Abstract
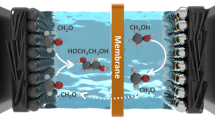
Electrosynthesis provides a method of driving organic reaction chemistry under ambient conditions with electricity. Pairing two reactions together enables the synthesis of two valuable chemicals with no waste product. Here we report the paired electrolysis of 4-methoxybenzyl alcohol to 4-methoxybenzaldehyde with the concomitant formation of 1-hexene from 1-hexyne in an electrochemical cell. These reaction chambers are separated by a dense palladium membrane that reduces protons formed at the anode to hydrogen atoms that can permeate through the palladium foil to hydrogenate 1-hexyne. The palladium membrane enables two reactions to be performed in distinct reaction conditions: hydrogenation in organic solvents and electrochemical oxidation in aqueous electrolyte. The starting materials in both chambers react quantitatively over 5 hours of electrolysis, and selectivities ≥95% can be achieved for 4-methoxybenzaldehyde and 1-hexene with control of reaction conditions. Exquisite control of the reaction kinetics and selectivities of each of the individual reactions is demonstrated.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Yan, M., Kawamata, Y. & Baran, P. S. Synthetic organic electrochemical methods since 2000: on the verge of a renaissance. Chem. Rev. 117, 13230–13319 (2017).
Frontana-Uribe, B. A., Daniel Little, R., Ibanez, J. G., Palma, A. & Vasquez-Medrano, R. Organic electrosynthesis: a promising green methodology in organic chemistry. Green. Chem. 12, 2099–2119 (2010).
Yoshida, J.-I., Kataoka, K., Horcajada, R. & Nagaki, A. Modern strategies in electroorganic synthesis. Chem. Rev. 108, 2265–2299 (2008).
Li, T., Cao, Y., He, J. & Berlinguette, C. P. Electrolytic CO2 reduction in tandem with oxidative organic chemistry. ACS Cent. Sci. 3, 778–783 (2017).
Llorente, M. J., Nguyen, B. H., Kubiak, C. P. & Moeller, K. D. Paired electrolysis in the simultaneous production of synthetic intermediates and substrates. J. Am. Chem. Soc. 138, 15110–15113 (2016).
Cha, H. G. & Choi, K.-S. Combined biomass valorization and hydrogen production in a photoelectrochemical cell. Nat. Chem. 7, 328–333 (2015).
You, B., Liu, X., Jiang, N. & Sun, Y. A general strategy for decoupled hydrogen production from water splitting by integrating oxidative biomass valorization. J. Am. Chem. Soc. 138, 13639–13646 (2016).
Paddon, C. A. et al. Towards paired and coupled electrode reactions for clean organic microreactor electrosyntheses. J. Appl. Electrochem. 36, 617 (2006).
Amemiya, F., Horii, D., Fuchigami, T. & Atobe, M. Self-supported paired electrosynthesis using a microflow reactor without intentionally added electrolyte. J. Electrochem. Soc. 155, E162–E165 (2008).
Zhang, L., Zha, Z., Wang, Z. & Fu, S. Aqueous electrosynthesis of carbonyl compounds and the corresponding homoallylic alcohols in a divided cell. Tetrahedron Lett. 51, 1426–1429 (2010).
Wicke, E., Brodowsky, H. & Züchner, H. in Hydrogen in Metals II (eds Alefeld, G. & Völkl, J.) 73–155 (Springer, Berlin, Heidelberg, 1978).
Dittmeyer, R., Höllein, V. & Daub, K. Membrane reactors for hydrogenation and dehydrogenation processes based on supported palladium. J. Mol. Catal. A Chem. 173, 135–184 (2001).
Dittmeyer, R., Svajda, K. & Reif, M. A review of catalytic membrane layers for gas/liquid reactions. Top. Catal. 29, 3–27 (2004).
Hatlevik, Ø. et al. Palladium and palladium alloy membranes for hydrogen separation and production: history, fabrication strategies, and current performance. Sep. Purif. Technol. 73, 59–64 (2010).
Shirasaki, Y. et al. Development of membrane reformer system for highly efficient hydrogen production from natural gas. Int. J. Hydrog. Energy 34, 4482–4487 (2009).
Itoh, N. & C. Xu, W. Selective hydrogenation of phenol to cyclohexanone using palladium-based membranes as catalysts. Appl. Catal. A 107, 83–100 (1993).
Nagamoto, H. & Inoue, H. The hydrogenation of 1,3-butadiene over a palladium membrane. Bull. Chem. Soc. Jpn 59, 3935–3939 (1986).
Shirai, M. & Arai, M. Hydrogenation of furan with hydrogen atoms permeating through a palladium membrane. Langmuir 15, 1577–1578 (1999).
Inoue, H., Abe, T. & Iwakura, C. Successive hydrogenation of styrene at a palladium sheet electrode combined with electrochemical supply of hydrogen. Chem. Commun. 0, 55–56 (1996).
Iwakura, C., Yoshida, Y. & Inoue, H. A new hydrogenation system of 4-methylstyrene using a palladinized palladium sheet electrode. J. Electroanal. Chem. 431, 43–45 (1997).
Gryaznov, V. M. Platinum metals as components of catalyst-membrane systems. Platin. Met. Rev. 36, 70–79 (1992).
Gryaznov, V. M., Ermilova, M. M. & Orekhova, N. V. Membrane–catalyst systems for selectivity improvement in dehydrogenation and hydrogenation reactions. Catal. Today 67, 185–188 (2001).
Farris, T. S. & Armor, J. N. Liquid-phase catalytic hydrogenation using palladium alloy membranes. Appl. Catal. A 96, 25–32 (1993).
Gryaznov, V. M. Hydrogen permeable palladium membrane catalysts. Platin. Met. Rev. 30, 68–72 (1986).
Niwa, S.-i. et al. A one-step conversion of benzene to phenol with a palladium membrane. Science 295, 105–107 (2002).
Iwakura, C., Ito, T. & Inoue, H. Construction of a new dehydrogenation system using a two-compartment cell separated by a palladized Pd sheet electrode. J. Electroanal. Chem. 463, 116–118 (1999).
Collins, J. P. et al. Catalytic dehydrogenation of propane in hydrogen permselective membrane reactors. Ind. Eng. Chem. Res. 35, 4398–4405 (1996).
Quicker, P., Höllein, V. & Dittmeyer, R. Catalytic dehydrogenation of hydrocarbons in palladium composite membrane reactors. Catal. Today 56, 21–34 (2000).
Criscuoli, A., Basile, A. & Drioli, E. An analysis of the performance of membrane reactors for the water–gas shift reaction using gas feed mixtures. Catal. Today 56, 53–64 (2000).
Uemiya, S., Sato, N., Ando, H. & Kikuchi, E. The water gas shift reaction assisted by a palladium membrane reactor. Ind. Eng. Chem. Res. 30, 585–589 (1991).
Gryaznov, V. Membrane catalysis. Catal. Today 51, 391–395 (1999).
Teschner, D. et al. The roles of subsurface carbon and hydrogen in palladium-catalyzed alkyne hydrogenation. Science 320, 86–89 (2008).
Aleksandrov, H. A., Kozlov, S. M., Schauermann, S., Vayssilov, G. N. & Neyman, K. M. How absorbed hydrogen affects the catalytic activity of transition metals. Angew. Chem. Int. Ed. 53, 13371–13375 (2014).
Zhao, S. et al. Multimodal study of the speciations and activities of supported Pd catalysts during the hydrogenation of ethylene. J. Phys. Chem. C 121, 18962–18972 (2017).
Armbrüster, M. et al. How to control the selectivity of palladium-based catalysts in hydrogenation reactions: the role of subsurface chemistry. ChemCatChem 4, 1048–1063 (2012).
Ceyer, S. T. The unique chemistry of hydrogen beneath the surface: catalytic hydrogenation of hydrocarbons. Acc. Chem. Res. 34, 737–744 (2001).
Teschner, D. et al. Understanding palladium hydrogenation catalysts: when the nature of the reactive molecule controls the nature of the catalyst active phase. Angew. Chem. Int. Ed. 47, 9274–9278 (2008).
Gao, D. et al. Switchable CO2 electroreduction via engineering active phases of Pd nanoparticles. Nano Res. 10, 2181–2191 (2017).
Sheng, W. et al. Electrochemical reduction of CO2 to synthesis gas with controlled CO/H2 ratios. Energy Environ. Sci. 10, 1180–1185 (2017).
Min, X. & Kanan, M. W. Pd-catalyzed electrohydrogenation of carbon dioxide to formate: high mass activity at low overpotential and identification of the deactivation pathway. J. Am. Chem. Soc. 137, 4701–4708 (2015).
Zhao, Z. et al. Synthesis of stable shape-controlled catalytically active β-palladium hydride. J. Am. Chem. Soc. 137, 15672–15675 (2015).
Güther, W. & Vielstich, W. Investigation on the electrocatalytic dehydrogenation of cho-compounds in acidic media via a palladium membrane. Electrochim. Acta 27, 811–816 (1982).
Bagotzky, V. S. & Vassiliev, Y. B. Absorption of organic substances on platinum electrodes. Electrochim. Acta 11, 1439–1461 (1966).
Ward, T. L. & Dao, T. Model of hydrogen permeation behavior in palladium membranes. J. Memb. Sci. 153, 211–231 (1999).
Abys, J. A. in Modern Electroplating (eds Schlesinger, M. & Paunovic, M.) 327–368 (John Wiley & Sons, Hoboken, 2011).
Badalyan, A. & Stahl, S. S. Cooperative electrocatalytic alcohol oxidation with electron-proton-transfer mediators. Nature 535, 406–410 (2016).
Acknowledgements
The authors are grateful to the Canadian Natural Science and Engineering Council (RGPIN 337345-13), Canadian Foundation for Innovation (229288), Canadian Institute for Advanced Research (BSE-BERL-162173), Canada Research Chairs and Google for financial support. Thank you to Y. Ling in the University of British Columbia Mass Spectrometry Centre for assistance with the gas chromatography–mass spectrometer instrument and to G. Owen in the Centre for High-Throughput Phenogenomics for assistance with scanning electron microscope imaging.
Author information
Authors and Affiliations
Contributions
R.S.S., B.P.M. and C.P.B. devised the concept. R.S.S. and R.S.D. performed preliminary proof-of-concept experiments. R.S.S. performed paired electrolysis experiments. V.A.C. helped with characterization. R.S.S. and C.P.B. wrote the manuscript. C.P.B. supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–10
Rights and permissions
About this article
Cite this article
Sherbo, R.S., Delima, R.S., Chiykowski, V.A. et al. Complete electron economy by pairing electrolysis with hydrogenation. Nat Catal 1, 501–507 (2018). https://doi.org/10.1038/s41929-018-0083-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-018-0083-8
This article is cited by
-
Electrochemical hydrogenation and oxidation of organic species involving water
Nature Reviews Chemistry (2024)
-
Indirect H2O2 synthesis without H2
Nature Communications (2024)
-
Electrochemistry-assisted selective butadiene hydrogenation with water
Nature Communications (2023)
-
Doubling the efficiency of electrosynthesis
Nature Catalysis (2023)
-
Electrocatalytic dual hydrogenation of organic substrates with a Faradaic efficiency approaching 200%
Nature Catalysis (2023)