Abstract
Positive adjustment to chronic diseases reduces psychiatric comorbidity and enhances quality of life. Very little is known about the benefit of internet-based and mobile-based Cognitive Behavioral Therapy (IM-CBT) on physical outcomes and its reciprocal interactions with psychiatric outcomes, the active therapeutic elements, and effect moderators among people with major chronic medical conditions. In this systematic review and meta-analysis (PROSPERO: CRD42022265738), CINAHL of Systematic Reviews, MEDLINE, PsycINFO, PubMed, Web of Science are systematically searched up to 1 June 2022, for randomized controlled trials (RCTs) comparing IM-CBT against non-CBT control condition(s) among people with chronic disease(s). Primary outcomes include improvements in psychiatric symptoms (depressive, anxiety, PTSD symptoms, general psychological distress) from baseline to post-intervention and follow-ups. Secondary outcomes include improvements in physical distress (physical symptoms, functional impairment, self-rated ill health, objective physiological dysfunction). Among 44 RCTs (5077 patients with seven different chronic diseases), IM-CBT improves depressive symptoms, anxiety symptoms, and general psychological distress at post-intervention and across follow-ups, and improves physical distress and functional impairment at post-intervention. Preliminary evidence suggests that behavioral modification and problem-solving could be necessary components to reduce psychiatric symptoms in IM-CBT, whereas cognitive restructuring, psychoeducation, and mindfulness elements relate to reduced physical distress. IM-CBT shows stronger benefits in chronic pain, cancer, arthritis, and cardiovascular disease, relative to other conditions. Changes in psychiatric symptoms and physical distress prospectively predict each other over time. IM-CBT is an effective intervention for comprehensive symptom management among people with chronic diseases.
Similar content being viewed by others
Introduction
Chronic diseases are responsible for not only deaths but also years lived with disability, a common expansion of morbidity1. Growing numbers of people live with chronic ill health and compromised quality of life over the past decades1, among which one-third experience multiple conditions2. Interventions for mental health are also prioritized to be integrated into the management of chronic medical conditions3,4,5,6. Those patients are 2-3 times more likely to have comorbid mental ill health such as depressive/anxiety disorders relative to the general population3,7. Comorbid physical and psychiatric conditions could jointly predict poorer prognosis3,5 and add financial and psychosocial burden3,6,8. With the ever-increasing burden on the healthcare system, digitalizing the management of chronic conditions9,10,11 could overcome practical barriers such as immune compromise, mobility difficulties, shortage of clinical personnel, and health disparity12,13.
The clinical benefits of specialized psychological treatment namely Cognitive Behavioral Therapy (CBT) delivered across the internet and/or mobile devices [Internet-based and mobile-based CBT (IM-CBT)] for people with chronic diseases should be rigorously reviewed. IM-CBT has been shown to be as effective as face-to-face CBT14,15 and increase the accessibility of care for underserved patients16,17. Two meta-analyses of different chronic diseases18,19 and one systematic review of people with rheumatic conditions20 have documented the effectiveness of internet-based CBT in reducing psychiatric and/or physical symptoms. However, previous work did not comprehensively study how IM-CBT effects might differ across various diagnostic conditions and/or health outcomes. More importantly, very little is known about the therapeutic elements specifically responsible for the improved clinical outcomes, the effect moderators, and the reciprocity between mental health outcomes and secondary physical health outcomes.
This systematic review and meta-analysis aims to examine the effectiveness of IM-CBT in reducing psychiatric symptoms among patients across most common chronic medical conditions in randomized controlled trials. In-depth analyses were also conducted on active CBT treatment components, the influence of patient-related/treatment-related factors, and the prospective association between resulting psychiatric symptoms and physical distress.
IM-CBT relates to reduced psychiatric symptoms and physical distress, with the two improvements prospectively predicting each other. Among the CBT components, behavioral modification and problem-solving reduce psychiatric symptoms whereas cognitive restructuring, psychoeducation, and mindfulness reduce physical distress. IM-CBT benefits patients with chronic pain, cancer, arthritis, and cardiovascular disease more, psychologically and physically, relative to those with other diseases. Our results attest the clinical utility of IM-CBT for patients with chronic diseases.
Results
The study selection process is shown in Fig. 1. This study included 44 eligible RCTs21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64 reporting 48 IM-CBT-to-control comparisons among a total of 5077 patients (2728 in intervention, 2349 in control groups). Descriptive information on included studies is summarized in Table 1 and Supplementary Tables 1–5.
Included studies
Twelve studies were conducted in North America (US, Canada)22,29,34,36,38,51,52,54,56,60,63,64, 20 in Europe (Netherlands, Sweden, UK, Ireland, Germany, Norway)21,24,25,26,27,31,35,37,39,40,43,44,45,47,53,55,58,59,61,62, 10 in Australia23,28,30,32,33,41,46,48,49,50, and 2 in Asia (Japan, Korea)42,57. Three (6.82%), 24 (54.55%), and 17 (38.64%) studies were assessed to have low, some, and high risks of overall bias, respectively (Supplementary Table 6).
Included patients had a mean age of 47.61 (SD = 13.27) years (range = 11–91 years, based on retrievable information in n = 24 studies) (Supplementary Table 1). Proportions of females ranged 28.81–100%. Chronic diseases included chronic pain (n = 19, 43.18%), cancer (n = 7, 15.91%), arthritis (n = 6, 13.64%), cardiovascular disease (n = 4, 9.09%), diabetes (n = 2, 4.55%), HIV (n = 1, 2.27%), multiple sclerosis (n = 1, 2.27%), and different chronic diseases (n = 4, 9.09%). Comorbid physical or psychiatric conditions were reported in 23 (52.27%) studies. Complementary treatments for either physical or comorbid psychiatric conditions were reported in 22 (50.00%) studies. For details, see Supplementary Table 1.
Interventions across studies were predominantly delivered through web-based modules (n = 39, 88.64%), alongside videoconferences (n = 3, 6.82%) and mobile apps (n = 3, 6.82%); they were generally short in duration (<12 sessions)19 (n = 35, 79.55%) and guided12 (n = 32, 72.73%). Control groups included active (n = 21 studies, 47.73%) or non-active (n = 24, 54.55%) types. The active control condition included information/education (k = 10 comparisons), discussion forum (k = 5), relaxation (k = 2), attention control (scheduled contact) (k = 2), supportive therapy (k = 1), computerized cognitive remediation therapy (k = 1), and lifestyle management (k = 1). For details, see Supplementary Tables 1 and 4. The first follow-up was conducted 8–36 weeks after the intervention ended, whereas the last follow-up 12–48 weeks.
Effectiveness of IM-CBT
IM-CBT exhibited a small-to-moderate effect on decreased depressive symptoms, anxiety symptoms, and general psychological distress across all timepoints: at post-intervention (depressive symptoms, g = 0.448, 95% CI [0.309, 0.587], p < 0.001; anxiety symptoms, g = 0.322, 95% CI [0.193, 0.451], p < 0.001; general psychological distress, g = 0.623, 95% CI [0.229, 1.016], p = 0.002) (Figs. 2–4)21,22,23,24,25,26,27,28,29,30,31,32,33,34,36,37,38,39,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64, first follow-up (depressive symptoms, g = 0.319, 95% CI [0.142, 0.497], p < 0.001; anxiety symptoms, g = 0.171, 95% CI [0.020, 0.322], p = 0.027; general psychological distress, g = 0.581, 95% CI [0.195, 0.968], p = 0.003) (Figs. 5–7)21,23,24,29,31,36,37,39,50,52,54,55,58,59,60,61,62, and last-follow-up (depressive symptoms, g = 0.357, 95% CI [0.207, 0.507], p < 0.001; anxiety symptoms, g = 0.321, 95% CI [0.162, 0.481], p < 0.001; general psychological distress, g = 0.673, 95% CI [0.180, 1.165], p = 0.007) (Figs. 8–10)23,29,37,58,60,61.
The effects on decreased PTSD symptoms23,39,40 and combined depressive and anxiety symptoms21,35,39,40 were significant at follow-up(s) only: at first follow-up (PTSD symptoms, g = 0.867, 95% CI [0.453, 1.282], p < 0.001; combined depressive and anxiety symptoms, g = 0.241, 95% CI [0.020, 0.461], p = 0.032), and last-follow-up (PTSD symptoms, g = 0.576, 95% CI [0.024, 1.128], p = 0.041).
Effect sizes of the positive associations of IM-CBT with decreased physical symptoms (g = 0.184) (Fig. 11) and functional impairment (g = 0.284) (Fig. 12) were small-to-moderate and only at post-intervention21,22,24,25,26,27,29,31,32,33,34,35,36,37,38,39,41,43,47,50,51,52,53,54,56,57,58,59,60,62,63.
Results are summarized in Table 2. A complete list of all forest plots is available in Supplementary Figure 1. No significant differences in the effect of IM-CBT on decreased psychiatric outcomes were found across timepoints, whereas the effect on decreased physical distress was present only at post-intervention (Table 3).
Reciprocity between changes in psychiatric symptoms and changes in physical distress
Decreased psychiatric symptoms at post-intervention prospectively predicted decreased physical distress at follow-ups, B = 0.761, 95% CI [0.405, 1.118], p < 0.00121,24,35,36,37,39,50,51,54,58,59,60,62. Likewise, decreased physical distress at post-intervention prospectively predicted decreased psychiatric symptoms at follow-ups, B = 1.456, 95% CI [0.597, 2.314], p = 0.00121,24,35,36,37,39,50,52,54,58,59,60,62. The results showed bidirectional positive associations (Fig. 13).
Note. a The regression of averaged effect sizes of physical distress (at follow-ups) on averaged effect sizes of psychiatric symptoms (at post-intervention). b The regression of averaged effect sizes of psychiatric symptoms (at follow-ups) on averaged effect sizes of physical distress (at post-intervention). A figure with regressions for n = 58 studies (including 44 studies here and another 14 studies included in the Supplementary Information only) is available in Supplementary Fig. 3.
Core therapeutic elements of IM-CBT affecting clinical responses
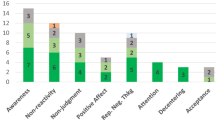
Therapeutic elements within individual studies are summarized in Supplementary Table 3. With the exception of two studies without either behavioral modification (473 patients)30 or problem-solving (45 patients)51, the majority of studies (ks = 46 comparisons) included both elements. IM-CBT was effective for reducing psychiatric symptoms when the protocols included behavioral modification (g = 0.442, 95% CI [0.322, 0.561], p < 0.001) or problem-solving (g = 0.433, 95% CI [0.311, 0.556], p < 0.001). IM-CBT protocols were equally effective for reducing psychiatric symptoms with or without cognitive restructuring, psychoeducation, or mindfulness (all ps ≤ 0.001). Meanwhile, IM-CBT significantly reduced physical distress when the protocols included cognitive restructuring (g = 0.212, p < 0.001)21,24,25,26,31,32,33,35,36,37,38,41,43,49,50,51,52,53,56,57,59,60,63,64, psychoeducation (g = 0.176, p < 0.001)21,22,24,25,26,27,31,32,33,34,35,36,38,39,41,43,47,49,52,53,54,56,57,58,59,60,62,63, or mindfulness (g = 0.173, p < 0.001)21,22,25,26,27,29,32,33,34,35,38,39,41,43,49,53,54,56,57,59,60, but the effects were non-significant without these elements. The effects on reducing psychiatric symptoms or physical distress were independent of the total number of therapeutic elements (ps ≥ 0.407). Results are summarized in Table 3.
Likely candidates for patient-related and treatment-related moderators
IM-CBT related to decreased psychiatric symptoms among patients diagnosed with chronic pain (g = 0.391, p < 0.001)24,25,26,27,29,32,33,35,38,39,47,51,52,53,57,58,59,62,63, cancer (g = 0.495, p = 0.014)21,23,28,36,42,43,48, arthritis (g = 0.402, p = 0.010)37,45,50,54,56,60, cardiovascular disease (g = 0.504, p < 0.001)41,44,55,61, and different chronic diseases (g = 0.566, p = 0.001)34,40,46,64, but not those with diabetes, multiple sclerosis, or HIV (ps ≥ 0.154). Decreased symptoms were reported by patients who were not on concurrent psychotherapies for psychiatric condition(s) (g = 0.396, p < 0.001)21,22,23,24,25,26,27,28,29,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64 but not those receiving psychotherapies (p = 0.136). IM-CBT effects were observed independent of patients’ gender and presence of comorbidity (ps ≥ 0.287). Age as a potential moderator was not analyzed because most studies were conducted among people with a wide range of age (11–91 years, information retrievable from n = 24 studies)23,27,29,31,32,33,34,35,37,38,39,40,42,44,45,46,47,50,51,52,53,57,58,62 while only mean age (SD) was available for all included studies.
The effect sizes for the IM-CBT effects on decreased psychiatric symptoms were larger with fewer than 12 sessions (g = 0.481, p < 0.001)21,22,23,24,25,26,27,28,29,31,32,33,34,36,37,38,39,40,44,45,46,47,48,49,50,51,52,53,54,55,58,59,60,61,62 relative to ≥12 sessions (g = 0.186, p = 0.034) but independent of guidance (p = 0.814). Effects were significant only among interventions delivered through web-based modules (g = 0.448, p < 0.001), relative to interventions delivered through videoconference34,36,57 and mobile app22,40,42. Results are summarized in Table 3. Factors associated with stronger IM-CBT effect sizes on decreased physical distress included specific diagnosis (i.e., chronic pain), absence of complementary treatments for physical/psychiatric conditions, fewer than 12 sessions, and lower frequency of the intervention (Table 3).
Level of confidence in the evidence
IM-CBT effects on psychiatric symptoms were not influenced by methodological factors including overall risk of bias, attrition at post-intervention, and utilization of intention-to-treat analysis, but the effect sizes were larger when comparisons involved non-active (g = 0.535, p < 0.001) than active (g = 0.299, p < 0.001) control groups, p = 0.047. Meanwhile, the effects on physical distress were significant only when studies showed some-to-high risks of overall bias, low-to-moderate (≤20%) attrition rate, utilization of intention-to-treat analysis, and/or non-active control groups (Table 3). No significant publication bias was found on most outcomes among the pooled studies (Supplementary Table 7 and Supplementary Fig. 2). We performed a sensitivity analysis by replicating all analyses after including 14 studies with non-synchronized CBTs delivered through telephone or self-help materials and found largely consistent results (Supplementary Tables 8–9 and Supplementary Fig. 3).
Discussion
This study is a comprehensive and up-to-date systematic review and meta-analysis on the effects of internet-based and mobile-based Cognitive Behavioral Therapy (IM-CBT) on psychiatric symptoms, physical distress, and the reciprocity between improved mental and physical health outcomes among people with chronic diseases. We specifically investigated the therapeutic elements and effect moderators. Our conclusions were based on 44 RCTs (48 comparisons) conducted across 11 countries, with a total of 5077 patients affected by seven different chronic diseases. We found immediate and/or sustained effects of IM-CBT on reducing psychiatric symptoms and physical distress, with improved psychiatric symptoms and physical distress prospectively positively predicting each other. Behavioral modification and problem-solving benefited psychiatric symptoms, whereas cognitive restructuring, psychoeducation, and mindfulness benefited physical distress. Diagnoses of chronic diseases and lower intervention intensity moderated the clinical benefits of IM-CBT, suggesting the need to investigate its impact in more diverse chronic diseases and the cautions in applying different therapeutic elements among different patients.
This meta-analysis went beyond previous studies by robustly validating the clinical benefits of IM-CBT with more types of chronic diseases, more varied psychiatric and physical outcomes, and longer durations of follow-up. Telemedicine and digital psychotherapeutic interventions9,65,66,67 have become more common in recent years68,69. With comparable effectiveness as face-to-face CBT14,15, IM-CBT could contribute added values over its conventional counterparts9,10,11,65,66.
This meta-analysis confirmed the benefits of IM-CBT on the mental health of people with chronic diseases. The effectiveness of CBT for alleviating depressive and/or anxiety symptoms has been attested among people with chronic pain70, rheumatoid arthritis71, COPD72, cardiovascular disease73, and kidney disease74. Adding to previous evidence on the IM-CBT effects on decreased depressive and anxiety symptoms, this study reported some of the first evidence that suggests sustainability of the positive effect over time and across other psychiatric conditions including PTSD symptoms and general psychological distress. The effects remained significant controlling for study quality. It is important to note, however, that included studies focused on depression, anxiety, PTSD, and general psychological distress only although we intended to review studies with all kinds of psychiatric conditions. Beyond the conventional IM-CBT approaches examined within the current review, a previous meta-analysis on technology-based acceptance and commitment therapy (20 articles/interventions)75 reported intervention effects for functioning and acceptance-related outcomes, whereas another systematic review on internet-based mindfulness-based interventions (11 articles on 10 interventions)76 reported intervention effects for psychiatric symptoms, coping, and/or quality of life. It warrants further investigation, however, whether the clinical benefits of conventional IM-CBT and its extensions are uniformly comparable or domain-specific70,77. Taken together, current and previous evidence generally supported the potential of IM-CBT and its third-wave extensions among patients with chronic diseases.
This study assessed comprehensive dimensions of physical distress, namely physical symptoms, functional impairment, self-rated ill health, and objective physiological dysfunction. Existing evidence on physical health benefits of CBT in chronic diseases has been relatively mixed – both positive70,71,78,79 and null72,77 effects have been reported among limited scopes of chronic diseases. Similarly, mixed findings have been identified on the physical health benefits of IM-CBT19,20. Although our moderator analyses suggested that IM-CBT could ameliorate physical symptoms and functional impairment, the significant effects could be attributable to methodological factors such as comparisons with non-active control groups21,24,25,31,32,33,37,38,39,43,47,49,50,51,52,54,57,58,60,63 and some-to-high risk of bias21,22,24,25,26,27,29,31,32,33,34,35,36,38,39,43,47,49,50,53,54,56,57,58,59,60,62,63,64. Non-significant effects could be attributable to high attrition26,35,36,39,53. Additionally, in order to obtain a complete picture on IM-CBT effects on physical distress, our current analysis maximally included all available data categorized under the four pre-specified subcategories, despite potential heterogeneity across specific outcomes. Further meta-analytic reviews are therefore warranted to look into IM-CBT effects on specific individual outcomes under physical distress.
Our findings showed that IM-CBT-related decrease in psychiatric symptoms and physical distress positively predicted each other in the long run, adding to existing cross-sectional evidence on the positive associations in previous empirical studies or meta-analyses19,20,70,71,72,77,78,79,80. The reciprocity suggested that the two dimensions of health are complementary to and benefit each other in the long run. Common psychiatric and physical conditions share etiology and maintenance factors5. Symptom and treatment management plans could consider psychiatric and physical conditions as a larger syndrome towards a holistic symptom management for people with chronic diseases81.
While CBT practically involves skill sets that could be theoretically classified into different categories82, most if not all previous studies overlooked the heterogeneity in therapeutic elements across interventions that share the same label of CBT18,19,20. This could limit a full understanding on the therapeutic mechanism(s) of IM-CBT (or CBT in general) responsible for clinical benefits on physical and mental health83.
We observed that two therapeutic elements, namely behavioral modification and problem-solving, were most commonly adopted across included RCTs (i.e., 42 out of the 44 studies included both). As such, cautions are warranted in interpreting these two factors as potential moderators of IM-CBT effects on psychiatric symptoms. While our analyses could not fully confirm on an empirical level that the two components are necessary conditions to ensure the benefits on physical and mental health of people with chronic diseases, theoretically, the clinical implications of behavioral modification and problem-solving have been documented in existing literature. For example, subordinate strategies within behavioral modification such as behavioral contracting and physical exercise could enhance activity level and healthy lifestyle, which in turn serve as protective factors of mental health5,84. Problem-solving, denoting systematic procedures to identify and address everyday life problems and enhance coping skills, has been found to decrease depressive symptoms among older adults with physical conditions85 and among psychiatric patients in the primary care setting86.
Cognitive restructuring, psychoeducation, and mindfulness were shown to be important therapeutic components for reducing physical distress. Cognitive restructuring replaces negative and inaccurate thoughts with more realistic and adaptive ones87. Relatedly, psychoeducation equips people with knowledge on chronic diseases and guides them to be aware of disease-related cognition and behaviors88. Both could increase health literacy and relieve psychological burden, leaving these people with more motivation and energy necessary for symptom management such as medication adherence and health-promoting behaviors3. In addition, mindfulness, as the ability or practice to observe one’s present sensations, thoughts, and feelings with an open and nonjudgmental attitude89, has been found to improve pain and fatigue, blood pressure, and weight control among people with different chronic diseases, although uncertainties exist in its mechanism, variability, and consistency across different modalities90.
The effects of IM-CBT in reducing psychiatric symptoms were more established among chronic pain, cancer, arthritis, and cardiovascular disease, but not diabetes, multiple sclerosis, and HIV. However, it should be noted that the latter three conditions have been investigated by fewer studies22,30,31,49.
The significant IM-CBT effects among interventions delivered via web-based modules but not videoconferences and mobile apps could be due to the fact that it was the predominant format adopted across eligible studies. However, because there were few studies on interventions delivered via videoconferences and mobile apps, which also tended to be less methodologically reliable (i.e., absence of intention-to-treat analysis and/or high attrition rates in most of them) compared to those delivered via web-based modules, we were not able to fully assess the impact of delivery platform on IM-CBT effects. More systematic investigation is needed on whether there is true advantage of delivering IM-CBT over particular types of platforms.
Surprisingly, the effects of IM-CBT on reducing psychiatric symptoms and physical distress were stronger with fewer sessions (<12 sessions) and thus shorter intervention durations. The effects for physical outcomes were similarly contingent upon fewer intervention sessions and lower intervention frequency. We followed these up with chi-squared tests, and noticed that on a methodological level, interventions with longer duration (≥12 sessions) and higher frequency tended to include no guidance (i.e., absence of therapists’ active provision of intervention, feedback, and/or support). Interventions with higher frequency were also more likely to include active control groups (e.g., information/education, discussion forum), and interventions with more sessions were more likely to target patients with physical and/or psychiatric comorbidity. These variations across the RCTs in this review in terms of design and quality suggest the importance of considering the multidimensional sources of therapeutic benefits. We found that active control group was a significant moderator. Based on the common factors theory91, a part of the IM-CBT effects could be protocol-nonspecific, and thus frequent engagement in the active control activities could be inversely related to psychiatric symptoms or physical distress over a period of time. In addition, our findings could call for more attention and empirical investigation to reconsider whether the effects of IM-CBT vary, positively, as functions of treatment duration and/or frequency. The association between intervention duration (number of sessions) and outcome could be curvilinear instead of linear, meaning a possible diminishing marginal benefit after an optimal number of sessions92. Our findings were indeed consistent with previous evidence suggesting lower dose as a cost-effective design93,94. Short intervention with frequent breaks has been suggested to be useful for accommodating fatigue in CBT for adolescents with chronic diseases88. Lower intervention intensity has also been recommended for people with poorer general health95, such as those with chronic diseases in the current meta-analysis. Frequent reminders on the intervention could inadvertently result in notification fatigue and increase non-adherence that has been observed in digital interventions among patients with chronic diseases65. In the current study, we observed that non-adherence (different from attrition) information was insufficiently reported and thus we could not include this variable in the formal analyses. Taken together, these observations invite an open discussion on optimizing the prescription of IM-CBT in order to maximize its clinical benefits for patients with chronic diseases.
This quantitative synthesis considered a wide range of chronic diseases and examined a large number of psychiatric and physical outcomes within IM-CBT for people with chronic diseases, as well as the positive prospective associations between physical and mental health outcomes. Effective individual therapeutic elements for reducing psychiatric symptoms and physical distress were identified, and patient-related/treatment-related moderators affecting the clinical responses were examined. Our evidence points to a clear direction for developing a holistic support care service for these people.
This meta-analysis has some limitations. We pooled the results despite the technical and clinical variations that exist across the included studies. Effect sizes for physical distress were synthesized under four subcategories (i.e., physical symptoms, functional impairment, self-rated ill health, objective physiological dysfunction) although there could be disparity in specific outcomes under each subcategory. Our moderator analyses were conducted on composite constructs of psychiatric symptoms and physical distress instead of the specific constructs. These procedures were applied in order to maximize the number of comparisons. Still, the pooled effect sizes and the moderator analyses might be restricted by the existing number of studies on certain outcomes or features. Some outcomes at follow-up timepoints were missing for synthesis, and differences across subgroups could be left undetected due to a lack of statistical power in the analysis of small samples. Finally, the existing evidence base is biased towards high-income countries/regions, restricting generalizability of the findings to less developed parts of the world.
In conclusion, internet-based and mobile-based cognitive behavioral therapy (IM-CBT) could be implemented in clinical settings in order to produce meaningful benefits on reducing psychiatric symptoms and physical distress among patients with chronic diseases. It is likely that the positive effects of IM-CBT on physical and mental health reciprocally benefit each other in the long run. IM-CBT could be particularly beneficial for people within some chronic diseases, while specific therapeutic elements could be key drivers of clinical benefits. It is important for medical scientists and clinicians to consider the fundamental driving forces of positive therapeutic changes in patients, as quality matters more than quantity in IM-CBT. The present findings could also be applicable to psychological services amid large-scale disasters, such as the COVID-19 pandemic, natural hazards, and wars, when physical comorbidities are more likely, restrictions are put on mobility, or the physical environment is not conducive to face-to-face interventions.
Method
Search strategy and selection criteria
This systematic review and meta-analysis was conducted according to the Preferred Reporting Items for Systematic review and Meta-analysis (PRISMA) guidelines96, and was pre-registered on PROSPERO (CRD42022265738). Any deviations were outlined and explained in Supplementary Note 1. Searches were performed in CINAHL of Systematic Reviews, MEDLINE, PsycINFO, PubMed, and Web of Science from inception through June 1, 2022, using combined variations of the following keyword categories: chronic diseases, cognitive behavioral therapy, psychiatric symptoms, study design. The detailed search algorithm is documented in Supplementary Note 2.
E.T.F.Y., T.K.L., and P.B.S. selected the articles and extracted data; disagreements were resolved through discussion with T.J.T., L.K.Y.M., and W.K.H. Only English articles published in peer-reviewed journals were considered. The current study reviewed randomized clinical trials that compared psychiatric symptoms between IM-CBT and non-CBT control condition(s) among patients diagnosed with chronic diseases listed on ICD-11 for ≥3 months. Because it is not quite possible to include the great variety of chronic diseases in one single systematic review/meta-analysis, we generated a list of common chronic diseases by referring to leading causes of disability-adjusted life years in the Global Burden of Disease Study 2015 in The Lancet1. This study was set out to focus on the more conventional types of CBT, which focus more on modifying and controlling behaviors, thoughts and emotions, relative to the third-wave extensions, which alternatively focus more on acceptance and mindfulness approaches97. In practice, their boundaries could be less clear-cut, and therefore in cases where interventions included a mix of cognitive-behavioral and third-wave elements, our key criterion to decide whether the interventions were eligible was whether they were predominantly defined by the cognitive and/or behavioral elements as opposed to the third-wave elements. Studies were also excluded if the treatment group contained any in-person psychosocial interventions.
Quality assessment
Included articles were assessed by E.T.F.Y., T.K.L., and P.B.S. using the revised Cochrane risk-of-bias tool for randomized trials (RoB 2)98, and were categorized into low risk, some concerns, or high risk (Supplementary Table 6).
Outcome measures
Primary outcomes included improvements in psychiatric symptoms (i.e., depressive, anxiety, and PTSD symptoms, general psychological distress) from baseline to (1) post-intervention, (2) first follow-up, and (3) last follow-up. When a study included multiple instruments for the same psychiatric outcome, only one scale was chosen based on hypothesized frequency of use99. Secondary outcomes included improvements in physical distress (i.e., physical symptoms, functional impairment, self-rated ill health, objective physiological dysfunction) from baseline to different timepoints. If studies included multiple treatment/control arms, each eligible comparison was separately considered, with the sample size of the treatment/control arm divided correspondingly to avoid double counting100.
Quantitative synthesis on effectiveness
To statistically account for any baseline differences, we calculated the Hedge’s g (0.2 = small, 0.5 = moderate, 0.8 = large) with 95% CI for each outcome based on the change score from baseline to post-intervention (or to follow-ups) between the intervention and control groups101. Correlations between scores within the same group was set at 0.7102. If insufficient baseline data was reported (2 studies, 4.55%), Hedge’s g was calculated based on cross-sectional comparison(s) between the intervention and control groups. Group means and standard deviations, if not readily available for quantitative syntheses, were converted from other statistics (Supplementary Note 3). In addition, the Q and I2 (25% = low, 50% = moderate, 75% = high) indices were calculated to indicate the presence and the degree of heterogeneity across results. Analyses with a random-effects approach were performed using Comprehensive Meta-Analysis version 3.0.
The prospective associations between changes in psychiatric symptoms and changes in physical distress were examined in two meta-regressions, one regressing the effect size of physical distress at follow-ups on that of psychiatric symptoms at post-intervention and one regressing the effect size of psychiatric symptoms at follow-ups on that of physical distress at post-intervention.
Moderator effects
In the subgroup analyses, demographic and medical characteristics of the patients and the characteristics and methodology of the included interventions were investigated with Q-tests and meta-regressions: psychiatric symptoms/physical distress, demographics, medical profile, complementary treatments, intervention delivery platform, presence of guidance (i.e., therapeutic input in the form of therapists’ active provision of intervention, feedback, and/or support)12, intervention duration/frequency, therapeutic elements (i.e., behavioral modification, cognitive restructuring, problem-solving, psychoeducation, mindfulness; Supplementary Note 4)82, assessment schedule, control type, overall risk of bias, attrition rate, and the use of intention-to-treat analysis. To address dependency issues, multiple effect sizes from the same source were averaged in all meta-analytic procedures103.
Certainty of the evidence
Risk of publication bias was assessed using funnel plots and the Egger test of asymmetry104. In cases of significant asymmetry, results were statistically adjusted with the trim-and-fill method105.
Data availability
W.K.H. has full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. All study materials are available from the corresponding author upon reasonable request.
References
GBD. 2015 DALYs & HALE Collaborators. Global, regional, and national disability-adjusted life-years (DALYs) for 315 diseases and injuries and healthy life expectancy (HALE), 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet 388, 1603–1658 (2016).
Hajat, C. & Stein, E. The global burden of multiple chronic conditions: a narrative review. Prev. Med. Rep. 12, 284–293 (2018).
Naylor, C. et al. Long-term conditions and mental health: the cost of co-morbidities. The King’s Fund and Centre for Mental Health. London, UK. 1–32 (2012).
Ngo, V. K. et al. Grand challenges: integrating mental health care into the non-communicable disease agenda. PLoS Med. 10, e1001443 (2013).
O’Neil, A. et al. A shared framework for the common mental disorders and non-communicable disease: key considerations for disease prevention and control. BMC Psychiatry 15, 15 (2015).
Stein, D. J. et al. Integrating mental health with other non-communicable diseases. BMJ 364, l295 (2019).
Daré, L. O. et al. Co-morbidities of mental disorders and chronic physical diseases in developing and emerging countries: a meta-analysis. BMC Public Health 19, 304 (2019).
Armbrecht, E. et al. Economic and humanistic burden associated with noncommunicable diseases among adults with depression and anxiety in the United States. J. Med. Econ. 23, 1032–1042 (2020).
Andersson, G. & Titov, N. Advantages and limitations of internet‐based interventions for common mental disorders. World Psychiatry 13, 4–11 (2014).
Barbosa, W., Zhou, K., Waddell, E., Myers, T. & Dorsey, E. R. Improving access to care: telemedicine across medical domains. Annu. Rev. Public Health 42, 463–481 (2021).
Bendig, E. et al. Internet-based interventions in chronic somatic disease. Dtsch. Ärztebl 115, 659–665 (2018).
Karyotaki, E. et al. Internet-based cognitive behavioral therapy for depression: a systematic review and individual patient data network meta-analysis. JAMA Psychiatry 78, 361–371 (2021).
Sanders, L. M. E-health care: promise or peril for chronic illness. J. Pediatr. 195, 15 (2018).
Andersson, G., Cuijpers, P., Carlbring, P., Riper, H. & Hedman, E. Guided Internet‐based vs. face‐to‐face cognitive behavior therapy for psychiatric and somatic disorders: a systematic review and meta‐analysis. World Psychiatry 13, 288–295 (2014).
Carlbring, P., Andersson, G., Cuijpers, P., Riper, H. & Hedman-Lagerlöf, E. Internet-based vs. face-to-face cognitive behavior therapy for psychiatric and somatic disorders: an updated systematic review and meta-analysis. Cogn. Behav. Ther. 47, 1–8 (2018).
Ebert, D. D. et al. Internet- and mobile-based psychological interventions: applications, efficacy, and potential for improving mental health. Eur. Psychol. 23, 167–187 (2018).
Kumar, V., Sattar, Y., Bseiso, A., Khan, S. & Rutkofsky, I. H. The effectiveness of internet-based cognitive behavioral therapy in treatment of psychiatric disorders. Cureus 9, e1626 (2017).
Mehta, S., Peynenburg, V. A. & Hadjistavropoulos, H. D. Internet-delivered cognitive behaviour therapy for chronic health conditions: a systematic review and meta-analysis. J. Behav. Med. 42, 169–187 (2019).
van Beugen, S. et al. Internet-based cognitive behavioral therapy for patients with chronic somatic conditions: a meta-analytic review. J. Med. Internet Res. 16, e2777 (2014).
Terpstra, J. A., van der Vaart, R., Ding, H. J., Kloppenburg, M. & Evers, A. W. Guided internet-based cognitive-behavioral therapy for patients with rheumatic conditions: a systematic review. Internet Interv. 26, 100444 (2021).
Atema, V. et al. Efficacy of internet-based cognitive behavioral therapy for treatment-induced menopausal symptoms in breast cancer survivors: results of a randomized controlled trial. J. Clin. Oncol. 37, 809–822 (2019).
Barroso, J., Madisetti, M. & Mueller, M. A feasibility study to develop and test a cognitive behavioral stress management mobile health application for HIV-related fatigue. J. Pain. Symptom Manag. 59, 242–253 (2020).
Beatty, L., Koczwara, B. & Wade, T. Evaluating the efficacy of a self-guided web-based CBT intervention for reducing cancer-distress: a randomised controlled trial. Support. Care Cancer 24, 1043–1051 (2016).
Buhrman, M., Fältenhag, S., Ström, L. & Andersson, G. Controlled trial of internet-based treatment with telephone support for chronic back pain. Pain 111, 368–377 (2004).
Buhrman, M., Nilsson-Ihrfeldt, E., Jannert, M., Ström, L. & Andersson, G. Guided internet-based cognitive behavioural treatment for chronic back pain reduces pain catastrophizing: a randomized controlled trial. J. Rehabil. Med. 43, 500–505 (2011).
Buhrman, M. et al. Guided internet-delivered cognitive behavioural therapy for chronic pain patients who have residual symptoms after rehabilitation treatment: randomized controlled trial. Eur. J. Pain 17, 753–765 (2013).
Buhrman, M. et al. Individualized guided internet-delivered cognitive-behavior therapy for chronic pain patients with comorbid depression and anxiety. Clin. J. Pain 31, 504–516 (2015).
Chambers, S. K. et al. Web-delivered cognitive behavioral therapy for distressed cancer patients: randomized controlled trial. J. Med. Internet Res. 20, e42 (2018).
Chiauzzi, E. et al. painACTION-back pain: a self-management website for people with chronic back pain. Pain Med. 11, 1044–1058 (2010).
Clarke, J. et al. A web-based cognitive behavior therapy intervention to improve social and occupational functioning in adults with type 2 diabetes (the springboard trial): randomized controlled trial. J. Med. Internet Res. 21, e12246 (2019).
Cooper, C. L. et al. Computerised cognitive behavioural therapy for the treatment of depression in people with multiple sclerosis: external pilot trial. Trials 12, 259 (2011).
Dear, B. F. et al. The pain course: a randomised controlled trial of a clinician-guided internet-delivered cognitive behaviour therapy program for managing chronic pain and emotional well-being. Pain 154, 942–950 (2013).
Dear, B. F. et al. The Pain Course: a randomised controlled trial examining an internet-delivered pain management program when provided with different levels of clinician support. Pain 156, 1920–1935 (2015).
Doorley, J. D. et al. Feasibility randomized controlled trial of a mind–body activity program for older adults with chronic pain and cognitive decline: the virtual “Active Brains” study. Gerontologist 62, 1082–1094 (2022).
Dowd, H. et al. Comparison of an online mindfulness-based cognitive therapy intervention with online pain management psychoeducation. Clin. J. Pain 31, 517–527 (2015).
Ferguson, R. J. et al. A randomized trial of videoconference‐delivered cognitive behavioral therapy for survivors of breast cancer with self‐reported cognitive dysfunction. Cancer 122, 1782–1791 (2016).
Ferwerda, M. et al. A tailored-guided internet-based cognitive-behavioral intervention for patients with rheumatoid arthritis as an adjunct to standard rheumatological care: results of a randomized controlled trial. Pain 158, 868–878 (2017).
Friesen, L. N. et al. Examination of an internet-delivered cognitive behavioural pain management course for adults with fibromyalgia: a randomized controlled trial. Pain 158, 593–604 (2017).
Gasslander, N. et al. Tailored internet-based cognitive behavioral therapy for individuals with chronic pain and comorbid psychological distress: a randomized controlled trial. Cogn. Behav. Ther. 51, 408–434 (2022).
Geirhos, A. et al. Feasibility and potential efficacy of a guided internet-and mobile-based CBT for adolescents and young adults with chronic medical conditions and comorbid depression or anxiety symptoms (youthCOACHCD): a randomized controlled pilot trial. BMC Pediatr. 22, 69 (2022).
Glozier, N. et al. Internet-delivered cognitive behavioural therapy for adults with mild to moderate depression and high cardiovascular disease risks: a randomised attention-controlled trial. PLoS One 8, e59139 (2013).
Ham, K. et al. Preliminary results from a randomized controlled study for an app-based cognitive behavioral therapy program for depression and anxiety in cancer patients. Front. Psychol. 10, 1592 (2019).
Hummel, S. B. et al. Efficacy of internet-based cognitive behavioral therapy in improving sexual functioning of breast cancer survivors: results of a randomized controlled trial. J. Clin. Oncol. 35, 1328–1340 (2017).
Johansson, P. et al. An internet-based cognitive behavioral therapy program adapted to patients with cardiovascular disease and depression: randomized controlled trial. JMIR Ment. Health 6, e14648 (2019).
Lundgren, J. G. et al. The effect of guided web-based cognitive behavioral therapy on patients with depressive symptoms and heart failure: a pilot randomized controlled trial. J. Med. Internet Res. 18, e5556 (2016).
Migliorini, C., Sinclair, A., Brown, D., Tonge, B. & New, P. A randomised control trial of an internet-based cognitive behaviour treatment for mood disorder in adults with chronic spinal cord injury. Spinal Cord 54, 695–701 (2016).
Mourad, G. et al. Guided Internet-delivered cognitive behavioural therapy in patients with non-cardiac chest pain–a pilot randomized controlled study. Trials 17, 352 (2016).
Murphy, M. J. et al. Randomised controlled trial of internet‐delivered cognitive behaviour therapy for clinical depression and/or anxiety in cancer survivors (iCanADAPT early). Psycho‐Oncol. 29, 76–85 (2020).
Newby, J. et al. Web-based cognitive behavior therapy for depression in people with diabetes mellitus: a randomized controlled trial. J. Med. Internet Res. 19, e7274 (2017).
O’moore, K. A. et al. Internet cognitive-behavioral therapy for depression in older adults with knee osteoarthritis: a randomized controlled trial. Arthritis Care Res. 70, 61–70 (2018).
Palermo, T. M., Wilson, A. C., Peters, M., Lewandowski, A. & Somhegyi, H. Randomized controlled trial of an internet-delivered family cognitive–behavioral therapy intervention for children and adolescents with chronic pain. Pain 146, 205–213 (2009).
Palermo, T. M. et al. Internet-delivered cognitive-behavioral treatment for adolescents with chronic pain and their parents: a randomized controlled multicenter trial. Pain 157, 174–185 (2016).
Peters, M. L. et al. Happy despite pain: a randomized controlled trial of an 8-week internet-delivered positive psychology intervention for enhancing well-being in patients with chronic pain. Clin. J. Pain 33, 962–975 (2017).
Shigaki, C. L. et al. RAHelp: an online intervention for individuals with rheumatoid arthritis. Arthritis Care Res. 65, 1573–1581 (2013).
Simblett, S. K. et al. Computerized cognitive behavioral therapy to treat emotional distress after stroke: a feasibility randomized controlled trial. JMIR Ment. Health 4, e6022 (2017).
Stinson, J. N. et al. An internet-based self-management program with telephone support for adolescents with arthritis: a pilot randomized controlled trial. J. Rheumatol. 37, 1944–1952 (2010).
Taguchi, K. et al. Clinical effectiveness and cost-effectiveness of videoconference-based integrated cognitive behavioral therapy for chronic pain: randomized controlled trial. J. Med. Internet Res. 23, e30690 (2021).
Thesen, T. et al. Effectiveness of internet-based cognitive behavioral therapy with telephone support for noncardiac chest pain: randomized controlled trial. J. Med. Internet Res. 24, e33631 (2022).
Trautmann, E. & Kröner-Herwig, B. A randomized controlled trial of internet-based self-help training for recurrent headache in childhood and adolescence. Behav. Res. Ther. 48, 28–37 (2010).
Trudeau, K. J. et al. A randomized controlled trial of an online self-management program for adults with arthritis pain. J. Behav. Med. 38, 483–496 (2015).
Westas, M., Lundgren, J., Andersson, G., Mourad, G. & Johansson, P. Effects of internet-delivered cognitive behavioural therapy adapted for patients with cardiovascular disease and depression: a long-term follow-up of a randomized controlled trial at 6 and 12 months posttreatment. Eur. J. Cardiovasc. Nurs. 21, 559–567 (2022).
Wiklund, T. et al. Internet-delivered cognitive behavioral therapy for insomnia comorbid with chronic pain: randomized controlled trial. J. Med. Internet Res. 24, e29258 (2022).
Williams, D. A. et al. Internet-enhanced management of fibromyalgia: a randomized controlled trial. Pain 151, 694–702 (2010).
Wilson, M. et al. Engaging adults with chronic disease in online depressive symptom self-management. West. J. Nurs. Res. 40, 834–853 (2018).
Karekla, M. et al. Best practices and recommendations for digital interventions to improve engagement and adherence in chronic illness sufferers. Eur. Psychol. 24, 49–67 (2019).
Maisto, M. et al. Digital interventions for psychological comorbidities in chronic diseases—a systematic review. J. Pers. Med. 11, 30 (2021).
Aboujaoude, E., Salame, W. & Naim, L. Telemental health: a status update. World Psychiatry 14, 223–230 (2015).
Rodriguez, J. A., Shachar, C. & Bates, D. W. Digital inclusion as health care—supporting health care equity with digital-infrastructure initiatives. N. Engl. J. Med. 386, 1101–1103 (2022).
Tuckson, R. V., Edmunds, M. & Hodgkins, M. L. Telehealth. N. Engl. J. Med. 377, 1585–1592 (2017).
Khoo, E. L. et al. Comparative evaluation of group-based mindfulness-based stress reduction and cognitive behavioural therapy for the treatment and management of chronic pain: a systematic review and network meta-analysis. Evid. Based Ment. Health 22, 26–35 (2019).
Shen, B. et al. Effects of cognitive behavioral therapy for patients with rheumatoid arthritis: a systematic review and meta-analysis. Psychol. Health Med. 25, 1179–1191 (2020).
Ma, R. C., Yin, Y. Y., Wang, Y. Q., Liu, X. & Xie, J. Effectiveness of cognitive behavioural therapy for chronic obstructive pulmonary disease patients: a systematic review and meta‐analysis. Complement. Ther. Clin. Pract. 38, 101071 (2020).
Reavell, J., Hopkinson, M., Clarkesmith, D. & Lane, D. A. Effectiveness of cognitive behavioral therapy for depression and anxiety in patients with cardiovascular disease: a systematic review and meta-analysis. Psychosom. Med. 80, 742–753 (2018).
Ng, C. Z. et al. A systematic review and meta-analysis of randomized controlled trials of cognitive behavioral therapy for hemodialysis patients with depression. J. Psychosom. Res. 126, 109834 (2019).
Herbert, M. S. et al. Technology-supported Acceptance and Commitment Therapy for chronic health conditions: a systematic review and meta-analysis. Behav. Res. Ther. 148, 103995 (2022).
Russell, L., Ugalde, A., Milne, D., Austin, D. & Livingston, P. M. Digital characteristics and dissemination indicators to optimize delivery of internet-supported mindfulness-based interventions for people with a chronic condition: systematic review. JMIR Ment. Health 5, e9645 (2018).
Farver-Vestergaard, I., Jacobsen, D. & Zachariae, R. Efficacy of psychosocial interventions on psychological and physical health outcomes in chronic obstructive pulmonary disease: a systematic review and meta-analysis. Psychother. Psychosom. 84, 37–50 (2015).
Hanlon, I., Hewitt, C., Bell, K., Phillips, A. & Mikocka‐Walus, A. Systematic review with meta‐analysis: online psychological interventions for mental and physical health outcomes in gastrointestinal disorders including irritable bowel syndrome and inflammatory bowel disease. Aliment. Pharmacol. Ther. 48, 244–259 (2018).
Bernard, P. et al. Cognitive behavior therapy combined with exercise for adults with chronic diseases: systematic review and meta-analysis. Health Psychol. 37, 433–450 (2018).
Harkness, E. et al. Identifying psychosocial interventions that improve both physical and mental health in patients with diabetes: a systematic review and meta-analysis. Diabetes Care 33, 926–930 (2010).
Fisher, E. B. et al. Conceptual perspectives on the co-occurrence of mental and physical disease: diabetes and depression as a model. In: Sartorius, N., Holt, R. I. G. & Maj, M. (eds.) Comorbidity of Mental and Physical Disorders 179, 1–14 (Karger Publishers, 2015).
Beck, J. S. Cognitive Behavior Therapy: Basics and Beyond (Guilford Publications, 2020).
Wasil, A. R., Venturo-Conerly, K. E., Shingleton, R. M. & Weisz, J. R. A review of popular smartphone apps for depression and anxiety: assessing the inclusion of evidence-based content. Behav. Res. Ther. 123, 103498 (2019).
Cuijpers, P., Van Straten, A. & Warmerdam, L. Behavioral activation treatments of depression: a meta-analysis. Clin. Psychol. Rev. 27, 318–326 (2007).
Frost, R., Bauernfreund, Y. & Walters, K. Non-pharmacological interventions for depression/anxiety in older adults with physical comorbidities affecting functioning: systematic review and meta-analysis. Int. Psychogeriatr. 31, 1121–1136 (2019).
Zhang, A., Park, S., Sullivan, J. E. & Jing, S. The effectiveness of problem-solving therapy for primary care patients’ depressive and/or anxiety disorders: a systematic review and meta-analysis. J. Am. Board. Fam. Med. 31, 139–150 (2018).
Traeger, L. Cognitive restructuring. In: Encyclopedia of Behavioral Medicine. https://doi.org/10.1007/978-3-030-39903-0166 (Springer International Publishing, 2020).
Morey, A. & Loades, M. E. How has cognitive behaviour therapy been adapted for adolescents with comorbid depression and chronic illness? a scoping review. Child Adolesc. Psychiatry Ment. Health 26, 252–264 (2021).
Kabat-Zinn, J. & Hanh, T. N. Full Catastrophe Living: Using the Wisdom of Your Body and Mind to Face Stress, Pain, and Illness (Delta, 2009).
Zhang, D., Lee, E. K., Mak, E. C., Ho, C. Y. & Wong, S. Y. Mindfulness-based interventions: an overall review. Br. Med. Bull. 138, 41–57 (2021).
Cuijpers, P., Reijnders, M. & Huibers, M. J. The role of common factors in psychotherapy outcomes. Annu. Rev. Clin. Psychol. 15, 207–231 (2019).
Robinson, L., Delgadillo, J. & Kellett, S. The dose-response effect in routinely delivered psychological therapies: a systematic review. Psychother. Res. 30, 79–96 (2020).
Donkin, L. et al. Rethinking the dose-response relationship between usage and outcome in an online intervention for depression: randomized controlled trial. J. Med. Internet Res. 15, e2771 (2013).
Venkatesan, A., Forster, B., Rao, P., Miller, M. & Scahill, M. Improvements in depression outcomes following a digital cognitive behavioral therapy intervention in a polychronic population: retrospective study. JMIR Form. Res. 6, e38005 (2022).
Bruijniks, S. J. et al. Individual differences in response to once versus twice weekly sessions of CBT and IPT for depression. J. Consult. Clin. Psychol. 90, 5–17 (2022).
Moher, D., Liberati, A., Tetzlaff, J. & Altman, D. G. & PRISMA Group*. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann. Intern. Med. 151, 264–269 (2009).
Cattie, J. E., Buchholz, J. L. & Abramowitz, J. S. Cognitive therapy and cognitive-behavioral therapy. In: Messer, S. B. & Kaslow, N. J. (eds.) Essential Psychotherapies: Theory and Practice (4th ed.) 142–182 (The Guilford Press, 2019).
Higgins, J. P. T., Savović, J., Page, M. J., Elbers, R. G. & Sterne, J. A. C. Chapter 8: Assessing risk of bias in a randomized trial. In: Higgins, J. P. T., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M. J. & Welch, V. A. (eds.) Cochrane Handbook for Systematic Reviews of Interventions (Version 6.3). www.training.cochrane.org/handbook (2022).
Vita, A. et al. Effectiveness, core elements, and moderators of response of cognitive remediation for schizophrenia: a systematic review and meta-analysis of randomized clinical trials. JAMA Psychiatry 78, 848–858 (2021).
Higgins, J. P.T., Eldridge, S. & Li, T. Chapter 23: Including variants on randomized trials. In: Higgins, J. P. T., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M. J. & Welch, V. A. (eds.) Cochrane Handbook for Systematic Reviews of Interventions (Version 6.3). www.training.cochrane.org/handbook (2022).
Deeks, J. J., Higgins, J. P. T. & Altman, D. G. Chapter 10: Analysing data and undertaking meta-analyses. In: Higgins, J. P. T., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M. J. & Welch, V. A. (eds.) Cochrane Handbook for Systematic Reviews of Interventions (Version 6.3). www.training.cochrane.org/handbook (2022).
Rosenthal, R. Meta-Analytic Procedures for Social Research (Rev ed.) (Sage, 1991).
Borenstein, M., Hedges, L. V., Higgins, J. P. & Rothstein, H. R. Introduction to Meta-Analysis. (John Wiley & Sons, 2021).
Egger, M., Smith, G. D., Schneider, M. & Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 315, 629–634 (1997).
Duval, S. & Tweedie, R. Trim and fill: a simple funnel‐plot–based method of testing and adjusting for publication bias in meta‐analysis. Biometrics 56, 455–463 (2000).
Schulz, K. F. & Grimes, D. A. Sample size slippages in randomised trials: exclusions and the lost and wayward. Lancet 359, 781–785 (2002).
Acknowledgements
This work was supported by Research Grants Council, University Grants Committee, Hong Kong SAR, China [grant numbers C7069-19 GF and 18600320 (W.K.H.)]. The funding source had no role in any process of our study.
Author information
Authors and Affiliations
Contributions
Authorship of this article complies with inclusion & ethics in global research. All authors of this article meet criteria for authorship. Authors T.J.T. and T.K.L. contributed equally to this work. T.J.T.: Conceptualization, Methodology, Formal analysis, Data curation, Writing—Original draft, Writing—Review & Editing; T.K.L.: Formal analysis, Investigation, Data curation, Writing—Review & Editing; E.T.F.Y.: Investigation, Data curation, Writing—Review & Editing; H.L.: Methodology, Formal analysis, Writing—Review & Editing; Phoenix Bibha Shris: Investigation, Writing—Review & Editing; L.K.Y.M.: Writing—Review & Editing; T.M.C.L.: Writing—Review & Editing; W.K.H.: Conceptualization, Methodology, Formal analysis, Data curation, Writing—Original draft, Writing—Review & Editing, Supervision, Project administration, Funding acquisition.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tao, T.J., Lim, T.K., Yeung, E.T.F. et al. Internet-based and mobile-based cognitive behavioral therapy for chronic diseases: a systematic review and meta-analysis. npj Digit. Med. 6, 80 (2023). https://doi.org/10.1038/s41746-023-00809-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41746-023-00809-8
This article is cited by
-
A randomized controlled trial of a self-led, virtual reality-based cognitive behavioral therapy on sick role adaptation in colorectal cancer patients: study protocol
BMC Cancer (2024)
-
Efficacy of a cognitive-behavioral digital therapeutic on psychosocial outcomes in rheumatoid arthritis: randomized controlled trial
npj Mental Health Research (2024)