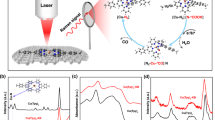
Abstract
The electrocatalytic reduction of CO2 offers a sustainable route to the many carbon fuels and feedstocks that society relies on. [fac-Mn(bpy)(CO)3Br] (bpy, 2,2-bipyridine) is one of the most promising and intensely studied CO2 reduction electrocatalysts. However, the catalytic mechanism remains experimentally unproven and many key intermediates of the prototypical catalyst have not been observed. Here we report the use of vibrational sum-frequency generation spectroscopy to study the catalytic intermediates during CO2 reduction in situ at the electrode surface. We explore the complex applied-potential and acid-dependent mechanistic pathways and provide evidence of the theoretically derived mechanisms. Demonstrating the ability to detect the key species that are only transiently present at the electrode surface is important as the need for an improved mechanistic understanding is a common theme throughout the field of molecular electrocatalysis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
Raw data for all figures within the paper are freely available from the University of Liverpool Research Data Catalogue at https://doi.org/10.17638/datacat.liverpool.ac.uk/533.
References
Dey, S. et al. Molecular electrocatalysts for the oxygen reduction reaction. Nat. Rev. Chem. 1, 0098 (2017).
DuBois, D. L. Development of molecular electrocatalysts for energy storage. Inorg. Chem. 53, 3935–3960 (2014).
Costentin, C., Robert, M. & Savéant, J.-M. Catalysis of the electrochemical reduction of carbon dioxide. Chem. Soc. Rev. 42, 2423–2436 (2013).
Lee, K. J., Elgrishi, N., Kandemir, B. & Dempsey, J. L. Electrochemical and spectroscopic methods for evaluating molecular electrocatalysts. Nat. Rev. Chem. 1, 0039 (2017).
Shen, Y. R. Surface properties probed by second-harmonic and sum-frequency generation. Nature 337, 519–525 (1989).
Shen, Y. R. Basic theory of surface sum-frequency generation. J. Phys. Chem. C 116, 15505–15509 (2012).
Lambert, A. G., Davies, P. B. & Neivandt, D. J. Implementing the theory of sum frequency generation vibrational spectroscopy: a tutorial review. Appl. Spectrosc. Rev. 40, 103–145 (2005).
Rey, N. G. & Dlott, D. D. Studies of electrochemical interfaces by broadband sum frequency generation. J. Electroanal. Chem. 800, 114–125 (2017).
Baldelli, S. Probing electric fields at the ionic liquid−electrode interface using sum frequency generation spectroscopy and electrochemistry. J. Phys. Chem. B 109, 13049–13051 (2005).
Liu, W.-T. & Shen, Y. R. In situ sum-frequency vibrational spectroscopy of electrochemical interfaces with surface plasmon resonance. Proc. Natl Acad. Sci. USA 111, 1293–1297 (2014).
Tadjeddine, A. et al. Sum and difference frequency generation at the electrochemical interface. Phys. Status Solidi 175, 89–107 (1999).
Anfuso, C. L. et al. Orientation of a series of CO2 reduction catalysts on single crystal TiO2 probed by phase-sensitive vibrational sum frequency generation spectroscopy (PS-VSFG). J. Phys. Chem. C 116, 24107–24114 (2012).
Ge, A. et al. Surface-induced anisotropic binding of a rhenium CO2-reduction catalyst on rutile TiO2(110) surfaces. J. Phys. Chem. C 120, 20970–20977 (2016).
Anfuso, C. L., Ricks, A. M., Rodríguez-Córdoba, W. & Lian, T. Ultrafast vibrational relaxation dynamics of a rhenium bipyridyl CO2–reduction catalyst at a Au electrode surface probed by time-resolved vibrational sum frequency generation spectroscopy. J. Phys. Chem. C 116, 26377–26384 (2012).
Wang, J. et al. Short-range catalyst–surface interactions revealed by heterodyne two-dimensional sum frequency generation spectroscopy. J. Phys. Chem. Lett. 6, 4204–4209 (2015).
Neri, G., Donaldson, P. M. & Cowan, A. J. The role of electrode–catalyst interactions in enabling efficient CO2 reduction with Mo(bpy)(CO)4 as revealed by vibrational sum-frequency generation spectroscopy. J. Am. Chem. Soc. 139, 13791–13797 (2017).
Tory, J., Setterfield-Price, B., Dryfe, R. A. W. & Hartl, F. [M(CO)4 (2,2′-bipyridine)] (M = Cr, Mo, W) complexes as efficient catalysts for electrochemical reduction of CO2 at a gold electrode. ChemElectroChem 2, 213–217 (2015).
Bourrez, M., Molton, F., Chardon-Noblat, S. & Deronzier, A. [Mn(bipyridyl)(CO)3Br]: an abundant metal carbonyl complex as efficient electrocatalyst for CO2 reduction. Angew. Chem. Int. Ed. 50, 9903–9906 (2011).
Stanbury, M., Compain, J.-D. & Chardon-Noblat, S. Electro and photoreduction of CO2 driven by manganese–carbonyl molecular catalysts. Coord. Chem. Rev. 361, 120–137 (2018).
Sampson, M. D. & Kubiak, C. P. Manganese electrocatalysts with bulky bipyridine ligands: utilizing Lewis acids to promote carbon dioxide reduction at low overpotentials. J. Am. Chem. Soc. 138, 1386–1393 (2016).
Ngo, K. T. et al. Turning on the protonation-first pathway for electrocatalytic CO2 reduction by manganese bipyridyl tricarbonyl complexes. J. Am. Chem. Soc. 139, 2604–2618 (2017).
Walsh, J. J., Neri, G., Smith, C. L. & Cowan, A. J. Electrocatalytic CO2 reduction with a membrane supported manganese catalyst in aqueous solution. Chem. Commun. 50, 12698–12701 (2014).
Walsh, J. J. et al. Improving the efficiency of electrochemical CO2 reduction using immobilized manganese complexes. Faraday Discuss. 183, 147–160 (2015).
Reuillard, B. et al. Tuning product selectivity for aqueous CO2 reduction with a Mn(bipyridine)–pyrene catalyst immobilized on a carbon nanotube electrode. J. Am. Chem. Soc. 139, 14425–14435 (2017).
Hartl, F., Rossenaar, B. D., Stor, G. J. & Stufkens, D. Role of an electron-transfer chain reaction in theunusual photochemical formation of five-coordinated anions [Mn(CO)3 (α-diimine)] from fac-[Mn(X)(CO)3 (α-diimine)] (X = halide) at low temperatures. Recl. Trav. Chim. Pays-Bas 114, 565–570 (1995).
Bourrez, M. et al. Pulsed-EPR evidence of a manganese(ii) hydroxycarbonyl intermediate in the electrocatalytic reduction of carbon dioxide by a manganese bipyridyl derivative. Angew. Chem. Int. Ed. 53, 240–243 (2014).
Rossenaar, B. D. et al. Electrochemical and IR/UV−vis spectroelectrochemical studies of fac-[Mn(X)(CO)3(iPr-DAB)]n (n = 0, X = Br, Me, Bz; n = +1, X = THF, MeCN, nPrCN, P(OMe)3; iPr-DAB = 1,4-diisopropyl-1,4-diaza-1,3-butadiene) at variable temperatures: relation between electrochemical and photochemical generation of [Mn(CO)3(α-diimine)]–. Organometallics 16, 4675–4685 (1997).
Franco, F. et al. Local proton source in electrocatalytic CO2 reduction with [Mn(bpy-R)(CO)3Br] complexes. Chem. Eur. J. 23, 4782–4793 (2017).
Hartl, F., Rosa, P., Ricard, L., Le Floch, P. & Záliš, S. Electronic transitions and bonding properties in a series of five-coordinate ‘16-electron’ complexes [Mn(CO)3(L2)]– (L2 = chelating redox-active π-donor ligand). Coord. Chem. Rev. 251, 557–576 (2007).
Hawecker, J., Lehn, J.-M. & Ziessel, R. Electrocatalytic reduction of carbon dioxide mediated by Re(bipy)(CO)3Cl (bipy = 2,2′-bipyridine). J. Chem. Soc. Chem. Commun. 0, 328–330 (1984).
Riplinger, C., Sampson, M. D., Ritzmann, A. M., Kubiak, C. P. & Carter, E. A. Mechanistic contrasts between manganese and rhenium bipyridine electrocatalysts for the reduction of carbon dioxide. J. Am. Chem. Soc. 136, 16285–16298 (2014).
Riplinger, C. & Carter, E. A. Influence of weak Brønsted acids on electrocatalytic CO2 reduction by manganese and rhenium bipyridine catalysts. ACS Catal. 5, 900–908 (2015).
Lam, Y. C., Nielsen, R. J., Gray, H. B. & Goddard, W. A. A Mn bipyrimidine catalyst predicted to reduce CO2 at lower overpotential. ACS Catal. 5, 2521–2528 (2015).
Grills, D. C. et al. Mechanism of the formation of a Mn-based CO2 reduction catalyst revealed by pulse radiolysis with time-resolved infrared detection. J. Am. Chem. Soc. 136, 5563–5566 (2014).
Lagutchev, A., Lozano, A., Mukherjee, P., Hambir, S. A. & Dlott, D. D. Compact broadband vibrational sum-frequency generation spectrometer with nonresonant suppression. Spectrochim. Acta A 75, 1289–1296 (2010).
Bishop, D. M. The vibrational Stark effect. J. Chem. Phys. 98, 3179–3184 (1993).
Ohno, P. E., Wang, H. & Geiger, F. M. Second-order spectral lineshapes from charged interfaces. Nat. Commun. 8, 1032 (2017).
Sampson, M. D. et al. Manganese catalysts with bulky bipyridine ligands for the electrocatalytic reduction of carbon dioxide: eliminating dimerization and altering catalysis. J. Am. Chem. Soc. 136, 5460–5471 (2014).
Borovkov, V. Y., Kolesnikov, S. P., Kovalchuk, V. I. & D’Itri, J. L. Probing adsorption sites of silica-supported platinum with 13C16O + 12C16O and 13C18O + 12C16O mixtures: a comparative Fourier transform infrared investigation. J. Phys. Chem. B 109, 19772–19778 (2005).
Scheiring, T., Kaim, W. & Fiedler, J. Geometrical and electronic structures of the acetyl complex Re(bpy)(CO)3(COCH3) and of [M(bpy)(CO)4](OTf), M = Mn, Re. J. Organomet. Chem. 598, 136–141 (2000).
Franco, F., Pinto, M. F., Royo, B. & Lloret-Fillol, J. A highly active N-heterocyclic carbene Mn(i) complex for selective electrocatalytic CO2 reduction to CO. Angew. Chem. Int. Ed. 57, 4603–4606 (2018).
Smieja, J. M. & Kubiak, C. P. Re(bipy-tBu)(CO)3Cl-improved catalytic activity for reduction of carbon dioxide: IR-spectroelectrochemical and mechanistic studies. Inorg. Chem. 49, 9283–9289 (2010).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Perdew, J., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Grimme, S. Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J. Comput. Chem. 27, 1787–1799 (2006).
Amft, M., Lebègue, S., Eriksson, O. & Skorodumova, N. V. Adsorption of Cu, Ag, and Au atoms on graphene including van der Waals interactions. J. Phys. Condens. Matter 23, 395001 (2011).
Wu, Y. et al. Electrode–ligand interactions dramatically enhance CO2 conversion to CO by the [Ni(cyclam)](PF6)2 catalyst. ACS Catal. 7, 5282–5288 (2017).
Keith, J. A., Grice, K. A., Kubiak, C. P. & Carter, E. A. Elucidation of the selectivity of proton-dependent electrocatalytic CO2 reduction by fac-Re(bpy)(CO)3Cl. J. Am. Chem. Soc. 135, 15823–15829 (2013).
Acknowledgements
We are grateful to C. Smith (University of Liverpool) for the synthesis of 1. This work was carried out at the Ultra facility of the UK Central Laser Facility during experiments 15130005, 16130016 and 16230052. A.J.C. and G.N. acknowledge support from EPSRC (EP/K006851/1, EP/P034497/1 and EP/N010531/). G.T. acknowledges support from EPSRC (EP/I004483/1, EP/K013610/1, EP/P022189/1 and EP/P022189/1). This work made use of the ARCHER (via the UKCP Consortium, EPSRC UK EP/K013610/1 and EP/P022189/1) and UK Materials and Molecular Modelling Hub (EPSRC UK EP/P020194/1) High-Performance Computing facilities.
Author information
Authors and Affiliations
Contributions
G.N., A.J.C., P.M.D. and J.J.W. carried out the experimental work. G.T. carried out the computational work. A.J.C. and G.T. wrote the manuscript. All the authors contributed to the editing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Supplementary Figures 1–15, Supplementary Tables 1–22, Supplementary References
Rights and permissions
About this article
Cite this article
Neri, G., Walsh, J.J., Teobaldi, G. et al. Detection of catalytic intermediates at an electrode surface during carbon dioxide reduction by an earth-abundant catalyst. Nat Catal 1, 952–959 (2018). https://doi.org/10.1038/s41929-018-0169-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-018-0169-3