Abstract
The survival outcomes of the FLAURA trial support osimertinib as the new standard of care for untreated patients harboring activating mutations in the epidermal growth factor receptor (EGFR). Despite the initial response, disease progression invariably occurs. Although uncommon, BRAF V600E mutation arises as a unique mechanism of resistance, and thus far, no prospective studies are available to support concurrent EGFR/BRAF blockade. We report a case of impressive radiological and ctDNA response under dabrafenib, trametinib, and osimertinib in an advanced EGFR-mutant lung adenocarcinoma patient who developed BRAF V600E as one of the acquired resistance mechanisms to second-line osimertinib. Moreover, the patient experienced remarkable clinical improvement and good tolerance to combination therapy. The present case suggests the importance of prospective studies evaluating both efficacy and safety of the combination in later line settings and points towards the potential of ctDNA to monitor resistance mechanisms and treatment benefit in clinical practice.
Similar content being viewed by others
Introduction
The use of osimertinib to target epidermal growth factor receptor (EGFR) has become the standard of care in untreated EGFR-mutant non–small cell lung cancer (NSCLC) patients. Although osimertinib can be highly active, showing more durable outcomes than first-generation tyrosine kinase inhibitors (TKI)1, most tumors invariably become resistant, limiting its long-term clinical benefit. The heterogeneity of resistance mechanisms to osimertinib, including EGFR C797S mutation, EGFR, and MET amplifications, off-target mutations in PIK3CA, KRAS, and HER2 as well as histologic transformation2,3,4,5,6,7,8,9, has stimulated routine performance of repeated biopsies to identify specific underlying mechanisms of resistance throughout the treatment course, and to guide the development of novel therapeutic strategies to overcome and prevent acquired resistance (AR)7,10,11,12,13.
Combined targeted therapy (TT) strategies have been increasingly addressed in prospective clinical trials14,15,16,17. A phase Ib/II trial reported 47% ORR with a combination of capmatinib (MET inhibitor) plus gefitinib in patients progressing to EGFR TKI and presenting MET dysregulation14. Likewise, SAVANNAH (NCT03778229)15 is an ongoing phase II trial designed to evaluate the efficacy of combination savolitinib and osimertinib in post-osimertinib progression/MET-positive patients. The biomarker-matched study ORCHARD (NCT03944772)16 is also underway to assess the efficacy of several osimertinib-based combinations following disease progression under frontline osimertinib.
BRAF mutations and fusions (i.e. AGK-BRAF, ESYT2-BRAF) have recently emerged as additional mechanisms of AR to third-generation EGFR TKI2,18,19,20. Studies demonstrating the efficacy of concurrent inhibition of EGFR and BRAF3 or MEK21 in pre-clinical models have raised clinicians’ expectations about overcoming AR by combining TT. Nevertheless, reports of successful combinations of TT for patients harboring BRAF-driven AR to osimertinib are very limited19,22,23,24 and no prospective data regarding efficacy and safety of BRAF/MEK/EGFR concurrent inhibition are available, with chemotherapy-based regimens remaining the treatment of choice in this unfavorable scenario. Similarly, PIK3CA mutations may also mediate AR to second-line osimertinib in 4–11%, but no clinical reports suggesting potential benefits of blocking these alterations in NSCLC are available6,9,25,26,27.
The detection of circulating tumor DNA (ctDNA) using liquid biopsies allows noninvasive real-time monitoring of treatment response and early detection of AR to TT, anticipating radiological response and treatment failure28,29. The widespread availability of highly specific and sensitive techniques to quantify ctDNA makes the longitudinal assessment of patients with NSCLC under TKI therapy very attractive. Nonetheless, for NSCLCs, ctDNA evaluation in routine clinical practice is currently limited to the detection of EGFR exon 19 deletion, EGFR L858R and T790M activating mutations29.
A man with metastatic lung adenocarcinoma harboring an EGFR mutation, who had progressed to erlotinib due to the emergence of the EGFR T790M mutation started osimertinib and remained on treatment for 15 months, then developing disease progression (PD). Sequential liquid biopsies were collected to monitor treatment response and disclosed the emergence of BRAF V600E and PIK3CA E545K resistance mutations 4 months before clinical progression. With this result and considering few available reports in the literature, our patient started a triple therapy with osimertinib, dabrafenib (BRAF inhibitor), and trametinib (MEK inhibitor).
Results
Case report
A 50-year-old non-smoker man was diagnosed with a tubule-papillary lung adenocarcinoma metastatic to bones and soft tissue (stage IVB - AJCC 8th edition) in July 2016. Several hypermetabolic bone lesions were observed on staging 18-fluorodeoxyglucose positron-emission tomography (18F-FDG PET-CT) scan at diagnosis. A baseline Next Generation Sequencing (NGS) assay (TruSightTumor™ - Illumina®) of a soft tissue metastasis revealed the presence of an activating EGFR exon 19 deletion (EGFR E746_A750del). No concurrent alterations in BRAF or in PIK3CA were identified at that time. ALK d5f3 immunohistochemistry and fluorescent in situ hybridization (FISH) for ROS1 and RET resulted negative.
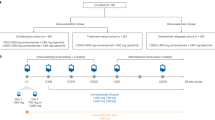
In September 2016, the patient started on erlotinib 150 mg once daily (OD), achieving complete metabolic response in February 2017. We started to perform serial blood sample collections for ctDNA analysis in May 2017. In October 2017, oligo-progression (oligo-PD) was observed at the primary site and treated with stereotactic body radiation therapy (SBRT; 3 × 16 Gy). In November 2017, we identified the EGFR T790M and EGFR E746_A750del mutations in patient’s plasma using droplet digital PCR (ddPCR) and these results were confirmed in December 2017 with the Foundation ACT® ctDNA assay. Figure 1a shows all systemic and focal therapies of this patient since diagnosis.
a Timeline displaying systemic and focal therapies since September 2016. b ctDNA levels in serial plasma samples based on fractional abundance of EGFR T790M, EGFR del19 (E746_A750 del), BRAF V600E, and PIK3CA E545K mutations. #2.4 CN results: EGFR del19 WT/RNAseP copy number ratio. c Paired 18F-FDG PET-CT scan maximum intensity projection (MIP) images displaying tumor burden variations. PD progression of disease, SBRT stereotatic body radiotherapy, Pembro pembrolizumab, CT chemotherapy, DTO dabrafenib, trametinib and osimertinib.
Erlotinib was kept until January 2018 when disease progression in the bones was observed and the fractional abundance (FA) of EGFR E746_A750del and EGFR T790M were 12.0 and 2.3%, respectively (Fig. 1b). The treatment was promptly switched to osimertinib 80 mg OD. Bone partial response (PR) was observed 2 months later, along with a significant drop in the FA of EGFR E746_A750del and EGFR T790M mutations in the plasma (Fig. 1b).
In July 2018, the patient developed oligo-PD in T3 vertebrae, which was treated with SBRT (1 × 20 Gy). At that point, ctDNA analysis revealed an increase in the FA of the EGFR E746_A750del in the plasma, but not in EGFR T790M, suggesting genetic heterogeneity between metastatic lesions. In December 2018, a new oligo-PD in the left iliac bone was detected by 18F-FDG PET-CT scan (Fig. 1c). At that time, ctDNA analysis revealed the emergence of BRAF V600E (FA: 0.4%) and PIK3CA E545K (FA: 0.9%) mutations, as well as a significant increase in the FA of the EGFR E746_A750del (FA: 7.5%) in the plasma. These mutations were also detected in a specimen from an iliac bone biopsy using a NGS assay (TruSightTumor™ - Illumina® - EGFR del19 allelic fraction 81.1%, EGFR del19 amplification – 12 copies, BRAF V600E allelic fraction 17.7% and PIK3CA E545K allelic fraction 32.7%). Even though the patient was treated with SBRT (1 × 18 Gy), he developed new bone metastases in March 2019 (Fig. 1b), consistent with the significant increase in the FA of EGFR E746_A750del, BRAF V600E and EGFR T790M mutations (Fig. 1b). At that specific timepoint and in April 2019, assessments of PIK3CA E545K plasma levels were not obtained due to limited amount of cfDNA.
Between April and September 2019, systemic treatment with carboplatin plus pemetrexed and pembrolizumab provided modest clinical benefit, reducing tumor burden and controlling the disease (Fig. 1b). Due to a new PD observed in October 2019, the treatment was switched to docetaxel monotherapy and, subsequently, to vinorelbine after progression under docetaxel; nonetheless, the patient experienced symptomatic PD in bones, lymph nodes, and lung in late January 2020 (Fig. 1c, 2a–d). He came to the clinic with a Karnofsky performance status of 70%, complaining of fatigue, appetite loss, and severe pain in the hips (despite regular use of 10 mg buprenorphine patch and oxycodone plus acetaminophen PO), which had been preventing him from performing his daily activities due to the inability to stay seated. At that point, high levels of all three resistance mutations were detected in the circulating DNA (PIK3CA E545K FA: 13.2%, BRAF V600E FA: 12.3%, and EGFR T790M FA: 5.3%; Fig. 1b). We also observed an exponential increase in the number of copies of EGFR E746_A750del, suggesting gene amplification (Fig. 1b).
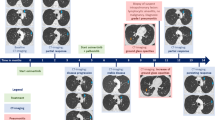
a–d baseline imaging (January/2020) showing: hypermetabolic spiculated mass (orange circle) in the left-superior lobe measuring 4.4 × 3.3 cm (SUVmax: 9.4); multiple hilar, mediastinal, retroperitoneal, and iliac hypermetabolic lymph nodes (red arrow) measuring up to 2.7 cm (SUVmax: 12.8); several hypermetabolic bone lesions throughout axial and appendicular skeleton (yellow circle); a left iliac bone lesion with signs of periosteal reaction and adjacent soft-tissue infiltration (SUVmax: 11.4). e–h first response evaluation imaging (April/2020) showing considerable partial response in the lung mass, measuring 3.8 × 2.5 cm (SUVmax: 2.7), as well as in several bone lesions, especially in the left-iliac bone (SUVmax: 4.8); complete response in lymph nodes. i–l third response evaluation imaging showing disease progression in the left ischium (SUVmax: 12.5; previous SUVmax: 5.9) (October/2020). SUVmax maximum standard uptake value.
In February 2020, after careful consideration due to patient’s good performance status, normal organ functions, severe pain, motivation and the emergence of BRAF V600E as an AR mechanism, he started on dabrafenib 75 mg twice daily (BID), trametinib 1 mg OD and osimertinib 80 mg OD. The use of a PI3K inhibitor was not considered an option due to the absence of reports in the literature suggesting efficacy of these drugs combined with osimertinib to treat advanced NSCLC patients. Within 2 weeks of treatment, the patient achieved complete resolution of the severe bone pain in the hips with no further need of opioid administration, as well as appetite gain and marked improvement in quality of life, which turned possible for him to resume his daily activities. As adverse events (AE), he experienced grade 1 fatigue, dysgeusia, fever, and nausea, all managed with symptomatic medication. Complete resolution of the fever occurred spontaneously within two weeks of treatment. An attempt to increase dosages of dabrafenib to 150 mg BID and trametinib 2 mg OD resulted unsuccessful due to persistent grade 2 fatigue. In April 2020, a new 18F-FDG PET-CT scan disclosed a complete response in lymph nodes and a dramatic PR in the lung and bones (PERCIST 1.1 evaluating 5 target lesions: Δ- 67%; Figs. 1c and 2e–h). Detection of EGFR T790M, BRAF V600E, and PIK3CA E545K mutations became negative in April 2020, while EGFR E746_A750del despite marginally positive, presented marked reduction (Fig. 1b) suggesting that combination dabrafenib, trametinib, and osimertinib might be an effective strategy to overcome PIK3CA E545K and BRAF V600E-driven resistance to osimertinib in advanced EGFR-mutant NSCLC patients. The patient remained in response until October 2020, when he developed asymptomatic bone PD in lumbar spine, left ischium, and right iliac bone, 8 months after starting this combination therapy (Figs. 1c and 2i–l). A FoundationOne®Liquid CDx plasma NGS disclosed the following alterations: CHCHD3-BRAF fusion, BRAF V600E, EGFR E746_750del, EGFR amplification, PIK3CA E545K, MAP2K2 (MEK2) C125S, MTAP rearrangement intron 5, TP53 V197M, and TP53 S241A. Owing to the considerable clinical benefit, good tolerance and lack of systemic treatment options, we decided to keep the patient on treatment and increase the doses of dabrafenib (150 mg BID) and trametinib (2 mg OD) every other day. He remains asymptomatic and tolerating well the proposed dose adjustment.
Discussion
Several studies have recently highlighted the importance of considering genes of interest within the context of commonly co-occurring mutations9,30. For example, as described by Blakely et al. through performing a cfDNA NGS analysis of 1,122 advanced stage EGFR-mutant NSCLCs, in ~93% of the patients, at least one more variables with known or likely known functional properties were present, disclosing the molecular complexity of this oncogenic driver and suggesting an association of co-occuring genomic alterations with TKI response and clinical outcomes9. In addition, Roper and colleagues reported the identification of at least two co-existing AR mechanisms in 73% of patients treated with osimertinib, as well as 6–23 different subclones per individual in a phylogenetic analysis performed in multiple metastatic sites of 15 individuals30. In the same publication, the authors also called the attention for a high incidence of acquired EGFR amplifications in post-osimertinib patients, which suggests maintenance of EGFR central role in the setting of progression, as previously reported by our group28 and also detected in the present case.
Although uncommon, BRAF V600E mediates AR in approximately 3% of the patients under second-line osimertinib6,20 and little is known about the efficacy of combined TT in this population3,21,22,23. Existing reports lack details regarding objective responses using standardized radiological criteria (i.e., RECIST 1.1; PERCIST) and the dynamics of resistance mechanisms through longitudinal ctDNA measurements22,23. Huang and colleagues22 described a case of an EGFRdel19/T790M + NSCLC patient who developed BRAF V600E-driven AR after second-line osimertinib and achieved stable disease under dabrafenib 150 mg BID, trametinib 1 mg OD and osimertinib 80 mg OD, with ongoing disease control 7.4 months after. Zhou and colleagues23 also reported their experience with dabrafenib 150 mg BID, trametinib 2 mg OD and osimertinib 80 mg OD leading to tumor reduction within 6 weeks of treatment, along with grade 2 rash and decreased appetite as AEs. Similarly, Meng et al. reported two cases treated with this triple regimen31. The first patient discontinued therapy after one month due to severe pneumonitis; the second one presented tumor response under dabrafenib 50 mg BID, trametinib 0.5 mg OD, and osimertinib 80 mg OD, with progression-free survival of 14 months. According to the authors, this reduced dose was prescribed owing to a grade 2 pyrexia, nausea, and vomiting under higher dabrafenib and trametinib doses. Dagogo-Jack and colleagues19 also described a successful case of combined EGFR/MAP kinase pathway blockade with osimertinib 80 mg OD and trametinib 1 mg OD; as treatment-related AEs, their patient experienced grade 2 diarrhea and fatigue, along with grade 1 rash and gastrointestinal bleeding. Nevertheless, it is worth highlighting the rapid clinical improvement, the remarkable radiologically confirmed objective response, as well as the good tolerance observed in this case even using only half standard dose of dabrafenib and trametinib approved for NSCLCs harboring BRAF V600E mutations. Since data regarding the efficacy of these combined approaches, the optimal drug association and dosing, as well as the toxicity profile are conflicting and largely unknown, further investigation into the mechanistic basis of this association represents an important priority. The Table 1 summarizes the toxicities arising under osimertinib plus BRAF/MEK inhibition and reported in the literature.
Liquid biopsy is emerging as an important diagnostic and predictive tool in the treatment of NSCLCs. The accurate identification of predictive genetic alterations is important for both patients’ management and the understanding of clonal evolution and AR to different therapies9,29,32. Also, circulating biomarkers from multiple disease sites better reflect systemic tumor burden, including alterations from genetically different metastatic lesions, which may be missed with single-site tissue biopsies8. Here, we were able to identify and monitor multiple concurrent mechanisms of resistance throughout the entire patient’s treatment. As shown in Fig. 1b, variations in blood ctDNA levels exhibited a positive correlation with imaging findings, even in situations involving focal treatments for oligo-PD. The observed concordance between radiological PD or response and ctDNA measurements also points towards its great potential to be incorporated into clinical practice to anticipate radiologic findings in a more effective manner.
Along with BRAF V600E mutation, the presence of PIK3CA E545K mutation is also associated with AR to osimertinib6,9,25,27. However, the clinical implication of concomitant targeting of EGFR and PI3K remains unclear. Whereas an alternative argument for the observed response could be a rechallenge following a long time upon off-osimertinib, in our opinion, this is an unlikely explanation33. Interestingly, the presence of a BRAF fusion along with a MEK2 mutation identified through plasma NGS in the setting of PD might represent a mechanism of resistance to this triple regimen34. Nonetheless, since pretreatment blood samples had not been analyzed using a similar methodology, this hypothesis deserves careful interpretation.
Increased understanding of the relationship of concurring genomic alterations in EGFR-mutant NSCLC may enable new therapeutic opportunities following disease progression to osimertinib. Here, we reported an impressive objective response to dabrafenib, trametinib, and osimertinib with concordant decrease in plasma ctDNA levels in a metastatic lung cancer patient harboring EGFR E746_A750del, BRAF V600E, and PIK3CA E545K activating mutations. This case report leads to a greater understanding of the currently limited literature regarding the management of EGFR-mutant NSCLC patients with acquired BRAFV600E mutation, since it reports a successful attempt to target both alterations concurrently while providing concordant and interesting data of serial ctDNA assessments throughout the entire treatment. Further investigation to optimize the efficacy and mitigate the toxicity profile of this drug association represents an important issue.
Methods
Patient
This study was approved by Hospital Sírio-Libanês Ethics Committee (HSL-RC 2020-16). The patient provided written informed consent for blood collection, ctDNA analysis and publication of this report.
Sample collection and plasma DNA extraction
Serial blood samples were collected between May 2017 and April 2020 (Fig. 1b). Peripheral blood (20 ml) was collected into tubes containing EDTA (BD, Franklin Lakes, New Jersey). Plasma was separated from the blood within 2 hours of blood collection, as previously described28. cfDNA was extracted using QIAamp MinElute Virus Vacuum Kit (Qiagen, Hilden, Germany) and stored at −80 °C.
ctDNA-ddPCR
Cell-free DNA (cfDNA) was quantified using the RNase P Copy Number Reference Assay (Life Technologies, Carlsbad, California). A total of 3000 genome-equivalents (~10 ng of cfDNA) were analyzed per assay to achieve a detection sensitivity of 0.2%. This detection limit has been assessed by using cell line-derived genomic DNA. A total of 10 ng of input DNA with varying proportions of mutant DNA was serially diluted into wild-type DNA to obtain samples with a mutant abundance of 1%, 0.5%, 0.1%, and 0.05% and subjected to droplet digital PCR (ddPCR). ddPCR was used to quantify the circulating levels of the EGFR activating mutation (EGFR E746_A750 del) and of the resistance mutations (EGFR T790M, PIK3CA E545K, and BRAF V600E). Probes and primers were obtained from BioRad (EGFR E746_A750 del #10041170, EGFR T790M #10040782, PIK3CA E545K #10041188, and BRAF V600E #10040779; Hercules, California). ddPCR was performed on the QX200 Droplet Digital PCR System, and data were analysed using QuantaSoft software (Bio-Rad). ctDNA quantification is presented as fractional abundance (FA–the proportion of the mutant allele in total cfDNA).
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
The datasets that support the findings of this study are not publicly available in order to protect patient privacy. The data will be made available on reasonable request. For data access requests regarding the liquid biopsy (ctDNA quantification) data, please contact Dr. Franciele Knebel, email address: fhknebel@mochsl.org.br. For data access requests regarding the PET-CT high resolution images and PERCIST calculations, please contact Dr. José Marin, email address:jfgmarin@yahoo.com.br. For data access requests regarding the summary of the toxicities arising under osimertinib plus BRAF/MEK inhibition, please contact the corresponding author Dr. Maurício Ribeiro, email address: mauricio.fsaribeiro@hsl.org.br. The data generated and analysed during this study are described in the following metadata record: https://doi.org/10.6084/m9.figshare.1347594635.
References
Ramalingam, S. S. et al. Overall survival with osimertinib in untreated, egfr-mutated advanced NSCLC. N. Engl. J. Med. 382, 41–50 (2020).
Oxnard, G. R. et al. Assessment of resistance mechanisms and clinical implications in patients with EGFR T790M-positive lung cancer and acquired resistance to osimertinib. JAMA Oncol. 4, 1527–1534 (2018).
Ho, C. C. et al. Acquired BRAF V600E mutation as resistant mechanism after treatment with osimertinib. J. Thorac. Oncol. 12, 567–572 (2017).
Ahn, S. et al. Transformation to small cell lung cancer of pulmonary adenocarcinoma: clinicopathologic analysis of six cases. J. Pathol. Transl. Med. 50, 258–263 (2016).
Thress, K. S. et al. Acquired EGFR C797S mutation mediates resistance to AZD9291 in non-small cell lung cancer harboring EGFR T790M. Nat. Med. 21, 560–562 (2015).
Leonetti, A. et al. Resistance mechanisms to osimertinib in EGFR-mutated non-small cell lung cancer. Br. J. Cancer 121, 725–737 (2019).
Ricordel, C., Friboulet, L., Facchinetti, F. & Soria, J. C. Molecular mechanisms of acquired resistance to third-generation EGFR-TKIs in EGFR T790M-mutant lung cancer. Ann. Oncol. 29, i28–i37 (2018).
Piper-Vallillo, A. J., Sequist, L. V. & Piotrowska, Z. Emerging treatment paradigms for EGFR-mutant lung cancers progressing on osimertinib: a review. J. Clin. Oncol. JCO1903123 (2020). https://doi.org/10.1200/JCO.19.03123. Epub ahead of print.
Blakely, C. M. et al. Evolution and clinical impact of co-occurring genetic alterations in advanced-stage EGFR-mutant lung cancers. Nat. Genet. 49, 1693–1704 (2017).
Oxnard, G. R. et al. TATTON: a multi-arm, phase Ib trial of osimertinib combined with selumetinib, savolitinib, or durvalumab in EGFR-mutant lung cancer. Ann. Oncol. 31, 507–516 (2020).
Sequist, L. V. et al. Osimertinib plus savolitinib in patients with EGFR mutation-positive, MET-amplified, non-small-cell lung cancer after progression on EGFR tyrosine kinase inhibitors: interim results from a multicentre, open-label, phase 1b study. Lancet Oncol. 21, 373–386 (2020).
Arulananda, S. et al. Combination osimertinib and gefitinib in C797S and T790M EGFR-mutated non-small cell lung cancer. J. Thorac. Oncol. 12, 1728–1732 (2017).
Piotrowska, Z. et al. Landscape of acquired resistance to osimertinib in EGFR-mutant NSCLC and clinical validation of combined EGFR and RET inhibition with osimertinib and BLU-667 for acquired RET fusion. Cancer Discov. 8, 1529–1539 (2018).
Wu, Y. L. et al. Phase Ib/II study of capmatinib (INC280) plus gefitinib after failure of epidermal growth factor receptor (EGFR) inhibitor therapy in patients with EGFR-mutated, MET factor-dysregulated non-small-cell lung cancer. J. Clin. Oncol. 36, 3101–3109 (2018).
Oxnard, G. R. et al. SAVANNAH: a Phase II trial of osimertinib plus savolitinib for patients (pts) with EGFR-mutant, MET-driven (MET+), locally advanced or metastatic non-small cell lung cancer (NSCLC), following disease progression on osimertinib. J. Clin. Oncol. 37, TPS9119–TPS9119 (2019).
Yu, H. et al. P2.01-22 ORCHARD: a phase II platform study in patients with advanced NSCLC who have progressed on first-line osimertinib therapy. J. Thorac. Oncol. 14, S647 (2019).
Yang, J. C. H., Ellers-Lenz, B., Straub, J., Johne, A. & Wu, Y. L. 536TiP - INSIGHT 2: tepotinib plus osimertinib in patients with EGFR-mutant NSCLC having acquired resistance to EGFR TKIs due to MET-amplification: a phase II trial in progress study. Ann. Oncol. 30, ix181 (2019).
Vojnic, M. et al. Acquired BRAF rearrangements induce secondary resistance to EGFR therapy in EGFR-mutated lung cancers. J. Thorac. Oncol. 14, 802–815 (2019).
Dagogo-Jack, I. et al. Response to the combination of osimertinib and trametinib in a patient with EGFR-mutant NSCLC harboring an acquired BRAF fusion. J. Thorac. Oncol. 14, e226–e228 (2019).
Guibert, N. et al. Amplicon-based next-generation sequencing of plasma cell-free DNA for detection of driver and resistance mutations in advanced non-small cell lung cancer. Ann. Oncol. 29, 1049–1055 (2018).
Ohashi, K. et al. Lung cancers with acquired resistance to EGFR inhibitors occasionally harbor BRAF gene mutations but lack mutations in KRAS, NRAS, or MEK1. Proc. Natl Acad. Sci. USA 109, E2127–E2133 (2012).
Huang, Y., Gan, J., Guo, K., Deng, Y. & Fang, W. Acquired BRAF V600E mutation mediated resistance to osimertinib and responded to osimertinib, dabrafenib, and trametinib combination therapy. J. Thorac. Oncol. 14, e236–e237 (2019).
Zhou, F., Zhao, W., Chen, X., Zhang, J. & Zhou, C. Response to the combination of dabrafenib, trametinib and osimertinib in a patient with EGFR-mutant NSCLC harboring an acquired BRAF(V600E) mutation. Lung Cancer 139, 219–220 (2020).
Xie, Z. et al. Lung adenocarcinoma harboring concomitant EGFR mutations and BRAF V600E responds to a combination of osimertinib and vemurafenib to overcome osimertinib resistance. Clin. Lung Cancer. S1525-7304(20)30190-X (2020). https://doi.org/10.1016/j.cllc.2020.06.008. Epub ahead of print.
Gelatti, A. C. Z., Drilon, A. & Santini, F. C. Optimizing the sequencing of tyrosine kinase inhibitors (TKIs) in epidermal growth factor receptor (EGFR) mutation-positive non-small cell lung cancer (NSCLC). Lung Cancer 137, 113–122 (2019).
Schoenfeld, A. J. et al. Tumor analyses reveal squamous transformation and off-target alterations as early resistance mechanisms to first-line osimertinib in EGFR-mutant lung cancer. Clin. Cancer Res. 26, 2654–2663 (2020).
Le, X. et al. Landscape of EGFR-dependent and -independent resistance mechanisms to osimertinib and continuation therapy beyond progression in EGFR-mutant NSCLC. Clin. Cancer Res. 24, 6195–6203 (2018).
Knebel, F. H. et al. Sequential liquid biopsies reveal dynamic alterations of EGFR driver mutations and indicate EGFR amplification as a new mechanism of resistance to osimertinib in NSCLC. Lung Cancer 108, 238–241 (2017).
Kilgour, E., Rothwell, D. G., Brady, G. & Dive, C. Liquid biopsy-based biomarkers of treatment response and resistance. Cancer Cell 37, 485–495 (2020).
Roper, N. et al. Clonal evolution and heterogeneity of osimertinib acquired resistance mechanisms in EGFR mutant lung cancer. Cell Rep. Med. 1, 100007 (2020).
Meng, P. et al. Combined osimertinib, dabrafenib and trametinib treatment for advanced non-small-cell lung cancer patients with an osimertinib-induced BRAF V600E mutation. Lung Cancer 146, 358–361 (2020).
Piotrowska, Z. et al. Heterogeneity underlies the emergence of EGFRT790 wild-type clones following treatment of T790M-positive cancers with a third-generation EGFR inhibitor. Cancer Discov. 5, 713–722 (2015).
Metro, G. et al. Successful response to osimertinib rechallenge after intervening chemotherapy in an EGFR T790M-positive lung cancer patient. Clin. Drug Investig. 38, 983–987 (2018).
Van Allen, E. M. et al. The genetic landscape of clinical resistance to RAF inhibition in metastatic melanoma. Cancer Discov. 4, 94–109 (2014).
Ribeiro, M. et al. Metadata supporting the article: impressive response to combination dabrafenib, trametinib, and osimertinib in a metastatic EGFR-mutant/BRAF V600E lung adenocarcinoma patient. figshare https://doi.org/10.6084/m9.figshare.13475946 (2020).
Acknowledgements
We thank the clinical research team at Hospital Sírio-Libanês for their assistance collecting samples. We thank Ernande Xavier dos Santos and Elisângela Monteiro for the outstanding technical support. We also thank Mrs. Dina Binzagr and Vivian Hannud for supporting the Translational Research Program at Hospital Sírio Libanês. This work was supported by Ludwig Cancer Research and Fundação de Amparo à Pesquisa do Estado de São Paulo – FAPESP. Lastly, we would like to thank the patient and his family for consenting to the present publication.
Author information
Authors and Affiliations
Contributions
Conceptualization by M.F.S.A.R., F.H.K., J.V.M.A., A.A.C., and A.K.; Methodology by F.H.K. and A.A.C.; Resources br M.F.S.A.R., F.H.K., A.A.C., and J.V.M.A.; Writing of the first draft by M.F.S.A., F.H.K., and J.V.M.A.; Review, editing and approval of the final draft by all authors; Supervision by A.A.C. and A.K.; Funding acquisition by F.H.K. and A.A.C. M.F.S.A.R. and F.H.K. are co-first authors of the present work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ribeiro, M.F.S.A., Knebel, F.H., Bettoni, F. et al. Impressive response to dabrafenib, trametinib, and osimertinib in a metastatic EGFR-mutant/BRAF V600E lung adenocarcinoma patient. npj Precis. Onc. 5, 5 (2021). https://doi.org/10.1038/s41698-021-00149-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41698-021-00149-4
This article is cited by
-
Therapeutic strategies for EGFR-mutated non-small cell lung cancer patients with osimertinib resistance
Journal of Hematology & Oncology (2022)
-
Dabrafenib/osimertinib/trametinib
Reactions Weekly (2021)