Abstract
The air–liquid (a–l) interfacial chemistry of glyoxal is of great interest in atmospheric chemistry. We present molecular imaging of glyoxal and hydrogen peroxide (H2O2) dark aging using in situ time-of-flight secondary ion mass spectrometry (ToF-SIMS). More organic peroxides and cluster ions are observed at the a–l interface in dark aging compared to UV aging. Cluster ions formed with more water molecules in dark aging indicate that the aqueous secondary organic aerosol (aqSOA) could form hydrogen bond with water molecules, suggesting that aqSOAs at the aqueous phase are more hydrophilic. Thus the interfacial aqSOA in dark aging could increase hygroscopic growth. Strong contribution of cluster ions and large water clusters in dark aging indicates change of solvation shells at the a–l interface. The observation of organic peroxides and cluster ions indicates that the aqueous surface could be a reservoir of organic peroxides and odd hydrogen radicals at night. Our findings provide new understandings of glyoxal a–l interfacial chemistry and fill in the gap between field measurements and the climate model simulation of aqSOAs.
Similar content being viewed by others
Introduction
Secondary organic aerosol (SOA) formation occurring at the air–liquid (a–l) interface is important in atmospheric chemistry and its impact on global climate modeling.1,2,3 SOA is defined as the organic mass produced by gas-to-particle partition and oxidation of vapors of semivolatile compounds or volatile organic compounds (VOCs) in the atmosphere.4 SOA formed in the aqueous phase such as cloud, fog, and aqueous aerosols is termed as aqSOA.5 Given the chemical and physical properties (e.g., surface tension, surface density, and reactivity) of the a–l interface, interfacial chemistry is critical in SOA formation, acting as a reactive sink for trace gases (e.g., NOx, O3, and SO2) and enhancing aerosol growth.6 The a–l chemistry profoundly affects the aerosol’s ability to serve as cloud condensation nuclei (CCN) and ice nuclei, and mitigate the earth’s radiation budget.7,8
Glyoxal, the smallest di-carbonyl compound with a lifetime ranging from hours to a day, has diverse sources during the oxidation of VOCs from anthropogenic and biogenic activities. Its solubility leads to a high uptake coefficient on cloud, fog, or aqueous aerosols.9 Thus multiphase chemistry takes place at the aqueous surface, offering a significant source of SOA in the atmosphere. For instance, glyoxal contributed up to 15% of SOA mass in Mexico city.10 Hydrogen peroxide (H2O2) plays a vital role in the nonradical oxidation of aldehydes and dialdehydes, contributing to the SOA burden in addition to radical oxidation pathway.6 Recent observation of hydroxyhydroperoxides (α-HHPs) from glyoxal and H2O2 oxidation improved the SOA simulation.11,12
To the best of our knowledge, a handful of studies were conducted to investigate the aqSOA formation at the air–water interfaces, suggesting its effect on global aerosol budgets.13,14,15 Most studies were performed using either bulk solution or chamber experiments coupled with NMR,16 ESI-MS,17 FT-ICR-MS,18 or DESI-MS for product detection.19 Schweitzer proposed that surface chemistry of glyoxal protonation occurred prior to uptake by aqueous droplet like formaldehyde.20 The formation of chemisorbed surface complex of aldehyde was hypothesized but not verified.21 Nevertheless, the SOA formation pathways at the a–l interface are still poorly understood.22 Obtaining in situ molecular speciation and distribution at the a–l interface is a daunting task due to the lack of advanced in situ analytical tools. The large gap between modeling and observation is identified due to difficulties in direct observation and interpretation of the interfacial reaction mechanism complicated with factors such as night-time chemistry or incomplete surface speciation.23,24 Scientific questions, such as what the reaction mechanism is and what the potential effects on the aerosol growth are during dark aging, remain unanswered.
We developed a unique liquid–vacuum interfacial analysis technique,25,26,27,28,29 namely system for analysis at the liquid–vacuum interface (SALVI), and enabled in situ time-of-flight secondary ion mass spectrometry (ToF-SIMS). We recently demonstrated that in situ liquid SIMS can be used to study the a–l interface in photochemical ultraviolet (UV) aging conditions simulated by the vacuum–liquid interface.30,31 Furthermore, weak interaction formation, such as ion pairs and ion solvation at the interface was monitored by in situ liquid SIMS.32
We explore interfacial chemistry relevant to aqSOA formation in dark aging in this work (Supplementary Fig. 1). We find the following during dark aging: (1) the a–l surface will be more hydrophilic with increased dark-aging time, (2) more cluster ions form at the aqueous surface in dark aging compared with UV aging, and (3) SOA species with higher oxidizing capacity are generated at the aqueous surface in dark aging.
Results
Temporal evolution of dark-aging products
Without UV irradiation, glyoxal undergoes oxidation by H2O2 with a rate constant of (1.67 ± 0.80) × 10−4 M−1 s−1.33 Although reactions are slower in dark aging than photochemical aging, carboxylic acids, organic peroxides, and other products are detected (Table 1).34 This finding is confirmed by in situ observations in Supplementary Figs. 2–13. Here normalized intensity is used to illustrate the trend of products formed during dark aging. In Fig. 1, the oxidation products (e.g., formic acid m/z− 45 CHO2−, glyoxylic acid m/z− 73 C2HO3−) are observed to form quickly within the first 2 h of dark reactions.35 Glyoxylic acid (m/z− 73 C2HO3−) forms by oxygen (O) addition to the glyoxal molecule or the Baeyar–Villiger rearrangement of the hydroperoxides generated in subsequent reactions.36,37 The O atom in H2O2 could nucleophilically attack the carbonyl group, and contribute a H atom to the carbonyl group.38 We find that 2-hydroxy-2-hydroperoxyethanal (HHPE m/z− 91 C2H3O4−) is produced following this mechanism in the early dark-aging time (≤2 h). HHPE is difficult to detect using bulk approaches because it is unstable in dilute solutions.39,40 Zhao et al. reported HHP formation in dark reactions of glyoxal and H2O2 in the aqueous phase.11,12 HHPs are recently detected by APCI-MS/MS to study the SOA.41,42 In situ detection of hydroperoxides suggests the oxidizing capacity of the aqueous surface exists even without UV light. The high Henry’s law constant indicates that the HHPs has a high potential to partition into the aqueous phase,43 further facilitating the uptake of VOCs and other trace gases onto the aqueous surface of clouds, fogs, or wet aerosols.
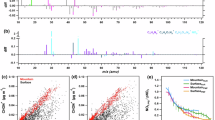
Evolution of different products in dark aging reactions observed in the SIMS negative ion mode: a oxidation products, b oligomers, c cluster ions, and d water clusters. FA formic acid, GA glyoxylic acid, HHPE 2-hydroxy-2-hydroperoxyethanal. Normalization is calculated using total ion intensities of the products observed in this study. The error bars in the plot indicate the standard deviation of three replicates
The sensitive liquid SIMS observation also allows identification of more organic compounds useful for model modification. The temporal profiles of oxidation products are consistent with two-dimensional (2D) images shown in Supplementary Figs. 15a and 16a. As dark aging persists, aldehyde groups and hydroperoxides can undergo interreactions (e.g., peroxyhemiacetal (PHA) formation, carbonyl addition reaction, and hydration) to form high molecular weight oligomers in the acidic aqueous environment.44,45,46 The pH measurements of all samples are listed in Supplementary Table 1. These dark aging samples were acidic. The acidic environment could play an important role in catalyzing surface reactions for aqSOA formation.20 The oligomers (e.g., m/z− 189 C6H5O7−, m/z− 257 C6H9O11−, m/z− 311 C6H15O14−) are postulated to form by PHA reaction of hydroperoxides and carboxylic acids (e.g., m/z 74 C2H2O3, 76 C2H4O3, 110 C2H6O5, m/z 90 C2H2O4, 92 C2H4O4, see Supplementary Table 2), implying that the aqueous surface chemistry could promote the organic peroxide production in dark. Such phenomenon was previously established mainly from the gas phase oxidation, but not in the aqueous phase.47 Our dry SIMS analysis of the same conditions in dark does not show existence of cluster ion or water cluster peaks,31,48 further illustrating the necessity to study the a–l chemistry using in situ approaches.
In addition, cluster ions formed via noncovalent bonding (e.g., hydrogen bond, van der Waals force) are observed at the aqueous surface. We have demonstrated that the Bi3+ primary ion beam was softer at the liquid surface than the solid surface.49,50 The fragmentation and cluster ionization induced by the primary ion beam could be reduced. Supplementary Fig. 2 shows a SIMS spectral comparison of DI water and dark aging samples after 2 and 6 h reactions, confirming that cluster ions are formed as a result of dark aging not ion fragmentation in liquid SIMS. The cluster ions show different evolution trends, for instance, large molecular weight cluster ions (e.g., m/z− 335 [C6H5O15···H2O]−, 387 [C7H13O17···H2O]−) peak around 6 h; and smaller cluster ions (e.g., m/z− 139 [C2HO6···H2O]−, 175 [C2H5O8···H2O]−, 203 [C2H6O5···C2H5O4]−) at 1 h. The appearance of cluster ions (i.e., m/z− 139 C2H3O7−, 175 C2H7O9−, 203 C4H11O9−) correlates with water clusters, glyoxal hydrates (e.g., m/z− 94 C2H6O4−), and several glyoxal derived HHPs (e.g., m/z− 91 C2H3O4−, 110 C2H6O5). Furthermore, a large amount of cluster ions (e.g., m/z+ 263 [C2H7O5···C4H8O6]+, m/z+ 281 [C6H15O11···H2O]+, m/z+ 299 [C6H15O11···2H2O]+, m/z+ 317 [C6H15O11···3H2O]+ etc.) are identified by an addition of m/z 18 (H2O) in the aqueous phase as a result of dark aging (Fig. 1c, Supplementary Figs. 5 and 6). Such cluster ion formation in dark aging indicates that organics have high water adsorption ability at the aqueous surface. These observations suggest that glyoxal oxidation products in dark can evolve via weak intermolecular interactions with either water molecules or other small organic compounds that have hydrophilic functional groups (e.g., –OOH, –OH). Our results substantiate the effect of cluster ion formation from oxygenated molecules on nucleation and aerosol growth.51,52
The sharp decrease of cluster ions and carboxylic acids in Fig. 1 can be explained from the principal component analysis (PCA) results (Fig. 2). The 3 h sample is separated from the 2 h one in the scores plot (Fig. 2a). Corresponding reduction in loadings (Fig. 2b) indicates that the main contributors to positive PC1, including formic acid, glyoxylic acid, small oligomers (e.g., m/z− 121, C2HO6−, 133 C4H5O5−, 151 C4H7O6−), and small cluster ions (e.g., m/z− 175 C2H7O9−, 203 C4H11O9−, 265 C4H9O13−), have changed to different ones in the negative PC1, including large oligomers and large cluster ions (e.g., m/z− 335 C6H17O16−, 387 C7H15O18−). Water clusters reach their highest intensities at different dark-aging times. For example, the small water cluster (m/z− 161 (H2O)8OH−) reaches its maximum at 3 h, while the larger ones do not show an obvious trend (Supplementary Fig. 21b). Water is an indispensable media in most nucleation processes in the atmosphere, however the effect of cluster formation involving water molecules on particle or cloud formation is still unclear.53 Our direct observation of water cluster and cluster ion formation during dark aging enhances the understanding of the role of water molecules in aqSOA formation at the atmospheric aqueous surface including fog, cloud, or aqueous aerosols.
SIMS spectral PCA of dark aging reactions in the negative ion mode. a PC1 vs. PC3 score plots, b PC1 and PC3 loading plots in the m/z− range of 1–300 and c 301–500. Red color bars depict the location of water clusters, green carboxylic acids, blue oligomers, cyan cluster ions, black color corresponds to glyoxal ([C2H2O2]−)
Principal component analysis (PCA) of dark aging
ToF-SIMS spectral PCA is performed to investigate if there is a time dependence on glyoxal dark aging. The PC1 (67.58%) vs. PC3 (6.32%) scores plot in the negative ion mode is interesting (Fig. 2). The initial dark process (e.g., 30 min, 1 h, 2 h) is dominated by the formation of oxidation products (e.g., m/z− 45 CHO2−, 73 C2HO3−, 91 C2H3O4−) and supported by high positive PC1 loadings. Glyoxylic acid may be produced in two ways,36,37 and the primary path for glyoxylic acid formation is carbonyl group oxidation based on in situ SIMS results. Consequently, glyoxylic acid reacts with H2O2 to generate formic acid (m/z− 45, CHO2−), a main product from the acidic environment in dark.35
Along with oxidation products, some small oligomers (e.g., m/z− 121 C2HO6−, 133 C4H5O5−, 151 C4H7O6−, 167 C4H7O7−, 185 C4H9O8−) are produced in 1 and 2 h reactions. The small oligomers are mostly related to glyoxal hydration products and first generation products from the reaction of HHPs and species with aldehyde function groups. In the occurrence of HHPs and other reducing compounds (e.g., glyoxal, glyoxylic acid), PHA formation and aldol condensation in the acidic aqueous environment can take place in the continued dark process (e.g., 3 and 4 h), leading to a large amount of organic peroxide formation (e.g., m/z− 303 C7H11O13−, 307 C6H11O14−, 311 C6H15O14−, 317 C6H5O15−, 319 C6H7O15−, 337 C7H13O15−, 353 C7H13O16−). Highly abundant organic peroxides are also found in SOA from previous studies.54,55 More organic peroxides related to glyoxal and H2O2 dark oxidation are identified at the aqueous surface in this work (Supplementary Tables 2 and 3). HHPs contribute to the atmospheric oxidation capacity and the elevated SOA loading in aerosols by peroxy radical (RO2) oxidation.56 It is worth noting that cluster ions (e.g., m/z+ 263 [C2H7O5···C4H8O6]+, 281 [C6H15O11···H2O]+, 299 [C6H15O11···2H2O]+, and 317 [C6H15O11···3H2O]+) are more likely to form in the longer dark time reactions (≥4 h). Cluster ions are also observed in the positive spectral and confirmed by spectral PCA (Supplementary Figs. 19 and 20). Similarly, most of the cluster ions are related to the hydrophilic species at the aqueous surface,57 indicating that the organics can form hydrogen bonding more effectively with water molecules at the surface compared with UV aging. This finding suggests that the SOA at the aqueous surface become more hydrophilic with increased dark-aging time. This was postulated by a recent perspective discussing the role of water in atmospheric chemistry.58 Water plays a vital role in the molecular arrangement at the aqueous surface to advance interfacial reactions.59,60 Additional 2D PCA results are depicted in Supplementary Fig. 17.
Evolution of the mixing state of dark-aging products
Aerosol particles are known to vary from solid, semisolid, and liquid phase under different relative humidity, temperature, and chemical composition conditions.61 Thus the aerosol-mixing state, in terms of hygroscopicity, has been a hot topic to investigate aerosol evolution. The aerosol surface-mixing state can affect the adsorption of water and other hydrophilic compounds (e.g., HNO3, HCl) onto existing particles.62,63 The cluster ions formed at the interface can undergo collisions with ions or particles in the atmosphere, leading to charge–exchange of the aerosol, which in turn affects the aerosol’s ability to act as CCN.64
In this work, 2D and three-dimensional (3D) images are reconstructed to show the spatial distribution of chemical species (Fig. 3 and Supplementary Fig. 21). In the initial dark aging (≤2 h), HHPE and carboxylic acids (e.g., m/z− 45 CHO2−, 73 C2HO3− in Fig. 3a) are likely to be an inhomogeneous mixture. However, they appear well mixed as dark aging continues, indicating the dark aging process facilitates HHPs and carboxylic acids. Organic acids formed from H2O2 oxidation of VOCs account for substantial formation of aqSOA. The evolution of the mixing state of oligomers (e.g., m/z− 309 C6H13O14−, m/z− 329 C6H17O15−, m/z− 369 C6H23O16−) (Fig. 3b, c) in dark aging shows the oligomer growth via PHA formation and aldol condensation catalyzed by acids. Such oligomers are a type of organic peroxides, exhibiting their potential importance in water soluble organic compound formation and acting as reservoirs of odd hydrogen radicals (e.g., OH, HO2).65
Normalized 3D images of important peaks in the negative ion mode. a oxidation products m/z− 45 CHO2−, 73 C2HO3−, 91 C2H3O4−, b oligomers I m/z− 217 C4H9O10−, 239 C6H7O10−, 261 C6H13O11−, c oligomers II m/z− 309 C6H13O14−, 329 C6H17O15−, 369 C7H13O17−, d cluster ions m/z− 103 C3H3O4−, 175 C2H7O9−, 265 C4H9O13−, and e water clusters m/z− 215 (H2O)11OH−, 287 (H2O)15OH−, 359 (H2O)19OH−. The brightness of color dots indicates relative intensity of a product. Lighter color means lower intensity and darker higher intensity
Cluster ions are substantial products in dark aging, including but not limited to m/z− 103 [C2H2O···CHO2]−, 175 [C2HO6···3H2O]−, 265 [C2HO6··· C2H6O6···H2O]−, or m/z+ 319 [C6H15O11···3H2O]+, 353 [C6H15O11···5H2O]+, and 389 [C6H15O11···7H2O]+ (see Supplementary Fig. 21c, d). Furthermore, cluster ions formed with more water molecules have higher relative intensities in the longer dark reactions, implying their increased contribution to surface hygroscopicity of SOA with increased aging time. The concurring increase of water molecules in cluster ions with increased aging time suggests that the formation of cluster ions in dark-aging account for the water molecule uptake of organic peroxides in the heterogeneous surface reactions and have an impact on the cluster formation involving organic peroxides (Supplementary Fig. 21e).
Comparison between UV and dark aging
We find that time enhances the oligomer formation and large water clusters in the glyoxal and H2O2 interfacial photochemistry in our recent work. Here we compare the effect of time on dark and photochemical aging of glyoxal oxidation in the aqSOA formation using spectral PCA (Fig. 4 and Supplementary Figs. 18 and 19). In the negative ion mode, PC1 and PC2 are the main components, explaining 65% of the data. The reactions at different times are well separated by PC1, while the two types of aging conditions (dark vs. photochemical) are isolated by PC2 (Fig. 4). In general, small water clusters ((H2O)nOH−, 1 ≤ n ≤ 13), small oligomers (e.g., m/z− 133 C4H5O5−, 189 C6H5O7−, 261 C6H13O11), and cluster ions (e.g., m/z− 315 C6H19O14−, 333 C6H21O15−, 369 C6H23O16−, 387 C7H15O18−, and 405 C7H17O19−) are more likely to form in dark aging (Supplementary Figs. 19, 22 and 23); while oligomers with relatively higher mass (e.g., m/z− 329 C9H13O13−, 347 C9H15O14−, 365 C12H13O13−, and 383 C12H15O14−) and large water clusters ((H2O)nOH−, 14 ≤ n ≤ 43) are more likely to form in UV aging (Fig. 4). The formation of more cluster ions implies that the aqSOA at the surface in dark aging tends to be more hydrophilic (e.g., m/z+ 261 [C2H5O5···C4H8O6]+, 263 [C2H7O5···C4H8O6]+, 281 [C6H15O11···H2O]+, 299 [C6H15O11···2H2O]+, 317 [C6H15O11···3H2O]+, 335 [C6H15O11···4H2O]+, 353 [C6H15O11···5H2O]+, and m/z+ (353 + 18n) [C6H15O11···(5 + n)H2O]+, 1 ≤ n ≤ 9) shown in Supplementary Fig. 19a. The cluster ion growth with water molecules is stronger in dark aging than in UV aging (e.g., m/z+ 331 [C9H13O12···H2O]+, 349 [C9H13O12···2H2O]+, 367 [C9H13O12···3H2O]+, 385 [C9H13O12···4H2O]+, 403 [C9H13O12···5H2O]+, and m/z+ (403 + 18n) [C6H15O11···(5 + n)H2O]+, 1 ≤ n ≤ 3, Supplementary Fig. 18). The comparison of cluster ions formed between water clusters and organics between UV and dark aging shows that the dark aging is favorable for cluster ion formation in this route than UV. Cluster ions with higher molecular mass are formed by incorporating more water clusters in dark. This finding suggests that the organics in dark aging are more likely to form hydrogen bonding with water molecules than those in UV aging. This phenomenon could be related to different solvation shells at the aqueous surface between dark and UV aging.66 Previous theoretical calculations have shown that the configuration of clusters in the aqueous phase is similar to their gas phase counterparts only for small cluster sizes. The external medium is expected to stabilize ion–water cluster interaction in larger clusters, and it also plays an important role in the structure of the hydrated ion.67 In contrast, water molecules tend to form larger water clusters and they peak between 2 to 4 h in UV aging (Supplementary Fig. 14). This difference indicates that dark aging facilitates the SOA hygroscopic growth at the aqueous surface. The formation of large amount of oligomers (e.g., m/z− 329 C9H13O13−, 347 C9H15O14−, 383 C9H17O15−, 419 C9H19O17−) in UV aging is mainly related to radical reactions, hemiacetal formation, glyoxal self-oligomerization, and hydration driven by the acidic aqueous environment. Few techniques can provide direct chemical information and 2D/3D mapping at the a–l interface. More importantly, our direct a–l imaging results could help elucidate the findings of aerosol hygroscopicity variation in field campaigns.68,69
Selected peak spectral PCA in the negative ion mode comparing UV and dark-aging products after different aging times. a PC1 vs. PC2 score plots, b PC1 and PC2 loading plots in the m/z range of 1–300 and c 301–500. Dark aging samples are shaded in gray. Red color bars depict the location of water clusters, green carboxylic acids, blue oligomers, and cyan cluster ions. The black bar corresponds to glyoxal ([C2H2O2]−)
Discussion
The different product formation in dark and UV aging suggests two distinctive oxidation mechanisms. In dark aging, formation of more cluster ions with more water molecules indicates that weak intermolecular interactions (e.g., hydrogen bonding, van de Waals force) play an important role. In UV aging, the observation of more oligomers suggests that the covalent bonds (e.g., O–O, C–O, C–C) are generated at the aqueous surface due to photochemistry (Fig. 5). When exposed to solar radiation, organic peroxides, and their cluster ions can produce H2O2 and other small organics as weak O–O bonds undergo photolysis easily,12,70 thus initiating chain reactions under UV. Our new findings imply dark aging can act as a source of aqSOA and influence the glyoxal and H2O2 aqueous surface photochemistry after sunrise. Our findings confirm that dissimilar types of products formed in UV and dark-aging processes and they lead to different surface properties and affects the aerosol potential as CCNs. The aqueous surface in dark is more hydrophilic than in UV aging, which could enhance water uptake of aldehydes at the surface after sunset.
Glyoxal and H2O2 could form organic peroxides and other high molecular weight oligomers via oxidation, PHA formation and aldol condensation in dark aging, and contribute to the aqSOA loading. These products ensure dark aging as a source of OH and HO2 radicals and significantly contributes to the global SOA formation.71,72,73 The observation of more organic peroxides in dark aging suggests that more adverse health effects of SOAs on human beings may exist at night time.34,74 Cluster ions formed with more water molecules and organic peroxides in dark reactions indicate that they can serve as a source of peroxides when exposed to UV, providing a new source for glyoxal and H2O2 photochemistry. Furthermore, the observation of more hydrophilic cluster ions suggests enhanced aqSOA hydrophilicity and hygroscopicity at night. Hydrophilicity represents the water uptake ability and influences the growth ability of the aqueous surface. Upon hygroscopic growth, aerosol size and composition change, thus affecting the atmospheric oxidizing capacity, optical properties, and their ability to behave as a CCN, all playing a role in the Earth’s radiative effect. Our results expand the a–l interfacial reaction mechanisms and enhance the interfacial molecular understanding of the evolution of the aqueous surface of atmospheric aerosols.
Methods
SALVI enabled liquid SIMS characterization
Details about the SALVI fabrication and installation were described in our previous papers.25,26,27,28,29 Our unique microfluidic design ensures that water does not spill out from the aperture and high vacuum is maintained during analysis.26,27 After oxidation at different time intervals, the device was assembled on the sample stage and analyzed in a ToF-SIMS V-100 instrument (IONTOF GmbH, Münster, Germany). In order to map the organic compounds, a 25 KeV Bi3+ primary ion beam was used; multiple 2-µm diameter apertures were drilled through the SiN membrane for direct observation of liquid surfaces.25,31,49
Dark and UV-aging experiment
Aqueous solution of glyoxal (5 mM) and hydrogen peroxide (20 mM) were prepared in DI water. Then solutions were injected into the device by a syringe pump. The dark and UV reaction times were 0.5, 1, 2, 3, 4, 6, and 8 h, respectively. Devices filled with glyoxal and hydrogen peroxide mixtures were stored in a petri dish covered by a clean aluminum foil to simulate dark aging (Supplementary Fig. 1). For UV aging, the device was set 10 cm away from a light source.30 As soon as the desired aging time was reached, the device was immediately put into ToF-SIMS for analysis. More detailed information is described in Supplementary Methods.
Data analysis
The SIMS raw data was mass calibrated by following the criteria listed in our previous studies.48,49 After conducting mass calibration, the spectral and 2D image PCA were done to investigate the surface reaction mechanism. In situ liquid SIMS is limited to unit mass resolution due to instrument constraints.49 Detailed dry sample analysis was conducted to ensure confidence in peak identification.30,31 2D and 3D image visualization was also conducted to show the spatial distribution of surface species. Details of data analysis could be found in SI.28
Data availability
The in situ liquid ToF-SIMS data are uploaded to the PNNL portal and available per request.
References
Rossignol, S. et al. Atmospheric photochemistry at a fatty acid-coated air-water interface. Science 353, 699–702 (2016).
Vaida, V. Atmospheric radical chemistry revisited sunlight may directly drive previously unknown organic reactions at environmental surfaces. Science 353, 650–650 (2016).
Donaldson, D. J. & Vaida, V. The influence of organic films at the air-aqueous boundary on atmospheric processes. Chem. Rev. 106, 1445–1461 (2006).
Volkamer, R., Ziemann, P. J. & Molina, M. J. Secondary organic aerosol formation from acetylene (C2H2): seed effect on SOA yields due to organic photochemistry in the aerosol aqueous phase. Atmos. Chem. Phys. 9, 1907–1928 (2009).
Gilardoni, S. et al. Direct observation of aqueous secondary organic aerosol from biomass-burning emissions. Proc. Natl Acad. Sci. USA 113, 10013–10018 (2016).
Herrmann, H. et al. Tropospheric aqueous-phase chemistry: kinetics, mechanisms, and its coupling to a changing gas phase. Chem. Rev. 115, 4259–4334 (2015).
Gacita, M. S., Longo, K. M., Freire, J. L. M., Freitas, S. R. & Martin, S. T. Impact of mixing state and hygroscopicity on CCN activity of biomass burning aerosol in Amazonia. Atmos. Chem. Phys. 17, 2373–2392 (2017).
Taylor, N. F. et al. Hygroscopic growth of water soluble organic carbon isolated from atmospheric aerosol collected at US national parks and Storm Peak Laboratory. Atmos. Chem. Phys. 17, 2555–2571 (2017).
Kampf, C. J. et al. Effective Henry’s law partitioning and the salting constant of glyoxal in aerosols containing sulfate. Environ. Sci. Technol. 47, 4236–4244 (2013).
Volkamer, R. et al. A missing sink for gas‐phase glyoxal in Mexico City: formation of secondary organic aerosol. Geophys. Res. Lett. 34, L19807 (2007).
Zhao, R., Lee, A. K. Y. & Abbatt, J. P. D. Investigation of aqueous-phase photooxidation of glyoxal and methylglyoxal by aerosol chemical ionization mass spectrometry: observation of hydroxyhydroperoxide formation. J. Phys. Chem. A 116, 6253–6263 (2012).
Zhao, R., Lee, A. K. Y., Soong, R., Simpson, A. J. & Abbatt, J. P. D. Formation of aqueous-phase alpha-hydroxyhydroperoxides (alpha-HHP): potential atmospheric impacts. Atmos. Chem. Phys. 13, 5857–5872 (2013).
Tinel, L. et al. Mechanistic insights on the photosensitized chemistry of a fatty acid at the air/water interface. Environ. Sci. Technol. 50, 11041–11048 (2016).
Bernard, F., Ciuraru, R., Boréave, A. & George, C. Photosensitized formation of secondary organic aerosols above the air/water interface. Environ. Sci. Technol. 50, 8678–8686 (2016).
Fu, H. B. et al. Photosensitized production of atmospherically reactive organic compounds at the air/aqueous interface. J. Am. Chem. Soc. 137, 8348–8351 (2015).
Chalbot, M.-C. G. & Kavouras, I. G. Nuclear magnetic resonance spectroscopy for determining the functional content of organic aerosols: a review. Environ. Pollut. 191, 232–249 (2014).
Pratt, K. A., Fiddler, M. N., Shepson, P. B., Carlton, A. G. & Surratt, J. D. Organosulfates in cloud water above the Ozarks’ isoprene source region. Atmos. Environ. 77, 231–238 (2013).
Mazzoleni, L. R. et al. Identification of water-soluble organic carbon in non-urban aerosols using ultrahigh-resolution FT-ICR mass spectrometry: organic anions. Environ. Chem. 9, 285–297 (2012).
Laskin, J. et al. Chemical analysis of complex organic mixtures using reactive nanospray desorption electrospray ionization mass spectrometry. Anal. Chem. 84, 7179–7187 (2012).
Schweitzer, F., Magi, L., Mirabel, P. & George, C. Uptake rate measurements of methanesulfonic acid and glyoxal by aqueous droplets. J. Phys. Chem. A 102, 593–600 (1998).
Jayne, J. T., Worsnop, D. R., Kolb, C. E., Swartz, E. & Davidovits, P. Uptake of gas-phase formaldehyde by aqueous acid surfaces. J. Phys. Chem. 100, 8015–8022 (1996).
Liss, P. S., Liss, P. S. & Duce, R. A. The Sea Surface and Global Change. (Cambridge University Press, UK and New York, NY, USA, 2005).
Li, J. Y. et al. Observational constraints on glyoxal production from isoprene oxidation and its contribution to organic aerosol over the Southeast United States. J. Geophys. Res. Atmos. 121, 9849–9861 (2016).
Knote, C. et al. Simulation of semi-explicit mechanisms of SOA formation from glyoxal in aerosol in a 3-D model. Atmos. Chem. Phys. 14, 6213–6239 (2014).
Yao, J. et al. Two coexisting liquid phases in switchable ionic liquids. Phys. Chem. Chem. Phys. 19, 22627–22632 (2017).
Yang, L., Yu, X. Y., Zhu, Z. H., Thevuthasan, T. & Cowin, J. P. Making a hybrid microfluidic platform compatible for in situ imaging by vacuum-based techniques. J. Vac. Sci. Technol. A 29, 061101 (2011).
Yang, L., Yu, X. Y., Zhu, Z. H., Iedema, M. J. & Cowin, J. P. Probing liquid surfaces under vacuum using SEM and ToF-SIMS. Lab. Chip. 11, 2481–2484 (2011).
Hua, X. et al. Two-dimensional and three-dimensional dynamic imaging of live biofilms in a microchannel by time-of-flight secondary ion mass spectrometry. Biomicrofluidics 9, 031101 (2015).
Yu, J., Zhou, Y., Hua, X., Zhu, Z. & Yu, X.-Y. In situ characterization of hydrated proteins in water by SALVI and ToF-SIMS. J. Vis. Exp. 108, e53708 (2016).
Sui, X. et al. Deciphering the aqueous chemistry of glyoxal oxidation with hydrogen peroxide using molecular imaging. Phys. Chem. Chem. Phys. 19, 20357–20366 (2017).
Sui, X. et al. ToF-SIMS characterization of glyoxal surface oxidation products by hydrogen peroxide: a comparison between dry and liquid samples. Surf. Interface Anal. 50, 927–938 (2018).
Yu, X. Y. et al. Mesoscopic structure facilitates rapid CO2 transport and reactivity in CO2 capture solvents. J. Phys. Chem. Lett. 9, 5765–5771 (2018).
Schöne, L. & Herrmann, H. Kinetic measurements of the reactivity of hydrogen peroxide and ozone towards small atmospherically relevant aldehydes, ketones and organic acids in aqueous solutions. Atmos. Chem. Phys. 14, 4503–4514 (2014).
Carlton, A. G. et al. Atmospheric oxalic acid and SOA production from glyoxal: results of aqueous photooxidation experiments. Atmos. Environ. 41, 7588–7602 (2007).
Lee, A. K. Y., Zhao, R., Gao, S. S., Abbatt, J. P. D. & Aqueous-Phase, O. H. Oxidation of glyoxal: application of a novel analytical approach employing aerosol mass spectrometry and complementary off-line techniques. J. Phys. Chem. A 115, 10517–10526 (2011).
Strukul, G. Transition metal catalysis in the Baeyer–Villiger oxidation of ketones. Angew. Chem. Int. Ed. 37, 1198–1209 (1998).
Ogata, Y. & Sawaki, Y. Kinetics of the Baeyer–Villiger reaction of aromatic ketones with perbenzoic acid. J. Org. Chem. 37, 2953–2957 (1972).
Mucha, M. & Mielke, Z. Photochemistry of the glyoxal-hydrogen peroxide complexes in solid argon: Formation of 2-hydroxy-2-hydroperoxyethanal. Chem. Phys. Lett. 482, 87–92 (2009).
Gäb, S. et al. Formation of alkyl and hydroxyalkyl hydroperoxides on ozonolysis in water and in air. Atmos. Environ. 29, 2401–2407 (1995).
Wang, H., Huang, D., Zhang, X., Zhao, Y. & Chen, Z. Understanding the aqueous phase ozonolysis of isoprene: distinct product distribution and mechanism from the gas phase reaction. Atmos. Chem. Phys. 12, 7187–7198 (2012).
Zhao, Z. X., Xu, Q., Yang, X. Y. & Zhang, H. F. Heterogeneous ozonolysis of endocyclic unsaturated organic aerosol proxies: implications for criegee intermediate dynamics and later-generation reactions. ACS Earth Space Chem. 3, 344–356 (2019).
Zhou, S., Rivera-Rios, J. C., Keutsch, F. N. & Abbatt, J. P. Identification of organic hydroperoxides and peroxy acids using atmospheric pressure chemical ionization–tandem mass spectrometry (APCI-MS/MS): application to secondary organic aerosol. Atmos. Meas. Tech. 11, 3081–3089 (2018).
O’Sullivan, D. W., Lee, M., Noone, B. C. & Heikes, B. G. Henry’s law constant determinations for hydrogen peroxide, methyl hydroperoxide, hydroxymethyl hydroperoxide, ethyl hydroperoxide, and peroxyacetic acid. J. Phys. Chem. 100, 3241–3247 (1996).
Claeys, M. et al. Formation of secondary organic aerosols from isoprene and its gas-phase oxidation products through reaction with hydrogen peroxide. Atmos. Environ. 38, 4093–4098 (2004).
Hall, W. A. & Johnston, M. V. Oligomer formation pathways in secondary organic aerosol from MS and MS/MS measurements with high mass accuracy and resolving power. J. Am. Soc. Mass Spectrom. 23, 1097–1108 (2012).
Yee, L. D. et al. Secondary organic aerosol formation from low-NO x photooxidation of dodecane: Evolution of multigeneration gas-phase chemistry and aerosol composition. J. Phys. Chem. A 116, 6211–6230 (2012).
Lim, Y. B. & Turpin, B. J. Laboratory evidence of organic peroxide and peroxyhemiacetal formation in the aqueous phase and implications for aqueous OH. Atmos. Chem. Phys. 15, 12867–12877 (2015).
Fu, Y. et al. Does interfacial photochemistry play a role in the photolysis of pyruvic acid in water? Atmos. Environ. 191, 36–45 (2018).
Zhou, Y. F. et al. Improving the Molecular Ion Signal Intensity for In Situ Liquid SIMS Analysis. J. Am. Soc. Mass Spectrom. 27, 2006–2013 (2016).
Yu, X. et al. An investigation of the beam damage effect on in situ liquid secondary ion mass spectrometry analysis. Rapid Commun. Mass. Sp. 31, 2035–2042 (2017).
Riccobono, F. et al. Oxidation products of biogenic emissions contribute to nucleation of atmospheric particles. Science 344, 717–721 (2014).
Kirkby, J. et al. Ion-induced nucleation of pure biogenic particles. Nature 533, 521–526 (2016).
Bourgalais, J. et al. Low temperature kinetics of the first steps of water cluster formation. Phys. Rev. Lett. 116, 113401 (2016).
Epstein, S. A., Blair, S. L. & Nizkorodov, S. A. Direct photolysis of a-pinene ozonolysis secondary organic aerosol: effect on particle mass and peroxide content. Environ. Sci. Technol. 48, 11251–11258 (2014).
Mertes, P., Pfaffenberger, L., Dommen, J., Kalberer, M. & Baltensperger, U. Development of a sensitive long path absorption photometer to quantify peroxides in aerosol particles (Peroxide-LOPAP). Atmos. Meas. Tech. 5, 2339–2348 (2012).
Krechmer, J. E. et al. Formation of low volatility organic compounds and secondary organic aerosol from isoprene hydroxyhydroperoxide low-NO oxidation. Environ. Sci. Technol. 49, 10330–10339 (2015).
Pivokonsky, M. et al. The impact of interactions between algal organic matter and humic substances on coagulation. Water Res. 84, 278–285 (2015).
Vaida, V. Perspective: water cluster mediated atmospheric chemistry. J. Chem. Phys. 135, 020901 (2011).
Gerber, R. B. et al. Computational studies of atmospherically-relevant chemical reactions in water clusters and on liquid water and ice surfaces. Acc. Chem. Res. 48, 399–406 (2015).
Garczarek, F. & Gerwert, K. Functional waters in intraprotein proton transfer monitored by FTIR difference spectroscopy. Nature 439, 109–112 (2006).
Liu, Y. C. et al. Submicrometer particles are in the liquid state during heavy haze episodes in the urban atmosphere of Beijing, China. Environ. Sci. Technol. Lett. 4, 427–432 (2017).
Jiang, R. X. et al. Comparison of aerosol hygroscopicity and mixing state between winter and summer seasons in Pearl River Delta region, China. Atmos. Res. 169, 160–170 (2016).
Pan, X. et al. Real-time observational evidence of changing Asian dust morphology with the mixing of heavy anthropogenic pollution. Sci. Rep. 7, 335 (2017).
Harrison, R. G. Cloud formation and the possible significance of charge for atmospheric condensation and ice nuclei. Space Sci. Rev. 94, 381–396 (2000).
Hua, W. et al. Atmospheric hydrogen peroxide and organic hydroperoxides during PRIDE-PRD'06, China: their concentration, formation mechanism and contribution to secondary aerosols. Atmos. Chem. Phys. 8, 6755–6773 (2008).
Petersen, M. K., Iyengar, S. S., Day, T. J. F. & Voth, G. A. The hydrated proton at the water liquid/vapor interface. J. Phys. Chem. B 108, 14804–14806 (2004).
Merchant, S., Dixit, P. D., Dean, K. R. & Asthagiri, D. Ion-water clusters, bulk medium effects, and ion hydration. J. Chem. Phys. 135, 054505 (2011).
Yan, Y. et al. Hygroscopic behavior of water-soluble matter in marine aerosols over the East China Sea. Sci. Total Environ. 578, 307–316 (2017).
Zhang, C. et al. Insights into the formation of secondary organic carbon in the summertime in urban Shanghai. J. Environ. Sci. 72, 118–132 (2018).
Badali, K. M. et al. Formation of hydroxyl radicals from photolysis of secondary organic aerosol material. Atmos. Chem. Phys. 15, 7831–7840 (2015).
Monod, A. et al. Photooxidation of methylhydroperoxide and ethylhydroperoxide in the aqueous phase under simulated cloud droplet conditions. Atmos. Environ. 41, 2412–2426 (2007).
Kamboures, M. A., Nizkorodov, S. A. & Gerber, R. B. Ultrafast photochemistry of methyl hydroperoxide on ice particles. Proc. Natl Acad. Sci. USA 107, 6600–6604 (2010).
Roehl, C. M., Marka, Z., Fry, J. L. & Wennberg, P. O. Near-UV photolysis cross sections of CH3OOH and HOCH2OOH determined via action spectroscopy. Atmos. Chem. Phys. 7, 713–720 (2007).
Platt, S. M. et al. Two-stroke scooters are a dominant source of air pollution in many cities. Nat. Commun. 5, 3749 (2014).
Acknowledgements
This work was supported by funding from the Pacific Northwest National Laboratory (PNNL) Materials Synthesis and Simulation across Scales (MS3) Initiative Laboratory Directed Research and Development (LDRD) and the Earth and Biological Sciences Directorate (EBSD) Mission Seed LDRD. J.C. was supported by the National Natural Science Foundation of China (no. 21527814), the Ministry of Science and Technology of China (nos. 2016YFC0202700, 2016YFE0112200 2014BAC22B01), Marie Skłodowska-Curie Actions (690958-MARSU-RISE-2015). F.Z. was grateful for the support from China Scholarship Council and PNNL Alternate Sponsored Fellowship. The research was conducted at W. R. Wiley Environmental Molecular Sciences Laboratory (EMSL), a National Science User Facility sponsored by the Office of Biological and Environmental Research (OBER).
Author information
Authors and Affiliations
Contributions
X.Y.Y. designed the project. F.Z. and X.Y. conducted the experiment. F.Z. and X.Y.Y. analysed the data and wrote the paper. F.Z. and Z.Z. did peak identification. All authors discussed the results and commented on the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, F., Yu, X., Chen, J. et al. Dark air–liquid interfacial chemistry of glyoxal and hydrogen peroxide. npj Clim Atmos Sci 2, 28 (2019). https://doi.org/10.1038/s41612-019-0085-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41612-019-0085-5
This article is cited by
-
ToF-SIMS evaluation of PEG-related mass peaks and applications in PEG detection in cosmetic products
Scientific Reports (2024)