Abstract
In the mouse embryo, the transition from the preimplantation to the postimplantation epiblast is governed by changes in the gene regulatory network (GRN) that lead to transcriptional, epigenetic, and functional changes. This transition can be faithfully recapitulated in vitro by the differentiation of mouse embryonic stem cells (mESCs) to epiblast-like cells (EpiLCs), that reside in naïve and formative states of pluripotency, respectively. However, the GRN that drives this conversion is not fully elucidated. Here we demonstrate that the transcription factor OCT6 is a key driver of this process. Firstly, we show that Oct6 is not expressed in mESCs but is rapidly induced as cells exit the naïve pluripotent state. By deleting Oct6 in mESCs, we find that knockout cells fail to acquire the typical morphological changes associated with the formative state when induced to differentiate. Additionally, the key naïve pluripotency TFs Nanog, Klf2, Nr5a2, Prdm14, and Esrrb were expressed at higher levels than in wild-type cells, indicating an incomplete dismantling of the naïve pluripotency GRN. Conversely, premature expression of Oct6 in naïve cells triggered a rapid morphological transformation mirroring differentiation, that was accompanied by the upregulation of the endogenous Oct6 as well as the formative genes Sox3, Zic2/3, Foxp1, Dnmt3A and FGF5. Strikingly, we found that OCT6 represses Nanog in a bistable manner and that this regulation is at the transcriptional level. Moreover, our findings also reveal that Oct6 is repressed by NANOG. Collectively, our results establish OCT6 as a key TF in the dissolution of the naïve pluripotent state and support a model where Oct6 and Nanog form a double negative feedback loop which could act as an important toggle mediating the transition to the formative state.
Similar content being viewed by others
Introduction
During early mammalian development, cells of the pre-implantation epiblast have the capacity to differentiate into all the cell types of the organism, a property known as pluripotency. This feature persists in the post-implantation epiblast and is present until the time of gastrulation, when the three embryonic germ layers emerge1. Pluripotency exists in a continuum of states but has recently been subdivided into three subtypes that are linked to the different stages of epiblast development. Thus, pre-implantation epiblast cells are referred to be in a naïve pluripotent state, while early and late post-implantation epiblast cells display formative and primed pluripotency, respectively2. These different subtypes are characterized by changes in their transcriptional, epigenetic, metabolic as well as functional properties3.
In mice, the transition between the naïve and formative states can be faithfully recapitulated in vitro with the use of mouse embryonic stem cells (mESCs). They represent an invaluable tool for investigating the mechanisms underlying early mammalian development, as they possess the capacity to self-renew indefinitely while retaining naïve pluripotency4. Importantly, they can be induced to differentiate to epiblast-like cells (EpiLCs), a transitory cell population obtained after the removal of the media components that support naive pluripotency. EpiLCs are in a formative state of pluripotency, similar to the early post-implantation epiblast, and are characterized by the downregulation of the naive transcription factors (TFs) Esrrb and Klf4, the expression of markers such as Fgf5 and Dnmt3A, X-chromosome inactivation in female cells and a state-specific epigenetic landscape3,5. The conversion from mESCs to EpiLCs thus constitutes an excellent model for studying the molecular mechanisms of pluripotency conversion and changes in cell identity.
Central to these developmental transitions are the gene regulatory networks (GRNs) that orchestrate the precise control of gene expression, guiding cellular fate decisions. GRNs comprise a complex interplay of transcription factors (TFs), signaling molecules, and epigenetic modifiers that act in concert to regulate the dynamic patterns of gene expression during development6,7. An important aspect of GRNs are the intricate architectural features known as "network motifs", i.e., the recurring patterns of gene interactions within the regulatory networks8. Each network motif performs a defined information processing function within the network and some of them, such as the double negative feedback loop, can lead to “toggle switches” between different cell states9. Thus, delineating the circuitry of interactions among different TFs is key to understanding the molecular mechanisms that govern cell fate decisions.
Among the key regulatory components, Oct4, Sox2, and Nanog constitute the core pluripotency TFs. They act by activating the expression of other pluripotency-associated factors and simultaneously repressing lineage-specific genes, while also upregulating their own gene expression and that of each other10,11. While Oct4 and Sox2 are fundamental for the formation of the inner cell mass (ICM) and the maintenance of the pluripotent state of mESCs, Nanog is important for the acquisition of pluripotency but dispensable once it is achieved12. Interestingly, although Nanog is expressed in mESCs and in the late post-implantation like epiblast stem cells (EpiSCs), its expression is transiently downregulated as cells differentiate to EpiLCs5,10,13. Conversely, other TFs are necessary for a proper induction of formative pluripotency. Although both Oct4 and Otx2 are expressed in naïve mESCs, their reorganization to new enhancers has been shown to be required for the correct acquisition of a formative identity14. In addition, Otx2 as well as Foxd3 promote the transition to formative pluripotency by coordinating the silencing of naïve genes and the activation of early post-implantation epiblast markers15,16. Interestingly, as is the case of Oct4, Otx2, and Foxd3, most of the TFs shown to be important in the formative GRN are also expressed in mESCs.
Here, we show that the TF Oct6 is not expressed in ground-state mESCs but is rapidly induced as cells transition to formative pluripotency. Oct6 is a well-established marker of formative and primed pluripotency14 and is important during the neural differentiation of mESCs17. However, its role in the transition to EpiLCs has not been extensively addressed. We show that the absence of Oct6 prevented the correct dismantling of the naïve TF circuitry in differentiating cells, partially blocking the morphological transformation observed in EpiLCs. Furthermore, premature expression of Oct6 in naïve cells was sufficient to induce a morphology akin EpiLCs, while inducing formative genes like Sox3, Dnmt3A, FGF5 and repressing Nanog in a bistable manner. Our results suggest that Oct6 promotes the exit from naïve pluripotency through the repression of Nanog and the induction of formative specific genes. Collectively, our results show that Oct6 and Nanog form a double negative feedback loop which could act as a toggle switch important for the transition to the formative state.
Results
Oct6 is one of the earliest TFs induced during the exit from naïve pluripotency
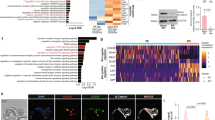
To shed light on the conversion of GRNs from naïve to formative pluripotency, we initially focused on the TFs induced at early stages of differentiation, since they could be crucial to GRN restructuring. Based on a thorough study performed by Yang and collaborators18, we identified TFs that were upregulated in the first 6 h of the transition and remained highly expressed at 48 h (Fig. S1A). This preliminary analysis revealed known facilitators of formative pluripotency, such as Otx2, Foxd3 and c-Myc15,16,19. Other TFs that exhibited this trend include the retinoic acid receptors Rarg and Rxrg, as well as Myrf, Ar, and Aff3. Interestingly, among these early-induced genes, Oct6 (also known as Pou3f1) was the one that displayed the most important induction when comparing mESCs vs. 48 h EpiLCs, and it was significantly upregulated only 2 h after the onset of differentiation (Fig. 1A). In contrast, other formative markers such as Fgf5, Otx2, and Dnmt3A were upregulated at later time points. The rapid induction of Oct6 transcription was accompanied by a significant decrease of the naïve TF Klf4 expression at 2 h, whereas other naïve TFs such as Nanog, Esrrb, and Tbx3 were downregulated at later time points. Quantitative immunofluorescence showed that OCT6 was not detected in naïve pluripotent ground state conditions and that its levels increased gradually as cells entered formative pluripotency, with all cells expressing this TF at 48 h of differentiation (Fig. 1B, Fig. S1B). In light of these results, we decided to assess the role of Oct6 in the dissolution of naïve pluripotency.
Loss of Oct6 affects the transition to formative pluripotency. (A) RT-qPCR analysis of naïve and formative pluripotency markers during the first hours of EpiLC differentiation. Results are presented as mean ± SEM for three independent replicates. Different letters indicate significant differences between time points (p < 0.05). (B) Quantification of nuclear OCT6 during EpiLC induction. The violin plot shows the distribution of OCT6 nuclear intensity for each timepoint and the circles show the mean fluorescence for individual cells. (C) Diagram of Oct6 genomic locus showing the sgRNA binding region for CRISPR/Cas9 mediated KO generation. (D) Representative immunoblot showing OCT6 depletion in Oct6 KO EpiLCs. GAPDH was used as a loading control. (E) Representative immunofluorescence of OCT6 in WT and KO 48 h EpiLCs. DAPI was used as a nuclear stain. Scale bar: 50 µm. (F) Morphological differences between WT and Oct6 KO cells induced to differentiate to EpiLCs. Brightfield micrographs of WT and Oct6 KO EpiLCs. Scale bar: 50 µm. (G) Left. Representative images of DAPI stained nuclei of WT and Oct6 KO EpiLCs. Right. Distribution of the distances to the nearest neighbor cells for all nuclei in the representative images (see Materials and Methods). (H) Heatmap of differentially expressed genes between WT and Oct6 KO EpiLCs as obtained from Deseq2 analysis. (I) Examples of DE gene expression for naïve pluripotency genes (top) and formative genes (bottom) from the RNA-seq results. (J) RT-qPCR analysis of Nanog in WT and Oct6 KO EpiLCs. Results are as presented as mean ± SEM for three independent replicates. (K) Representative immunofluorescence of NANOG in WT and Oct6 KO EpiLCs. Scale bar: 100 µm. (L) Flow cytometry analysis of Nanog expression showing higher expression in Oct6 KO cells compared to WT EpiLCs.
We then conducted an analysis of the Oct6 promoter by examining previously published ChIP-seq experiments to identify potential cis-regulatory elements (CRE) and likely regulators influencing Oct6 expression14,16,18,20,21,22. We discovered two CREs located 10 and 12 kb upstream of the transcription start site, which we refer to as CRE#1 and CRE#2, respectively (Fig. S2). In mESCs maintained in 2i + LIF, the TFs OCT4, OTX2, NANOG, and ESRRB bind only to CRE#2 and not to CRE#1. Interestingly, EpiLCs presented a reorganization of OCT4 and OTX2 binding, with their occupancy extended to CRE#1. Moreover, this regulatory element exhibited an increased signal of the active enhancer marks H3K27ac and H3K4me1, along with open chromatin detected by ATAC-seq. These analyses collectively suggest that CRE#2 acts as a regulatory sequence specific to naïve pluripotency, potentially involved in repressing Oct6 expression, while CRE#1 may function as an enhancer specific to EpiLCs.
Absence of Oct6 negatively affects the transition to formative pluripotency
To directly assess the role of Oct6 in the transition to EpiLCs, we generated an Oct6 knockout line (Oct6-KO) using CRISPR/Cas9 in 46C mESCs (Fig. 1C, Fig. S3A). Western blot and immunofluorescence analysis confirmed the lack of expression of OCT6 protein after induction to EpiLCs (Fig. 1D,E). Importantly, KO cells cultured in ground state conditions did not show any changes in morphology or the expression of the pluripotency transcription factor Nanog (Fig. S3B,C).
Oct6 has been previously reported as a key player in neural progenitor cell (NPC) differentiation. We thus evaluated the ability of the Oct6-KO cells to differentiate to the neural lineage by taking advantage of 46C mESCs line, that expresses GFP under the control of the neural marker Sox1. As expected, Oct6-KO cells produced significantly lower rates of SOX1-GFP + cells than WT 46C cells after 6 days of differentiation (~ 60% vs. ~ 90% SOX1-GFP + cells, respectively) (Fig. S3D). These data are consistent with Oct6 promoting neural induction, although its expression is not fundamental for the differentiation of NPCs.
We then addressed if Oct6-KO cells were affected in their differentiation capacity to EpiLCs. The transition from naïve to formative pluripotency is accompanied by important morphological changes. mESCs in the naïve ground state grow as tightly packed colonies with a dome shape. Upon EpiLC induction, cells quickly undergo a morphological conversion that includes flattening, diminished cell–cell interactions, and the formation of cellular protrusions14,23. Interestingly, we noticed that upon differentiation, Oct6-KO cells exhibited a more compact morphology with considerably fewer protrusions compared to parental WT cells (Fig. 1F). Indeed, quantification of the distribution of distances to each cell’s nearest neighbor both for WT and KO EpiLCs confirmed that Oct6-KO cells were more tightly packed and did not show colony-detached cells as in the case of WT EpiLCs (Fig. 1G). Thus, the absence of Oct6 impairs the phenotypic changes observed as mESCs exit naïve pluripotency.
To further assess the role of Oct6 during differentiation we performed an RNA sequencing (RNA-seq) experiment on WT and Oct6-KO EpiLCs. A total of 292 genes were differentially expressed (DE) with at least 1 log twofold change (Fig. 1H, Table S1). Functional annotation analysis of the DE genes with Gene Ontology (GO) revealed significant enrichment for the biological process term “regulation of cell motility” (GO:2000145) and the cellular component terms “plasma membrane bounded cell projection” (GO:0120025) and “cell leading edge” (GO:0031252), in agreement with the phenotypic effects previously observed (Table S2). Importantly, the GO term “cell differentiation” (GO:0030154) was also highly enriched, suggesting that KO EpiLCs may display alterations in their differentiation capacity. Among the DE genes, we found that key TFs associated with naïve pluripotency such as Esrrb, Klf2, Nr5a2, Dppa2, Tdh, Zic3, and Prdm14 showed higher expression levels in KO cells compared to WT EpiLCs (Fig. 1I, Fig. S3E). Interestingly, although not detected in the RNA-seq data due to the stringency of the analysis, assessment of the master pluripotency regulator Nanog also showed a slight but significant upregulation in KO EpiLCs, both at the mRNA and protein level (Fig. 1J–L). On the other hand, genes associated with the transition to formative pluripotency or neural differentiation such as Zic2, Sox1, Nestin, Foxp1, and Hoxa124 were less expressed in KO EpiLCs. Supporting the role of Oct6 in the changes in cell morphology, we also observed differences in genes associated with cell attachment such as Vimentin, E-Cadherin, N-Cadherin and Claudins 5, 6, 7 and 9 (Fig. S3E,F). In summary, our results indicate that the absence of Oct6 impairs the correct acquisition of the GRN associated with formative pluripotency.
We next intended to assess the genome-wide binding of OCT6 in EpiLCs. However, there are currently no suitable Oct6 antibodies for ChIP (see the Discussion). Therefore, we analyzed previously published data, where Matsuda et al. evaluated OCT6 binding in epiblast stem cells (EpiSCs) by overexpressing a tagged version of Oct625. Although EpiLCs and EpiSCs represent different developmental stages of post-implantation epiblast development, we reasoned that these data could be useful to infer which of the DE genes in our RNA-seq experiment could be direct targets of OCT6. Indeed, among the 292 DE genes detected in our RNA-seq, 114 were associated with OCT6 ChIP binding peaks in EpiSCs (Fig. S4A). This number was almost three times higher than the ~ 40 genes that would be expected out of chance, as evaluated by a bootstrap analysis. Interestingly, among the DE genes of our RNA-seq that contained OCT6 peaks in EpiSCs we found the naïve pluripotency expressed genes Dppa2, Prdm14, Nr5a2, and Vim and the EpiLCs induced genes Zic2, Zic3, Sox1, Nes, and Foxp1 (Fig. S4B). Overall, this analysis validated our RNA-seq results and identified genes that are potentially regulated by Oct6 at the transcriptional level by direct binding to their loci.
Overexpression of Oct6 in naïve cells represses Nanog and induces morphological changes associated with formative cells
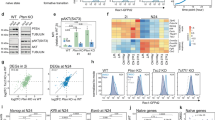
To further study the role of Oct6 in the transition from naïve to formative pluripotency, we analyzed the effect of its premature expression in the pluripotent ground state. To that end, we engineered a new cell line where KO cells were complemented with a construct that allows the doxycycline (Dox) inducible expression of an HA-tagged version of OCT6, together with the fluorescent protein mCherry via a self-cleaving peptide (Fig. 2A). As expected, the addition of Dox induced the expression of the Oct6 and mCherry transgenes, while allowing the in vivo observation of red fluorescence in OCT6 overexpressing cells (Fig. 2B). Quantitative immunofluorescence confirmed that mCherry signal was a good proxy of OCT6 expression since their levels were highly and linearly correlated throughout several orders of magnitude (Fig. 2C). Importantly, induction of transgenic OCT6 expression in differentiating KO cells rescued the morphological effect observed previously, as seen by the reappearance of diminished cell–cell interactions and cell protrusions (Fig. 2D).
Overexpression of Oct6 in undifferentiated cells promotes morphological and transcriptional changes associated with formative pluripotency. (A) Diagram of the Oct6-P2A-mCherry overexpressing construct and the experimental design. (B) Treatment with Dox in 2i + LIF medium induces the expression of mCherry and a morphological transformation reminiscent of formative cells. Scale bar: 200 µm. (C) The plot shows the linear correlation of mean nuclear intensity fluorescence of mCherry and OCT6 obtained from OCT6 immunostaining of Dox treated cells. Each circle shows the data of one individual nucleus. (D) Overexpression of Oct6 in KO EpiLCs rescues the morphological differences observed between WT and KO EpiLCs. Scale bar: 50 µm. (E) RT-qPCR analysis of naïve and formative markers upon Dox treatment. Results are as presented as mean ± SEM for three independent replicates. (F) Representative immunofluorescence showing how Dox treatment induces the repression of NANOG in Oct6-P2A-mCherry expressing cells. Scale bar: 100 µm. (G) Quantification of immunofluorescence experiments of mCherry and NANOG in untreated or 24, 48, and 72 h Dox treated cells in 2i + LIF medium. The violin plots show the distribution of mCherry or NANOG nuclear intensity for each time point. The circles show the mean fluorescence for individual cells. (H) The plot shows the correlation between Oct6-P2A-mCherry and NANOG nuclear intensity expression in untreated or Dox-treated cells in 2i + LIF after immunostaining experiments. The circles show the mean fluorescence for individual cells. Data for 24, 48, and 72 h Dox treatment was pooled since they behaved similarly.
We next evaluated the effect of Oct6 expression in ground state conditions. While dox untreated cells formed typical compact dome-shaped colonies, the addition of Dox for 72 h induced a pronounced morphological change similar to the observed upon differentiation, with flattened colonies, diminished cell–cell interactions, and the formation of cellular protrusions (see Fig. 2B). These changes could be observed as early as 24 h after Dox treatment. As an additional control, no morphological changes were observed when using an inducible cell line that only overexpressed mCherry (Fig. S5A). Gene expression analysis showed that induction of Oct6 expression in 2i + LIF media upregulated the formative marker genes Sox3, Dnmt3A, Fgf5, Foxp1, Brn2, Zic2, Zic3, and endogenous Oct6, while not affecting Otx2 nor the general pluripotency marker Oct4 (Fig. 2E, Fig. S5B). Of the formative marker genes, only Dnmt3A lacks OCT6-binding peaks in EpiSCs, suggesting that the remaining genes could be direct targets of Oct6 (see Fig. S4B). Importantly, no significant changes were observed in the expression of the naïve pluripotency markers Esrrb, Rex1, Prdm14, Klf4, and Tbx3.
To gain a deeper insight into the regulatory effect of Oct6, we performed quantitative immunofluorescence experiments against key TFs regulated in this transition. OCT4, SOX2, and KLF4 showed similar protein levels between Dox treated and untreated mESCs, with only a slight reduction in the mean expression (Fig. S5C). In the case of SOX3, consistent with the gene expression data, we detected an important upregulation in a subset of OCT6 overexpressing cells, further reinforcing the regulatory link between these genes. We were particularly interested in the analysis of Nanog, not only because of its increased transcript levels in KO versus WT EpiLCs, but also because we discovered that NANOG and OCT6 proteins are expressed in a mutually exclusive manner in WT EpiLCs (Fig. S6A). Strikingly, while more than 90% of untreated cells expressed high levels of NANOG, we observed that overexpression of OCT6 repressed the expression of NANOG at the protein level in more than 70% of the cells, both at 24, 48 and 72 h of Dox (Fig. 2F, Fig. S6B). This effect was positively correlated with OCT6 levels and displayed a bistable switch-like behavior (Fig. 2G). As a result, cells that were mCherry negative were more than 80% NANOG positive, while mCherry expression two-fold above background levels already showed more than 40% NANOG negative cells, suggesting that mild OCT6 expression is sufficient to repress NANOG (Fig. 2H). For cells expressing mCherry at levels beyond four-fold above background, more than 90% of cells were NANOG negative. Interestingly, the fact that NANOG exhibited a clear bistabe “ON–OFF” expression might indicate the existence of a repressive feedback loop between these TFs.
We next wondered whether the repression of NANOG was reversible after releasing the induction of OCT6. To answer this, we removed Dox from OCT6-induced cultures and analyzed mCherry and NANOG protein levels at different time points after washing the cells (Fig. S7A). Interestingly, as early as 24 h after Dox removal, mCherry fluorescence returned to background levels and NANOG expression was restored (Fig. S7B). We did not observe appreciable differences in the levels of apoptosis among the experimental conditions, indicating that NANOG + /OCT6- derived from previously NANOG-/OCT6 + cells (Fig. S8). The morphological changes induced by Dox were also reverted and colonies re-acquired their typical highly packed dome shape. Overall, these results indicate that OCT6 expression in naïve culture conditions induces a reversible non-physiological identity with similarities to the formative pluripotent state.
OCT6 represses Nanog at the transcriptional level
To further analyze the dose dependency of OCT6 in NANOG repression, we took advantage of the expression of mCherry and analyzed 24 h Dox-treated cells by flow cytometry, gating them into 3 populations, Low, Med, and High, all of them with the same number of cells (Fig. 3A). In agreement with our previous results, ~ 75% of cells in the mCherry-low population displayed high NANOG levels, while these percentages decreased to ~ 35% and ~ 2% in the mCherry-Med and mCherry-High populations, respectively (Fig. 3B). By sorting these 3 cell populations, we confirmed the correlation between mCherry fluorescence and the mRNA levels of Oct6, mCherry, and the endogenous Sox3 mRNAs (Fig. 3C). Interestingly, mCherry expression showed an inverse relation with Nanog mRNA levels, suggesting that OCT6 repressed Nanog at the transcriptional level. Even though the mCherry-Med population contained 65% of cells with reduced NANOG protein levels, we only observed a roughly two-fold reduction in its mRNA levels. This could be explained by the contribution of the remaining ~ 35% fully NANOG positive cells, which can mask the downregulation when analyzing the mean Nanog mRNAs levels in the entire population. To complement these findings, we conducted a luciferase reporter assay to assess the impact of OCT6 expression on the Nanog promoter. Remarkably, induction of Oct6 using Dox resulted in a significant decrease in Nanog reporter activity, observed in both undifferentiated and 24 h differentiating cells (Fig. S9A). These findings provide compelling evidence of OCT6's regulatory influence on the Nanog promoter, further supporting the notion of regulation at the transcriptional level. To reinforce these results, we performed single-molecule RNA-FISH experiments to detect individual Nanog mRNAs in OCT6 overexpressing cells. We first validated this method by analyzing undifferentiated mESCs and 24 h EpiLCs, which confirmed homogeneous expression of Nanog transcripts in naïve cells and a significant reduction as cells transited towards the formative state (Fig. S9B). After this validation, we evaluated the distribution of Nanog transcripts in undifferentiated cells induced to express Oct6 while simultaneously detecting NANOG and OCT6 proteins. As expected, cells with low levels of OCT6 protein were positive for Nanog transcripts and expressed NANOG at the protein level. Interestingly, cells with high levels of OCT6 protein did not present neither NANOG protein nor Nanog mRNAs (Fig. 3D). In summary, our results demonstrate that OCT6 expression in naïve ground state conditions represses Nanog at the transcriptional level. Together with the over-expression of formative-specific transcription factors such as Sox3 and the de novo DNA methyltransferase Dnmt3A, Oct6 might ultimately induce a shift in the naïve GRN that partially sets it to a formative configuration similar to the early post-implantation epiblast.
Oct6 represses Nanog at the transcriptional level. (A) Flow cytometry sorting of 24 h Dox treated cells into populations with low, medium (med), and high levels of mCherry. (B) Left. Correlation between mCherry and NANOG by flow cytometry. The different mCherry expressing subpopulations are shown. Right. The proportion of NANOG high, med, and low in the different mCherry expressing subpopulations. (C) RT-qPCR analysis of exogenous Oct6, mCherry, Sox3, and Nanog in Oct6 overexpressing cells. Untreated or Dox-treated cells (bulk population) are shown in black and statistically compared using a student’s t-test. The different mCherry expressing subpopulations are shown in shades of red and were compared using random block ANOVA. Different letters indicate significant differences (p < 0.05). (D) Simultaneous single-molecule RNA FISH against Nanog cytoplasmic transcripts and immunostaining against Nanog and Oct6 proteins. The insets show example cells with high and low Oct6 transgene expression. Scale bar: 20 µm.
Oct6 and Nanog form a double negative feedback loop with positive autoregulation
Our results so far demonstrated that OCT6 and NANOG are expressed in a mutually exclusive manner in WT EpiLCs and that OCT6 activates its own transcription while repressing the expression of Nanog. Moreover, Nanog is known to present positive autoregulation26,27. This led us to hypothesize that these two TFs could constitute a double negative feedback loop that could act as a toggle switch to initiate the dissolution of the naïve pluripotent state. Indeed, we have previously shown that NANOG binds to CRE#2 in Oct6’s promoter along with OCT4, OTX2 and ESRRB (see Fig. S2). To assess if OCT6 might also bind to the Nanog locus, we analyzed the aforementioned work by Matsuda et al. in EpiSCs. While the ChIP-seq signal in Nanog locus exhibited relatively low intensity, we observed a discrete accumulation of reads at the 5 kb distal enhancer located upstream of the Nanog TSS (Fig. S9C). This distal enhancer, recognized as a crucial CRE, is known to be targeted by other key pluripotency transcription factors28,29. This subtle observation prompted us to consider that OCT6 might bind to Nanog’s promoter in EpiLCs.
Finally, we evaluated if NANOG inhibited the expression of Oct6 by generating a cell line capable of expressing a Nanog transgene under the control of Dox (Fig. 4A). As in the case of Oct6 overexpression, this cell line also expresses mCherry via a self-cleaving peptide. To confirm the correct behavior of this line, we first differentiated them for 48 h in the presence or absence of Dox and confirmed that they expressed mCherry and NANOG in a highly correlated fashion (Fig. 4B,C). Next, we evaluated whether Dox-treated EpiLCs showed reduced levels of OCT6. Our results show that while mCherry negative cells exhibited normal levels of OCT6, mCherry expressing cells did not express this transcription factor, indicating that NANOG repressed Oct6 in a cell-autonomous fashion (Fig. 4D,E). Overall, our results indicate that these genes could constitute a toggle-switch circuit important for the correct dissolution of naïve pluripotency and the transition to a post-implantation epiblast-like phenotype.
Oct6 and Nanog repress each other forming a double negative feedforward loop network motive. (A) Diagram of the Nanog-P2A-mCherry overexpressing construct and the experimental design. (B) Treatment with Dox during EpiLC differentiation induces the expression of mCherry. Scale bar: 50 µm. (C) The plot shows the correlation of mean nuclear intensity fluorescence of mCherry and NANOG obtained from NAONG immunostaining of Dox-treated EpiLCs. Each circle shows the data of one individual nucleus. (D) Immunostaining of OCT6 showing that Dox-treated Nanog-P2A-mCherry overexpressing cells do not express OCT6. Conversely, Dox-treated cells not expressing Nanog-P2A-mCherry normally express OCT6. Scale bar: 50 µm. (E) The plot shows the correlation of mean nuclear intensity fluorescence of Nanog-P2A-mCherry and OCT6, obtained from OCT6 immunostaining of Dox-treated EpiLCs. Each circle shows the data of one individual nucleus. (F) Proposed model of transcriptional circuitry between Nanog and Oct6 that regulates the transition from naïve to formative pluripotency.
Discussion
In this study, we identified OCT6 as an important TF in the regulation of the formative pluripotent state as well as in dismantling the previous naïve identity. OCT6 belongs to the POU domain family of TFs, which are known to play critical roles in embryonic development and cellular differentiation. Previous studies have implicated Oct6 in various biological processes, including neural development, axon regeneration, and glial cell differentiation30,31. Recently, it was shown that Oct6 can replace Oct4 in reprogramming human cells but fails to do so in mouse cells19. Whether this difference relies on the different types of pluripotency presented by human and mouse PSCs, i.e., naïve vs. primed, remains to be fully investigated. Indeed, Oct6 has recently been shown to block the differentiation of porcine iPSCs, which display a primed pluripotency phenotype32. However, its precise role in the context of mouse PSC differentiation and the dissolution of naïve pluripotency circuitry remains largely unexplored.
Here, to identify key effectors of the conversion from naïve to formative pluripotency, we first searched for TFs that were rapidly induced and displayed high expression throughout the first 48 h of differentiation, when formative identity is established. Using this strategy, we identified Oct6 as a gene that is induced as early as 2 h after the onset of differentiation, with its expression peaking at 24 and 48 h of EpiLC induction. We show that OCT6 protein is expressed homogeneously in EpiLCs, reaching its maximum at 48 h. Importantly, OCT6 was not detected in ground state pluripotency when cells are cultured under defined conditions (N2B27 + 2i + LIF). This finding challenges previous reports that indicate that OCT6 can be detected in naïve cells17,33. This inconsistency might be attributed to the culture method, as mESCs cultured in undefined conditions containing serum result in heterogeneous populations where naïve cells coexist with primed and differentiated cells13,34. OCT6 expression in these culture conditions could thus be restricted to the primed pluripotency compartment. Additionally, we discovered that several commercial antibodies are not specific for OCT6 and may detect other members of the Pou family of TFs (data not shown), underscoring the need for careful assessment of OCT6 expression. Overall, the fact that Oct6 is induced during the transition to the formative state is consistent with its expression being first detected at the egg-cylinder stage of the mouse embryo35.
We then investigated the function of Oct6 during the transition to formative pluripotency by generating an Oct6 KO mESC line. The absence of OCT6 resulted in cells organized as densely packed colonies more reminiscent of the naïve phenotype, as opposed to the typical formative morphology characterized by reduced cell–cell interactions and protrusions14,23. Indeed, KO cells showed altered expression of genes associated with motility and cell morphology, such as Vim, E-Cad, N-Cad, and claudin proteins. Importantly, transcriptome analysis revealed that key naïve TFs, including Nanog, Klf2, Esrrb, Prdm14, Nr5a2, Dppa2, and Tdh10,34,36, were not properly downregulated in KO cells, indicating inefficient dismantling of the naïve GRN circuitry. We also observed reduced expression of genes normally induced in EpiLCs, such as Zic2 and Foxp137,38, and early neural genes weakly expressed in EpiLCs, including Nes and Sox1. Collectively, our findings support a critical role for Oct6 in the transition from naïve to formative pluripotency.
Other groups have recently analyzed the effect of reducing OCT6 levels during mESCs differentiation, either by knockout or knockdown strategies. Kalkan and collaborators observed that Oct6 KO mESCs did not delay the exit from naïve pluripotency39; however, this was only evaluated by the expression of a Rex1 reporter at 26 h of differentiation. Consistent with this, we did not observe differences in the expression of Rex1 when comparing WT and KO 48 h EpiLCs. On the other hand, Zhu and collaborators showed that Oct6 is important for the neural differentiation of mESCs specifically during the transition from epiblast stem cells (EpiSCs) to NPCs17. The impairment in neural differentiation was later confirmed by another group using Oct6 KO mESCs40. Our results are consistent with this work, as Oct6 KO cells only generated 60% SOX1-GFP + cells compared to 90% generated by the parental WT cell line. However, the fact that the formative state was affected in KO cells and that the naïve circuitry was not completely dismantled could also explain the reduced efficiency of neural differentiation.
An important limitation of our study was the inability to perform genome-wide binding analysis of OCT6 in EpiLCs, as there are no suitable antibodies for chromatin immunoprecipitation (ChIP). Despite our efforts to evaluate various antibodies we were unable to obtain specific signals when analyzing different genomic locations. We also attempted the Cut and Run technique41 with our engineered cell line expressing HA-Oct6, using both OCT6 and HA-specific antibodies, but unfortunately, specific signals were not detected. Interestingly, Matsuda et al. successfully captured OCT6 binding to the genome by overexpressing a tagged version of this TF in EpiSCs, a slightly more advanced stage of postimplantation epiblast development25. By comparing their data with our RNA-seq analysis, we found that 39% of the differentially expressed genes between WT and KO EpiLCs presented OCT6 binding peaks, a proportion significantly higher than the 14% expected by chance. Although this analysis strongly validates our results, future research should address diverse strategies to map the binding of OCT6 to the genome in EpiLCs.
The absence of OCT6 had a significant impact on the transition to formative pluripotency, although it did not completely hinder the dissolution from the naïve state. Interestingly, Oct6 KO mice are not embryonic lethal and only die within the first few days after birth42, suggesting the presence of compensatory mechanisms during early development. To investigate the specific role of Oct6 within the pluripotency GRN, we examined the effects of its premature expression in the naïve state. Our findings demonstrate that expression of Oct6 in 2i + LIF media induces a morphological change similar to that observed in WT EpiLCs, while activating formative genes such as Sox3, FGF5, Dnmt3A, and the endogenous Oct6. Notably, Sox3 and FGF5 contained OCT6 binding peaks in EpiSCs, indicating their potential direct regulation by this TF. Strikingly, the majority of cells overexpressing OCT6 repressed the expression of the core pluripotency gene Nanog, evidenced at both the transcriptional and protein levels, without affecting OCT4, SOX2, and KLF4. This finding is noteworthy, as downregulation of Nanog is necessary to allow the exit from the naïve ground state12,43,44. Although OCT6 overexpression displayed high heterogeneity and a wide range of expression, we observed that OCT6 levels immediately above background were sufficient to repress Nanog. Furthermore, we observed a subtle ChIP-seq signal of OCT6 binding to Nanog’s distal enhancer in EpiSCs, indicating that this TF might directly bind Nanog regulatory regions in EpiLCs. Interestingly, NANOG exhibited a bistable "ON–OFF" behavior within the "low OCT6" population, which is characteristic of double negative feedback loop subcircuits. To study this possibility, we evaluated Nanog and Oct6 expression in WT EpiLCs, which showed that these two proteins were expressed in a mutually exclusive manner. This prompted us to study whether NANOG also repressed Oct6. Our analysis of published data revealed that NANOG binds to the Oct6 promoter at a CRE regulated by other key TFs that potentially inhibits Oct6 expression in the undifferentiated state. Importantly, we observed that overexpression of NANOG in differentiating cells blocked the induction of OCT6, consistent with a recent study45. By causally demonstrating the mutual repression between Nanog and Oct6, along with their positive autoregulation, we propose that these two TFs form a double negative feedback loop (Fig. 4F), serving as a toggle switch crucial for the transition to the formative state and providing robustness to the GRN of naïve and formative pluripotency.
Materials and methods
Cell culture
46C mESCs and the transgenic lines derived from them were cultured using the chemically defined medium N2B27 with 1000 U/ml LIF (Millipore), 1 µM PD0325901 (Tocris) and 3 µM CHIR99021 (Tocris), hereafter called ‘2i + LIF medium’. N2B27 formulation is a 1:1 mix of DMEM-F12 (Invitrogen Cat# 12634) and neurobasal medium (Invitrogen Cat# 21103), supplemented with N2 (Invitrogen Cat# 17502), B27 supplement without Vitamin A (Invitrogen Cat# 12587), 2 mM GlutaMAX (Invitrogen Cat# 35050), 40 μg/ml of BSA (Invitrogen Cat# 15260), 10 μg/ml insulin (Sigma-Aldrich Cat# I9278), 0.1 mM β mercaptoethanol (Invitrogen Cat# 21985). Cells were maintained on 0,1% gelatin-coated dishes, passaged every 2–3 days using TrypLE (Gibco) and cultured at 37 °C in a 5% CO2 (v/v) incubator. Cells were cultured without antibiotics and routinely assessed for mycoplasma contamination by PCR. To induce differentiation from the naïve ground state to EpiLCs, 50.000 cells/cm2 were plated in 15 µg/ml Fibronectin (Thermo Fisher Scientific) coated dishes in the “EpiLC” medium. This medium consisted of base N2B27 supplemented with 1% KSR, 12 µg/ml bFGF, and 20 µg/ml Activin A (all from Thermo Fisher Scientific). Cells were differentiated for 24 or 48 h, as indicated in the main text. 46C mESCs were a kind gift from Dr. Austin Smith.
Generation of Oct6 KO 46C mESCs
46C mESCs Oct6 knockout cell line was constructed using CRISPR/Cas9. We designed two specific sgRNAs targeting the 5’ region within the Oct6 coding sequence to generate an indel next to the initiation codon. sgRNAs were designed using CHOPCHOP V346. Sense and antisense oligonucleotides were annealed and cloned into a BbsI-linearized pX330-GFP-Puro plasmid47. Recombinant plasmids were validated by DNA sequencing. Wild type 46C mESCs were independently transfected with the sgRNA plasmids using Linear Polyethylenimine (PEI, Polysciences) with a DNA/PEI ratio of 1:3 and selected with 3 μg/ml puromycin from 24 to 72 h post-transfection. After limiting dilution, 12 individual clonal lines were obtained for each sgRNA, amplified, and cryopreserved. Knockout generation for each clone was evaluated by genomic DNA sequencing of a PCR-amplified Oct6 locus. A clone with a homozygous insertion of a nucleotide within the Cas9 cutting site was chosen and further validated by Western Blotting and immunofluorescence, as indicated in the main text. Sequences for sgRNA sequences and KO evaluation primers are listed in Table S3.
Generation of Oct6-P2A-mCherry, Nanog-P2A-mCherry and mCherry overexpressing cell lines
To overexpress Oct6 in an inducible manner we designed a piggyBac plasmid that could be integrated into the cell's genome through a transposable recombination approach. The sequence coding for Oct6-P2A-mCherry was designed in silico, fully synthesized by SynBio Technologies, and cloned into a piggyBac target vector. This plasmid expresses the TET repressor together with a delta-puro antibiotic resistance gene. It also contains a TET-ON promoter followed by an AgeI and NotI restriction site where the Oct6 cassette was cloned. The HA-Oct6-P2A-mCherry cassette contains the following elements in the 5′–3′ orientation: an AgeI site followed by the Kozak sequence, an hemagglutinin (HA) tag fused to the Oct6 coding sequence in the amino termini, a HindIII site, the P2A self-cleaving peptide sequence, a MluI sequence and the mCherry coding sequence followed by a stop codon. This plasmid, named ePB-HA-Oct6-P2A-mCherry, was co-transfected into 46C Oct6 KO mESCs together with the piggyBac transposase at a ratio of 2:1, respectively, using PEI, as described previously. After puromycin selection for 10 days, clonal cell lines were generated by limiting dilution, amplified, and cryopreserved. To evaluate the expression of Oct6-P2A-mCherry cells were treated with Dox at concentrations ranging from 50 ng/ml to 2000 ng/ml. The concentration of 200 ng/ml of Dox and cell clone #3 was chosen as they produced the most homogeneous mCherry expression.
To generate the Nanog-P2A-mCherry cell line we followed a similar strategy. Briefly, the Nanog coding sequence was synthesized by SynBio Technologies and cloned into the piggybac plasmid constructed previously, between the AgeI and HindIII sites. WT 46C cells were co-transfected with this plasmid and the piggyBac transposase as previously described. Cells were treated with puromycin for 10 days to select stable transfectants. mCherry and Nanog expression was verified by Dox treatment at 200 ng/ml.
To generate the control mCherry inducibly expressed cell line, we modified the ePB-HA-Oct6-P2A-mCherry plasmid to remove the Oct6 coding sequence flanked by the AgeI and MluI sites. We exchanged this sequence with a short in vitro annealed sequence containing the AgeI and MluI overhangs and an ATG sequence to initiate the translation of the mCherry transgene. This plasmid was co-transfected into WT 46C mESCs with the piggyBac transposase as previously described. Cells were treated with puromycin for 10 days to select stable transfectants. mCherry expression was verified by Dox treatment at 200 ng/ml.
Quantitative real-time PCR
Total RNA was extracted with Trizol (Thermo Fisher Scientific) following the manufacturer’s instructions, treated with DNAse (Thermo Fisher Scientific), and reverse transcribed using MMLV reverse transcriptase (Promega). Quantitative PCR was performed in a StepOne Real-Time PCR system (Applied Biosystems). Primer efficiency and N0 values were determined by LinReg software48. Gene expression was normalized to the geometrical mean of GAPDH and PGK1 housekeeping genes, data was then log-transformed and relativized to the average of the biological replicates for the 2i + LIF condition. Statistical significance for qPCR data was analyzed by either Student-T test or randomized block design ANOVA. In the latter case, comparisons between means were assessed using the Tukey test. Plots were generated either with R or with Graphpad 8.
Quantitative immunofluorescence
For immunofluorescence experiments, cells were grown on 12 mm coverslips coated for 30 min with Geltrex (Thermo Fisher Scientific). Cells were fixed for 15 min with 4% paraformaldehyde, permeabilized with 0.1% Triton X-100 PBS (PBST), and blocked with 3% normal donkey serum in PBST. Primary antibodies (Table S4) were added in block solution, incubated at 4 °C overnight, and then washed three times in PBST for 30 min. Secondary antibodies and DAPI were incubated in block solution at room temperature for 30 min. Samples were washed as before, rinsed with distilled water, and mounted and imaged at 20X on an EVOS fluorescence microscope with adequate parameters (Thermo Fisher Scientific).
For quantifications, we followed a pipeline that included background subtraction, automatic nuclear segmentation using Stardist49, and nuclear fluorescence intensity measurements for different channels with custom Fiji/ImageJ Macros. Measurement tables were then analyzed using custom R scripts with the tidyverse library. For nearest-neighbor analysis, the central nuclear position of each cell was obtained using Fiji/ImageJ macros and data was analyzed in Python with the scikit-learn library.
Single molecule RNA-FISH
smRNA-FISH was performed using Stellaris RNA-FISH probes (Biosearch Technologies) that we designed specifically to recognize Nanog mRNAs (Table S4). For smRNA-FISH alone (no immunofluorescence), we followed the manufacturer’s protocol for adherent cells. Briefly, cells were fixed with PFA 4%, permeabilized with cold ethanol 70% for 24 h, and then treated 2 times for 3 min with FISH Wash Buffer. The wash buffer consisted of 2X sodium citrate buffer (SSC, Roche) and 5% deionized formamide (Sigma-Aldrich) in nuclease-free water. Nanog Stellaris FISH Probes Quasar® 670 were added to the hybridization buffer (HB) and incubated with coverslips containing fixed cells for 16 h at 37 °C. HB consisted of 0.1% dextran sulfate (Sigma-Aldrich), 2X SSC, and 10% deionized formamide in nuclease-free water. After incubations, cells were rinsed once with Wash Buffer and incubated with the same solution at 37 °C for 30 min. After two rinses with nuclease-free PBS, nuclei were stained with DAPI and mounted into glass slides using Prolong Diamond Antifade Mountant (Thermo Fisher Scientific).
For immunofluorescence coupled with smRNA-FISH, we first followed the regular immunofluorescence protocol for the desired antigens. After washing the secondary antibodies, cells were re-fixed with 4% PFA for 15 min and the smRNA-FISH protocol was initiated and performed as described above.
FISH images were captured in an Axio Observer Z1 epifluorescence microscope (Zeiss) using an EC Plan-Neofluar 63x/1.25 Oil M27 and the ZEISS Axiocam 702 mono camera. Z-stacks encompassing the entire volume of the cells were taken with maximum Z resolution. For the smRNA-FISH channel, each Z-stack was exposed for 3 s with maximum LED intensity. After obtaining the images, a Laplacian of Gaussian transformation was applied to the smRNA FISH channel, and a maximum intensity projection (MIP) was generated and merged with the MIPs of the other fluorescence channels.
Western blot analysis
Protein samples were collected with RIPA buffer, resolved on 10% SDS–polyacrylamide gels, and transferred to PVDF membranes (Amersham). Membranes were blocked for 1 h at room temperature in TBST 1% BSA and primary antibodies (Table S4) were incubated overnight at 4 °C in block. Peroxidase-bound secondary antibodies were incubated for 1 h at room temperature. Membranes were revealed with ECL in a G-Box System (Syngene).
Flow cytometry
Flow cytometric analysis of unfixed Sox1 + neural precursor cells or fixed immunostaining against Nanog was performed in an Accuri C6 benchtop cytometer (BD). Immunostaining was performed with similar steps as for immunofluorescence, except that cells were previously trypsinized, fixed, and stained with primary and secondary antibodies in suspension for 1 h each. Cell sorting of mCherry + live mESCs was performed on a BD FACS Aria FUSION flow cytometer. Sorted populations were immediately treated with Trizol reagent for posterior RNA extraction.
Analysis of the Nanog-Luciferase reporter
To evaluate firefly luciferase activity in response to Oct6 expression, Nanog5P reporter (Addgene #16,337) was co-transfected together with the ePB-HA-Oct6-P2A-mCherry plasmid into undifferentiated mESCs. Transfected cells were either maintained in this state or differentiated for 24 h, either with or without Dox. Luciferase activity was analyzed using as previously reported50.
RNA-seq and bioinformatic analysis
For the analysis of TFs induced during the early stages of EpiLC differentiation, processed RNA-seq data from Yang et (GSE117896) were downloaded from the GREIN database51. Raw data was fed to the into DESeq2 for differential expression analysis (fdr < 0.1) between 0 and 6 h of EpiLC induction. Among the upregulated genes, TFs were selected using the Biomart package in R, filtering by the GO term GO:0003700 “DNA-binding transcription factor activity”.
For the RNA-seq experiments performed in this work, WT and Oct6 KO cells were differentiated to EpiLCs for 48 h in 35 mm dishes as described previously, in three biological replicates. Total RNA was extracted with Trizol and its integrity was evaluated by agarose gel electrophoresis. Samples were sent to Macrogen Inc for library construction and sequencing. Libraries were prepared with TruSeq stranded total RNA with Ribo-Zero Gold and sequenced on an Illumina NovaSeq6000 platform to deliver 30 M 100 bp pair-end reads for each sample. Quality checked reads (FastQC) were processed in Fastp tool to remove any leftover adapters and low-quality ends. Processed reads were mapped to the mouse reference genome (GRCm39/mm39) using STAR aligner52 in two-pass mode, producing gene abundance quantification as well. Raw counts were then fed into DESeq2 for differential expression analysis (fdr < 0.1). Graphics and statistical analyses were performed in R. Raw data will be made available upon publication of the manuscript. The list of DE genes is available in Table S1.
For the analysis of published ChIP-seq data, we used the ChIP-atlas web service53. Bigwig and peak-called data for Oct6 EpiSCs binding profiles (SRX1410928) were downloaded from ChIP-atlas and analyzed in R using the dplyr and rtracklayer libraries. Peaks were assigned to nearby genes using GREAT54. Crossing our RNA-seq database with Oct6 EpiSCs binding profiles determined that 114 out of 292 DEG contained Oct6 peaks. To ascertain the number of genes with Oct6 peaks out of 292 random genes we used a bootstrap strategy in R with 10.000 iterations55, which allowed us to determine the random distribution of Oct6 binding for that number of genes.
Statistical analysis
Experimental results are presented as mean ± standard error of the mean (SEM) of at least three biological replicates or a representative experiment of the independent replicates. Statistical significance between groups was analyzed using either paired Student’s t-test or randomized block design (RBD) ANOVA. Residuals fitted normal distribution and homogeneity of variance. Comparisons between means were assessed using the Tukey test. Statistical analysis was performed using Infostat Software56.
Data availability
Raw RNA-seq data were deposited in Gene Expression Omnibus (GEO) under reference number GSE237157.
References
Osorno, R. et al. The developmental dismantling of pluripotency is reversed by ectopic Oct4 expression. J. Cell Sci. 125, e1–e1 (2012).
Smith, A. Formative pluripotency: The executive phase in a developmental continuum. Dev. Camb. 144, 365–373 (2017).
Morgani, S., Nichols, J. & Hadjantonakis, A. K. The many faces of Pluripotency: In vitro adaptations of a continuum of in vivo states. BMC Dev. Biol. 17, 10–12 (2017).
Nichols, J. & Smith, A. Naive and primed pluripotent states. Cell Stem Cell 4, 487–492 (2009).
Hayashi, K., Ohta, H., Kurimoto, K., Aramaki, S. & Saitou, M. Reconstitution of the mouse germ cell specification pathway in culture by pluripotent stem cells. Cell 146, 519–532 (2011).
Davidson, E. H. & Erwin, D. H. Gene regulatory networks and the evolution of animal body plans. Science 311, 796–797 (2006).
Davidson, E. H. & Levine, M. S. Properties of developmental gene regulatory networks. Proc. Natl. Acad. Sci. U. S. A. 105, 20063–20066 (2008).
Alon, U. An introduction to systems biology: Design principles of biological circuits. second edition. By Uri Alon. A Chapman & Hall Book. Boca Raton (Florida): CRC Press (Taylor & Francis Group). $143.96 (hardcover); $55.96 (paper). xviii + 324 p.; ill.; index. Q. Rev. Biol. 96, 40–40 (2021).
Alon, U. Network motifs: Theory and experimental approaches. Nat. Rev. Genet. 8, 450–461 (2007).
Hackett, J. A. & Azim Surani, M. Regulatory principles of pluripotency: From the ground state up. Cell Stem Cell 15, 416–430 (2014).
Yeo, J.-C. & Ng, H.-H. The transcriptional regulation of pluripotency. Cell Res. 23, 20–32 (2013).
Chambers, I. et al. Nanog safeguards pluripotency and mediates germline development. Nature 450, 1230–1234 (2007).
Gu, K.-L. et al. Pluripotency-associated miR-290/302 family of microRNAs promote the dismantling of naive pluripotency. Cell Res. 26, 1–17 (2016).
Buecker, C. et al. Reorganization of enhancer patterns in transition from naive to primed pluripotency. Cell Stem Cell 14, 838–853 (2014).
Acampora, D. et al. Functional antagonism between OTX2 and NANOG specifies a spectrum of heterogeneous identities in embryonic stem cells. Stem Cell Rep. 9, 1642–1659 (2017).
Bleckwehl, T. et al. Enhancer-associated H3K4 methylation safeguards in vitro germline competence. Nat. Commun. 12, 5771 (2021).
Zhu, Q. et al. The transcription factor Pou3f1 promotes neural fate commitment via activation of neural lineage genes and inhibition of external signaling pathways. eLife 2014, 1–21 (2014).
Yang, P. et al. Multi-omic profiling reveals dynamics of the phased progression of pluripotency article multi-omic profiling reveals dynamics of the phased progression of pluripotency. Cell Syst. 8, 427-445.e10 (2019).
Kim, K. P. et al. Reprogramming competence of OCT factors is determined by transactivation domains. Sci. Adv. 6, 1–17 (2020).
Atlasi, Y. et al. Epigenetic modulation of a hardwired 3D chromatin landscape in two naive states of pluripotency. Nat. Cell Biol. 21, 568–578 (2019).
Chen, A. F. et al. GRHL2-dependent enhancer switching maintains a pluripotent stem cell transcriptional subnetwork after exit from naive pluripotency. Cell Stem Cell 23, 226–238. https://doi.org/10.1016/j.stem.2018.06.005 (2018).
Narita, T. et al. Enhancers are activated by p300/CBP activity-dependent PIC assembly, RNAPII recruitment, and pause release. Mol. Cell 81, 2166-2182.e6 (2021).
Waisman, A. et al. Cell cycle dynamics of mouse embryonic stem cells in the ground state and during transition to formative pluripotency. Sci. Rep. 9, 8051 (2019).
Ying, Q.-L., Stavridis, M., Griffiths, D., Li, M. & Smith, A. Conversion of embryonic stem cells into neuroectodermal precursors in adherent monoculture. Nat. Biotechnol. 21, 183–186 (2003).
Matsuda, K. et al. ChIP-seq analysis of genomic binding regions of five major transcription factors highlights a central role for ZIC2 in the mouse epiblast stem cell gene regulatory network. Dev. Camb. 144, 1948–1958 (2017).
Boyer, L. A. et al. Core transcriptional regulatory circuitry in human embryonic stem cells. Cell 122, 947–956 (2005).
Saunders, A., Faiola, F. & Wang, J. Concise review: Pursuing self-renewal and pluripotency with the stem cell factor Nanog. Stem Cells 31, 1227–1236 (2013).
Blinka, S., Reimer, M. H., Pulakanti, K. & Rao, S. Super-enhancers at the Nanog locus differentially regulate neighboring pluripotency-associated genes. Cell Rep. 17, 19–28 (2016).
Levasseur, D. N., Wang, J., Dorschner, M. O., Stamatoyannopoulos, J. A. & Orkin, S. H. Oct4 dependence of chromatin structure within the extended Nanog locus in ES cells. Genes Dev. 22, 575–580 (2008).
Kuhlbrodt, K., Herbarth, B., Sock, E., Hermans-Borgmeyer, I. & Wegner, M. Sox10, a novel transcriptional modulator in glial cells. J. Neurosci. 18, 237–250 (1998).
Mistri, T. K. et al. Selective influence of Sox2 on POU transcription factor binding in embryonic and neural stem cells. EMBO Rep. 16, 1177–1191 (2015).
Yang, X.-C. et al. OCT6 inhibits differentiation of porcine-induced pluripotent stem cells through MAPK and PI3K signaling regulation. Zool. Res. 43, 911–922 (2022).
Suzuki, N., Rohdewohld, H., Neuman, T., Gruss, P. & Schöler, H. R. Oct-6: A POU transcription factor expressed in embryonal stem cells and in the developing brain. EMBO J. 9, 3723–3732 (1990).
Kolodziejczyk, A. A. et al. Single cell RNA-sequencing of pluripotent states unlocks modular transcriptional variation. Cell Stem Cell 17, 471–485 (2015).
Zwart, R., Broos, L., Grosveld, G. & Meijer, D. The restricted expression pattern of the POU factor Oct-6 during early development of the mouse nervous system. Mech. Dev. 54, 185–194 (1996).
Du, J., Chen, T., Zou, X., Xiong, B. & Lu, G. Dppa2 knockdown-induced differentiation and repressed proliferation of mouse embryonic stem cells. J. Biochem. (Tokyo) 147, 265–271 (2010).
Hackett, J. A. et al. Tracing the transitions from pluripotency to germ cell fate with CRISPR screening. Nat. Commun. 9, 4292 (2018).
Smith, A. & Smith, A. Mapping the Route from Naive Pluripotency to Lineage Specification (2014).
Kalkan, T. et al. Complementary activity of ETV5, RBPJ, and TCF3 drives formative transition from naive pluripotency. Cell Stem Cell 24, 785-801.e7 (2019).
Li, G. et al. Dysregulation of the SIRT1/OCT6 axis contributes to environmental stress-induced neural induction defects. Stem Cell Rep. 8, 1270–1286 (2017).
Kaya-Okur, H. S. et al. CUT&Tag for efficient epigenomic profiling of small samples and single cells. Nat. Commun. 10, 1930 (2019).
Jaegle, M. et al. The POU factor Oct-6 and Schwann cell differentiation. Science 273, 507–510 (1996).
Abranches, E., Bekman, E. & Henrique, D. Generation and characterization of a novel mouse embryonic stem cell line with a dynamic reporter of Nanog expression. PLoS ONE 8, e59928 (2013).
Liu, X. et al. USP21 deubiquitylates Nanog to regulate protein stability and stem cell pluripotency. Signal Transduct. Target. Ther. 1, 16024 (2016).
Barral, A. et al. Nanog regulates Pou3f1 expression at the exit from pluripotency during gastrulation. Biol. Open 8, 367 (2019).
Labun, K. et al. CHOPCHOP v3: Expanding the CRISPR web toolbox beyond genome editing. Nucleic Acids Res. 47, W171–W174 (2019).
Waisman, A. et al. Inhibition of Cell Division and DNA Replication Impair Mouse-Naive Pluripotency Exit. J Mol Biol 429, 2802–2815 (2017).
Ruijter, J. M. et al. Amplification efficiency: Linking baseline and bias in the analysis of quantitative PCR data. Nucleic Acids Res. 37, e45 (2009).
Schmidt, U., Weigert, M. & Broaddus, C. Cell Detection with Star-Convex Polygons Vol. 2 (Springer International Publishing, 2018).
Francia, M. et al. SUMO conjugation susceptibility of Akt/ protein kinase B affects the expression of the pluripotency transcription factor Nanog in embryonic stem cells. PLoS ONE 16, 1–20 (2021).
Mahi, N. A., Najafabadi, M. F., Pilarczyk, M., Kouril, M. & Medvedovic, M. GREIN: An Interactive Web Platform for Re-analyzing GEO RNA-seq Data. Sci. Rep. 9, 7580 (2019).
Dobin, A. et al. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21. https://doi.org/10.1093/bioinformatics/bts635 (2013).
Zou, Z., Ohta, T., Miura, F. & Oki, S. ChIP-Atlas 2021 update: A data-mining suite for exploring epigenomic landscapes by fully integrating ChIP-seq, ATAC-seq and Bisulfite-seq data. Nucleic Acids Res. 50, W175–W182 (2022).
McLean, C. Y. et al. GREAT improves functional interpretation of cis-regulatory regions. Nat. Biotechnol. 28, 495–501 (2010).
Efron, B. & Tibshirani, R. J. An Introduction to the Bootstrap. Refrig. Air Cond. 57, 436 (1993).
di Rienzo, J. A. et al. Infostat—Sofware Estadístico (Universidad Nacional de Córdoba, 2013).
Acknowledgements
We would like to thank Dr. Hernán Grecco and his lab for their help with single-molecule RNA-FISH experiments. We are also grateful to Alejandra Ventura and Gabriel Neiman for the important discussions that contributed to this work. The following grants provided financial support to this work: FONCYT: PICT-2011-1927, PICT-2015-1469, and PID-2014-0052; CONICET: PIP2015-2017, PICT-2018-02672, PICT-2018-00836, PICT-2018-01722, PICT-2020-03553, PICT2020-03523. We would also like to thank Fundación FLENI and Fundación Perez Companc for their continuous support.
Author information
Authors and Affiliations
Contributions
A.W. conceived, performed, and analyzed most of the experiments. D.S. and A.W. constructed the Nanog overexpressing cell line and analyzed the overexpression of Nanog in differentiating cells. A.W. and F.S. performed the Oct6 Dox reversion experiments and helped with many immunofluorescence experiments. M.F. performed the luciferase reporter experiments. A.L., C.B., B.P., R.B., and G.A. contributed to proteomic and immunofluorescence analyses. A.L.G., A.S., J.S., and A.W. oversaw bioinformatic analyses. L.M., G.S., and A.G. were fundamental in their contribution to useful discussions. AW and SM discussed all the experiments and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Waisman, A., Sevlever, F., Saulnier, D. et al. The transcription factor OCT6 promotes the dissolution of the naïve pluripotent state by repressing Nanog and activating a formative state gene regulatory network. Sci Rep 14, 10420 (2024). https://doi.org/10.1038/s41598-024-59247-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-59247-5
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.