Abstract
To maintain current cognitive function and access greater cognitive reserves, nonpharmacological interventions may be a viable alternative for older adults with or without cognitive impairment. This study aimed to compare different nonpharmacological interventions for enhancing global cognition, including mind–body exercise, physical exercise, non-invasive brain stimulation, cognitive training intervention (CTI), acutherapy (ACU), meditation, and music therapy, by applying a network meta-analysis (NMA). Sixty-one randomized controlled trials evaluating the efficacy of interventions on global cognition in older adults with or without mild cognitive decline were selected. An NMA was conducted to compare the efficacy of different nonpharmacological interventions. The NMA revealed that mind–body exercise (standardized mean difference, 1.384; 95% confidence interval, 0.777–1.992); ACU (1.283; 0.478–2.088); meditation (0.910; 0.097–1.724); non-invasive brain stimulation (1.242; 0.254–2.230); CTI (1.269; 0.736–1.802); and physical exercise (0.977; 0.212–1.742), showed positive effects compared to passive controls. There were no significant differences between the efficacies of other interventions. Nonpharmacological interventions may potentially enhance and maintain global cognition through various pathways, such as memorizing movements and enhancing brain plasticity by reducing stress in the older adult population. Additional studies are needed to clarify the impact of other variables, including intervention methods or psychological variables.
Similar content being viewed by others
Introduction
The global population is experiencing rapid aging. The World Health Organization (WHO) predicts that the proportion of older adults (aged > 60 years) will nearly double, from 12 to 22%, between 2015 and 20501. With an increasingly aging population, cognitive deterioration has become a widespread phenomenon2. Current estimates indicate that almost 47 million people worldwide have dementia, with an expected increase to 75 million by 20303. This disease imposes a significant economic burden on society, with the total global societal cost of dementia estimated at US$ 818 billion in 2015 and projected to reach US$ 1 trillion annually by 20304.
To maintain current cognitive function and access greater cognitive reserves, nonpharmacological interventions could serve as a viable alternative to pharmacological interventions for older adults with or without cognitive impairment. Nonpharmacological interventions are likely to be more affordable to develop and implement than pharmacological treatments, while also producing minimal side effects, even when lacking significant efficacy5.
Substantial evidence has accumulated from neuroimaging studies showing that nonpharmacological intervention is as effective as pharmacological intervention among older adults6,7. Therefore, nonpharmacological interventions, including primary and secondary prevention programs, can be applied in older adults in the early stages of cognitive decline8.
Given the benefits of nonpharmacological interventions, recent research has focused on their efficacy, including cognitive training intervention (CTI)9,10, music therapy11,12, acutherapy (ACU), such as acupuncture and acupressure,13,14, meditation15,16,17, physical exercise18,19, mind–body exercise15,20, and non-invasive brain stimulation21,22,23, for preventing cognitive decline in older adults with or without cognitive impairment. Review and meta-analysis studies of these interventions have demonstrated their effectiveness in maintaining or improving cognitive abilities.
A recent meta-analysis of prospective studies including patients with neurodegenerative diseases reported a small-to-moderate efficacy of CTI when compared to control conditions in global cognitive function in addition to various cognitive domains including, memory, and executive function10,24. Musical interventions can help prevent neurodegeneration25,26. One meta-analysis demonstrated the beneficial but minimal efficacy of music therapy on global cognition in patients with dementia27. A previous meta-analysis demonstrated that ACU intervention was more effective than Western medication for improving global cognition in individuals with mild cognitive impairment (MCI)13,28,29. The results of another meta-analysis indicated that non-invasive brain stimulation (transcranial magnetic stimulation [TMS] and transcranial direct current stimulation [tDCS]) has a positive influence, with a small to moderate effect size, in ameliorating cognitive decline in healthy older adults and patients with Alzheimer’s disease (AD)22,23,30.
Additionally, a meta-analysis reported that physical exercise was associated with a 28% reduction in incident dementia31. Furthermore, a growing number of meta-analyses have suggested that mind–body exercise may improve cognitive function in older adults15,20,32. Moreover, previous research has suggested that meditation may contribute to cognitive well-being and emotional balance by enhancing brain regions associated with interoception and attention33,34.
These studies suggested that nonpharmacological intervention may enhance cognitive function by increasing the levels of growth factors, modulating inflammatory cytokines, oxidative stress, and autophagy, and attenuating amyloid-beta pathology35,36,37,38.
While these studies provide an overview of the efficacy and safety of nonpharmacological interventions for global cognition in older patients, they predominantly compare only two interventions (i.e., control condition vs. nonpharmacological intervention), which allows for traditional pairwise meta-analysis. Consequently, a significant knowledge gap exists regarding the relative efficacy of each intervention for cognitive decline. To address this gap, we conducted a network meta-analysis (NMA) to compare the efficacy of several interventions39. The NMA for Pharmacoeconomics and Outcome Research strongly recommends comparing the efficacy of different treatment modalities, as endorsed by the International Society for Pharmacoeconomics and Outcome Research (ISPOR)40.
Recently, NMAs have compared various nonpharmacological interventions, including physical exercise, music therapy, CTI, and nutritional therapy, in older patients with MCI or AD41,42,43. They found that physical exercise or cognitive stimulation had more significant positive effects on global cognition compared to the other interventions. However, because these studies were conducted exclusively in patients with cognitive impairment, the results primarily pertained to the effectiveness of cognitive rehabilitation rather than cognitive improvement or maintenance. To date, no NMA has been conducted on older adults with minimal or no cognitive impairment to compare the effects of all interventions on global cognition.
Therefore, we performed an NMA of randomized controlled trials to examine the relative effectiveness of seven interventions—CTI, music therapy, non-invasive brain stimulation, ACU intervention, meditation, physical exercise, and mind–body exercise—on the improvement of global cognition among older adults with minimal or no cognitive impairment.
Methods
Study protocol registration
This study adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines44. This NMA was conducted as per the registered protocol in PROSPERO (CRD42023401854). Supplementary Table 1 presents our results in accordance with the PRISMA-NMA checklist.
Search strategy and study selection
We conducted a systematic literature search of electronic databases including Google Scholar, MEDLINE, PubMed, the Cochrane Central Register of Controlled Trials (CENTRAL), EMBASE, Web of Science, PsycINFO, and ProQuest Dissertations, covering the period from inception to May 2023. The following keywords were applied: (cognition OR global cognition OR cognitive improvement OR cognitive function OR cognitive impairment OR mild cognitive impairment OR MCI) AND (elderly OR elder OR old) AND (randomized OR random OR randomly OR randomization OR randomization OR RCT OR RCTs) AND ((non-pharmacological treatment OR non-pharmacological therapy OR non-pharmacological intervention) OR (Mind–body exercise OR, Baduanjin OR Qi gong OR Tai chi OR Taiji) OR (meditation OR mindfulness OR MBSR) OR (music therapy OR music intervention OR music treatment) OR (non-invasive brain stimulation OR tDCS OR rTMS OR TMS) OR (cognitive training intervention OR cognitive training) OR (acupuncture OR acupressure OR acumassage OR acupoint OR acupoint) OR (exercise OR physical exercise OR fitness OR resistance exercise OR aerobic OR strengthening exercises)). Additionally, we manually reviewed the reference lists of identified publications and relevant articles suggested by meta-analyses and systematic reviews45. No restrictions were placed on the language and country of publication as well as the sex or ethnicity of the participants.
Inclusion and exclusion criteria
To extract data from selected articles, we applied the PICOS (population, intervention, comparison, outcome, and study design) approach.
Population
The participants included were healthy older adults or patients with MCI aged 55 years or above. We excluded individuals with moderate-to-severe cognitive impairment (dementia or Parkinson’s disease) or neurological/psychiatric disorders (severe AD, epilepsy, schizophrenia, or multiple sclerosis).
Intervention
We included studies that administered structured and conceptualized nonpharmacological interventions, such as CTI, non-invasive brain stimulation, music treatment, mind–body exercise, meditation, and ACU, to healthy older adults and patients with MCI. We excluded studies comparing the efficacy of different treatment approaches within the same intervention category (e.g., Zen meditation vs. Vipassana meditation or tDCS vs. rTMS). Additionally, studies combining multiple interventions were excluded due to difficulties in isolating the efficacy of each intervention from the combined effects (e.g., CTI combined with meditation vs. CTI) (Table 1).
Comparison
All studies had to include other types of nonpharmacological interventions or control groups. The control group was defined as a passive control group that did not receive any intervention (e.g., waiting list, treatment, or care as usual) or a control group that was provided with other activities (e.g., exercise or sham intervention).
Outcome
The studies must have evaluated global cognition46,47 using measures such as the Mini-Mental State Examination (MMSE)48, the Montreal Cognitive Assessment (MoCA)49, the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS)50, the Cambridge Cognitive Examination (CAMCOG)51, the Mattis Dementia Rating Scale52, and the Alzheimer’s Disease Assessment Scale-Cognitive subscale (ADAS-Cog)53.
Study design: This NMA identified randomized controlled trials (RCTs) that assessed the efficacy of nonpharmacological interventions on global cognition. Case reports and case series were excluded.
Data extraction
We created a predefined data extraction sheet formed on a pilot extraction with highly related references, and two research assistants extracted the relevant information by applying this template. For data synthesis, a data extraction form included the information such as title, authors, publication year, participants’ characteristics, including age, sex, sample size, and presence of cognitive impairment (i.e., healthy or MCI), nonpharmacological intervention details, types of control conditions, intervention duration, assessments of global cognition, and pre- and post-intervention scores for global cognition. This NMA only included the studies reporting the mean or standard deviation of pre- or post-treatment measurements. If multiple post-intervention scores were reported at different follow-up time points, only the evaluation immediately following the completion of the intervention phase was considered. The extracted data were cross-checked and consensus was reached by discussion; any disagreements were adjudicated by a third party.
Quality appraisal
Two independent reviewers conducted full-text screening of the included articles and evaluated the quality of individual studies using the Cochrane risk-of-bias tool for RCTs54. Quality appraisal considered various bias domains, including selection bias (random sequence generation and allocation concealment), reporting bias (selective reporting), performance bias (blinding of participants and personnel), detection bias (blinding of outcome assessment), attrition bias (incomplete outcome data), and other sources of bias. These items were rated as low, high, or unclear. Quality appraisals were cross-checked, and any disagreements between the reviewers were resolved through discussion or adjudication by a third party.
Summary measure
Calculation of effect size
We conducted an NMA that comprised 60 studies utilizing the pretest–posttest control (PPC) design. The PPC design offers a more efficient framework for estimating treatment effects compared to pretest–posttest comparison group or posttest-only control group designs. Therefore, we excluded studies with other study designs from the analyses. Intervention efficacies were evaluated using Cohen’s d effect size. To determine the d value, we applied the effect size estimate suggested by Morris55. Effect size was calculated using the mean pre-post change in the intervention group minus the mean pre-post change in the control group, standardized by the pooled pre-intervention standard deviation, as follows:55,56
Mpost-I, Mpre-I, Mpost-c, and Mpre-c are the mean scores of the intervention and control groups for the posttest and pretest, respectively. Additionally, SDpooled and Cp are the pooled standard deviation and the bias correction, respectively.
D values of 0.2–05 are considered small, those of 05–0.8 are considered moderate, and those > 0.8 are considered large57.
Network meta-analysis
As outlined in the study protocol, we categorized seven nonpharmacological interventions and two control conditions as follows: (1) CTI, (2) non-invasive brain stimulation, (3) music therapy, (4) ACU, 5) mind–body exercise, (6) meditation, (7) physical exercise, (8) passive control, and (9) sham intervention. Following categorization, we performed an NMA using a frequentist random-effects model with the net-meta R package (version 8.0)58 to compare the efficacy between interventions. The network plot chart displayed nodes representing each intervention and control condition, with lines indicating direct comparisons between two interventions or control conditions. The width of each line was proportional to the number of studies reporting pairwise comparisons58.
Heterogeneity between studies was assessed using Q statistics and I2, which describe the proportion of total variability in effect estimates across studies attributable to heterogeneity rather than sampling error58,59. A P value < 0.05 for the Q statistic indicated significant heterogeneity60. To rank the interventions based on their efficacy for each outcome, P-scores ranging from 0 to 1 were obtained by estimating the effect sizes of pairwise intervention comparisons using the surface under the cumulative ranking curve (SUCRA)61. A higher P-score indicated a more effective intervention, with 0 or 1 representing the worst or best intervention, respectively58.
Additionally, we performed a post-hoc subgroup analysis comparing MCI and healthy older adult groups.
Assessment of inconsistency
To assess the possible sources of inconsistency, we employed two methods: net heat plot and node-splitting analysis. The net heat plot is a matrix visualization method used to identify inconsistencies within the NMA62. In this plot, each gray box represents the contribution of the direct estimates to the network estimates, with the size of the box indicating the magnitude of the contribution. The color of the box indicates changes in inconsistency, with blue or warm colors representing an increase or decrease in inconsistency, respectively62. Additionally, we conducted a node-splitting analysis to evaluate the inconsistency between direct and indirect estimates in the forest plots of the NMA63. To address publication bias, we examined the adjusted funnel plots. Egger’s test was used to test the asymmetry of the funnel plot64.
Results
Study selection
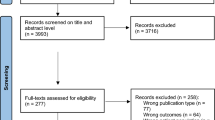
Figure 1 illustrates the flow diagram depicting the selection of the included studies. Initially, our search yielded 9663 papers related to our research terms. Of them, 9135 duplicate and irrelevant papers were excluded during the title/abstract screening. Additionally, 202 papers were omitted because of unavailability of data or the presence of adjunctive treatment. Through manual searching, four additional studies were identified. Ultimately, 61 RCTs were included in the NMA (Fig. 1 and Table 2). Two studies compared individual intervention arms (CTI vs. acupressure and CTI vs. Meditation) (Table 2).
Study characteristics
Table 2 presents the characteristics of the included studies. All included studies were conducted and published between January 2005 and December 2023. A total of 5458 older adults (1626 healthy individuals and 3832 patients with MCI) participated in the 61 RCTs included in the analysis. Among these, 2772 and 2686 participants were randomly assigned to the intervention (i.e., Qigong, music therapy, CTI, non-invasive brain stimulation, ACU, and meditation) and control conditions (passive control, exercise, and sham intervention), respectively. The age range of the participants in the RCTs was 55–89 years, with a mean age of 70.12 years. Three studies did not provide specific age information.
The mean duration of Qigong, non-invasive brain stimulation, CTI, ACU, meditation, and music therapy was 72.73 (SD = 34.72), 40.08 (SD = 24.63), 46.28 (SD = 27.87), 57.33 (SD = 45.18), 50.67 (SD = 34.56), and 40.80 (SD = 25.98) days, respectively. Two studies did not report the duration. The mean session duration for Qigong, non-invasive brain stimulation, CTI, ACU, meditation, and music therapy was 44.23 (SD = 17.27), 29.00 (SD = 10.25), 63.60 (SD = 37.54), 27.50 (SD = 3.54), 30.80 (SD = 20.28), and 85.00 (SD = 65.57) minutes, respectively.
Network meta-analysis results for global cognition
Figure 2a displays the network diagram of 60 studies comparing eight intervention groups with passive controls, including mind–body exercise, CTI, non-invasive brain stimulation, ACU, meditation, music therapy, exercise, and sham intervention. All intervention groups had at least one passive control group. The forest plot in Fig. 2b represents the relative efficacy of each intervention compared with passive control for global cognition.
The result of network meta-analysis for global cognition. (A) The network graph representing treatment arms included in the network for global cognition; the thickness of the lines shows the number of studies. (B) Random effects model forest plot for comparison of each intervention arm vs. passive control. (C) Ranking of medications for disruptive symptoms using SUCRA values. (D) Comparison of the included interventions: standardized mean differences (95% CI). Each cell represents the effect of the column-defining intervention compared to the row-defining intervention. BS Non-invasive brain stimulation, CI Confidence interval, CTI Cognitive training intervention, Ex Exercise, MBE Mind–Body exercise, Me Meditation, MT Music therapy, PC Passive control, SI Sham intervention, SUCRA Surface Under the Cumulative Ranking curve.
When combined in the NMA, six interventions were significantly more effective than passive control for global cognition: mind–body exercise, with a standardized mean difference (SMD) of 1.384 (95% CI 0.777–1.992); ACU, with 1.283 (95% CI 0.478–2.088); non-invasive brain stimulation, with 1.242 (95% CI 0.254–2.230); CTI, with 1.269 (95% CI 0.736–1.802); exercise, with 0.977 (95% CI 0.212–1.742); and meditation with 0.910 (95% CI 0.097–1.724) (Fig. 2b).
The ranking order of intervention efficacy for global cognition is as follows: mind–body exercise (P score = 0.7499), ACU (P-score = 0.6671), CTI (P-score = 0.6643), brain stimulation (P-score = 0.6432), music therapy (P-score = 0.5622), exercise (P-score = 0.4366), meditation (P-score = 0.4183), and sham intervention (P-score = 0.3441). Passive controls are ranked last in improving global cognition (P-score = 0.0143) (Fig. 2c).
Detailed results of the NMAs with different references are presented in Fig. 2d. Despite having the highest efficacy for global cognition compared to placebo, mind–body exercise did not show significant superiority over other interventions in direct and indirect comparisons. No significant differences in global cognition were observed between the interventions, except in comparison to passive controls (Fig. 2d).
In the results of subgroup analyses, the MCI and healthy groups responded to interventions differently (Supplementary Figure s1). In the MCI group, ACU intervention, CTI, non-invasive brain stimulation, and mind–body exercise were significantly effective compared to the passive control. However, in the healthy control group, mind–body exercise, ACU intervention, CTI, and meditation showed significant efficacy for global cognition.
Quality assessment
We conducted a quality assurance analysis using Review Manager software version 5.4 (Nordic Cochrane Center, Copenhagen, Denmark). Supplementary Figures S2 and S3 present a summary of the risk of bias across studies and the methodological features of each study.
The network quality analysis for global cognition indicated no evidence of heterogeneity (Q = 39.24, P = 0.9937, tau2 = 0.000, I2 = 0%). Additionally, no significant discrepancy was found between direct and indirect estimates of global cognition (Supplementary Tables S2 and S3).
The node-splitting method, which evaluates the incoherence in all pairs of direct and indirect estimates, revealed no significant inconsistency in the direct and indirect estimates (Supplementary Figure S4). Net heat plots were generated to visualize the consistency pattern in each comparison and detect important inconsistencies expressed as hot spots (red squares) (Supplementary Figure S5).
The risk of publication bias in the NMA was evaluated using the generated funnel plots (Supplementary Figure S6). Most studies exhibited a normal distribution, and no significant asymmetric pattern was observed in the funnel plots. Consequently, our study did not provide evidence of publication bias.
Discussion
This is the first RCT NMA to assess the individual efficacy of nonpharmacological interventions on global cognition in older adults with minimal or no cognitive decline. The NMA results showed that mind–body exercise, ACU intervention, meditation, non-invasive brain stimulation, CTI, and physical exercise had significant effects on improving global cognition compared to passive control conditions, with large effect sizes. The ranking order of intervention efficacy for global cognition is as follows: mind–body exercise, ACU, CTI, brain stimulation, music therapy, exercise, and meditation. However, no significant differences were found between the interventions, including the sham intervention (Fig. 2d).
Previous NMAs that focused on different interventions in older patients with AD or MCI also demonstrated the positive effect of physical exercise, ranking it higher than other interventions41,42,43. Consistent with these findings, our study showed that mind–body exercise was ranked first for improving global cognition (P-score = 0.7984). Mind–body exercises involve specific body postures, controlled breathing techniques, and mental concentration. Although there are several types of mind–body exercises (i.e., Qigong, Tai Chi, and Baduanjin), all consist of physical movement, breathing, and meditative exercises65. While the relationship between mind–body exercise and global cognition has not been systematically investigated, previous studies have suggested that the cognitive improvement observed after mind–body exercise may be attributed to cognitive stimulation and changes in stress-related physiological systems66,67. Compared to traditional exercises, mind–body exercises involve factors that provide additional cognitive stimulation66. Mind–body exercises require various higher-order cognitive functions beyond just memory training, such as attention, visual-spatial ability, perceptual speed, multitasking, and planning, in order to maintain postural stability66.
Both meditation and mind–body exercise have down-regulating effects on the sympathetic nervous system and the hypothalamic–pituitary–adrenal (HPA) axis activities triggered by stress68. Activation of the HPA axis in response to stress leads to increased secretion of cortisone, which can disrupt synaptic plasticity, damage hippocampal dendrites, and reduce neurogenesis in the adult brain, ultimately resulting in cognitive impairment69.
Large effect sizes (SMD > 0.8) were observed for ACU intervention, meditation, CTI, and non-invasive brain stimulation in improving global cognition. Although ACU interventions have not been extensively studied in meta-analyses of acupuncture, there is growing evidence supporting their cognitive improvement effects. ACU intervention at the acupoints “Baihui,” “Dazhui,” “Shenshu,” and “Zusanli” improved cognitive function, especially learning and memory, by repairing synaptic damage in patients with cerebral ischemia, and exerted neuroprotective effects by modulating the expression of CREB, BDNF, BCL2, and BAX genes70,71.
CTI has been validated and proven effective in improving and maintaining cognition and preventing dementia72. The prevailing hypothesis suggests that repeated practice with a set of tasks targeting specific cognitive domains (e.g., attention, memory, language, and executive function) may enhance functioning in those domains, leading to improvements in global cognition72. CTIs have the potential to activate neuroplasticity and increase cognitive reserve, which refers to the brain’s capacity to mitigate losses resulting from brain injury or degeneration73.
Non-invasive brain stimulation is an emerging tool for rehabilitating cognition in patients with neurodegenerative diseases, although its therapeutic mechanisms are not yet fully understood74. It has been hypothesized that high-frequency electric current stimulation induces various functional changes, including physiological (i.e., modulation of cortical excitability) and metabolic effects (i.e., enhanced neurotransmitter release), which in turn promote structural reorganization of the brain through neuroreparative processes75,76,77.
Music therapy is widely employed in clinical practice to benefit patients with dementia25. While the effect size is small (SMD < 0.3), numerous reviews and meta-analyses have suggested positive effects of music on cognitive function25,27,78. However, contrary to these findings, our study did not observe a significant effect of music therapy on global cognition compared to passive control conditions. This discrepancy may be attributed to sample heterogeneity, as previous studies focused on older people with moderate to severe cognitive impairment25,27,78. Music intervention appears to be more beneficial for individuals with cognitive impairment, as it primarily targets specific cognitive domains, particularly episodic memory, rather than global cognition. In daily life, the brain stores memories and emotions associated with significant episodes accompanied by music, which can trigger the retrieval of autobiographical memories11,79. Music may be particularly helpful for individuals with AD dementia who struggle with recalling salient autobiographical contents. However, the effect of improving global cognition in older adults with no or mild cognitive decline may not have reached statistical significance in our study.
Consistent with previous meta-analyses, our findings revealed that exercise intervention ameliorated global cognition in older adults, both with and without MCI. However, the relative effect size of exercise intervention ranked low on the list of interventions. This could be attributed to bias resulting from averaging the results of different exercise interventions. Multiple meta-analyses examining the effect of exercise intervention on global cognition in older adults with MCI, as well as in disease-free older adults, have found significant variations in effect sizes across studies43,80,81,82,83,84,85,86. The relationship between exercise intervention and global cognition may depend on the type of exercise being employed87. Previous studies have shown that different types of exercise can have varying effects on cognition, possibly due to different molecular mechanisms. For instance, the SMD for resistance exercise ranges from 0.41 to 0.71, while the SMD for aerobic exercise ranges from 0.13 to 0.5884,86,88,89. Therefore, it is important to consider the type of exercise when prescribing exercise intervention with the aim of preventing or decelerating cognitive deterioration.
Additionally, our subgroup analysis of the MCI group or healthy older group revealed some inconsistent results (Supplementary Figure S1). ACU intervention, CTI, and mind–body exercise were promising intervention types for preventing the decline of global cognition in both groups. However, the order of comparative effectiveness of these interventions varied across the two groups. Mind–body exercise was most likely to be the optimal intervention with a SUCRA value of 77.35% for the healthy older group. However, for the MCI group, ACU intervention had a slightly higher probability of being the most optimal intervention (SUCRA = 77.35%), higher than CTI (SUCRA = 77.21%) and mind–body exercise (SUCRA = 62.81%). This discrepancy might be attributed to the complexity of the mind–body exercise. Mind–body exercise combines various components including movement, breath control, and attention modulation. This characteristic might increase the complexity of implementing the intervention and reduce intervention fidelity (i.e., the consistency between plan and execution), especially for older adults with MCI35,90. Therefore, our results suggest that different treatments should be provided depending on the characteristics of the patient group.
In summary, our NMA found that mind–body exercise, ACU intervention, meditation, CTI, brain stimulation, and physical exercise were effective interventions for improving global cognition in older individuals, with a large effect size. However, music intervention did not show a significant effect in individuals with or without mild cognitive decline. Regarding the mechanism of action for each intervention, our study revealed that improvements in global cognition may occur through various pathways, such as memorizing movements and enhancing brain plasticity by reducing stress. Furthermore, the results of subgroup analyses demonstrated that intervention efficacy varied depending on the characteristics of the older adults. This suggests that healthcare professionals should consider patient characteristics when prescribing interventions to ensure the most effective treatment for their patients.
This study has some limitations. Firstly, there are some differences between the methods in this manuscript and those in the pre-registration. In the pre-registration, the original participants for the study were patients with dementia and MCI. However, for this study, dementia patients were excluded and people with normal cognitive function were included. As this study aimed to evaluate and compare the efficacies of nonpharmacological interventions alone, RCT studies involving both nonpharmacological and pharmacological interventions were excluded from the analyses. Most RCT studies of patients with dementia typically involve both types of interventions. Therefore, the studies on patients with dementia were excluded from this study. If the studies were collected according to the research steps initially proposed in the pre-registration, the sample size would be small, which could lead to an overestimated treatment efficacy91. Therefore, we broadened the sample group pool by including both individuals with normal function and patients with MCI. Secondly, the number of included studies, especially those investigating ACU or music interventions, was relatively small. Thirdly, although our review explored multiple nonpharmacological interventions for global cognition, we were unable to consider factors such as intervention length, duration, frequency, psychological status, current medication use, and participant sex ratios due to insufficient information. To identify the effects of these variables on global cognition, further NMAs will be needed using these variables as covariates. Fourthly, we specifically focused on older people with or without minor cognitive decline and excluded research involving those with other psychiatric or neurological disorders, which represent only a subset of the older population worldwide. Fifthly, we excluded studies that combined multiple interventions or interventions with pharmacological treatments due to limitations in statistical analysis. Finally, including studies utilizing various scales to measure global cognition may have introduced a potential limitation. However, previous studies have demonstrated a strong correlation among different measures of global cognition92.
Despite these limitations, our study demonstrated the efficacy of seven interventions and provided insights into the treatment mechanisms for improving global cognition in older adults with and without mild cognitive decline. Considering the concurrent use of medications, variations in drug metabolism, a higher risk of adverse effects, and the presence of comorbid medical conditions, this population may exhibit greater sensitivity to pharmacological interventions compared to younger individuals93. Based on our review, nonpharmacological interventions, particularly mind–body exercises, could be a potential strategy for enhancing and maintaining global cognition in the older population.
However, the generalizability of these findings and the ability to draw definitive conclusions regarding the efficacy of these interventions are limited. To gain a better understanding of how intervention methods or psychological variables impact global cognition, future studies should incorporate additional moderating variables in the design of RCTs, such as intervention length, duration, frequency, psychological status, current medication use, and participant sex ratios.
Data availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- AD:
-
Alzheimer’s disease
- ADAS-Cog:
-
Alzheimer’s Disease Assessment Scale-Cognitive subscale
- CAMCOG:
-
Cambridge Cognitive Examination
- CENTRAL:
-
Cochrane Central Register of Controlled Trials
- CTI:
-
Cognitive training intervention
- MMSE:
-
Mini-Mental State Examination
- MCI:
-
Mild cognitive impairment
- MoCA:
-
Montreal Cognitive Assessment
- NMA:
-
Network meta-analysis
- PRISMA:
-
Preferred Reporting Items for Systematic Reviews and Meta-Analyses
- RBANS:
-
Repeatable Battery for the Assessment of Neuropsychological Status
- RCT:
-
Randomized controlled trial
- tDCS:
-
Transcranial direct current stimulation
- TMS:
-
Transcranial magnetic stimulation
References
Organization WH. 2015. World report on ageing and health: World Health Organization; ISBN: 9241565047.
Daffner, K. R. Promoting successful cognitive aging: A comprehensive review. J. Alzheimer’s Dis. 19(4), 1101–1122 (2010).
Ijaopo, E. Dementia-related agitation: A review of non-pharmacological interventions and analysis of risks and benefits of pharmacotherapy. Transl. Psychiatry. 7(10), e1250 (2017).
Pratchett, T. A global assessment of dementia, now and in the future. Lancet Neurol. 14, 691 (2015).
Smart, C. M. et al. Non-pharmacologic interventions for older adults with subjective cognitive decline: Systematic review, meta-analysis, and preliminary recommendations. Neuropsychol. Rev. 27, 245–257 (2017).
Nguyen, L., Murphy, K. & Andrews, G. Cognitive and neural plasticity in old age: A systematic review of evidence from executive functions cognitive training. Ageing Res. Rev. 53, 100912 (2019).
Greenwood, P. M. & Parasuraman, R. Neuronal and cognitive plasticity: A neurocognitive framework for ameliorating cognitive aging. Front. Aging Neurosci. 2, 150 (2010).
Herholz, S. C., Herholz, R. S. & Herholz, K. Non-pharmacological interventions and neuroplasticity in early stage Alzheimer’s disease. Expert Rev. Neurotherapeutics. 13(11), 1235–1245 (2013).
Giuli, C., Papa, R., Lattanzio, F. & Postacchini, D. The effects of cognitive training for elderly: Results from my mind project. Rejuvenation Res. 19(6), 485–494 (2016).
Zhang, H. et al. Effect of computerised cognitive training on cognitive outcomes in mild cognitive impairment: A systematic review and meta-analysis. BMJ open. 9(8), e027062 (2019).
Matziorinis, A. M. & Koelsch, S. The promise of music therapy for Alzheimer’s disease: A review. Ann. N. Y. Acad. Sci. 1516(1), 11–17 (2022).
Wang, Z. et al. Music therapy improves cognitive function and behavior in patients with moderate Alzheimer’s disease. Int. J. Clin. Exp. Med. 11(5), 4808–4814 (2018).
Kim, H. et al. Cognitive improvement effects of electro-acupuncture for the treatment of MCI compared with Western medications: A systematic review and Meta-analysis. BMC Complement. Altern. Med. 19, 1–15 (2019).
Li, W., Wang, Q., Du, S., Pu, Y. & Xu, G. Acupuncture for mild cognitive impairment in elderly people: Systematic review and meta-analyses. Medicine 99(39), e22365 (2020).
Chan, J. S., Deng, K., Wu, J. & Yan, J. H. Effects of meditation and mind–body exercises on older adults’ cognitive performance: A meta-analysis. The Gerontologist 59(6), e782–e790 (2019).
Chen, Y. et al. Meditation treatment of Alzheimer disease and mild cognitive impairment: A protocol for systematic review. Medicine 99(10), e19313 (2020).
Newberg, A. B., Wintering, N., Khalsa, D. S., Roggenkamp, H. & Waldman, M. R. Meditation effects on cognitive function and cerebral blood flow in subjects with memory loss: a preliminary study. J. Alzheimer’s Dis. 20(2), 517–526 (2010).
de Asteasu, M. L. S., Martinez-Velilla, N., Zambom-Ferraresi, F., Casas-Herrero, A. & Izquierdo, M. Role of physical exercise on cognitive function in healthy older adults: A systematic review of randomized clinical trials. Ageing Res. Rev. 37, 117–134 (2017).
van Uffelen, J. G., Paw, M. J. C. A., Hopman-Rock, M. & van Mechelen, W. The effects of exercise on cognition in older adults with and without cognitive decline: A systematic review. Clin. J. Sport Med. 18(6), 486–500 (2008).
Wang, Y., Zhang, Q., Li, F., Li, Q. & Jin, Y. Effects of tai chi and Qigong on cognition in neurological disorders: A systematic review and meta-analysis. Geriatr. Nurs. 46, 166–177 (2022).
Birba, A. et al. Non-invasive brain stimulation: A new strategy in mild cognitive impairment?. Front. Aging Neurosci. 13(9), 16 (2017).
Chu, C.-S. et al. Cognitive effects and acceptability of non-invasive brain stimulation on Alzheimer’s disease and mild cognitive impairment: A component network meta-analysis. J. Neurol. Neurosurg. Psychiatry. 92(2), 195–203 (2021).
Hsu, W.-Y., Ku, Y., Zanto, T. P. & Gazzaley, A. Effects of noninvasive brain stimulation on cognitive function in healthy aging and Alzheimer’s disease: A systematic review and meta-analysis. Neurobiol. Aging 36(8), 2348–2359 (2015).
Gates, N. & Valenzuela, M. Cognitive exercise and its role in cognitive function in older adults. Curr Psychiatry Rep. 12, 20–27 (2010).
Moreno-Morales, C., Calero, R., Moreno-Morales, P. & Pintado, C. Music therapy in the treatment of dementia: A systematic review and meta-analysis. Front. Med. 7, 160 (2020).
Hyde, K. L. et al. The effects of musical training on structural brain development: A longitudinal study. Ann. N. Y. Acad. Sci. 1169(1), 182–186 (2009).
Fusar-Poli, L., Bieleninik, Ł, Brondino, N., Chen, X.-J. & Gold, C. The effect of music therapy on cognitive functions in patients with dementia: A systematic review and meta-analysis. Aging Mental Health 22(9), 1103–1112 (2018).
Kaptchuk, T. J. Acupuncture: Theory, efficacy, and practice. Ann. Intern. Med. 136(5), 374–383 (2002).
Li, Y.-W. et al. The autonomic nervous system: A potential link to the efficacy of acupuncture. Front. Neurosci. 16, 1038945 (2022).
Miniussi, C., Harris, J. A. & Ruzzoli, M. Modelling non-invasive brain stimulation in cognitive neuroscience. Neurosci. Biobehav. Rev. 37(8), 1702–1712 (2013).
Hamer, M. & Chida, Y. Physical activity and risk of neurodegenerative disease: A systematic review of prospective evidence. Psychol. Med. 39(1), 3–11 (2009).
Jahnke, R., Larkey, L., Rogers, C., Etnier, J. & Lin, F. A comprehensive review of health benefits of qigong and tai chi. Am. J. Health Promot. 24(6), e1–e25 (2010).
Hölzel, B. K. et al. Investigation of mindfulness meditation practitioners with voxel-based morphometry. Soc. Cognitive Affect. Neurosci. 3(1), 55–61 (2008).
Hart, J. Clinical applications for meditation: A review and recommendations. Altern. Complement. Ther. 13(1), 24–29 (2007).
Huang, X. et al. Comparative efficacy of various exercise interventions on cognitive function in patients with mild cognitive impairment or dementia: A systematic review and network meta-analysis. J. Sport Health Sci. 11(2), 212–223 (2022).
Sagud, M., Tudor, L. & Pivac, N. Personalized treatment interventions: Nonpharmacological and natural treatment strategies in Alzheimer’s disease. Expert Rev. Neurother. 21(5), 571–589 (2021).
Guo, T., Fang, J., Tong, Z. Y., He, S. & Luo, Y. Transcranial direct current stimulation ameliorates cognitive impairment via modulating oxidative stress, inflammation, and autophagy in a rat model of vascular dementia. Front. Neurosci. 14, 28 (2020).
Ribarič, S. Physical exercise, a potential non-pharmacological intervention for attenuating neuroinflammation and cognitive decline in Alzheimer’s disease patients. Int. J. Mol. Sci. 23(6), 3245 (2022).
Rouse, B., Chaimani, A. & Li, T. Network meta-analysis: An introduction for clinicians. Intern. Emerg. Med. 12, 103–111 (2017).
Jansen, J. P. et al. Interpreting indirect treatment comparisons and network meta-analysis for health-care decision making: report of the ISPOR task force on indirect treatment comparisons good research practices: Part 1. Value Health 14(4), 417–428 (2011).
Liang, J. H. et al. Comparison of multiple interventions for older adults with Alzheimer disease or mild cognitive impairment: A PRISMA-compliant network meta-analysis. Medicine 97(20), e10744 (2018).
Wang, Y. Q. et al. Effects of non-pharmacological therapies for people with mild cognitive impairment. A Bayesian network meta-analysis. Int. J. Geriatr. Psychiatry. 35(6), 591–600 (2020).
Xu, Z. et al. Comparative effectiveness of interventions for global cognition in patients with mild cognitive impairment: A systematic review and network meta-analysis of randomized controlled trials. Front. Aging Neurosci. 18(13), 653340 (2021).
Hutton, B. et al. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: Checklist and explanations. Ann. Intern. Med. 162(11), 777–784 (2015).
Vassar, M., Atakpo, P. & Kash, M. J. Manual search approaches used by systematic reviewers in dermatology. J. Med. Libr. Assoc. JMLA. 104(4), 302 (2016).
Vega-Ávila, G. C. et al. Rhythmic physical activity and global cognition in older adults with and without mild cognitive impairment: A systematic review. Int. J. Environ. Res. Public Health. 19(19), 12230 (2022).
Wouters, H., van Gool, W. A., Schmand, B., Zwinderman, A. H. & Lindeboom, R. Three sides of the same coin: Measuring global cognitive impairment with the MMSE, ADAS-cog and CAMCOG. Int. J. Geriatr. Psychiatry 25(8), 770–779 (2010).
Tombaugh, T. N. & McIntyre, N. J. The mini-mental state examination: A comprehensive review. J. Am. Geriatr. Soc. 40(9), 922–935 (1992).
Nasreddine, Z. S. et al. The montreal cognitive assessment, MoCA: A brief screening tool for mild cognitive impairment. J. Am. Geriatr. Soc. 53(4), 695–699 (2005).
Randolph, C., Tierney, M. C., Mohr, E. & Chase, T. N. The repeatable battery for the assessment of neuropsychological status (RBANS): Preliminary clinical validity. J. Clin. Experiment. Neuropsychol. 20(3), 310–319 (1998).
Roth, M. et al. CAMDEX: A standardised instrument for the diagnosis of mental disorder in the elderly with special reference to the early detection of dementia. Br. J. Psychiatry 149(6), 698–709 (1986).
Matteau, E. et al. Mattis dementia rating scale 2: Screening for MCI and dementia. Am. J. Alzheimer’s Dis. Dement® 26(5), 389–398 (2011).
Rosen, W. G., Mohs, R. C. & Davis, K. L. A new rating scale for Alzheimer’s disease. Am. J. Psychiatry 141(11), 1356–1364 (1984).
Cumpston, M., Li, T, Page, M, Chandler, J., Welch, V., Higgins, J. Updated guidance for trusted systematic reviews: a new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Db Syst Rev. 2019; 10: 000142
Morris, S. B. Estimating effect sizes from pretest-posttest-control group designs. Organizational Res. Method. 11(2), 364–386 (2008).
Becker, B. J. Synthesizing standardized mean-change measures. Br. J. Math. Stat. Psychol. 41(2), 257–278 (1988).
Sawilowsky, S. S. New effect size rules of thumb. J. Mod. Appl. Stat. Method. 8(2), 26 (2009).
Rücker, G. & Schwarzer, G. Ranking treatments in frequentist network meta-analysis works without resampling methods. BMC Med. Res. Methodol. 15(1), 1–9 (2015).
Tonin, F. S., Rotta, I., Mendes, A. M. & Pontarolo, R. Network meta-analysis: A technique to gather evidence from direct and indirect comparisons. Pharm. Pract. 15(1), 943 (2017).
Ribassin-Majed, L. et al. What is the best treatment of locally advanced nasopharyngeal carcinoma? An individual patient data network meta-analysis. J. Clin. Oncol. 35(5), 498 (2017).
Rosenberger, K. J., Duan, R., Chen, Y. & Lin, L. Predictive P-score for treatment ranking in Bayesian network meta-analysis. BMC Med. Res. Methodol. 21(1), 1–10 (2021).
Krahn, U., Binder, H. & König, J. A graphical tool for locating inconsistency in network meta-analyses. BMC Med. Res. Methodol. 13(1), 1–18 (2013).
Yu-Kang, T. Node-splitting generalized linear mixed models for evaluation of inconsistency in network meta-analysis. Value Health 19(8), 957–963 (2016).
Sterne, J. A. C. et al. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ. 343, d4002 (2011).
McCaffrey, R. & Fowler, N. L. Qigong practice: A pathway to health and healing. Holist. Nurs. Pract. 17(2), 110–116 (2003).
Zou, L., Loprinzi, P. D., Yeung, A. S., Zeng, N. & Huang, T. The beneficial effects of mind-body exercises for people with mild cognitive impairment: A systematic review with meta-analysis. Arch. Phys. Med. Rehabilit. 100(8), 1556–1573 (2019).
Gothe, N. P. & McAuley, E. Yoga and cognition: A meta-analysis of chronic and acute effects. Psychosom. Med. 77(7), 784–797 (2015).
Ross, A. & Thomas, S. The health benefits of yoga and exercise: A review of comparison studies. J. Altern. Complement. Med. 16(1), 3–12 (2010).
Russell-Williams, J. et al. Mindfulness and meditation: Treating cognitive impairment and reducing stress in dementia. Rev. Neurosci. 29(7), 791–804 (2018).
Wu, L., Dong, Y., Zhu, C. & Chen, Y. Effect and mechanism of acupuncture on Alzheimer’s disease: A review. Front. Aging Neurosci. 15, 1035376 (2023).
Wang, Y.-Y. et al. Effect of electroacupuncture of different acupoint groups on learning-memory ability and expression of IL-1β and TNF-α in hippocampus and prefrontal cortex in rats with Alzheimer’s disease. Acupunct. Res. 45(8), 617–622 (2020).
Mondini, S. et al. Cognitive reserve in dementia: Implications for cognitive training. Front. Aging Neurosci. 8, 84 (2016).
Shah, T. M., Weinborn, M., Verdile, G., Sohrabi, H. R. & Martins, R. N. Enhancing cognitive functioning in healthly older adults: A systematic review of the clinical significance of commercially available computerized cognitive training in preventing cognitive decline. Neuropsychol. Rev. 27(1), 62–80 (2017).
Pini, L. et al. Non-invasive brain stimulation in dementia: A complex network story. Neurodegener. Dis. 18(5–6), 281–301 (2019).
Dayan, E., Censor, N., Buch, E. R., Sandrini, M. & Cohen, L. G. Noninvasive brain stimulation: From physiology to network dynamics and back. Nat. Neurosci. 16(7), 838–844 (2013).
Hummel, F. C. & Cohen, L. G. Non-invasive brain stimulation: A new strategy to improve neurorehabilitation after stroke?. Lancet Neurol. 5(8), 708–712 (2006).
Aum, D. J. & Tierney, T. S. Deep brain stimulation: Foundations and future trends. Front. Biosci. Landmark. 23(1), 162–182 (2018).
Zhang, Y. et al. Does music therapy enhance behavioral and cognitive function in elderly dementia patients? A systematic review and meta-analysis. Ageing Res. Rev. 35, 1–11 (2017).
Juslin, P. N. & Västfjäll, D. Emotional responses to music: The need to consider underlying mechanisms. Behav. Brain Sci. 31(5), 559–575 (2008).
Ahn, J. & Kim, M. Effects of exercise therapy on global cognitive function and depression in older adults with mild cognitive impairment: A systematic review and meta-analysis. Arch. Gerontol. Geriatr. 106, 104855 (2023).
Biazus-Sehn, L. F., Schuch, F. B., Firth, J. & de Souza, S. F. Effects of physical exercise on cognitive function of older adults with mild cognitive impairment: A systematic review and meta-analysis. Arch. Gerontol. Geriatr. 89, 104048 (2020).
Wang, S. et al. Efficacy of different types of exercises on global cognition in adults with mild cognitive impairment: A network meta-analysis. Aging Clin. Experiment. Res. 31, 1391–1400 (2019).
Gates, N., Singh, M. A. F., Sachdev, P. S. & Valenzuela, M. The effect of exercise training on cognitive function in older adults with mild cognitive impairment: A meta-analysis of randomized controlled trials. Am. J. Geriatr. Psychiatry 21(11), 1086–1097 (2013).
Zhang, L. et al. Meta-analysis: Resistance training improves cognition in mild cognitive impairment. Int. J. Sports Med. 41(12), 815–823 (2020).
Zheng, G., Xia, R., Zhou, W., Tao, J. & Chen, L. Aerobic exercise ameliorates cognitive function in older adults with mild cognitive impairment: A systematic review and meta-analysis of randomised controlled trials. Br. J. Sports Med. 50(23), 1443–1450 (2016).
Song, D., Doris, S., Li, P. W. & Lei, Y. The effectiveness of physical exercise on cognitive and psychological outcomes in individuals with mild cognitive impairment: A systematic review and meta-analysis. Int. J. Nurs. Stud. 79, 155–164 (2018).
Barha, C. K., Galea, L. A., Nagamatsu, L. S., Erickson, K. I. & Liu-Ambrose, T. Personalising exercise recommendations for brain health: Considerations and future directions. Br. J. Sports Med. 51(8), 636–639 (2017).
Landrigan, J.-F., Bell, T., Crowe, M., Clay, O. J. & Mirman, D. Lifting cognition: A meta-analysis of effects of resistance exercise on cognition. Psychol. Res. 84(5), 1167–1183 (2020).
Shu, Y. et al. Cognitive gains of aerobic exercise in patients with ischemic cerebrovascular disorder: A systematic review and meta-analysis. Front. Cell Dev. Biol. 8, 582380 (2020).
Spijker, A. et al. Effectiveness of nonpharmacological interventions in delaying the institutionalization of patients with dementia: A meta-analysis. J. Am. Geriatr. Soc. 56(6), 1116–1128 (2008).
Nüesch E, Trelle S, Reichenbach S, Rutjes AW, Tschannen B, Altman DG, et al. Small study effects in meta-analyses of osteoarthritis trials: Meta-epidemiological study. Bmj. 2010; 341.
Duro, D., Simões, M. R., Ponciano, E. & Santana, I. Validation studies of the Portuguese experimental version of the montreal cognitive assessment (MoCA): Confirmatory factor analysis. J. Neurol. 257, 728–734 (2010).
Gramaglia, C. et al. Non-pharmacological approaches to depressed elderly with no or mild cognitive impairment in long-term care facilities. A systematic review of the literature. Front. Public Health 9, 685860 (2021).
Acknowledgements
This work was supported by the Korea Institute of Oriental Medicine (KSN2312022) and the Ministry of Health & Welfare in Republic of Korea (HF23C0010).
Author information
Authors and Affiliations
Contributions
Jiwoo Suk wrote the main manuscript text and analyzed the data. Jaeuk U. Kim and Gahye Kim collected the data and prepared figures and tables. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Seok, JW., Kim, G. & Kim, J.U. Comparative efficacy of seven nonpharmacological interventions on global cognition in older adults with and without mild cognitive impairment: a network meta-analysis of randomized controlled trials. Sci Rep 14, 8402 (2024). https://doi.org/10.1038/s41598-024-58232-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-58232-2
Keywords
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.