Abstract
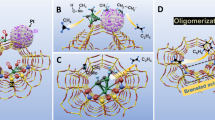
Methane constitutes the largest fraction of natural gas reserves and is a low-cost abundant starting material for the synthesis of value-added chemicals and fuel. Selective catalytic functionalization of methane remains a vital goal in the chemical sciences due to its low intrinsic reactivity. Borylation has recently emerged as a promising route for the catalytic functionalization of methane. A major challenge in this regard is selective borylation towards the monoborylated product that is more active than methane and can easily lead to over-functionalization. Herein, we report a highly selective microporous metal−organic-framework-supported iridium(iii) catalyst for methane borylation that exhibits a chemoselectivity of >99% (mono versus bis at 19.5% yield; turnover number = 67) for monoborylated methane, with bis(pinacolborane) as the borylation reagent in dodecane, at 150 °C and 34 atm of methane. The preference for the monoborylated product is ascribed to the shape-selective effect of the metal−organic framework pore structures.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cooper, J., Stamford, L., & Azapagic, A. Shale gas: a review of the economic, environmental, and social sustainability. Energy Technol. 4, 772–792 (2016).
Caballero, A. & Perez, P. J. Methane as raw material in synthetic chemistry: the final frontier. Chem. Soc. Rev. 42, 8809–8820 (2013).
Wang, B., Albarracin-Suazo, S., Pagan-Torres, Y. & Nikolla, E. Advances in methane conversion processes. Catal. Today 285, 147–158 (2017).
Alvarez-Galvan, M. C. et al. Direct methane conversion routes to chemicals and fuels. Catal. Today 171, 15–23 (2011).
Schwarz, H. Chemistry with methane: concepts rather than recipes. Angew. Chem. Int. Ed. 50, 10096–10115 (2011).
Gunsalus, N. J. et al. Homogeneous functionalization of methane. Chem. Rev. 117, 8521–8573 (2017).
Horn, R. & Schloegl, R. Methane activation by heterogeneous catalysis. Catal. Lett. 145, 23–39 (2015).
Periana, R. A. et al. Platinum catalysts for the high-yield oxidation of methane to a methanol derivative. Science 280, 560–564 (1998).
Periana, R. A., Mironov, O., Taube, D., Bhalla, G. & Jones, C. Catalytic, oxidative condensation of CH4 to CH3COOH in one step via CH activation. Science 301, 814–818 (2003).
Gunsalus, N. J. et al. Homogeneous functionalization of methane. Chem. Rev. 117, 8521–8573 (2017).
Mkhalid, I. A. I., Barnard, J. H., Marder, T. B., Murphy, J. M. & Hartwig, J. F. C−H activation for the construction of C−B bonds. Chem. Rev. 110, 890–931 (2010).
Cook, A. K., Schimler, S. D., Matzger, A. J. & Sanford, M. S. Catalyst-controlled selectivity in the C–H borylation of methane and ethane. Science 351, 1421–1424 (2016).
Smith, K. T. et al. Catalytic borylation of methane. Science 351, 1424–1427 (2016).
Weisz, P. B. & Frilette, V. J. Intracrystalline and molecular-shape-selective catalysis by zeolite salts. J. Chem. Phys. 64, 382 (1960).
Zaera, F. Shape-controlled nanostructures in heterogeneous catalysis. ChemSusChem 6, 1797–1820 (2013).
Wei, J., Floudas, C. A., Gounaris, C. E. & Somorjai, G. A. Search engines for shape selectivity. Catal. Lett. 133, 234–241 (2009).
Chen, N. Y., Kaeding, W. W. & Dwyer, F. G. Para-directed aromatic reactions over shape-selective molecular sieve zeolite catalysts. J. Am. Chem. Soc. 101, 6783–6784 (1979).
Yaghi, O. M. et al. Reticular synthesis and the design of new materials. Nature 423, 705–714 (2003).
Zhou, H. C., Long, J. R. & Yaghi, O. M. Introduction to metal–organic frameworks. Chem. Rev. 112, 673–674 (2012).
Lee, J. et al. Metal–organic framework materials as catalysts. Chem. Soc. Rev. 38, 1450–1459 (2009).
Warren, J. E. et al. Shape selectivity by guest-driven restructuring of a porous material. Angew. Chem. Int. Ed. 53, 4592–4596 (2014).
Cavka, J. H. et al. A new zirconium inorganic building brick forming metal organic frameworks with exceptional stability. J. Am. Chem. Soc. 130, 13850–13851 (2008).
Kanemitsu, H., Harada, R. & Ogo, S. A water-soluble iridium(iii) porphyrin. Chem. Commun. 46, 3083–3085 (2010).
Li, Q., Liskey, C. W. & Hartwig, J. F. Regioselective borylation of the C–H bonds in alkylamines and alkyl ethers. Observation and origin of high reactivity of primary C–H bonds beta to nitrogen and oxygen. J. Am. Chem. Soc. 136, 8755–8765 (2014).
Lawrence, J. D., Takahashi, M., Bae, C. & Hartwig, J. F. Regiospecific functionalization of methyl C−H bonds of alkyl groups in reagents with heteroatom functionality. J. Am. Chem. Soc. 126, 15334–15335 (2004).
Hartwig, J. F. Regioselectivity of the borylation of alkanes and arenes. Chem. Soc. Rev. 40, 1992–2002 (2011).
Chen, H., Schlecht, S., Semple, T. C. & Hartwig, J. F. Thermal, catalytic, regiospecific functionalization of alkanes. Science 287, 1995–1997 (2000).
Pump, E. et al. Exploiting confinement effects to tune selectivity in cyclooctane metathesis. ACS Catal. 7, 6581–6586 (2017).
Yuan, J., Fracaroli, A. M. & Klemperer, W. G. Convergent synthesis of a metal–organic framework supported olefin metathesis catalyst. Organometallics 35, 2149–2155 (2016).
Chen, T. et al. Confinement effects of metal–organic framework on the formation of charge-transfer tetrathiafulvalene dimers. Inorg. Chem. 55, 12758–12765 (2016).
Medders, G. R. & Paesani, F. Water dynamics in metal–organic frameworks: effects of heterogeneous confinement predicted by computational spectroscopy. J. Phys. Chem. Lett. 5, 2897–2902 (2014).
Gomez, D. A., Combariza, A. F. & Sastre, G. Confinement effects in the hydrogen adsorption on paddle wheel containing metal–organic frameworks. Phys. Chem. Chem. Phys. 14, 2508–2517 (2012).
Furukawa, H., Cordova, K. E., O’Keeffe, M. & Yaghi, O. M. The chemistry and applications of metal–organic frameworks. Science 341, 1230444 (2013).
Srivastan, S., Darwish, N. A., Gasem, K. A. M. & Robinson, R. L. Solubility of methane in hexane, decane, and dodecane at temperatures from 311 to 423 K and pressures to 10.4 MPa. J. Chem. Eng. Data 37, 516–520 (1992).
Darwish, N. A., Gasem, K. A. M. & Robinson, R. L. Solubility of methane in cyclohexane and in trans-decalin at temperatures from 323 to 423 K at pressures to 9.6 MPa. J. Chem. Eng. Data 43, 238–240 (1998).
Wang, Y. et al. Engineering catalytic coordination space in a chemically stable Ir-porphyrin MOF with a confinement effect inverting conventional Si–H insertion chemoselectivity. Chem. Sci. 8, 775–780 (2017).
Gonzalez, M. I., Bloch, E. D., Mason, J. A., Teat, S. J. & Long, J. R. Single-crystal-to-single-crystal metalation of a metal–organic framework: a route toward structurally well-defined catalysts. Inorg. Chem. 54, 2995–3005 (2015).
Hartwig, J. F. et al. Rhodium boryl complexes in the catalytic, terminal functionalization of alkanes. J. Am. Chem. Soc. 127, 2538–2552 (2005).
Hartwig, J. F. Borylation and silylation of C–H bonds: a platform for diverse C–H bond functionalizations. Acc. Chem. Res. 45, 864–873 (2012).
Tamura, H., Yamazaki, H., Sato, H. & Sakaki, S. Iridium-catalyzed borylation of benzene with diboron. Theoretical elucidation of catalytic cycle including unusual iridium(v) intermediate. J. Am. Chem. Soc. 125, 16114–16126 (2003).
Boller, T. M. et al. Mechanism of the mild functionalization of arenes by diboron reagents catalyzed by iridium complexes. Intermediacy and chemistry of bipyridine-ligated iridium trisboryl complexes. J. Am. Chem. Soc. 127, 14263–14278 (2005).
Acknowledgements
This work was supported as part of the Inorganometallic Catalyst Design Center, an Energy Frontier Research Center funded by the US Department of Energy, Office of Science, Basic Energy Sciences, under award DESC0012702. Use of the Advanced Photon Source at the Argonne National Laboratory is supported by the US Department of Energy, Office of Science, Basic Energy Sciences, under contract DE-AC-02-06CH11357. MRCAT operations are supported by the Department of Energy and the MRCAT member institutions. This work made use of the IMSERC, Jerome B. Cohen X-Ray Diffraction, EPIC and KECK II facilities of Northwestern University’s NUANCE Center, which has received support from the Soft and Hybrid Nanotechnology Experimental Resource (NSF ECCS-1542205), the MRSEC programme (NSF DMR-1121262) at the Materials Research Center, the International Institute for Nanotechnology, the Keck Foundation and the State of Illinois, through the International Institute for Nanotechnology.
Author information
Authors and Affiliations
Contributions
X.Z., M.D. and O.K.F. designed the experiments. X.Z. and Z.H. carried out the synthesis of the materials with help from P.L. and T.C.W. under the supervision of M.D. and O.K.F. M.F. and Z.H. performed the high-throughput catalysis under the supervision of M.D. and O.K.F. X.Z., Z.H. and D.Y. characterized the materials and analysed the data with help from L.R. under the supervision of M.D. and O.K.F. X.Z., Z.H., L.R., M.D. and O.K.F. wrote the manuscript with contributions from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods; Supplementary Figures 1–12; Supplementary Table 1; Supplementary References
Rights and permissions
About this article
Cite this article
Zhang, X., Huang, Z., Ferrandon, M. et al. Catalytic chemoselective functionalization of methane in a metal−organic framework. Nat Catal 1, 356–362 (2018). https://doi.org/10.1038/s41929-018-0069-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-018-0069-6
This article is cited by
-
Visible light-mediated intermolecular crossed [2+2] cycloadditions using a MOF-supported copper triplet photosensitizer
Nature Catalysis (2024)
-
Improvement of Theoretical Algorithm for Prediction of Carbon Nanotubes Catalytical Activity, Based on Quantum Chemical Semi-Empirical Approaches
Catalysis Letters (2023)
-
Design, synthesis and applications of functional zirconium-based metal-organic frameworks
Science China Chemistry (2023)
-
Installing a molecular truss beam stabilizes MOF structures
npj Computational Materials (2022)
-
Biocompatible MIP-202 Zr-MOF tunable sorbent for cost-effective decontamination of anionic and cationic pollutants from waste solutions
Scientific Reports (2021)