. Soc. 139, 18552–18557 (2017). Detection of CO 2 radical on Au substrate using SECM based on Hg/Pt ultra micro electrode. This is probably the initial experimental observation of CO 2 radical, which is extremely challenging due to the short lifetime of CO 2 radical and its reactivity with proton donors.
Lertanantawong, B. et al. Study of the underlying electrochemistry of polycrystalline gold electrodes in aqueous solution and electrocatalysis by large amplitude fourier transformed alternating current voltammetry. Langmuir 24, 2856–2868 (2008).
Zhang, J., Guo, S.-X., Bond, A. M. & Marken, F. Large-amplitude Fourier transformed high-harmonic alternating current cyclic voltammetry: kinetic discrimination of interfering faradaic processes at glassy carbon and at boron-doped diamond electrodes. Anal. Chem. 76, 3619–3629 (2004).
Guo, S. X., MacFarlane, D. R. & Zhang, J. Bioinspired electrocatalytic CO2 reduction by bovine serum albumin-capped silver nanoclusters mediated by [α‐SiW12O40]4−. ChemSusChem 9, 80–87 (2016).
Zhang, Y. et al. Direct detection of electron transfer reactions underpinning the tin-catalyzed electrochemical reduction of CO2 using Fourier-transformed ac voltammetry. ACS Catal. 7, 4846–4853 (2017).
Liu, M. et al. Enhanced electrocatalytic CO2 reduction via field-induced reagent concentration. Nature 537, 382–386 (2016). This work exploits the presence of charged reactant and intermediates, and demonstrates that CO 2 RR activity can be enhanced by altering the local concentration of reactant due to the effect of electric field concentration on sharp tipped surfaces.
Huang, S. et al. Molecular selectivity of graphene-enhanced raman scattering. Nano Lett. 15, 2892–2901 (2015).
Rehn, S. M. & Jones, M. R. New strategies for probing energy systems with in situ liquid-phase transmission electron microscopy. ACS Energy Lett. 3, 1269–1278 (2018).
Schneider, N. M. et al. Electron–water interactions and implications for liquid cell electron microscopy. J. Phys. Chem. C 118, 22373–22382 (2014).
Wu, J. et al. A metal-free electrocatalyst for carbon dioxide reduction to multi-carbon hydrocarbons and oxygenates. Nat. Commun. 7, 13869 (2016).
Ma, X., Li, Z., Achenie, L. E. K. & Xin, H. Machine-learning-augmented chemisorption model for CO2 electroreduction catalyst screening. J. Phys. Chem. Lett. 6, 3528–3533 (2015).
Ulissi, Z. W. et al. Machine-learning methods enable exhaustive searches for active bimetallic facets and reveal active site motifs for CO2 reduction. ACS Catal. 7, 6600–6608 (2017).
de Groot, F. High-resolution X-ray emission and x-ray absorption spectroscopy. Chem. Rev. 101, 1779–1808 (2001).
Artrith, N. & Kolpak, A. M. Understanding the composition and activity of electrocatalytic nanoalloys in aqueous solvents: a combination of dft and accurate neural network potentials. Nano Lett. 14, 2670–2676 (2014).
Handoko, A. D. et al. Elucidation of thermally induced internal porosity in zinc oxide nanorods. Nano Res. 11, 2412–2423 (2018).
Zaera, F. Infrared absorption spectroscopy of adsorbed CO: new applications in nanocatalysis for an old approach. ChemCatChem 4, 1525–1533 (2012).
Fahrenkrug, E., Alsem, D. H., Salmon, N. & Maldonado, S. Electrochemical measurements in in situ TEM experiments. J. Electrochem. Soc. 164, H358–H364 (2017).
Acknowledgements
This work was supported by the Institute of Materials Research and Engineering, A*STAR (IMRE/17-1R1211) and the National University of Singapore (R-143-000-A08-114).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Information
Supplementary Note 1, Supplementary Tables 1–3 and Supplementary References
Rights and permissions
About this article
Cite this article
Handoko, A.D., Wei, F., Jenndy et al. Understanding heterogeneous electrocatalytic carbon dioxide reduction through operando techniques. Nat Catal 1, 922–934 (2018). https://doi.org/10.1038/s41929-018-0182-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-018-0182-6
This article is cited by
-
Impact of palladium/palladium hydride conversion on electrochemical CO2 reduction via in-situ transmission electron microscopy and diffraction
Nature Communications (2024)
-
Advances in in situ/operando techniques for catalysis research: enhancing insights and discoveries
Surface Science and Technology (2024)
-
A US perspective on closing the carbon cycle to defossilize difficult-to-electrify segments of our economy
Nature Reviews Chemistry (2024)
-
Highly selective electrocatalytic alkynol semi-hydrogenation for continuous production of alkenols
Nature Communications (2023)
-
Dynamic imaging of interfacial electrochemistry on single Ag nanowires by azimuth-modulated plasmonic scattering interferometry
Nature Communications (2023)