Abstract
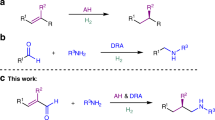
Dynamic kinetic resolutions (DKRs) allow for the conversion of both enantiomers of starting material into a single enantiomer of product, hence avoiding the 50% yield limit observed in traditional kinetic resolutions. Transition-metal-catalysed variants have become an important and useful method in asymmetric synthesis. Here we report an asymmetric hydrogenation of allylic alcohols using an Ir–N,P-ligand complex via DKR. In contrast to the many DKRs involving carbonyl reduction, this methodology allows for DKR during alkene reduction. Mechanistic studies support the hypothesis that racemization of the substrate is achieved by cleavage and reforming of the oxygen–carbon bond. Under the cooperative dynamic kinetic asymmetric hydrogenation, a broad range of chiral alcohols containing two stereogenic centres were produced with excellent diastereoselectivities (up to 95:5) and enantioselectivities (up to 99%).
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Peschiulli, A., Procuranti, B., O’Connor, C. J. & Connon, S. J. Synergistic organocatalysis in the kinetic resolution of secondary thiols with concomitant desymmetrization of an anhydride. Nat. Chem. 2, 380–384 (2010).
Binanzer, M., Hsieh, S.-Y. & Bode, J. W. Catalytic kinetic resolution of cyclic secondary amines. J. Am. Chem. Soc. 133, 19698–19701 (2011).
Taylor, J. E., Bull, S. D. & Williams, J. M. J. Amidines, isothioureas, and guanidines as nucleophilic catalysts. Chem. Soc. Rev. 41, 2109–2121 (2012).
Noyori, R. et al. Stereoselective hydrogenation via dynamic kinetic resolution. J. Am. Chem. Soc. 111, 9134–9135 (1989).
Huerta, F. F., Minidis, A. B. E. & Bäckvall, J.-E. Racemisation in asymmetric synthesis. Dynamic kinetic resolution and related processes in enzyme and metal catalysis. Chem. Soc. Rev. 30, 321–331 (2001).
Ebbers, E. J., Ariaans, G. J. A., Houbiers, J. P. M., Bruggink, A. & Zwanenburg, B. Controlled racemization of optically active organic compounds: prospects for asymmetric transformation. Tetrahedron 53, 9417–9476 (1997).
Hussain, I. & Bäckvall, J.-E. in Enzyme Catalysis in Organic Synthesis (eds Drauz, K., Gröger, H. and May, O.)1777–1806 (Wiley-VCH, Weinheim, 2012)..
Rachwalski, M., Vermue, N. & Rutjes, F. P. J. T. Recent advances in enzymatic and chemical deracemisation of racemic compounds. Chem. Soc. Rev. 42, 9268–9282 (2013).
Pellissier, H. Recent developments in organocatalytic dynamic kinetic resolution. Tetrahedron 72, 3133–3150 (2016).
Ikariya, T., Murata, K. & Noyori, R. Bifunctional transition metal-based molecular catalysts for asymmetric syntheses. Org. Biomol. Chem. 4, 393–406 (2006).
Steward, K. M., Gentry, E. C. & Johnson, J. S. Dynamic kinetic resolution of α-keto esters via asymmetric transfer hydrogenation. J. Am. Chem. Soc. 134, 7329–7332 (2012).
Ahn, Y., Ko, S.-B., Kim, M.-J. & Park, J. Racemization catalysts for the dynamic kinetic resolution of alcohols and amines. Coord. Chem. Rev. 252, 647–658 (2008).
Stürmer, R. Enzymes and transition metal complexes in tandem—a new concept for dynamic kinetic resolution. Angew. Chem. Int. Ed. Engl. 36, 1173–1174 (1997).
Shvo, Y., Czarkie, D., Rahamim, Y. & Chodosh, D. F. A new group of ruthenium complexes: structure and catalysis. J. Am. Chem. Soc. 108, 7400–7402 (1986).
Dinh, P. M., Howarth, J. A., Hudnott, A. R., Williams, J. M. J. & Harris, W. Catalytic racemization of alcohols: applications to enzymic resolution reactions. Tetrahedron Lett. 37, 7623–7626 (1996).
Larsson, A. L. E., Persson, B. A. & Bäckvall, J.-E. Enzymic resolution of alcohols coupled with ruthenium-catalyzed racemization of the substrate alcohol. Angew. Chem. Int. Ed. Engl. 36, 1211–1212 (1997).
Ohkuma, T., Ishii, D., Takeno, H. & Noyori, R. Asymmetric hydrogenation of amino ketones using chiral RuCl2(diphosphine)(1,2-diamine) complexes. J. Am. Chem. Soc. 122, 6510–6511 (2000).
Xie, J.-H., Zhou, Z.-T., Kong, W.-L. & Zhou, Q.-L. Ru-catalyzed asymmetric hydrogenation of racemic aldehydes via dynamic kinetic resolution: efficient synthesis of optically active primary alcohols. J. Am. Chem. Soc. 129, 1868–1869 (2007).
Xie, J.-H. et al. Highly enantioselective and diastereoselective synthesis of chiral amino alcohols by ruthenium-catalyzed asymmetric hydrogenation of α-amino aliphatic ketones. J. Am. Chem. Soc. 131, 4222–4223 (2009).
Liu, C., Xie, J.-H., Li, Y.-L., Chen, J.-Q. & Zhou, Q.-L. Asymmetric hydrogenation of α,α′-disubstituted cycloketones through dynamic kinetic resolution: an efficient construction of chiral diols with three contiguous stereocenters. Angew. Chem. Int. Ed. Engl. 52, 593–596 (2013).
Liu, S., Xie, J.-H., Wang, L.-X. & Zhou, Q.-L. Dynamic kinetic resolution allows a highly enantioselective synthesis of cis-α-aminocycloalkanols by ruthenium-catalyzed asymmetric hydrogenation. Angew. Chem. Int. Ed. Engl. 46, 7506–7508 (2007).
Hamada, Y. Diastereo- and enantioselective anti-selective hydrogenation of alpha-amino-beta-keto ester hydrochlorides and related compounds using transition-metal-chiral-bisphosphine catalysts. Chem. Rec. 14, 235–250 (2014).
Tao, X., Li, W., Li, X., Xie, X. & Zhang, Z. Diastereo- and enantioselective asymmetric hydrogenation of alpha-amido-beta-keto phosphonates via dynamic kinetic resolution. Org. Lett. 15, 72–75 (2013).
Ding, Z., Yang, J., Wang, T., Shen, Z. & Zhang, Y. Dynamic kinetic resolution of beta-keto sulfones via asymmetric transfer hydrogenation. Chem. Commun. 571–573 (2009).
Lin, H. et al. Enantioselective approach to (−)-hamigeran B and (−)-4-bromohamigeran B via catalytic asymmetric hydrogenation of racemic ketone to assemble the chiral core framework. Org. Lett. 18, 1434–1437 (2016).
Shi, L. et al. Enantioselective iridium-catalyzed hydrogenation of 3,4-disubstituted isoquinolines. Angew. Chem. Int. Ed. Engl. 51, 8286–8289 (2012).
Cram, D. J. & Kopecky, K. R. Studies in stereochemistry. XXX. Models for steric control of asymmetric induction. J. Am. Chem. Soc. 81, 2748–2755 (1959).
Reetz, M. T. Chelate- or nonchelate control in addition reactions of chiral α- and β-alkoxycarbonyl compounds. Angew. Chem. 96, 542–555 (1984).
Akashi, M., Arai, N., Inoue, T. & Ohkuma, T. Catalyst-controlled diastereoselection in the hydrogenation of heterocycloalkyl ketones. Adv. Synth. Catal. 353, 1955–1960 (2011).
Verendel, J. J., Pamies, O., Dieguez, M. & Andersson, P. G. Asymmetric hydrogenation of olefins using chiral Crabtree-type catalysts: scope and limitations. Chem. Rev. 114, 2130–2169 (2014).
Stork, G. & Kahne, D. E. Stereocontrol in homogeneous catalytic-hydrogenation via hydroxyl group coordination. J. Am. Chem. Soc. 105, 1072–1073 (1983).
Zhou, J. G. et al. Asymmetric hydrogenation routes to deoxypolyketide chirons. Chem. Eur. J. 13, 7162–7170 (2007).
Bernasconi, M., Ramella, V., Tosatti, P. & Pfaltz, A. Iridium- catalyzed asymmetric hydrogenation of 3,3-disubstituted allylic alcohols in ethereal solvents. Chem. Eur. J. 20, 2440–2444 (2014).
Crabtree, R. H. & Davis, M. W. Directing effects in homogeneous hydrogenation with [Ir(cod)(PCy3)(py)]PF6. J. Org. Chem. 51, 2655–2661 (1986).
Akai, S. et al. One-pot synthesis of optically active allyl esters via lipase–vanadium combo catalysis. Org. Lett. 12, 4900–4903 (2010).
Egi, M. et al. A mesoporous-silica-immobilized oxovanadium cocatalyst for the lipase-catalyzed dynamic kinetic resolution of racemic alcohols. Angew. Chem. Int. Ed. Engl. 52, 3654–3658 (2013).
Abate, A. et al. Chirality and fragrance chemistry: stereoisomers of the commercial chiral odorants muguesia and pamplefleur. J. Org. Chem. 70, 1281–1290 (2005).
Gualandi, A., Emma, M. G., Giacoboni, J., Mengozzi, L. & Cozzi, P. G. A highly stereoselective organocatalytic approach to lilial and muguesia. Synlett 24, 449–452 (2013).
Acknowledgements
The Swedish Research Council (VR) and Stiftelsen Olle Engkvist Byggmästare supported this work. J.L. thanks the Guangzhou Elite Scholarship Council for the PhD fellowship and Thishana Singh, School of Chemistry and Physics, University of Kwazulu-Natal, South Africa for proofreading and editing the manuscript.
Author information
Authors and Affiliations
Contributions
P.G.A. conceived and designed the experiments. J.L., S.K., J.Y. and J.-Q.L. performed experiments and prepared the Supplementary Information. P.G.A. and J.L. wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supporting Information
Supplementary Methods, Supplementary Figures 1–60, Supplementary References
Rights and permissions
About this article
Cite this article
Liu, J., Krajangsri, S., Yang, J. et al. Iridium-catalysed asymmetric hydrogenation of allylic alcohols via dynamic kinetic resolution. Nat Catal 1, 438–443 (2018). https://doi.org/10.1038/s41929-018-0070-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-018-0070-0
This article is cited by
-
Iridium-catalyzed enantioselective synthesis of chiral γ-amino alcohols and intermediates of (S)-duloxetine, (R)-fluoxetine, and (R)-atomoxetine
Communications Chemistry (2022)
-
Isolating contiguous Pt atoms and forming Pt-Zn intermetallic nanoparticles to regulate selectivity in 4-nitrophenylacetylene hydrogenation
Nature Communications (2019)
-
Iridium-catalysed enantioselective formal deoxygenation of racemic alcohols via asymmetric hydrogenation
Nature Catalysis (2019)
-
Enantioconvergent hydrogenations
Nature Catalysis (2018)