Abstract
Elevated remnant cholesterol (RC) is considered a risk factor for atherosclerotic cardiovascular disease, but the evidence on this association applies to the Chinese population with hypertension is limited. We aimed to explore the association between RC levels and carotid plaque in old adults with hypertension. 8523 hypertensive patients aged ≥ 60 years with serum lipids and carotid ultrasonography data were included in this community-based screening. Fasting RC was calculated as total cholesterol minus high-density lipoprotein cholesterol minus low-density lipoprotein cholesterol (LDLC). The associations of RC levels with carotid plaque risk were evaluated using Logistic regression and restricted cubic spline models. Carotid plaque was screened in 4821 (56.56%) subjects. After multivariable-adjusted, RC was significantly related to carotid plaque [Odd ratio (OR)] = 1.043 per 0.1 mmol/L increase, 95% confidence interval (CI): 1.030–1.056). The highest versus the lowest quartile of RC was 1.928 (1.673–2.223) for carotid plaque. A nonlinear association was found between serum RC levels and the risk of carotid plaque (P for nonlinearity < 0.001). Moreover, an RC > 0.78 mmol/L differentiated patients at a higher risk of carotid plaque compared to those at lower concentrations, regardless of whether LDLC was on target at 2.59 mmol/L. In old adults with hypertension, elevated RC was positively associated with carotid plaque, independent of LDLC and other conventional risk factors.
Similar content being viewed by others
Introduction
Carotid plaque formation is a significant risk factor for atherosclerotic cardiovascular disease (ASCVD), which is associated with future risk of stroke, transient ischemic attack, and acute myocardial infarction1,2. ~ 30% of Chinese have carotid plaques, and ~ 45% of the old adults are aged ≥ 60 years. The progression of carotid artery atherosclerosis increases more rapidly with age in Chinese than in Europeans3. Reducing low-density lipoprotein cholesterol (LDLC) can decrease atherosclerotic risk in clinical practice4, but a significant residual cardiovascular risk persists in individuals treated with statins5,6. This residual risk may be partly due to the increased concentration of remnant cholesterol (RC)7,8,9. RC is the cholesterol content of triglyceride-rich lipoproteins (TGRL). It comprises very low-density lipoprotein (VLDL) and intermediate-density lipoprotein in the fasting state and chylomicron remnants in the non-fasting state10. Like LDLC, RC can be trapped in the intima of the arterial wall, causing arterial intimal cholesterol deposition, vascular inflammation and subsequent plaque formation11,12. Several observational population-based studies have shown that elevated RC accelerated common carotid artery intima-media thickness (cIMT), promoted carotid plaque formation, and even increased the risk of atherosclerotic plaque rupture13,14,15,16, while these studies were mainly conducted in north China and the sample sizes were not large. Notably, an RC increase preceded the development of hypertension17, and dyslipidemia combined with hypertension could elevate atherosclerosis risk18,19,20. Limited studies have investigated the relationship between RC and carotid plaque in hypertension. Patients with hypertension are at higher risk of atherosclerosis risk21. Accurate identification of high-risk patients in the early stages of atherosclerosis events is necessary to control atherosclerosis progression timely.
Therefore, the present study aimed to evaluate the association between RC and the risk of carotid plaque in hypertension through a community-based screening. Additionally, we investigated whether the risk of carotid plaque associated with RC was independent of LDLC.
Methods
Study design and population
We conducted a study collecting individual-level data from a community-based screening of “High-Risk Stroke Population Screening and Intervention”. This screening was supported by the local Centers for Disease Control and Prevention (CDC) and the medical service community between May and July 2020 in Fuyang, Hangzhou. The participants with hypertension and/or diabetes ≥ 60 years old were selected from the service contractor of the family doctor of their community health service centres, and they would also be informed of the details of the screening, including a questionnaire survey, physical examination, and carotid ultrasonography, by their family doctor. Participants meeting the following criteria were excluded from the screening: (1) Mental disorders; (2) Long-term disability; (3) Refuse to attend follow-up. This screening included a sample of 15,561 participants according to the above exclusion criteria. For the present study, of the 11,433 participants with hypertension who were included, 2910 participants with incomplete information on carotid artery imaging, biochemical indexes or conventional risk factors were excluded. Finally, 8523 participants were included in this analysis (Supplementary Fig. 1).
Data collection
In this study, a trained community health worker collected detailed information on the risk factors of stroke using a standard questionnaire: demographic variables (sex and age), lifestyle (smoking and physical activity), histories of disease (stroke, transient ischemic attacks, and atrial fibrillation/valvular heart disease), and medication information. Smoking was defined as continuous or cumulative smoking for 6 months or more in a lifetime. Regular exercise was defined as exercise ≥ 3 times per week and moderate or above intensity exercise ≥ 30 min per time or engaged in moderate or heavy manual labour. The disease history records required a diagnosis from a middle or above-grade hospital. Height and weight were measured with participants wearing light dressing and without shoes. Body mass index (BMI) was calculated as weight/height2 (kg/m2). Blood pressure was measured using a standardized automatic electronic sphygmomanometer (HEM-741C; Omron, Tokyo, Japan).
Laboratory measurements
Blood samples were collected in the morning from each participant's antecubital vein who had fasted for at least 8 h.
Serum creatinine was measured by the alkaline picrate method. The estimated glomerular filtration rate (eGFR) was calculated as follows: eGFR = 175 × Scr−1.234 × age−0.179(if female, × 0.79), where Scr is serum creatinine concentration (in mg/dL) and age in years22. Fasting glucose was measured by glucose-oxidase method, total cholesterol (TC) by esterase oxidase-peroxidase, triglycerides (TG) by glycerol phosphate oxidase-peroxidase, and high-density lipoprotein cholesterol (HDLC) by direct measurement after phosphotungstate precipitation. LDLC was calculated by the Friedewald formula when plasma triglycerides were ≤ 4.0 mmol/L and otherwise measured directly23. RC was estimated from a standard lipid profile in the fasting state as TC minus LDLC minus HDLC. The above blood indicators were analyzed using the Roche Cobas automatic biochemical analyzer in an accredited central laboratory in the First People's Hospital of Fuyang (Hangzhou, China), and all laboratory equipment has been calibrated to reduce systematic error effectively.
Ultrasound examination
Carotid ultrasonography was performed by qualified sonographers who received unified training from the local medical service community, including learning the protocol of screening, unifying the operation procedure of carotid artery ultrasonography, and mastering emergency skills and methods. All sonographers had a professional certificate for ultrasound measurement awarded by the Ministry of Health of China and were blinded to the questionnaire results. High-resolution sonography machines (LOGIQ E9, Chicago, IL, USA) were used for carotid ultrasound with a 10-MHz probe. All participants were studied supine with their heads turned 45 degrees from the site being scanned. The carotid plaques were examined on the left and right sides, with each side measured at three different locations: common carotid artery, carotid bifurcation, and internal carotid artery, and the cIMT was measured. Carotid plaques were defined as focal structures encroaching into the arterial lumen of at least 0.5 mm or 50% of the surrounding cIMT value or demonstrating a thickness > 1.5 mm as measured from the intima-lumen interface to the media-adventitia interface24. The ultrasonography results were reviewed by two independent sonographers, and discrepancies were resolved by consensus.
Statistical analysis
Continuous variables were presented as the means and standard deviation (SD), and categorical variables were expressed as numbers (percentages). Values between groups were compared using variance (ANOVA) or independent t-test and Chi-square test or Fisher’s exact test, as appropriate. Logistic regressions were conducted to assess the association between remnant cholesterol levels and carotid plaque and to calculate the odds ratios (ORs) and 95% confidence intervals (CIs). We constructed 3 models: In model 1, we unadjusted; in model 2, we adjusted for sex and age; in model 3, we adjusted for sex, age, and a propensity score. The propensity score was calculated with a linear regression model entering RC as the dependent variable. The independent variables included smoking, regular exercise, history of stroke, history of transient ischemic attacks (TIA), history of atrial fibrillation /valvular heart disease, family history of stroke, diabetes, BMI, blood glucose, TG, LDLC, HDLC, eGFR, SBP, DBP, antihypertensive medications, and lipid-lowing medications. In these multivariable Logistic models, RC was grouped into quartiles: Q4, > 0.94 mmol/L; Q3, 0.65–0.94 mmol/L; Q2, 0.44–0.65 mmol/L; Q1, ≤ 0.44 mmol/L. RC was entered as a categorical variable with Q1 as the reference group or as a continuous variable per 0.1 mmol/L (3.89 mg/dL) increment. A restricted cubic spline with three knots at the 25th, 50th, and 75th centiles was conducted to flexibly model the association between the RC and carotid plaque risk with adjusted model 3. In addition, we calculated the predicted probabilities of the Logistic model adjusted with/without adjusting for RC, constructed receiver operating characteristic (ROC), and computed the areas under the curves (AUC) to assess the predictive power of each model. Subgroup analyses were also carried out using multivariable Logistic models stratified by traditional risk factors and summarized with forest plots. An interaction term within each subgroup was performed using the likelihood ratio test. For sensitivity analysis, we analyzed the association between RC levels and carotid plaque by excluding participants who were taking lipid-lowering medications. We also employed the E-value as a measure to evaluate the stability of Logistic regression models. This metric aids in assessing whether an observed association may be influenced by an unmeasured underlying factor. Statistical analysis was conducted using SAS 9.4 (SAS Institute Inc., Cary, NC, USA). Two-sided P-values < 0.05 were considered statistically significant.
Ethics approval and consent to participate
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Hangzhou Center for Disease Control and Prevention Research Ethics Committee (Ethics approval number: 2023-5). Informed consent was obtained from all individual participants included in the study.
Results
Participants characteristics
The characteristics of study participants were presented in RC quartiles in Table 1. The mean age was 69.91 ± 5.64 years, and 54.99% of participants were female. Participants in the highest quartiles of RC were more likely to be female and had a higher BMI, blood glucose, TC, and TG as compared to the other groups (all P < 0.001). The proportions of history of cardiovascular diseases, diabetes, and smoking were more frequent in participants with the lowest quartiles of RC (all P < 0.001), while the age of participants was relatively older in the lowest quartiles of RC. The median RC was 0.65 mmol/L (interquartile range, 0.44–0.94 mmol/L). Participants with carotid plaque had a higher level of TC, TG, LDLC, and RC and a lower level of HDLC (Supplemental Table 1). Smoking, history of atrial fibrillation/valvular heart disease, diabetes, TG, HDLC, LDLC, BMI, SBP, and DBP were independently associated with RC levels (Supplemental Table 2).
RC and carotid plaque risk
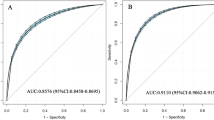
Multivariate Logistic regression analysis revealed that (Table 2), in model 1 (crude model), compared with Q1, participants with higher RC levels were significantly associated with an increased risk of carotid plaque (Q2: OR 1.555, 95% CI 1.378–1.754; Q3: OR 1.838, 95% CI 1.628–2.075; and Q4: OR 2.157, 95% CI 1.908–2.439). In the fully adjusted model (model 3), RC remained a significant risk factor for carotid plaque (Q2 vs. Q1: OR 1.489, 95% CI 1.316–1.683; Q3 vs. Q1: OR 1.721, 95% CI 1.516–1.953; and Q4 vs. Q1: OR 1.928, 95% CI 1.673–2.223; per 0.1 mmol/L increment: OR 1.043, 95%CI 1.030–1.056). In Fig. 1, the restricted cubic splines showed that higher levels of RC were associated with a progressively increased risk of carotid plaque (P-nonlinearity < 0.001). There was a substantial increase in the associated risk until around 0.75 mmol/L (29.18 mg/dL). The ROC analysis based on Logistic models without/with RC was presented in Fig. 2. The first model had an AUC of 0.605 (95% CI: 0.594–0.615). The second model included RC enabled a more accurate prediction of carotid plaque (AUC 0.617, 95% CI 0.606–0.627) and showed a significant model improvement (ΔAUC 0.012, 95% CI 0.007–0.017, P < 0.001 by DeLong’s test). Based on the combinations of each RC and LDLC cutoff value9, participants were categorized into four groups (Fig. 3). Compared to lower concentrations of RC within the same LDLC levels, even in participants with controlled LDLC, high RC was associated with a 66.0% higher risk of carotid plaque in RC&LDLC (> 0.78 & ≤ 2.59 mmol/L) group (OR 1.660, 95% CI 1.435–1.919).
Association of remnant cholesterol with carotid plaque. Red lines indicate hazard ratios, green line indicates the reference line (The minimum of RC as a reference point), and dashed lines indicate 95% CI from restricted cubic spline regression. ORs were calculated using Logistic regression analysis after adjustments for sex, age and a propensity score. RC remnant cholesterol; OR odds ratio; CI confidence interval.
Receiving operating characteristics curves for carotid plaque from Logistic regression models with and without remnant cholesterol in patients with hypertension. AUCs were performed containing: (1) sex, age and a propensity score, and (2) RC added. A comparison between the AUCs is noted in the bottom panel. RC remnant cholesterol; AUC areas under the curves.
Risk of carotid plaque based on categories of remnant cholesterol and LDLC. Data were adjusted for age, sex, smoking, regular exercise, history of stroke, history of TIA, history of atrial fibrillation/valvular heart disease, family history of stroke, diabetes, blood glucose, BMI, TG, HDLC, eGFR, SBP, DBP, antihypertensive medications, and lipid-lowing medications. OR odds ratio; CI confidence interval. Other abbreviations as in Table 1.
Subgroup analysis
Sex, age, BMI, smoking, regular exercise, diabetes, antihypertensive drug, hypertension control and eGFR levels were stratified for further analysis of the association between RC and carotid plaque (Fig. 4). Positive associations were found in various subgroups, except for associations stratified by antihypertensive medications. The higher risk of carotid plaque with RC levels persisted notably, regardless of sex, age, BMI, regular exercise, diabetes and eGFR levels. The association between RC level and carotid plaque risk was stronger in smoking and in participants with controlled hypertension.
Risk of carotid plaque for the highest vs. lowest quartile of remnant cholesterol in various subgroups adjusted for sex, age, smoking, regular exercise, history of stroke, history of TIA, history of atrial fibrillation/valvular heart disease, family history of stroke, diabetes, BMI, blood glucose, SBP, DBP, TG, LDLC, HDLC, eGFR, antihypertensive medications, and lipid-lowering medications other than variables for stratification. ORs for per 0.1 mmol/L increased in RC. P-value for interaction was calculated. Hypertension uncontrolled was defined as SBP ≥ 140 and/ or DBP ≥ 90 mmHg in this screening. OR odds ratio; CI confidence interval. Other abbreviations as in Table 1.
Sensitivity analysis
When excluding participants who were taking lipid-lowering medications, the results did not change substantially. In model 3, RC was positively correlated with the carotid plaque risk (per 0.1 mmol/L increment, OR 1.043, 95% CI 1.030–1.055). Compared with Q1, participants with higher RC levels were still significantly associated with an increased risk of carotid plaque (Q2: OR 1.487, 95% CI 1.315–1.681; Q3: OR 1.717, 95% CI 1.512–1.949; and Q4: OR 1.919, 95% CI 1.665–2.212) (Supplemental Table 3). We also calculated an E-value calculation to assess the potential impact of unaccounted confounders. An E-value of 1.17, with a lower bound of the 95% CI at 1.14. These confounding factors would need to have an OR of at least 1.17 to account for the observed association (Supplemental Fig. 2).
Discussion
Our results demonstrate that in older adults with hypertension, the association between RC and risk of carotid plaque was nonlinear. Higher RC levels were associated with an increased risk of carotid plaque, even in participants at the target range of LDLC levels. Adding the RC to a traditional risk model had an incremental effect on the discriminative ability of carotid plaque.
The association between high RC and ASCVD has been explored in numerous studies25, which indicated that elevated RC levels could increase the risk of ASCVD as well as coronary heart disease and stroke. The evidence for the risk of carotid plaque, as a surrogate marker of ASCVD risk26, with RC would be valuable. Cross-sectional studies showed that elevated fasting RC concentrations were positively associated with mean cIMT and maximum cIMT in 1496 patients with ischemic stroke13, and a high RC level indicated a higher risk of intra- and extra-cranial atherosclerosis in 3665 community residents16. Even among 767 youth, elevated serum RC levels were associated with increased cIMT27. Consistently, coronary artery disease patients with high RC are more likely to have atherosclerotic plaque and plaque rupture15,28. The above findings were in line with ours, whereas their sample size was not large, or the age distribution of the study population was different. The present study observed a nonlinear positive association between RC levels and carotid plaque risk in hypertension. Maintaining RC levels as low as possible was recommended to reduce the risk of carotid plaque. The effect of hypertension on RC-carotid plaque links was inconclusive. History of hypertension did not modify the relationship between RC and vascular disease13,29, while high RC levels could potentiate hypertension development and increase the risk of first-ever stroke, partially through the pathway of hypertension17,30. The increased carotid plaque risk associated with a high RC level in hypertension is due, in part, to BP-related atherogenic dyslipidemia. Hypertension is closely related to dyslipidemia, which is characterized by the increase of plasma triglyceride, VLDL, and atherogenic LDLC particles and the decrease of HLDC31. Interestingly, a study of 45 severe carotid artery stenosis patients found that cholesterol in the dense LDL and in the triglyceride-rich lipoprotein remnants, not LDLC, HDLC or TG levels, affected carotid plaque and carotid plaque inflammatory cellular composition32. On the one hand, TG is unlikely to be an independent risk factor for vascular disease33. Although according to the used Friedewald equation in the present study, RC equals TG divided by 5, TG could not directly cause atherosclerotic plaques. TG-rich chylomicrons are too large to enter the arterial wall and are phagocytosed by vascular macrophages34. Nordestgaard et al. reported that the harmful component in TRLs was cholesterol, not TG itself10. On the other hand, studies on the effect of elevated HDLC levels on vascular disease risk were inconsistent. Valentina et al. reported a nonlinear U-shaped association between HDLC levels and CVD risk in 6523 males with hypertension but not females35. Lipoproteomics revealed that medium and large HDL subclasses levels were negatively associated with higher cIMT, whereas very large HDL were positively associated with carotid plaque36. The increase in HDLC levels may fail to reduce vascular disease risk37. These findings supported that TG may just be a marker of elevated RC. Thus, the assessment of RC, an independent residual risk factor for hypertensive patients, could be more meaningful than TG or HDLC to provide additional information on the primary prevention of ASCVD.
In the present study, ROC analysis revealed that adding RC into a model resulted in a significant improvement in its discriminant ability of risk of carotid plaque in hypertensive patients. Our study implied that the assessment of RC may help improve the prognostic risk of future carotid plaque information in old adults with hypertension. In addition, we pointed out that the median RC was 0.65 mmol/l (interquartile range, 0.44–0.94 mmol/l), which was similar to that of the stroke patients but higher than that of the general population13,14. Ageing and hypertension may partly explain the relatively high RC concentrations. Future natural population cohort studies are needed to estimate the concentration distribution of RC to obtain a normal or low-risk physiological concentration range.
In this study, we also found that individuals with high RC concentration (> 0.78 mmol/L) had a higher risk of carotid plaque, regardless of whether LDLC concentration was or was not at optimal values (≤ 2.59 mmol/L). This finding was consistent with others from previous studies which were conducted in specific ethnic and disease groups25. Our conclusions extended these prior findings and implicated that for patients at the target LDLC levels, treatments to control RC to reduce the residual risk of ASCVD were more beneficial. In the subgroup analysis, we did not find an association between RC and carotid plaque among participants who did not take antihypertensive medications. We assumed the following possible explanations: First, the sample may be insufficient, with only 1.33% (114/8523). An analysis of this factor stratification may be underpowered. Additionally, with regards to hypertension control, given the specificity of the effects of antihypertensive medications on lipid metabolism38,39, the dosage and frequency should also be collected in future studies to evaluate the lipid metabolism in patients with hypertension comprehensively.
Potential mechanistic links
Previous observational and genetic epidemiological data have indicated a causal role of cholesterol content within TGRL and/or RC in the development of ASCVD. The strong effect of RC on ASCVD can be explained by the following mechanisms. Remnant lipoprotein cholesterol penetrates the endothelial layer and traps into the arterial intima. However, unlike LDLC, it can be absorbed directly by macrophages, which contributes to the formation of foam cells and atherosclerotic plaques40. RC is larger than LDLC and carries up to 40 times more cholesterol per particle, which may make RC more likely to cause arteriosclerosis than LDLC41. Elevated RC is related to low-grade inflammation11, causing arterial wall inflammation and a multilevel cellular immune response12. It is also related to the content of macrophages in the carotid artery, which is a marker of plaque instability32. RC is regarded as an indicator of endothelial vasomotor dysfunction and upregulates the expression of proatherothrombogenic molecules mediated by redox-sensitive mechanisms42. In summary, the combination of arterial intima accumulation, inflammation and endothelial vasomotor dysfunction could partially explain the increased risk of arterial plaque formation.
Merits and limitations
The main strength of this study was that we reported novel associations between RC and carotid plaque in hypertension in Chinese old adults. Our findings provided community-based evidence to support the hypothesis that RC was a risk factor for ASCVD and served as an explanation for the mechanism of RC-CVD links. Besides, RC could be obtained from the standard lipid profile without additional cost. This measurement was beneficial to be widely used in clinical practice to identify high-risk patients early.
Some limitations should be considered in the light of the present results. Firstly, the present study population was restricted to an old adults hypertensive sample in East China, which induced some selection bias and limited the generalizability of the present results. Secondly, given the nature of the cross-sectional study, the dynamic change effects of RC levels on carotid plaque formation were unable to be assess. A prospective cohort study was needed to clarify a cause-effect. Thirdly, we use fasting lipid samples to calculate RC. Thus, in the non-fasting state, it remains plausible that chylomicron remnants partially contribute to carotid plaque. However, RC was slightly but not clinically significant after a habitual meal. In this study, the strength of association between RC and carotid plaque may be underestimated by the use of fasting samples, but the association was still significant after the multivariate adjustment. Similar studies have also used fasting samples to calculate RC25, as in our study. Fourthly, carotid artery imaging was operated by a single sonographer, and we could not evaluate inter-rater variability for the measurements and the residual bias from diagnostic suspicion. Finally, some deficiencies may exist in the information on lifestyle factors, hypertension duration, and medication adherence. These could affect lipid metabolism and introduce residual bias.
Conclusions
In conclusion, elevated levels of RC were associated with carotid plaque in patients with hypertension independently of traditional cardiovascular risk factors. Our findings strengthened the evidence for the necessity of monitoring RC for vascular health in hypertension. RC may serve as a potential risk factor in community prevention and clinical intervention, even in people at optimal LDLC levels.
Data availability
The data presented in this study are available on request from the corresponding authors. The data are not publicly available due to ethical, legal, and privacy issues.
References
Hollander, M. et al. Carotid plaques increase the risk of stroke and subtypes of cerebral infarction in asymptomatic elderly: The Rotterdam study. Circulation 105(24), 2872–2877 (2002).
Jashari, F., Ibrahimi, P., Bajraktari, G., Wester, P. & Henein, M. Y. Carotid plaque echogenicity predicts cerebrovascular symptoms: A systematic review and meta-analysis. Eur. J. Neurol. 23(7), 1241–1247 (2016).
Clarke, R. et al. Burden of carotid artery atherosclerosis in Chinese adults: Implications for future risk of cardiovascular diseases. Eur. J. Prev. Cardiol. 24(6), 647–656 (2017).
Cholesterol Treatment Trialists’ (CTT) Collaboration et al. Efficacy and safety of more intensive lowering of LDL cholesterol: A meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet 376(9753), 1670–1681 (2010).
Wong, N. D. et al. Residual atherosclerotic cardiovascular disease risk in statin-treated adults: The Multi-Ethnic Study of Atherosclerosis. J. Clin. Lipidol. 11(5), 1223–1233 (2017).
Mora, S. et al. Determinants of residual risk in secondary prevention patients treated with high-versus low-dose statin therapy: The Treating to New Targets (TNT) study. Circulation 125(16), 1979–1987 (2012).
Jepsen, A. M. et al. Increased remnant cholesterol explains part of residual risk of all-cause mortality in 5414 patients with ischemic heart disease. Clin. Chem. 62(4), 593–604 (2016).
Sandesara, P. B. et al. The forgotten lipids: Triglycerides, remnant cholesterol, and atherosclerotic cardiovascular disease risk. Endocr. Rev. 40(2), 537–557 (2019).
Castañer, O. et al. Remnant cholesterol, not LDL cholesterol, is associated with incident cardiovascular disease. J. Am. Coll. Cardiol. 76(23), 2712–2724 (2020).
Nordestgaard, B. G. & Varbo, A. Triglycerides and cardiovascular disease. Lancet 384(9943), 626–635 (2014).
Varbo, A. et al. Elevated remnant cholesterol causes both low-grade inflammation and ischemic heart disease, whereas elevated low-density lipoprotein cholesterol causes ischemic heart disease without inflammation. Circulation 128(12), 1298–1309 (2013).
Bernelot Moens, S. J. et al. Remnant cholesterol elicits arterial wall inflammation and a multilevel cellular immune response in humans. Arterioscler. Thromb. Vasc. Biol. 37(5), 969–975 (2017).
Qian, S. et al. Remnant cholesterol and common carotid artery intima-media thickness in patients with ischemic stroke. Circ. Cardiovasc. Imaging 14(4), e010953 (2021).
Liu, B. et al. Association of remnant cholesterol and lipid parameters with new-onset carotid plaque in Chinese population. Front. Cardiovasc. Med. 9, 903390 (2022).
Gao, Y. et al. The relationship between residual cholesterol risk and plaque characteristics in patients with acute coronary syndrome: Insights from an optical coherence tomography study. Atherosclerosis 317, 10–15 (2021).
Wang, A. et al. Association of remnant cholesterol with intra- and extra-cranial atherosclerosis in Chinese community population. Atheroscler. Plus 46, 20–26 (2021).
Chen, M. M. et al. High remnant cholesterol level potentiates the development of hypertension. Front. Endocrinol. (Lausanne) 13, 830347 (2022).
Kostis, J. B. The importance of managing hypertension and dyslipidemia to decrease cardiovascular disease. Cardiovasc. Drugs Ther. 21(4), 297–309 (2007).
Si, X. B. & Liu, W. Relationship between blood lipid and arterial stiffness in hypertension. Clin. Invest. Med. 42(3), E47–E55 (2019).
Liu, J. et al. The role of blood pressure in carotid plaque incidence: interactions with body mass index, age, and sex-based on a 7-years cohort study. Front. Physiol. 12, 690094 (2021).
Cai, A. et al. Associations of systolic and diastolic blood pressure night-to-day ratios with atherosclerotic cardiovascular diseases. Hypertens. Res. 39(12), 874–878 (2016).
Zhang, L., Wang, F., Wang, L. et al. Prevalence of chronic kidney disease in China: A cross-sectional survey [published correction appears in Lancet 380 (9842), 650 (2012)]. Lancet 379 (9818), 815–822 (2012).
Friedewald, W. T., Levy, R. I. & Fredrickson, D. S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 18(6), 499–502 (1972).
Touboul, P. J. et al. Mannheim carotid intima-media thickness and plaque consensus (2004–2006–2011). An update on behalf of the advisory board of the 3rd, 4th and 5th watching the risk symposia, at the 13th, 15th and 20th European Stroke Conferences, Mannheim, Germany, 2004, Brussels, Belgium, 2006, and Hamburg, Germany, 2011. Cerebrovasc. Dis. 34(4), 290–296 (2012).
Chen, X. & Li, L. H. Remnant cholesterol, a valuable biomarker for assessing arteriosclerosis and cardiovascular risk: A systematic review. Cureus 15(8), e44202 (2023).
Zhu, G. et al. Carotid plaque imaging and the risk of atherosclerotic cardiovascular disease. Cardiovasc. Diagn. Ther. 10(4), 1048–1067 (2020).
Di Costanzo, A. et al. Elevated serum concentrations of remnant cholesterol associate with increased carotid intima-media thickness in children and adolescents. J. Pediatr. 232, 133-139.e1 (2021).
Lin, A. et al. Remnant cholesterol and coronary atherosclerotic plaque burden assessed by computed tomography coronary angiography. Atherosclerosis 284, 24–30 (2019).
Liu, J. et al. Association between remnant cholesterol and arterial stiffness in a Chinese community-based population: A cross-sectional study. Front. Cardiovasc. Med. 9, 993097 (2022).
Li, H. et al. Association and mediating mechanism between remnant cholesterol and first-ever stroke among the Chinese general population. Front. Neurosci. 17, 1161367 (2023).
Zicha, J., Kunes, J. & Devynck, M. A. Abnormalities of membrane function and lipid metabolism in hypertension: A review. Am. J. Hypertens. 12(3), 315–331 (1999).
Zambon, A. et al. Lipoprotein remnants and dense LDL are associated with features of unstable carotid plaque: A flag for non-HDL-C. Atherosclerosis 230(1), 106–109 (2013).
Emerging Risk Factors Collaboration et al. Major lipids, apolipoproteins, and risk of vascular disease. JAMA 302(18), 1993–2000 (2009).
Teramoto, R. et al. Molecular and functional characterization of familial chylomicronemia syndrome. Atherosclerosis 269, 272–278 (2018).
Trimarco, V. et al. High HDL (high-density lipoprotein) cholesterol increases cardiovascular risk in hypertensive patients. Hypertension 79(10), 2355–2363 (2022).
Blaauw, M. J. T. et al. Traditional cardiovascular risk factors are stronger related to carotid intima-media thickness than to presence of carotid plaques in people living with HIV. J. Am. Heart Assoc. 12(20), e030606 (2023).
Lincoff, A. M. et al. Evacetrapib and cardiovascular outcomes in high-risk vascular disease. N. Engl. J. Med. 376(20), 1933–1942 (2017).
Ames, R. P. Antihypertensive drugs and lipid profiles. Am. J. Hypertens. 1(4 Pt 1), 421–427 (1988).
Deshmukh, M., Lee, H. W., McFarlane, S. I. & Whaley-Connell, A. Antihypertensive medications and their effects on lipid metabolism. Curr. Diabetes Rep. 8(3), 214–220 (2008).
Varbo, A. & Nordestgaard, B. G. Remnant lipoproteins. Curr. Opin. Lipidol. 28(4), 300–307 (2017).
Joshi, P. H., Martin, S. S. & Blumenthal, R. S. The remnants of residual risk. J. Am. Coll. Cardiol. 65(21), 2276–2278 (2015).
Doi, H. et al. Remnant lipoproteins induce proatherothrombogenic molecules in endothelial cells through a redox-sensitive mechanism. Circulation 102(6), 670–767 (2000).
Acknowledgements
The authors thank the community health workers and participants of the present study for their important contributions.
Funding
This work was supported by the Hangzhou General Research Project on Agriculture and Social Development [Grant # 20201203B207], and Hangzhou Health Science and Technology Projects [Grant # A20230326].
Author information
Authors and Affiliations
Contributions
Z.Y. and H.Y. contributed equally. They were the main investigator of this study and draft the manuscript. C.J. and J.X. conceived the study and took part in study design. Z.C. and Y.Y. took part in data collection and statistical analysis. B.S. helped to draft the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yu, Z., Yang, H., Shou, B. et al. Remnant cholesterol and the risk of carotid plaque in hypertension: results from a community-based screening among old adults in Hangzhou, China. Sci Rep 14, 8407 (2024). https://doi.org/10.1038/s41598-024-58484-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-58484-y
Keywords
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.