Abstract
Hyaluronan (HA) accumulation in clear cell renal cell carcinoma (ccRCC) is associated with poor prognosis; however, its biology and role in tumorigenesis are unknown. RNA sequencing of 48 HA-positive and 48 HA-negative formalin-fixed paraffin-embedded (FFPE) samples was performed to identify differentially expressed genes (DEG). The DEGs were subjected to pathway and gene enrichment analyses. The Cancer Genome Atlas Kidney Renal Clear Cell Carcinoma (TCGA-KIRC) data and DEGs were used for the cluster analysis. In total, 129 DEGs were identified. HA-positive tumors exhibited enhanced expression of genes related to extracellular matrix (ECM) organization and ECM receptor interaction pathways. Gene set enrichment analysis showed that epithelial–mesenchymal transition-associated genes were highly enriched in the HA-positive phenotype. A protein–protein interaction network was constructed, and 17 hub genes were discovered. Heatmap analysis of TCGA-KIRC data identified two prognostic clusters corresponding to HA-positive and HA-negative phenotypes. These clusters were used to verify the expression levels and conduct survival analysis of the hub genes, 11 of which were linked to poor prognosis. These findings enhance our understanding of hyaluronan in ccRCC.
Similar content being viewed by others
Introduction
Global Cancer Statistics showed that approximately 430,000 new cases of kidney cancer (KC) were diagnosed in 2020, accounting for 2.2% of all human malignancies. It contributed to 180,000 deaths worldwide, making it the most lethal urological malignancy worldwide1, and the incidence of KC is increasing2. Renal cell carcinoma (RCC) is the most common type of kidney cancer, accounting for > 90% of primary kidney tumors3. The three most common histological subtypes of RCC are clear cell RCC (ccRCC), papillary RCC, and chromophobe RCC. Recently, more entities with molecularly defined pathogenesis have been identified4. RCC is often diagnosed incidentally, and one-third of the patients present with metastatic disease. Twenty percent of patients who undergo surgery for a primary tumor later develop metastases. Despite recent advances in systemic therapies, the prognosis of metastatic disease remains dismal5,6. Therefore, it is imperative to identify new biomarkers for disease detection, prognostication, and treatment.
Hyaluronan (HA) is a ubiquitous large glycosaminoglycan (GAG) found in the extracellular matrix (ECM), where it forms a pericellular coat surrounding cells and functions as a cushion7. It is composed of a variable number of repeating disaccharide units of N-acetyl-glucosamine (GlcNAc) and glucuronic acid (GlcUA), with an average molecular mass ranging from 1000 to 8000 kD8. The turnover of hyaluronan is rapid, and one-third of the hyaluronan mass undergoes turnover daily9. Hyaluronan is synthesized by the hyaluronan synthase enzymes HAS1, HAS2, and HAS310. Degradation is mediated mainly by the family of hyaluronidases (HYAL 1-4, PH20, CEMIP, and TMEM2)11,12,13. In addition to RCC, increased HA content has been associated with worse outcomes and more aggressive tumor growth in several human malignancies, including breast cancer, colon carcinoma, gastric carcinoma, thyroid cancer, pancreatic cancer, lung adenocarcinoma, lymphoma, hepatocellular carcinomas, and gliomas14,15,16,17,18,19,20,21,22,23. It is postulated that HA acts as a barrier that shields tumor cells from insults, such as therapeutic agents and the immune system, and could serve as a potential target for anticancer drugs24. The use of PEGylated human hyaluronidase (PEGPH20) has shown promising efficacy in sensitizing pancreatic cancer cells to radiotherapy and in improving the efficacy of anti-PD-1 therapy25,26.
In normal human kidneys, most HA is produced in the renal medulla, while the renal cortex, from which renal cell carcinomas usually arise, is hyaluronan poor27. Increased cortical HA content is associated with various non-neoplastic kidney diseases/conditions such as acute kidney injury, chronic kidney disease, allograft, and diabetic nephropathy28,29,30,31. To date, reports on HA in RCCs are limited. In our previous study, we showed that increased cellular hyaluronan conveys poor prognosis in patients with ccRCC14. In addition, a higher hyaluronan content was associated with a higher tumor grade. The individual molecules associated with HA have been studied on gene expression levels. Chi et al.32 showed that expression of HAS1 was increased in RCC tissue compared with adjacent normal tissue while HYAL1 expression was lower in ccRCC than in normal renal tissue32. Cai et al. found that HAS1-3 mRNA expression was higher in human ccRCC tissues than in adjacent normal tissues33. However, only HAS3 protein expression was higher. In conclusion, the results of expression studies are inconsistent, and the expression levels of different HA family proteins might not necessarily reflect overall HA levels. Therefore, to better understand the biological background of hyaluronan accumulation in ccRCC, we performed RNA sequencing of previously found hyaluronan-positive and-negative tumor cohorts. The aim of this study was to investigate the differences in RNA expression profiles and to find new potential hyaluronan-associated molecules.
Materials and methods
Patients and sample selection
A research flowchart of this study is shown in Fig. 1. Formalin-fixed and paraffin-embedded (FFPE) tissue samples from patients who underwent surgery for ccRCC in the period 2000–2013 at Kuopio University Hospital were collected from the Biobank of Eastern Finland. The study (Hyaluronan in Renal Cell Carcinoma, HARCC) was approved by the Ethics Committee of the Northern Savo Hospital District (379/2016, November 1st, 2016). The diagnostic samples were processed and diagnosed according to the routine protocol in the Department of Clinical Pathology. Hyaluronan staining and evaluation were performed as described by Jokelainen et al.14. We selected 48 hyaluronan-positive and 48 hyaluronan-negative tumor samples for RNA sequencing on the basis of tumor grade, sarcomatoid change, sex, survival, and metastasis status (Table 1). Three 1-mm-wide tissue cores were punched from each representative tumor block.
Research flowchart. The dashed line represents in silico analysis using the Cancer Genome Atlas Kidney Renal Clear Cell Carcinoma (TCGA-KIRC) data. HA Hyaluronan, ccRCC clear cell renal cell carcinoma, DEG differentially expressed gene, GO Gene Ontology, KEGG Kyoto Encyclopedia of Genes and Genomes, GSEA Gene Set Enrichment Analysis, STRING Search Tool for the Retrieval of Interacting Genes/Proteins, TRRUST Transcriptional Regulatory Relationships Unraveled by Sentence-based Text mining, NOJAH NOt Just Another Heatmap.
Next-generation sequencing
RNA was isolated by use of an RNeasy FFPE Kit (Qiagen), and deparaffinization was performed using 640–750 µL deparaffinization solution from the kit with 60–90 min incubation at 56 °C. RNA was eluted with 2′ 14 µL of RNase-free water. rRNA was removed by use of a QIASeq FastSelect (rRNA HMR, Qiagen), using 1 µg RNA or as much as could be handled by the kit (max 10 µL). RNA-sequencing libraries were constructed with a TruSeq Stranded mRNA Library Prep kit (Illumina) and 0.3 µL adapters with 30 PCR cycles. Barcodes were optimized by use of BARCOSEL software (http://ekhidna2.biocenter.helsinki.fi/barcosel/)34. Sequencing was performed using an Illumina Novaseq 6000 instrument. Adapter sequences, low-quality bases (q = 25), and short sequences (m = 30) were first trimmed using cutadapt (v.4.1)35 (https://cutadapt.readthedocs.io/en/stable/). The fourteenth (p14) patch release for the GRCh38 reference assembly and annotation was downloaded from https://ftp.ncbi.nlm.nih.gov/genomes/all/GCF/000/001/405/GCF_000001405.40_GRCh38.p14/. The STAR aligner (v2.7.9a_2021-06-25) (https://code.google.com/archive/p/rna-star/) with default parameters was used to map reads against the reference sequence36. Sorting and indexing of the alignment files were performed using Samtools (v.1.10) (https://www.htslib.org/)37.
Quality control and differentially expressed gene analysis
A significant portion of the mapped reads was concentrated on only a few genes in each sample, which necessitated an additional filtering step using Samtools (v. 1.16.1) to mark duplicated reads for removal using the no-multi-dup option. Gene counts were computed by use of R (v.4.1.1) (https://www.r-project.org/) and Rsubread (v.2.6.4) (https://bioconductor.org/packages/release/bioc/html/Rsubread.html) with multi-mapping and multi-overlapping reads removed38,39. In addition to the read-level quality control (QC) detailed in section "next-generation sequencing", further QC steps were performed according to the recommendations specified by Liu et al.40 for FFPE RNA-seq count data40. As FFPE samples were used, the total number of mapped reads in all samples was generally low, as expected. Therefore, the quality metric used for filtering the samples was the median sample-sample Spearman correlation after count normalization. Samples with a median correlation below 0.75 were removed, leaving 36 hyaluronan-positive and 41 hyaluronan-negative samples. Differential expression analysis was performed using edgeR (v.3.34.1) (https://bioconductor.org/packages/release/bioc/html/edgeR.html)41. Only protein-coding genes and genes with mean counts above 1 across all samples were included, leaving 10,633 genes for consideration. Count normalization was performed using the trimmed mean M-value method in edgeR. The results we plotted using the ggplot2 package (v.3.4.2) (https://ggplot2.tidyverse.org)42.
Gene Ontology and pathway enrichment analysis
Gene Ontology (GO) is a bioinformatics database established to provide simple annotation of gene products43,44. GO terms include biological processes (BP), cellular components (CC), and molecular functions (MF) of gene products. The Kyoto Encyclopedia of Genes and Genomes (KEGG) and REACTOME are free online databases containing information on biological pathways, molecular interactions, and reactions45,46,47,48. Database analysis was performed to investigate the molecular function of the identified DEGs using ToppGene (https://toppgene.cchmc.org/enrichment.jsp)49. ToppGene is a bioinformatics portal for gene-list enrichment analysis. The cutoff value for the false discovery rate (FDR) was set at p < 0.05. The Benjamini–Hochberg procedure was used to account for multiple testing. Results were plotted by SRplot (https://www.bioinformatics.com.cn/srplot), an online platform for data analysis and visualization50.
Gene set enrichment analysis
Gene Set Enrichment Analysis (GSEA) was performed to examine the gene expression profiles of hyaluronan-positive and hyaluronan-negative samples which had passed quality control steps51,52. The hyaluronan-positive phenotype was compared with the hyaluronan-negative phenotype. The trimmed mean of M values (TMM)-normalized count data and phenotype data were uploaded to GSEA software (build v.4.3.2.) (https://www.gsea-msigdb.org/gsea/index.jsp) and Human MSigDB h.all.v2023.1.Hs.symbols hallmark gene set was chosen. The number of permutations was set to 1000. All other run parameters were maintained at their default values.
Protein-to-protein network construction and subnetworks
The Search Tool for the Retrieval of Interacting Genes/Proteins (STRING) is a biological database of known and predicted protein–protein interactions (PPI) (https://string-db.org/)53. This includes the interactions derived from experiments and computationally predicted interactions. STRING (v.11.5) was used to predict interactions between the DEGs. Interactions with a combined score > 0.4 were considered statistically significant. The PPI network provided by STRING was imported into and visualized in Cytoscape (v.3.9.1.) (http://www.cytoscape.org)54. The Cytoscape plug-in MCODE (v.2.0.2) was used to identify highly interconnected regions in the network55. Settings used were as follows: Cluster Finding Haircut, Node Score Cutoff = 0.2, K-Core = 2, Max. Depth = 100.
Hub genes discovery and analysis
Hubgene analysis was performed using the CytoHubba (v.0.1) plug-in in Cytoscape56. Seven common algorithms (MCC, MNC, Degree, Closeness, Radiality, Stress, and EPC) were used to identify hub genes, and the UpSet intersection plot (by SRPlot) was used to identify common genes. The hub genes were then exported to GeneMANIA (http://www.genemania.org/), a predictor software used to identify other genes related to a set of input genes and internal associations in the gene sets57. Default methods were used to calculate connection weights. Lastly, we used Transcriptional Regulatory Relationships Unraveled by Sentence-based Text Mining (TRRUST) (v.2) to predict transcription factors (TFs) of hub genes (https://www.grnpedia.org/trrust/)58. TFs with adjusted P-value < 0.05 were considered significant. Subsequently, we used the TCGA-KIRC dataset to examine the expression of these TFs59.
In-silico TCGA heatmap and cluster analysis
To test the gene signature identified by DEG analysis with another dataset, the TCGA-KIRC RNA expression dataset was downloaded via the Bioconductor package TCGABiolinks (v.2.26.0) (https://bioconductor.org/packages/release/bioc/html/TCGAbiolinks.html) using R (v.4.2.2) and RStudio (2022.12.0 + 353)38,60,61. Phenotype data concerning tumor grade and patient survival were downloaded from the same source. Data from 537 ccRCC samples were collected after removing duplicates. The count values were TMM-normalized and log2(count + 1)-transformed. Genome-Wide Heatmap (GWH) analysis was performed using the NOJAH (NOt Just Another Heatmap) (v.1) interactive tool (https://github.com/bbisr-shinyapps/NOJAH/)62. A heatmap was plotted, using the set of 129 DEGs. The values selected for hierarchical clustering analysis parameters were the Z-score method for data normalization type, Euclidean for the distance method, and ward.D2 for the clustering method. Tumor phenotype data were combined into a heatmap, using NOJAH. Furthermore, the methylation status of TCGA samples, as described by Ricketts et al.63, was combined with the heatmap63. The observed distribution of tumor grade, patient survival, and methylation cluster within each heatmap cluster was tested using the chi-square test.
TCGA GSEA analysis
GSEA analysis was performed on TCGA-KIRC data using the newly discovered HA-positive and HA-negative phenotypes. TMM-normalized expression and phenotype data were uploaded to GSEA software (build v.4.3.2.) and Human MSigDB h.all.v2023.1.Hs.symbols hallmark gene set was chosen. The number of permutations was set to 1000. All other run parameters were maintained at their default values.
Hub gene expression analysis in silico
The mRNA expression levels of the identified hub genes were investigated, using the TCGA-KIRC dataset. RNA-seq data from 72 healthy renal tissues were downloaded using the TCGAbiolinks package (v.2.26.0) in R (v.4.2.2). TCGA samples identified belonging to “HA-negative” and “HA-positive” gene expression clusters were compared against each other and to normal renal tissue. The expression levels of the hub genes were TPM-normalized and log2-transformed for each cohort. The Mann–Whitney U-test was used to compare each group with the other two groups, and the expression levels were box-plotted using R package ggpubr (v.0.6.0) (https://rpkgs.datanovia.com/ggpubr/)64.
Prognostic value of hub genes
We used the Kaplan–Meier Plotter online database (http://kmplot.com/analysis/) containing TCGA-KIRC data from 530 patients to analyze the prognosis of hub genes65. Kaplan–Meier estimators were plotted, and hazard ratios were calculated for overall survival (OS) and disease-free survival (DFS). The samples were stratified into low- and high-expression groups based on the median cut-off (50%). Statistical significance was set at p < 0.05 and HR > 1.0 were considered significant.
Ethical considerations
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of the Northern Savo Hospital District (379/2016, November 1st, 2016).
Results
DEG identification
DEG analysis identified 129 differentially expressed genes between the hyaluronan-positive and hyaluronan-negative groups (Fig. 2). The full DEG list can be found in Supplementary Table S1. Of these genes, 97 were upregulated and 32 were downregulated in the hyaluronan-positive group compared with those in the hyaluronan-negative group. Only protein-coding genes were included, and the FDR was set at < 0.05. There were 53 genes with |log2 fold-change|≥ 1.
Functional analysis of DEGs
The ToppGene portal was used to perform GO, KEGG, and REACTOME enrichment analyses on the biological functions and pathways of the 129 DEGs identified. Gene ontology (GO) analysis revealed that these genes were enriched in 82 biological processes. Of these “tube development”, “cell adhesion”, and “extracellular matrix organization” were the most statistically significant. Of the 31 statistically significant cellular compartments (CC), “the external encapsulating structure”, “extracellular matrix”, and “collagen-containing extracellular matrix” were the most enriched. The most enriched molecular functions (N = 21) were “signaling receptor binding”, “peptidase inhibitor activity”, and “endopeptidase inhibitor activity” (Fig. 3). In terms of KEGG enrichment analysis, three pathways, “ECM receptor interaction” (FDR = 3.07E − 4), “glycosaminoglycan biosynthesis chondroitin sulfate” (FDR = 1.72E − 3) and “complement and coagulation cascades” (FDR = 4.0E − 2), were statistically significantly enriched. In the REACTOME pathway analysis, the top three enriched pathways were “extracellular matrix organization” (FDR = 1.36E − 8), “collagen formation” (p = 7.68E − 6), and “regulation of insulin-like growth factor transport and uptake by insulin-like growth factor-binding proteins” (7.77E − 6) (Fig. 3).
(A) ToppGene results of Gene Ontology (GO) and pathway analyses. The bars show the top three most significantly enriched GO terms from each subontology. (B) Bubble plot showing the 15 most significantly enriched pathways in the KEGG and REACTOME pathways. BP biological process, CC cellular compartment, MF molecular function, KEGG Kyoto Encyclopedia of Genes and Genomes.
Gene set enrichment analysis of DEGs
GSEA analysis showed that 8 gene sets were significantly enriched in the hyaluronan-positive phenotype at FDR < 0.25 and nominal p-value < 0.05. Four gene sets were significantly enriched at FDR < 0.25 and nominal p-value < 0.01. No gene sets were significantly enriched in the hyaluronan-negative phenotype. The pathways with the highest normalized enrichment scores (NES) in the hyaluronan-positive phenotype were epithelial-mesenchymal transition (NES 1.72, FDR = 0.034), coagulation (NES 1.67, FDR = 0.033), P53 pathway (NES 1.57, FDR = 0.075), apoptosis (NES 1.48, FDR = 0.153), MTORC1 signaling (NES 1.48, FDR = 0.127), apical surface (NES 1.46, FDR = 0.126), apical junction (NES 1.45, FDR = 0.125), and KRAS signaling up (NES 1.36, FDR = 0.166). The enrichment plots are shown in Fig. 4.
Protein-to-protein interaction network and subnetworks
A PPI network was constructed from 129 DEGs, using a minimum interaction score of 0.4. The network contained 127 nodes and 172 edges (PPI enrichment, p < 1.0E − 16). Fifty of the nodes were singletons with no connection to other nodes. The PPI network is shown in Supplementary Figure S1. The MCODE plug-in identified five closely connected subnetworks from the PPI network; the highly connected regions are shown in Supplementary Table S2. These networks contained 27 unique genes. ToppGene GO revealed that these genes were mostly associated with the molecular functions “endopeptidase inhibitor activity” and “peptidase inhibitor activity”, the biological processes “locomotion and cartilage development”, and the cellular components “extracellular matrix and external encapsulating structure”. The most enriched REACTOME pathway was “extracellular matrix organization” and the most enriched KEGG pathway was “glycosaminoglycan biosynthesis-chondroitin sulfate”.
Hub gene discovery and analysis
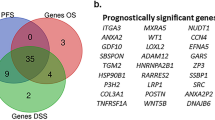
Using seven ranking algorithms of Cytoscape’s CytoHubba plug-in, we calculated the top 20 hub genes (Supplementary Table S3). The intersection of these results revealed 17 common hub genes: ANXA2, CD44, COL6A3, DCN, ENO2, GAPDH, HSP90B1, LOX, LRP1, MMP7, NCAM1, P4HB, SERPINE1, SERPINH1, SPP1, TGFBI, and TIMP1 (Fig. 5A). The full names and functions of the genes are listed in Table 2. All the common hub genes were overexpressed in HA-positive tumors compared with HA-negative tumors. The most enriched GO ontologies, as well as the KEGG and REACTOME pathways, did not differ significantly from those of the DEGs (Supplementary Dataset).
Next, we analyzed the common hub genes using the GeneMANIA database. The input gene list generated a composite network, with network weights for individual subnetworks of 51.05% for co-expression, 42.77% for physical interactions, 3.51% for colocalization, 2.64% for predicted, and 0.04% for pathways (Fig. 5B). GO functional enrichment analysis showed that nine of the 20 co-expressed genes were enriched in the biological processes of “extracellular matrix organization”, “extracellular structure organization”, and “external encapsulating structure organization”. Furthermore, 11/20 genes were involved in the REACTOME “extracellular matrix organization” pathway and 8/20 in “collagen formation” (Supplementary Dataset).
Finally, TFs that regulate hub genes were predicted using the TRRUST database. Eleven candidate regulators were identified, including CEBPA, CTNNB1, HDAC1, HIF1A, JUN, NFKB1, RELA, RUNX1, SP1, STAT3 and TWIST1 (Supplementary Table S4). We used publicly available TCGA-KIRC data to analyze the expression levels of these TFs in normal renal tissue as well as in inferred “HA-negative” and “HA-positive” phenotype TCGA samples (Supplementary Fig. S2). The analysis showed that TWIST1, RUNX1, CEBPA, and RELA were upregulated in carcinoma samples compared with normal renal tissues, whereas HIF1A was downregulated. The HA-positive phenotype had statistically higher expression of TWIST1, RUNX1, and CEBPA than did the HA-negative phenotype. Conversely, CTNNB1, HDAC1, RELA1, NFKB1, and STAT3 were downregulated in the HA-positive phenotype.
Signatures of differentially expressed genes identify prognostic groups in the TCGA-KIRC cohort
To explore our findings, we subjected TCGA-KIRC data to heatmapping and cluster analysis using the previously identified DEG set. The NOJAH tool was used for analysis. Heatmap plotting and cluster analysis revealed three distinct groups (Fig. 6A). A total of 151 samples had a gene expression profile similar to that of the HA-positive sequencing group, hereafter referred to as the HA-positive cluster. A total of 221 samples had a gene expression profile similar to that of the hyaluronan-negative sequencing group, hereafter referred to as the HA-negative cluster. The remaining samples (n = 165) were unclassifiable, based on our set of DEGs. Information about survival status, tumor pathological stage and tumor grade were available for each sample, and the hyaluronan-positive cluster was associated with death (Pearson chi-squared test, p < 0.001), higher tumor grade (Pearson chi-squared test, p < 0.001), higher stage (Pearson chi-squared test, p < 0.001) and methylation cluster 1 (Pearson chi-squared test p < 0.001). Grade 4 tumors tended to cluster in the HA-positive cluster. Comparison of the clinical characteristics between discovery set and TCGA clusters are shown in Supplementary Table S5. The Kaplan–Meier estimator showed worse prognosis for the HA-positive cluster in the TCGA data. There was no significant difference between the unclassifiable and HA-negative clusters (Fig. 6B).
TCGA GSEA analysis
GSEA revealed that 7 gene sets were significantly enriched in the HA-positive cluster at FDR < 0.25 and nominal p-value < 0.05. Two gene sets with similar confidence scores were enriched in the HA-negative cluster. The pathways with the highest positive NES values were IL6 JAK STAT3 signaling (NES 2.01, FDR = 0.066), epithelial-mesenchymal transition (NES 1.98, FDR = 0.047), inflammatory response (NES 1.97, FDR = 0.036), allograft rejection (NES 1.92, FDR 0.045), glycolysis (NES 1.68, FDR = 0.188), coagulation (NES 1.63, FDR = 0.205), and estrogen response late (NES 1.57, FDR = 0.177). The pathways with the lowest NES values were fatty acid metabolism (NES − 1.72, FDR 0.334) and β-catenin signaling (NES − 1.66, FDR 0.240). All pathways are available in the Supplementary Dataset.
Hub gene expression analysis
To analyze the hub gene expression pattern, we examined the relative mRNA expression levels in the TCGA-KIRC data. We compared the expression levels between the two phenotypes of HA-positive and HA-negative clusters that we identified in the heatmapping and cluster analysis. In addition, the mRNA expression levels of normal renal tissues were included in the analysis. The results showed that the following genes were significantly overexpressed in renal cell carcinomas with respect to normal renal tissue: ANXA2, CD44, COL6A3, ENO2, GAPDH, HSP90B1, LOX, LRP1, NCAM1, P4HB, SERPINE1, SERPINH1, TGFBI and TIMP1. SPP1 and DCN were significantly downregulated in carcinomas in comparison with normal renal tissue. All hub genes were overexpressed in the HA-positive cluster, consistent with our sequencing results (Supplementary Fig. S3).
Prognostic value of hub genes
Hub genes were subjected to survival analysis by KMplotter using the TCGA-KIRC data. This indicated that high expression levels of ANXA2, CD44, COL6A3, DCN, ENO2, GAPDH, MMP7, P4HB, SERPINH1, TGFBI, and TIMP1 were associated with unfavorable overall survival. No statistical difference in overall survival was observed with different expression levels of HSP90B1, LOX, LRP1, NCAM1, SERPINE1, and SPP1 (Supplementary Fig. S4 and S5). However, when subjected to disease-free survival (DFS) analysis, only the high expression levels of TGFBI and P4HB were statistically significant (Supplementary Fig. S6 and S7).
Discussion
In this study, we examined RNA expression levels in hyaluronan-positive and-negative tumors. To the best of our knowledge, this is the first study where tumoral hyaluronan accumulation has been studied through RNA sequencing. To date, hyaluronan has been studied in renal cell carcinoma in terms of individual protein and mRNA levels. However, the molecular pathways associated with hyaluronan accumulation have remained unclear.
DEG analysis revealed 129 genes with expression profiles that differed between the HA-positive and HA-negative cohorts. In total, 97 genes were up-regulated in the HA-positive group. These DEGs appeared to regulate pathways involved in the extracellular matrix, collagen formation, carbohydrate metabolism, and cellular adhesion. The most enriched gene ontology terms were related to “peptidase inhibitor activity”, “cell adhesion”, and “collagen-containing extracellular matrix”. GSEA revealed that the DEGs were enriched in similar pathways, such as the extracellular matrix, apical surface, and apical junction. These results are in line with previous findings that hyaluronan accumulation is associated with epithelial-mesenchymal transition, cell adhesion, and extracellular matrix organization7,98,99,100,101.
Among the DEGs, the hyaluronan-associated molecules CD44 and HABP2 were identified. CD44 is a hyaladherin, and HA-CD44 interaction has been shown to promote tumor cell survival and chemoresistance102. HABP2, on the other hand, is a serine protease that promotes migration, extravasation, tumor growth, and metastasis in lung cancer103. This protein also has a peculiar feature in that its expression is increased by low-molecular-weight HA (LMW-HA) and decreased by high-molecular-weight HA (HMW-HA)104. The absence of other hyaluronan-binding proteins from the DEG list, as well as hyaluronan-synthesizing and hyaluronan-degrading enzymes, is an interesting finding. This might reflect the previous observations that transcriptional regulation may not be the main driver of altered HA levels in RCC105. Therefore, alternative mechanisms (e.g., those related to the supply of HA substrates or regulation of translational and enzymatic activity) may explain HA accumulation in these tumors. RCC is a metabolically active disease, and tumors have been shown to increase the uptake and utilization of glucose and produce increased amount of pentose phosphate pathway (PPP) intermediates106. UDP-sugars, a type of PPP intermediate necessary for HA synthesis, have been shown to accumulate in breast carcinomas and strongly correlate with tumor HA levels independent of the mRNA levels of HA synthases107.
KEGG pathway “glycosaminoglycan biosynthesis chondroitin sulfate” and REACTOME pathway “glycosaminoglycan metabolism” were enriched in HA-positive tumors. This was mainly due to the overexpression of CHST11, DCN, CHPF, and CHST3, which are common to both pathways. These genes participate in the sulfation and biosynthesis of chondroitin sulfate (CS) as well as its organization in extracellular matrix108,109. CS has been observed to be elevated in breast cancer stroma, and increased CS levels are associated with poor differentiation status in hepatocellular carcinoma and in advanced stage and recurrent ovarian cancer110,111,112. Furthermore, there is significantly more CS in RCC tissues, and the CS biosynthesis pathway is upregulated in RCC compared with non-neoplastic kidney tissues113,114. The relationship between CS biosynthesis and HA accumulation in ccRCC remains unclear.
Gene set enrichment analysis showed that the mTORC1 signaling gene set was enriched in HA-positive tumors. Mechanistic target of rapamycin (mTOR) is a serine/threonine kinase involved in cellular growth, proliferation, and autophagy. mTOR activation plays a major role in RCC, and mTOR inhibitors have been used to treat metastatic RCC. Regrettably, mTOR inhibitors have low objective response rates, and tumors rapidly develop resistance115. Nevertheless, some patients with mTOR signaling-activating genomic alterations show long-lasting responses116. Therefore, tumors with HA accumulation may respond more favorably to mTOR inhibition.
Hub gene analysis identified 17 highly connected genes. These genes were involved in the same molecular pathways as DEGs, and 11 of them (ANXA2, CD44, COL6A3, DCN, ENO2, GAPDH, MMP7, P4HB, SERPINH1, TGFBI, and TIMP1) were correlated with poor prognosis in renal cell carcinoma at the mRNA level when the median cut-off was used. Many of the proteins encoded by these genes function in cell adhesion (CD44, TGFBI, COL6A3, SPP1, and NCAM1), participate in glycolysis (GAPDH and ENO2), exhibit protease activity (P4HB, HSP90B1, and TIMP1), and act as chaperones (SERPINE1 and SERPINH1) (Table 2). These proteins, besides CD44, have little to no known interaction with hyaluronan. However, LRP1 (a low-density lipoprotein receptor involved in endocytosis and regulation of signaling pathways) has been shown to interact with artificial sulfated hyaluronan in bone regeneration studies117. The protein expression of CD44 has been studied in renal cell carcinoma, and higher expression is associated with poor prognosis118. In addition, high protein expression of ANXA2, ENO2, P4HB, SERPINH1, TGFBI, and LRP1 is associated with poor prognosis in renal cell carcinoma67,89,92,119,120,121. Patraki and Cardille122 showed that MMP7 was more strongly expressed in high-grade RCC. However, no survival analysis was conducted. Furthermore, COL6A3 expression was shown to be higher in ccRCC metastases than in primary tumors70. Not surprisingly, the top 20 related genes identified by co-expression analysis were associated with the same biological processes as the hub genes. Hub genes and their co-expressing genes act as potential targets for downstream analyses.
Transcription factor analysis revealed that 11 genes were associated with our set of hub genes. Of notable interest is RUNX1, which is highly expressed in renal cell carcinoma compared with normal kidney tissue. Furthermore, RUNX1 expression was higher in the HA-positive cluster. RUNX1 has been shown to affect multiple biological processes, such as proliferation, apoptosis and differentiation, and lineage determination. In addition, its involvement as a fusion partner in acute myeloid leukemia (AML) is well-known123. Recently, it has been associated with other malignancies, such as promoting EMT in colorectal carcinoma by activating the Wnt/β-catenin signaling pathway124. RUNX1 has previously been associated with poor prognosis in renal cell carcinoma125. Moreover, Rooney et al.126 showed that deletion of RUNX1 in ccRCC cell lines reduced tumor cell growth and viability. RUNX1 deletion caused many alterations to biological pathways, notably cell adhesion and ECM modelling. Interestingly, the second most altered gene ontology observed was “eph and ephrin signaling”, which is a downstream target of the WNT signaling pathway. One of the genes with the most significantly altered expression was SERPINH1, one of the hub genes identified. Deletion of RUNX1 caused a significant reduction in SERPINH1 levels, indicating that RUNX1 is a positive regulator of SERPINH1. Interestingly, SERPINH1 has been shown to regulate EMT through Wnt/β-catenin signaling in gastric cancer127. The interplay between RUNX1, SERPINH1, and hyaluronan could offer insights into the molecular mechanisms underlying the hyaluronan-induced migratory phenotype and EMT.
To investigate our previous and present findings, we performed in silico heatmap clustering using the list of DEGs and TCGA-KIRC data14. Heatmap clustering identified two prognostically divergent clusters whose gene expression patterns corresponded to those of our cohorts. A gene expression pattern similar to that of our HA-positive sequencing group conveyed a poorer prognosis in the TCGA data. This cluster was enriched in tumors with a higher tumor grade and pathological stage. Additionally, it exhibited a methylation cluster that has previously been shown to be associated with the CpG island methylator (CIMP) phenotype and increased Wnt signaling pathway activity63. Motzer er al.128 molecularly categorized ccRCC into seven subtypes, using an integrated multi-omics approach. Two subtypes (clusters 1 and 6) were enriched with stromal transcriptional signatures, and cluster 6 contained a substantial proportion of sarcomatoid ccRCCs. Interestingly, cluster 1 showed enrichment of WNT signaling genes, in addition to high expression of genes related to the TGF-β, Hedgehog, and NOTCH signaling pathways. Regrettably, TCGA clinical data did not contain information regarding possible sarcomatoid changes. However, it could be deduced that since most of the grade 4 tumors were clustered in the HA-positive cluster, some of these were sarcomatoid. This is in line with our previous finding that HA accumulates in sarcomatoid carcinomas14.
In silico GSEA analysis of TCGA data revealed that genes in the HA-positive cluster were enriched in pathways associated with inflammation. These included “IL6/JAK/STAT3-signaling”, “inflammatory response”, and “allograft rejection”. It has been previously shown that hyaluronan-rich stroma and low-molecular-weight hyaluronan (LMW-HA) promote inflammation and cytokine production129,130. Kainulainen et al.131 showed that upregulation of proinflammatory genes in MV3 melanoma cells stimulated synthesis of a peritumoral HA coat. In addition, an increased number of HA-containing HYAL2+PD-L1+ myeloid-derived suppressor cells (MDSCs) have been observed in ccRCC, promoting HA degradation to LMW-HA, cancer-related inflammation, and immunosuppression132. Furthermore, HYAL2+ myeloid cells have been associated with HA degradation and angiogenesis in bladder cancer133. There is evidence that HA can modulate immune cell infiltration in the tumor microenvironment by binding to, polarizing, and recruiting macrophages. It is also well appreciated that immunity and angiogenesis are closely interlinked134,135. As RCC is one of the most immune-infiltrated tumors, it may be possible to improve the efficacy of cancer treatments, such as immunotherapy, by targeting HA25,136,137.
Our study had some limitations. The study design involving samples collected in the period 2000–2013 and their FFPE nature likely diminished the sensitivity and increased the variation. However, analyzing three punch cores from each tumor should compensate for the random scatter. Due to the inherent degradation of RNA, the count values were generally low, as expected. This phenomenon may have led to limited coverage of the full transcriptomic differences between the groups and posed limitation to the effective cross-validation of the dataset. In addition, the lack of independent known-label datasets with respect to hyaluronan status posed a hindrance, preventing us from leveraging such datasets to validate our results. Finally, owing to the relatively low number of underexpressed genes, GSEA analyses did not necessarily have sufficient statistical power to identify underexpressed gene sets.
Conclusions
Our study demonstrates that hyaluronan accumulation is associated with biological pathways related to the extracellular matrix, EMT, and cell–stroma interactions. The gene expression signature we discovered was associated with poor prognosis and a higher tumor grade in ccRCC. Whether the pathways identified in this study lead to hyaluronan accumulation or whether HA accumulation induces certain genes remains unclear. Identification of these pathways may open new avenues for hyaluronan research in renal cell carcinomas and other human malignancies. Further studies involving independent in silico and wet lab validation sets are required to validate these results.
Data availability
The data that support the findings of this study are available from the Biobank of Eastern Finland, but restrictions apply to the availability of these data, which were used under license for the current study and so are not publicly available. However, the data are available from the authors upon reasonable request and with permission from the Biobank of Eastern Finland. Please contact info@ita-suomenbiopankki.fi for access inquiries.
References
Sung, H. et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249 (2021).
Bukavina, L. et al. Epidemiology of renal cell carcinoma: 2022 update. Eur. Urol. 82, 529–542 (2022).
Hsieh, J. J. et al. Renal cell carcinoma. Nat. Rev. Dis. Primers 3, 66 (2017).
Alaghehbandan, R., Siadat, F. & Trpkov, K. What’s new in the WHO 2022 classification of kidney tumours?. Pathologica 115, 8–22 (2023).
Goodstein, T., Yang, Y., Runcie, K., Srinivasan, R. & Singer, E. A. Two is company, is three a crowd? Triplet therapy, novel molecular targets, and updates on the management of advanced renal cell carcinoma. Curr. Opin. Oncol. 35, 206–217 (2023).
Schiavoni, V. et al. Recent advances in the management of clear cell renal cell carcinoma: Novel biomarkers and targeted therapies. Cancers 15, 3207 (2023).
Garantziotis, S. & Savani, R. C. Hyaluronan biology: A complex balancing act of structure, function, location and context. Matrix Biol. 78–79, 1–10 (2019).
Cowman, M. K., Lee, H. G., Schwertfeger, K. L., McCarthy, J. B. & Turley, E. A. The content and size of hyaluronan in biological fluids and tissues. Front. Immunol. 6, 261 (2015).
Triggs-Raine, B. Biology of hyaluronan: Insights from genetic disorders of hyaluronan metabolism. World J. Biol. Chem. 6, 110 (2015).
Itano, N. et al. Three isoforms of mammalian hyaluronan synthases have distinct enzymatic properties. J. Biol. Chem. 274, 25085–25092 (1999).
Csoka, A. B., Frost, G. I. & Stern, R. The six hyaluronidase-like genes in the human and mouse genomes. Matrix Biol. 20, 499–508 (2001).
Spataro, S. et al. CEMIP (HYBID, KIAA1199): Structure, function and expression in health and disease. FEBS J. 16, 3946–3962 (2022).
Narita, T. et al. TMEM2 is a bona fide hyaluronidase possessing intrinsic catalytic activity. J. Biol. Chem. 299, 105120 (2023).
Jokelainen, O. et al. Cellular hyaluronan is associated with a poor prognosis in renal cell carcinoma. Urol. Oncol. 38(686), e11-686.e22 (2020).
Auvinen, P. et al. Hyaluronan in peritumoral stroma and malignant cells associates with breast cancer spreading and predicts survival. Am. J. Pathol. 156, 529–536 (2000).
Setälä, L. P. et al. Hyaluronan expression in gastric cancer cells is associated with local and nodal spread and reduced survival rate. Br. J. Cancer 79, 1133–1138 (1999).
Böhm, J. et al. Hyaluronan expression in differentiated thyroid carcinoma. J. Pathol. 196, 180–185 (2002).
Tahkola, K. et al. Stromal hyaluronan accumulation is associated with low immune response and poor prognosis in pancreatic cancer. Sci. Rep. 11, 12216 (2021).
Pirinen, R. et al. Prognostic value of hyaluronan expression in non-small-cell lung cancer: Increased stromal expression indicates unfavorable outcome in patients with adenocarcinoma. Int. J. Cancer 95, 12–17 (2001).
Hasselbalch, H., Hovgaard, D., Nissen, N. & Junker, P. Serum hyaluronan is increased in malignant lymphoma. Am. J. Hematol. 50, 231–233 (1995).
Li, J.-H. et al. Over expression of hyaluronan promotes progression of HCC via CD44-mediated pyruvate kinase M2 nuclear translocation. Am. J. Cancer Res. 6, 509–521 (2016).
Park, J. B., Kwak, H. & Lee, S. Role of hyaluronan in glioma invasion. Cell. Adh. Migr. 2, 202–207 (2008).
Ropponen, K. et al. Tumor cell-associated hyaluronan as an unfavorable prognostic factor in colorectal cancer. Cancer Res. 58, 342–347 (1998).
Hua, S. H., Viera, M., Yip, G. W. & Bay, B. H. Theranostic applications of glycosaminoglycans in metastatic renal cell carcinoma. Cancers 15, 266 (2023).
Blair, A. B. et al. Dual stromal targeting sensitizes pancreatic adenocarcinoma for anti-programmed cell death protein 1 therapy. Gastroenterology 163, 1267-1280.e7 (2022).
Seki, T. et al. PEGPH20, a PEGylated human hyaluronidase, induces radiosensitization by reoxygenation in pancreatic cancer xenografts. A molecular imaging study. Neoplasia 30, 100793 (2022).
Wells, A. F. et al. The localization of hyaluronan in normal and rejected human kidneys. Transplantation 50, 240–243 (1990).
Declèves, A. E. et al. Synthesis and fragmentation of hyaluronan in renal ischaemia. Nephrol. Dial. Transplant. 27, 3771–3781 (2012).
Akin, D., Ozmen, S. & Yilmaz, M. E. Kidney diseases: Hyaluronic acid as a new biomarker to differentiate acute kidney injury from chronic kidney disease. Iran. J. Kidney Dis. 11, 409–413 (2017).
Stenvinkel, P. et al. High serum hyaluronan indicates poor survival in renal replacement therapy. Am. J. Kidney Dis. 34, 1083–1088 (1999).
Jones, S., Jones, S. & Phillips, A. O. Regulation of renal proximal tubular epithelial cell hyaluronan generation: Implications for diabetic nephropathy. Kidney Int. 59, 1739–1749 (2001).
Chi, A. et al. Molecular characterization of kidney cancer: Association of hyaluronic acid family with histological subtypes and metastasis. Cancer 118, 2394–2402 (2012).
Cai, J.-L., Li, M. & Na, Y.-Q. Correlation between hyaluronic acid, hyaluronic acid synthase and human renal clear cell carcinoma. Chin. J. Cancer Res. 23, 59–63 (2011).
Somervuo, P. et al. BARCOSEL: A tool for selecting an optimal barcode set for high-throughput sequencing. BMC Bioinform. 19, 257 (2018).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J 17, 10–12 (2011).
Dobin, A. et al. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Li, H. et al. The sequence alignment/map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2022). https://www.R-project.org/.
Liao, Y., Smyth, G. K. & Shi, W. The R package Rsubread is easier, faster, cheaper and better for alignment and quantification of RNA sequencing reads. Nucleic Acids Res. 47, e47 (2019).
Liu, Y. et al. Quality control recommendations for RNASeq using FFPE samples based on pre-sequencing lab metrics and post-sequencing bioinformatics metrics. BMC Med. Genomics 15, 195 (2022).
Robinson, M. D. & Oshlack, A. A scaling normalization method for differential expression analysis of RNA-Seq data. Genome Biol. 11, R25 (2010).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Springer, 2016). https://ggplot2.tidyverse.org.
Ashburner, M. et al. Gene ontology: Tool for the unification of biology. Nat. Genet. 25, 25–29 (2000).
Aleksander, S. A. et al. The Gene Ontology knowledgebase in 2023. Genetics 224, 66 (2023).
Kanehisa, M. Toward understanding the origin and evolution of cellular organisms. Protein Sci. 28, 1947–1951 (2019).
Kanehisa, M. & Goto, S. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 28, 27–30 (2000).
Kanehisa, M., Furumichi, M., Sato, Y., Kawashima, M. & Ishiguro-Watanabe, M. KEGG for taxonomy-based analysis of pathways and genomes. Nucleic Acids Res. 51, D587–D592 (2023).
Gillespie, M. et al. The reactome pathway knowledgebase 2022. Nucleic Acids Res. 50, D687–D692 (2022).
Chen, J., Bardes, E. E., Aronow, B. J. & Jegga, A. G. ToppGene Suite for gene list enrichment analysis and candidate gene prioritization. Nucleic Acids Res. 37, 66 (2009).
Tang, D. et al. SRplot: A free online platform for data visualization and graphing. PLoS ONE 18, 66 (2023).
Subramanian, A. et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. 102, 15545–15550 (2005).
Mootha, V. K. et al. PGC-1α-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat. Genet. 34, 267–273 (2003).
Szklarczyk, D. et al. The STRING database in 2023: Protein–protein association networks and functional enrichment analyses for any sequenced genome of interest. Nucleic Acids Res. 51, D638–D646 (2023).
Shannon, P. et al. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 13, 2498–2504 (2003).
Bader, G. D. & Hogue, C. W. An automated method for finding molecular complexes in large protein interaction networks. BMC Bioinform. 4, 2 (2003).
Chin, C. H. et al. cytoHubba: Identifying hub objects and sub-networks from complex interactome. BMC Syst. Biol. 8, 66 (2014).
Warde-Farley, D. et al. The GeneMANIA prediction server: Biological network integration for gene prioritization and predicting gene function. Nucleic Acids Res. 38, 66 (2010).
Han, H. et al. TRRUST v2: An expanded reference database of human and mouse transcriptional regulatory interactions. Nucleic Acids Res. 46, D380–D386 (2018).
Creighton, C. J. et al. Comprehensivemolecular characterization of clear cell renal cell carcinoma. Nature 499, 43–49 (2013).
Colaprico, A. et al. TCGAbiolinks: An R/Bioconductor package for integrative analysis of TCGA data. Nucleic Acids Res. 44, e71 (2016).
RStudio Team. RStudio: Integrated Development Environment for R (RStudio, PBC, 2020). http://www.rstudio.com/.
Rupji, M., Dwivedi, B. & Kowalski, J. Nojah: Not just another heatmap for genome-wide cluster analysis. PLoS ONE 14, e0204542 (2019).
Ricketts, C. J. et al. The cancer genome atlas comprehensive molecular characterization of renal cell carcinoma. Cell Rep. 23, 313-326.e5 (2018).
Kassambara, A. ggpubr: 'ggplot2' Based Publication Ready Plots. R package version 0.6.0 (2023). https://rpkgs.datanovia.com/ggpubr/.
Nagy, Á., Munkácsy, G. & Győrffy, B. Pancancer survival analysis of cancer hallmark genes. Sci. Rep. 11, 6047 (2021).
Bharadwaj, A., Bydoun, M., Holloway, R. & Waisman, D. Annexin A2 heterotetramer: Structure and function. Int. J. Mol. Sci. 14, 6259–6305 (2013).
Yang, S. F. et al. Annexin A2 in renal cell carcinoma: Expression, function, and prognostic significance. Urol. Oncol. 33(22), e11-22.e21 (2015).
Mesrati, M. H., Syafruddin, S. E., Mohtar, M. A. & Syahir, A. CD44: A multifunctional mediator of cancer progression. Biomolecules 11, 1850 (2021).
Cheng, B. et al. Cancer stem cell markers predict a poor prognosis in renal cell carcinoma: A meta-analysis oncotarget. Oncotarget 7, 65862–65875 (2016).
Zhong, T. et al. Key genes associated with prognosis and metastasis of clear cell renal cell carcinoma. PeerJ 10, e12493 (2022).
Wang, J. & Pan, W. The biological role of the collagen alpha-3 (Vi) chain and its cleaved c5 domain fragment endotrophin in cancer. Onco Targets Ther. 13, 5779–5793 (2020).
Neill, T., Schaefer, L. & Iozzo, R. V. Decorin: A guardian from the matrix. Am. J. Pathol. 181, 380–387 (2012).
Xu, Y. et al. DCN deficiency promotes renal cell carcinoma growth and metastasis through downregulation of P21 and E-cadherin. Tumor Biol. 37, 5171–5183 (2016).
Pan, J., Jin, Y., Xu, X., Wei, W. & Pan, H. Integrated analysis of the role of Enolase 2 in clear cell renal cell carcinoma. Dis. Markers 2022, 6539203 (2022).
Huppertz, I. et al. Riboregulation of enolase 1 activity controls glycolysis and embryonic stem cell differentiation. Mol. Cell 82, 2666-2680.e11 (2022).
Nicholls, C., Li, H. & Liu, J. P. GAPDH: A common enzyme with uncommon functions. Clin. Exp. Pharmacol. Physiol. 39, 674–679 (2012).
Li, C. X., Chen, J., Xu, Z. G., Yiu, W. K. & Lin, Y. T. The expression and prognostic value of RNA binding proteins in clear cell renal cell carcinoma. Transl. Cancer Res. 9, 7415–7431 (2020).
Wang, Y. & Wang, X. A pan-cancer analysis of heat-shock protein 90 Beta1(HSP90B1) in human tumours. Biomolecules 12, 66 (2022).
Añazco, C. et al. Dysregulation of lysyl oxidases expression in diabetic nephropathy and renal cell carcinoma. Curr. Drug Targets 22, 1916–1925 (2021).
Di Stefano, V. et al. Major action of endogenous lysyl oxidase in clear cell renal cell carcinoma progression and collagen stiffness revealed by primary cell cultures. Am. J. Pathol. 186, 2473–2485 (2016).
Lin, S. et al. Comprehensive analysis on the expression levels and prognostic values of LOX family genes in kidney renal clear cell carcinoma. Cancer Med. 9, 8624–8638 (2020).
Xing, P. et al. Roles of low-density lipoprotein receptor-related protein 1 in tumors. Chin. J. Cancer 35, 6 (2016).
Ahluwalia, P. et al. Prognostic and therapeutic implications of extracellular matrix associated gene signature in renal clear cell carcinoma. Sci. Rep. 11, 7561 (2021).
Liao, H. Y., Da, C. M., Liao, B. & Zhang, H. H. Roles of matrix metalloproteinase-7 (MMP-7) in cancer. Clin. Biochem. 92, 9–18 (2021).
Meng, N. et al. A comprehensive pan-cancer analysis of the tumorigenic role of matrix metallopeptidase 7 (MMP7) across human cancers. Front. Oncol. 12, 916907 (2022).
Van Acker, H. H., Capsomidis, A., Smits, E. L. & Van Tendeloo, V. F. CD56 in the immune system: More than a marker for cytotoxicity?. Front. Immunol. 8, 892 (2017).
Daniel, L. et al. Neural cell adhesion molecule expression in renal cell carcinomas: Relation to metastatic behavior. Hum. Pathol. 34, 528–532 (2003).
Parakh, S. & Atkin, J. D. Novel roles for protein disulphide isomerase in disease states: A double edged sword?. Front. Cell Dev. Biol. 3, 30 (2015).
Zhu, Z. et al. Overexpression of P4HB is correlated with poor prognosis in human clear cell renal cell carcinoma. Cancer Biomark. 26, 431–439 (2019).
Li, S. et al. Plasminogen activator inhibitor-1 in cancer research. Biomed. Pharmacother. 105, 83–94 (2018).
Duarte, B. D. P. & Bonatto, D. The heat shock protein 47 as a potential biomarker and a therapeutic agent in cancer research. J. Cancer Res. Clin. Oncol. 144, 2319–2328 (2018).
Qi, Y. et al. SERPINH1 overexpression in clear cell renal cell carcinoma: association with poor clinical outcome and its potential as a novel prognostic marker. J. Cell. Mol. Med. 22, 1224–1235 (2018).
Zhao, K., Ma, Z. & Zhang, W. Comprehensive analysis to identify SPP1 as a prognostic biomarker in cervical cancer. Front. Genet. 12, 732822 (2022).
Rabjerg, M. et al. Molecular characterization of clear cell renal cell carcinoma identifies CSNK2A1, SPP1 and DEFB1 as promising novel prognostic markers. APMIS 124, 372–383 (2016).
Shou, Y. et al. TIMP1 indicates poor prognosis of renal cell carcinoma and accelerates tumorigenesis via EMT signaling pathway. Front. Genet. 13, 648134 (2022).
Corona, A. & Blobe, G. C. The role of the extracellular matrix protein TGFBI in cancer. Cell. Signal. 84, 110028 (2021).
Du, G.-W. et al. Identification of transforming growth factor beta induced (TGFBI) as an immune-related prognostic factor in clear cell renal cell carcinoma (ccRCC). Aging 12, 8484–8505 (2020).
Zoltan-Jones, A., Huang, L., Ghatak, S. & Toole, B. P. Elevated hyaluronan production induces mesenchymal and transformed properties in epithelial cells. J. Biol. Chem. 6, 66 (2003).
Li, L. et al. Transforming growth factor-β1 induces EMT by the transactivation of epidermal growth factor signaling through HA/CD44 in lung and breast cancer cells. Int. J. Mol. Med. 36, 113–122 (2015).
Kouvidi, K. et al. Role of receptor for hyaluronic acid-mediated motility (RHAMM) in low molecular weight hyaluronan (LMWHA)-mediated fibrosarcoma cell adhesion. J. Biol. Chem. 286, 38509–38520 (2011).
Porsch, H. et al. Efficient TGFβ-induced epithelial-mesenchymal transition depends on hyaluronan synthase HAS2. Oncogene 32, 4355–4365 (2013).
Bourguignon, L. Y. W., Spevak, C. C., Wong, G., Xia, W. & Gilad, E. Hyaluronan-CD44 interaction with protein kinase Cε promotes oncogenic signaling by the stem cell marker nanog and the production of microRNA-21, leading to down-regulation of the tumor suppressor protein PDCD4, anti-apoptosis, and chemotherapy resistance in breast tumor cells. J. Biol. Chem. 284, 26533–26546 (2009).
Mirzapoiazova, T. et al. HABP2 is a novel regulator of hyaluronan-mediated human lung cancer progression. Front. Oncol. 5, 164 (2015).
Mambetsariev, N. et al. Hyaluronic acid binding protein 2 is a novel regulator of vascular integrity. Arterioscler. Thromb. Vasc. Biol. 30, 483–490 (2010).
Jin, C. & Zong, Y. The role of hyaluronan in renal cell carcinoma. Front. Immunol. 14, 1127828 (2023).
Lucarelli, G. et al. Metabolomic profile of glycolysis and the pentose phosphate pathway identifies the central role of glucose-6-phosphate dehydrogenase in clear cell-renal cell carcinoma. Oncotargets 6, 13371–13386 (2015).
Oikari, S. et al. UDP-sugar accumulation drives hyaluronan synthesis in breast cancer. Matrix Biol. 67, 63–74 (2018).
Seidler, D. G. & Dreier, R. Decorin and its galactosaminoglycan chain: Extracellular regulator of cellular function?. IUBMB Life 60, 729–733 (2008).
Mikami, T. & Kitagawa, H. Biosynthesis and function of chondroitin sulfate. Biochim. Biophys. Acta 1830, 4719–4733 (2013).
Suwiwat, S. et al. Expression of extracellular matrix components versican, chondroitin sulfate, tenascin, and hyaluronan, and their association with disease outcome in node-negative breast cancer. Clin. Cancer Res. 10, 2491–2498 (2004).
Pothacharoen, P. et al. Raised serum chondroitin sulfate epitope level in ovarian epithelial cancer. J. Biochem. 140, 517–524 (2006).
Lv, H. et al. Elevate level of glycosaminoglycans and altered sulfation pattern of chondroitin sulfate are associated with differentiation status and histological type of human primary hepatic carcinoma. Oncology 72, 347–356 (2008).
Batista, L. T. A. et al. Heparanase expression and glycosaminoglycans profile in renal cell carcinoma. Int. J. Urol. 19, 1036–1040 (2012).
Gatto, F. et al. Glycosaminoglycan profiling in patients’ plasma and urine predicts the occurrence of metastatic clear cell renal cell carcinoma. Cell Rep. 15, 1822–1836 (2016).
Pezzicoli, G. et al. Playing the devil’s advocate: Should we give a second chance to mTOR inhibition in renal clear cell carcinoma?—ie strategies to revert resistance to mtor inhibitors. Cancer Manag. Res. 13, 7623–7636 (2021).
Voss, M. H. et al. Tumor genetic analyses of patients with metastatic renal cell carcinoma and extended benefit from mTOR inhibitor therapy. Clin. Cancer Res. 20, 1955–1964 (2014).
Großkopf, H. et al. Identification of intracellular glycosaminoglycan-interacting proteins by affinity purification mass spectrometry. Biol. Chem. 402, 1427–1440 (2021).
Li, X. et al. Prognostic value of CD44 expression in renal cell carcinoma: A systematic review and meta-analysis. Sci. Rep. 5, 13157 (2015).
Chen, W. J. et al. ENO2 affects the EMT process of renal cell carcinoma and participates in the regulation of the immune microenvironment. Oncol. Rep. 49, 33 (2023).
Yamanaka, M. et al. BIGH3 is overexpressed in clear cell renal cell carcinoma. Oncol. Rep. 19, 865–874 (2008).
Feng, C., Ding, G., Ding, Q. & Wen, H. Overexpression of low density lipoprotein receptor-related protein 1 (LRP1) is associated with worsened prognosis and decreased cancer immunity in clear-cell renal cell carcinoma. Biochem. Biophys. Res. Commun. 503, 1537–1543 (2018).
Patraki, E. & Cardillo, M. R. Quantitative immunohistochemical analysis of matrilysin 1 (MMP-7) in various renal cell carcinoma subtypes. Int. J. Immunopathol. Pharmacol. 20, 687–705 (2007).
Lin, T. C. RUNX1 and cancer. J. Exp. Clin. Cancer Res. 38, 334 (2022).
Li, Q. et al. RUNX1 promotes tumour metastasis by activating the Wnt/β-catenin signalling pathway and EMT in colorectal cancer. J. Exp. Clin. Cancer Res. 38, 334 (2019).
Fu, Y., Sun, S., Man, X. & Kong, C. Increased expression of RUNX1 in clear cell renal cell carcinoma predicts poor prognosis. PeerJ 7, e7854 (2019).
Rooney, N. et al. RUNX1 is a driver of renal cell carcinoma correlating with clinical outcome. Cancer Res. 80, 2325–2339 (2020).
Shan, T. et al. SERPINH1 regulates EMT and gastric cancer metastasis via the Wnt/β-catenin signaling pathway. Aging 12, 3574–3593 (2020).
Motzer, R. J. et al. Molecular subsets in renal cancer determine outcome to checkpoint and angiogenesis blockade. Cancer Cell 38, 803-817.e4 (2020).
Witschen, P. M. et al. Tumor cell associated hyaluronan-CD44 signaling promotes pro-tumor inflammation in breast cancer. Cancers 12, 1325 (2020).
Donelan, W., Dominguez-Gutierrez, P. R. & Kusmartsev, S. Deregulated hyaluronan metabolism in the tumor microenvironment drives cancer inflammation and tumor-associated immune suppression. Front. Immunol. 13, 971278 (2022).
Kainulainen, K. et al. M1 macrophages induce protumor inflammation in melanoma cells through TNFR–NF-κB signaling. J. Invest. Dermatol. 142, 3041-3051.e10 (2022).
Kusmartsev, S., Kwenda, E., Dominguez-Gutierrez, P. R., Crispen, P. L. & O’Malley, P. High levels of PD-L1+ and Hyal2+ myeloid-derived suppressor cells in renal cell carcinoma. J. Kidney Cancer VHL 9, 1–6 (2022).
Dominguez-Gutierrez, P. R. et al. Hyal2 expression in tumor-associated myeloid cells mediates cancer-related inflammation in bladder cancer. Cancer Res. 81, 648–657 (2021).
Spinelli, F. M., Vitale, D. L., Demarchi, G., Cristina, C. & Alaniz, L. The immunological effect of hyaluronan in tumor angiogenesis. Clin. Transl. Immunol. 4, e52 (2015).
Karousou, E. et al. Hyaluronan in the cancer cells microenvironment. Cancers 15, 798 (2023).
Vuong, L., Kotecha, R. R., Voss, M. H. & Hakimi, A. A. Tumorss microenvironment dynamics in clear-cell renal cell carcinoma. Cancer Discov. 9, 1349–1357 (2019).
Lasorsa, F. et al. Cellular and molecular players in the tumor microenvironment of renal cell carcinoma. J. Clin. Med. 12, 3888 (2023).
Acknowledgements
We would like to thank Mr. Einari Niskanen PhD (University of Eastern Finland, UEF) for assistance with bioinformatics analyses and Ms. Ella Ikonen MSc (Biobank of Eastern Finland) for expert technical assistance. In addition, we would like to acknowledge the contribution of prof. Petri Auvinen’s team and the Helsinki Institute of Life Science Institute of Biotechnology with RNA sequencing. We acknowledge Professors emer. Raija and Markku Tammi (UEF) for their critical reading and comments on the manuscript. Computational analyses were performed on servers provided by the UEF Bioinformatics Center, University of Eastern Finland, Finland. The research reported in this publication was supported in part by the Biostatistics and Bioinformatics Shared Resource of Winship Cancer Institute of Emory University and NIH/NCI under the award number P30CA138292. The content is the sole responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. The results published here are in whole or part based upon data generated by the TCGA Research Network: https://www.cancer.gov/tcga.
Funding
Financial support for this work was provided by Kuopio University Hospital Catchment Area State Research Funding (VTR), Munuaissäätiö, The Finnish Medical Foundation, Ida Monti Foundation, Northern Savo Cancer Foundation, and Paavo Koistinen Foundation.
Author information
Authors and Affiliations
Contributions
Conceptualization, O.J., R.S. and T.N.; methodology O.J, R.S., T.N., T.J.R., V.F.; software, O.J, T.N. and T.J.R.; validation, O.J. and T.N.; formal analysis, O.J. and T.J.R.; investigation, O.J., R.S. and T.J.R.; resources, O.J., T.J.R., R.S., T.N. and V.F.; data curation, O.J. and T.J.R.; writing—original draft preparation, O.J., T.J.R.; writing—review and editing, O.J., T.J.R., V.F., R.S., S. P-S., T.N.; visualization, O.J. and T.J.R.; supervision, T.N., R.S., and V.F.; project administration, T.N.; funding acquisition, O.J. and T.N. All the authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jokelainen, O., Rintala, T.J., Fortino, V. et al. Differential expression analysis identifies a prognostically significant extracellular matrix–enriched gene signature in hyaluronan-positive clear cell renal cell carcinoma. Sci Rep 14, 10626 (2024). https://doi.org/10.1038/s41598-024-61426-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-61426-3
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.