Abstract
Feeding on plant-derived sugars is an essential component of mosquito biology, affecting key aspects of their lives such as survival, metabolism, and reproduction. Among mosquitoes, Aedes aegypti and Aedes albopictus are two invasive mosquito species in the US, and are vectors of diseases such as dengue fever, chikungunya, and Zika. These species live in heavily populated, urban areas, where they have high accessibility to human hosts as well as to plants in backyards and public landscapes. However, the range of plants that are suitable sugar hosts for these species remains to be described, despite the importance of understanding what plants may attract or repel mosquitoes to inform citizens and municipal authorities accordingly. Here, we tested whether Ae. aegypti and Ae. albopictus would sugar-feed on eleven commonly planted ornamental plant species. We confirmed feeding activity using the anthrone method and identified the volatile composition of plant headspace using gas-chromatography mass-spectroscopy. These chemical analyses revealed that a broad range of olfactory cues are associated with plants that mosquitoes feed on. This prompted us to use plant DNA barcoding to identify plants that field-caught mosquitoes feed on. Altogether, results show that native and invasive mosquito species can exploit a broader range of plants than originally suspected, including wild and ornamental plants from different phyla throughout the Spring, Summer and Fall seasons.
Similar content being viewed by others
Introduction
Phytophagy, the act of feeding on plants, is important for many insect species, including blood-sucking mosquitoes. Acquiring carbohydrates is essential for both males and females and can, in some species, constitute the sole source of food for adults (e.g., Toxorhynchites spp.). Males feed exclusively on plant-derived sugars and recently emerged females tend to seek for sugar before taking their first blood meal, which enhances egg production1. In addition to carbohydrates, it has been determined that mosquitoes also acquire vitamins, amino acids, and salts from plant nectar2,3,4.
To locate a sugar meal, mosquitoes are driven by several cues, including visual and olfactory (e.g., plant-emitted semiochemicals)5. Volatile odorant compounds (VOCs) are perceived via odorant receptors present primarily on the antennae, which are fine-tuned to specific volatiles (either independently or in combination) and elicit attractive or repellent responses6,7. Terpenes and benzenoids are common compound classes in flower scent profiles that drive mosquito attraction. Some mosquito species have been shown to be attracted to flower semiochemicals like linalool, (Z)-3-hexen-1-ol, and benzaldehyde and repelled by compounds such as β-myrcene and limonene5,8. In addition, several studies have identified volatile compounds from fruits and plants that mosquitoes locate and feed on in the field9,10. However, a more comprehensive understanding of the semiochemicals associated with plants that are suitable sources of nectar to mosquitoes is critical to the development of new and efficient disease vector control tools, a pressing undertaking, as current control efforts are being challenged by increased insecticide resistance11. As an example, attractive toxic sugar baits (ATSBs) containing mixtures of attractive odorants and toxic compounds, take advantage of mosquitoes’ natural requirement to feed on sugar. Interestingly, these traps bypass preexisting problems that conventional control strategies have faced, such as insecticide resistance12,13. ATSBs have been successful in capturing several mosquito species, including Aedes aegypti, Aedes albopictus, Aedes japonicus, Culex pipiens, Culex quinquefasciatus, and Anopheles gambiae14,15,16,17,18,19.
Among disease vectors that are of particular concern are Ae. aegypti and Ae. albopictus mosquitoes. These species are responsible for spreading dengue, chikungunya and Zika viruses, all of which have high global incidence as vaccines and/or treatments remain unavailable20. According to a recent study by Leta et al.20, a total of 215 countries and territories exhibit environments that are suitable for Ae. aegypti and Ae. albopictus habitation. In the context of climate change and global warming, the geographic distribution of these mosquitoes might widen and potentially spread diseases in new areas, making it crucial to develop control strategies for both species21. This world-wide distribution further suggests that these species are capable of feeding on a broad range of plant hosts. However, their sugar feeding behavior is relatively understudied compared to their host-seeking and blood-feeding behaviors, primarily because pathogens are transmitted to humans and animals when an infected female bites a blood-host. Yet it appears crucial to study this behavior, as it bears the potential for the development of new tools for vector surveillance and control.
Resources that mosquitoes use in populated urban areas to obtain a sugar meal and how invasive species adapt to local ornamental plants remain poorly understood. Some ornamental plants (e.g., Ligustrum quihoui, Pittosporum tobira, Loropetalum chinense) have been shown to increase survivorship in Ae. albopictus and could therefore contribute to their ability to successfully transmit pathogens by increasing their overall fitness22. Moreover, ornamental plant abundance has been shown to directly affect the population distributions of both Ae. aegypti and Ae albopictus during field experiments in Huixtla, Chiapas, Mexico for Ae. aegypti and Guangzhou, China and Long Island, New York, USA for Ae. albopictus23,24,25. Individuals from both species exhibited a higher proportion of sugar feeding when collected from an urban area with a higher amount of blooming ornamental plants. Altogether, this supports the hypothesis that ornamental species, by providing nectar to mosquitoes, might greatly influence their fitness and could consequently increase the risk of pathogen transmission in heavily populated areas. However, the breadth of ornamental plants that can serve as a source of carbohydrates to mosquitoes remains to be fully understood.
In this context, we first examined mosquito landing and feeding on different ornamental plant species that are commonly found in nurseries and backyards in the United States, with varying flower shape, size, color, scent, and nectar contents. We then analyzed the scent profile of each of these plants using gas-chromatography coupled with mass-spectrometry (GC–MS) to test whether attractive plants would share common chemical compounds. Because these results revealed that a broad range of chemical combinations are associated with suitable plant hosts, we used plant DNA barcoding on field-caught mosquitoes to identify plants that they are feeding on. Collectively, our results indicate that a broader than suspected range of plants can serve as a source of carbohydrates and the implications of these results for mosquito control are discussed herein.
Results
Plant visitation assays
We first selected 11 commercially available ornamental plants from a local nursery (Christiansburg, VA, USA) to determine whether mosquitoes would land and feed on some of the species, selected for their diversity in flower morphology and color: wave Petunia, red Impatiens, marigold, Celosia, butterfly bush, Guara, Lantana, Mexican heather, Scaevola, goldenrod and yarrow. We performed cage landing assays on three individual plants for each species and, independently, recorded the numbers of landings and feedings by males and females of Ae. aegypti and Ae. albopictus.
Landings
When observed during a two-hour window centered on their spontaneous locomotor activity peak, Ae. aegypti visited 10 of the 11 plants tested, although low numbers of landings (< 6) were observed for 8 of these plants. We noted a higher number of visitations for the goldenrod and yarrow ornamental plant species (Fig. 1B; Generalized Linear Model: p = 0.0002 and p = 0.0027, respectively). Goldenrod had the highest number of landings from Ae. aegypti, while red impatiens and marigold had the least (one male “M” and one female “F”, respectively). Overall, we did not notice differences in landing activity between Ae. aegypti males and females (Generalized Linear Model: p = 0.7046), which is likely due to the low number of landings and number of replicates. Across all visitation assays, Ae. aegypti landed less on the plants compared to Ae. albopictus, although overall the mosquito species was not a significant predictor of the number of landings (Generalized Linear Mixed Model: p > 0.1). Ae. albopictus visited all of the 11 tested plants, but only 5 plant species received > 5 landings.
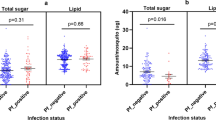
Mosquito sugar-feeding behavior on common ornamentals. (A) The standard layout for a plant visitation assay. Mosquitoes are released in a cage containing the ornamental flower [1], a GoPro aimed at the flower [2], a water cup topped with a soaked paper towel [3], and an iButton temperature/humidity recording device [4]. (B) Total number of observed landings by either Ae. aegypti males (light orange) and females (dark orange) or Ae. albopictus males (light blue) and females (dark blue) on the eleven different tested ornamental flower species (n = 3 × 20 mosquitoes (10M + 10F) per plant per mosquito species). (C) Total number of observed feedings. Ornamental flowers with a large ‘X’ indicate no activity from either species. (D) Negative (yellow) and positive (green) anthrone tests for the consumption of sugar. (E) Percentage of male and female Ae. aegypti and Ae. albopictus that tested positive for the consumption of fructose. (F) Total carbohydrate concentrations (µg) as determined by the anthrone method. Red impatiens and Ae. albopictus data is not present in these results (E, F), as an Ae. albopictus colony had not been established at the point of testing red impatiens.
Feedings
By visual examination of the video recordings, we observed Ae. aegypti feeding on five ornamental plant species: butterfly bush, Guara, Lantana, goldenrod, and yarrow (Fig. 1C). Although the difference was not significant due to the low number of feeding events we observed (Generalized Linear Model: p = 0.101), female Ae. aegypti tended to feed more than males. We did not observe Ae. aegypti feeding on the wave petunia, red impatiens, marigold, Celosia, or Scaevola, which coincided with the low number of landings observed (Fig. 1B). On the other hand, Ae. albopictus fed on 8 out of the 11 tested plants, which coincides with the higher number of landings observed in this species. In contrast to Ae. aegypti, we observed a tendency for more total feeding events from male Ae. albopictus compared to females (Generalized Linear Model: p = 0.0594), although low sample sizes prevent us from concluding on sex differences. Finally, we did not observe any feedings from either mosquito species on Mexican heather, which corresponded with the low landing activity (Fig. 1B).
Looking at the proportion of visually-observed feeding events over the number of observed landings, we found that all plant species but the Mexican heather, Scaevola, and wave Petunia, were significant predictors of the proportion of feeding (Generalized Linear Model: p < 0.05). In Ae. aegypti, the proportion of feedings observed on butterfly bush was significantly higher than the next largest proportion of feedings observed on goldenrod (80% and 30%, respectively; Generalized Linear Model: p = 0.0065), and all the other plants Ae. aegypti fed on (Generalized Linear Model: p < 0.05). The butterfly bush was also the plant that elicited most feedings per landings in Ae. albopictus, and the proportion of feedings on this plant was significantly higher than on Celosia, goldenrod, Guara and Lantana, marigold, Scaevola, and yarrow (Generalized Linear Model: p < 0.0065).
Survival
Because the tested plants likely differ in the quality and quantity of carbohydrates available to mosquitoes, we next, assessed the impact of the sugar meal for each ornamental plant species. Specifically, we quantified mosquito survival following the plant visitation assays. Across the eleven ornamental species and for both Ae. aegypti and Ae. albopictus, females tended to exhibit higher average survival rates than males, although these differences were not statistically significant (Fig. S1A, B; Generalized Linear Model: p > 0.0893). In addition, the plant species was not a significant predictor of mosquito survival (Generalized Linear Model: p > 0.4152), which could either be due to the low sample size, or suggest that mosquitoes had ample energy reserves before the experiments.
Carbohydrate content assays
Because mosquitoes may land and feed on plants outside of their peak activity time, we also used the anthrone colorimetric assay to identify successful sugar feedings that could have been missed by limiting our visual observations during mosquitoes’ peak activity time.
Qualitative analysis
First, the cold anthrone protocol was used to identify mosquitoes that were positive for fructose (Fig. 1E). Overall, Ae. albopictus exhibited a higher proportion of sugar feeding compared to Ae. aegypti (7.5% and 3.3%, respectively; Generalized Linear Model: p = 0.0028). No difference was observed between male and female Ae. aegypti, but male Ae. albopictus fed significantly more than females (11.9% and 4.45%, respectively; Generalized Linear Model: p = 0.0293). Overall, the plant species butterfly bush, Celosia, goldenrod, and Guara were significant predictors of the proportion of sugar feeding (Generalized Linear Model: p < 0.001, p = 0.033, p = 0.023, and p = 0.037). In addition, only Ae. albopictus tested positive for fructose when exposed to Scaevola, goldenrod, and yarrow, and only Ae. aegypti tested positive when exposed to butterfly bush and Lantana. No positive tests from either mosquito species were found for marigold and Mexican heather. It is worth noting that for some assays, such as goldenrod with Ae. aegypti, multiple feeding events were visually observed, but no mosquitoes tested positive for fructose consumption.
Quantitative analysis
The total amount of carbohydrates among the fructose-positive mosquitoes (n = 50) was quantified using the warm anthrone protocol (Fig. 1F). While the average sugar content of mosquitoes testing negative (n = 933) was below 30 µg (female Ae. aegypti: 16.9 ± 2.10 µg; male Ae. aegypti: 20.4 ± 5.51 µg; female Ae. albopictus: 9.95 ± 1.42 µg; male Ae. albopictus: 28.0 ± 13.7 µg), this amount increased by 1.43 folds in males and 11.15 folds in females Ae. albopictus, and by 3.28 folds in males and 6.62 folds in females Ae. aegypti. In negative-testing mosquitoes, no difference was found between species and sexes (Fig. S2A and S2B; Generalized Linear Model: p > 0.18). Overall, the sugar contents of positive-testing individuals of the two species were not significantly different (Generalized Linear Model: p = 0.3677), although carbohydrate concentrations tended to be higher in Ae. aegypti than Ae. albopictus. The plant species were not significant predictors of the mosquito sugar content either (Generalized Linear Model: p > 0.12), which could be explained by the low number of positive mosquitoes in our assays and likely due to the low number of replicates. Anecdotally, we found the highest carbohydrate concentration values in a female Ae. albopictus (541.43 µg ± 124.90 µg) and a male Ae. aegypti (301.43 µg ± 100.43 µg) that visited the wave petunia, followed by a female Ae. aegypti that visited Lantana (249.29 µg ± 39.71 µg).
GC–MS analysis of plant odor
To identify whether plants visited by mosquitoes shared common chemical cues, headspace collections and GC–MS analyses were conducted to resolve the VOC composition of the scent of each ornamental species (Fig. 2, Table 1). We found that terpenoids including linalool and aromatics such as nonanal were in abundance across each ornamental species tested. Nonanal had the highest relative concentration in the wave petunia (44.1%), red impatiens (35.7%) and Guara (57.8%). Other aliphatic compounds, including 2-hexanal and santolina triene, were present in small concentrations in all ornamental species except goldenrod. In Lantana, Mexican heather and Scaevola, ß-ocimene comprised the majority of the scent profile, representing 42.7%, 79.2%, and 56.9% of the scent composition, respectively. Benzaldehyde and limonene were found frequently across most of the ornamental species at varying concentrations. Benzaldehyde was found at its highest relative abundance in Lantana (29.6%), while limonene was found at high abundance in the scent of marigold (17.7%) and goldenrod (21.6%). ß-bisabolene was present only in the scent of Celosia but exhibited the highest relative peak abundance (30.8%). Germacrene D was a major constituent of the goldenrod scent (15%), and was present at smaller concentrations in marigold, Lantana and yarrow. We found that ɑ-pinene was in high abundance in the scent of goldenrod (25%) and was one of most abundant compounds in marigold (16.3%) and yarrow (7.7%). ɑ-farnesene was abundant only in the butterfly bush, making up 87.8% of the total scent’s peak area abundance. ß-phellandrene was found at small abundance in goldenrod, Lantana, and marigold, but had the highest relative peak abundance in yarrow (28%), with cis-verbenol being the second most abundant compound (26.8%). Finally, caryophyllene was the dominant compound in the scent profile of the marigold (20.5%) and was present at lower concentrations in Scaevola (8.4%) and yarrow (3.2%).
Example chromatograms for each of the eleven plants tested in the study. Pictures of the ornamental species are displayed on the right of each chromatogram. Scale: x-axis: au, arbitrary units, y-axis: time. Numbers above peaks within the chromatograms correspond to: 1, ɑ-pinene; 2, camphene; 3, β-pinene; 4, β-myrcene; 5, ɑ-phellandrene; 6, D-limonene; 7, β-ocimene; 8, linalool; 9, nonanal; 10, verbenol; 11, endo-borneol; 12, bornyl acetate; 13, aristolene; 14, caryophyllene; 15, γ-elemene; 16, germacrene D; 17, hexenal; 18, benzaldehyde; 19, β-phellandrene; 20, γ-terpinene; 21, p-cymene; 22, myroxide; 23, methyl salicylate; 24, β-farnesene; 25, humulene; 26, benzeneacetaldehyde; 27, eucalyptol; 28, m-ethylacetophenone; 29, p-cymen-7-ol; 30, γ-chlorobutyrophenone; 31, 5-octen-1-ol (Z); 32, β-bisabolene; 33, benzyl alcohol; 34, ɑ-farnesene. Bottom right: Nonmetric multidimensional scaling (NMDS) plot (stress = 0.1) of the chemical composition of the scent of all the plant species tested. Each dot represents a sample from a single individual plant. The ellipses represent the SD around the centroid of their respective cluster. Differences in scent composition and emission rate are significantly different between species (composition: ANOSIM, R = 0.93, p = 0.001).
We conducted an NMDS (non-metric multidimensional scaling) analysis to compare the scent profiles of each ornamental plant species based on the chemical compounds and their relative abundance (Fig. 2). Most of the ornamental species exhibited a distinct clustering of samples, indicating a unique and distinct scent composition (ANOSIM, R = 0.93, p = 0.001) (stress = 0.1), despite the aforementioned overlap in certain chemicals. Of note, marigold, Lantana and goldenrod samples showed individual clustering and separated from the main cluster. The scent profile of these three plants contains volatile compounds that are highly abundant relative to other ornamentals and, interestingly, goldenrod and Lantana samples clustered close together, while the marigold samples were isolated in the bottom-left quadrant of the NMDS. This positioning also reflects the behavior of the mosquitoes as neither species obtained carbohydrates from marigold.
Though the chemical profiles of plant scents are not enough to predict mosquito feeding behavior on their own, the broad range of chemical signatures associated with plants that were visited and fed on by mosquitoes suggests that an even broader range of plants than suspected could serve as sources of carbohydrates in the wild.
Plant DNA barcoding
The aforementioned laboratory experiments identified commercially available ornamental plants that can be visited and fed upon by mosquitoes. However, these plants' short seasonal blooming windows did not allow a wide coverage of the full range of plants potentially available to mosquitoes in the field. In addition, the breadth of chemical signatures found in plant hosts suggests an even broader range of suitable plant hosts. To circumvent limitations associated with the laboratory-based testing of individual plants, we relied on plant DNA barcoding to identify the plant species that field-caught mosquitoes fed upon.
Carbohydrate analysis
A total of 2360 mosquitoes were collected in Blacksburg, VA, USA, during the field collection season (June-October). The number of collected mosquitoes varied by week, with a large peak occurring in mid-July (Fig. 3A). We identified a total of five species: Ae. albopictus (N = 1527; 54.5%), Culex pipiens (N = 574; 24.3%), Anopheles punctipennis (N = 29; 1.2%), Ae. vexans (N = 93; 3.9%), and Ae. triseriatus (N = 96; 4.1%) (Fig. 3B). The overall fructose positivity rate was 38% (N = 903 positive tests; Fig. 3C). Throughout the season, this rate generally ranged between 25 and 50% but did not gradually decrease, even towards the end of the season in October (Fig. 3D). In addition, the fructose positivity rate varied across trap sites and ranged between 26 and 46% (Fig. 3E). The majority of mosquitoes captured were females (females: N = 1992; 84%; males: N = 368; 16%). However, we found that males had a higher proportion of positive sugar tests (N = 164; 45%) compared to females (N = 739; 37%) (Fig. 3C). Over 50% of collected mosquitoes were Ae. albopictus and their abundance varied by site (Fig. S3A, 3F), and ranged between 40 and 161, with the exception of trap site 8, where the highest number of Ae. albopictus mosquitoes were trapped (N = 670; Fig. 3F). The proportion of Ae. albopictus testing positive for fructose consumption ranged between 24 and 50% across the ten trap sites (Fig. 3G). (Fig. 3B).
Mosquito sugar-feeding activity in urban areas. (A) Total mosquitoes captured across the ten trapping locations per week of the candidate plant host study. (B) The proportion of different mosquito species captured across all trapping locations. (C) The proportion of males (light green) and females (dark green) captured across the trapping locations (left) and the proportion of positive sugar-feeding tests for all males and females. (D) The average proportion of positive sugar-feeding tests per week of the field season for males (light green) and females (dark green). (E) The average proportion of positive sugar-feeding tests across the field season for each trap site. (F) The total number of Ae. albopictus captured per week during the candidate plant host study. (G) The proportion of positive sugar tests per trap site given by male (light green) and female (dark green) Ae. albopictus.
Candidate host plants
A total of 75 mosquitoes contained plant DNA that was amplified using the rbcLa primers while 144 samples were amplified using trnH primers. From these 219 amplifications, we identified 26 unique candidate host plants from 18 different families (Table 2). Interestingly, most of these plants are commonly planted as ornamentals in gardens (e.g., Prunus spp., Acer spp., Carya illinoinensis). Eighteen of the 26 candidate host plant sequences were fully assembled; the remaining 8 did not assemble and were identified based on the forward or reverse primer sequences. Most of the plant candidates were identified from DNA that was extracted from Ae. albopictus females. However, some were also determined for male Ae. albopictus and female Cx. pipiens, An. punctipennis, Ae. japonicus and Ae. vexans. Plant species identified from multiple mosquitoes' crops include Trifolium repens (rbcLa, trnH; N = 12), Prunus spp. (rbcLa; N = 18), Drosera filiformis (rbcLa; N = 2), Plantago spp. (trnH; N = 5) and Acer spp. (rbcLa, trnH; N = 4). Most plant species were identified from mosquitoes during specific weeks of the season (Table 2). For example, we found that mosquitoes fed on Oxalis dillenii in July and on Drosera filiformis only in August. However, species like Trifolium repens and Prunus virginiana were identified from mosquitoes captured throughout the entire field season. When using rbcLa primers, the genus Prunus was identified from the crop of both male and female Ae. albopictus and female Cx. pipiens. For each Prunus result, several species consistently appeared with the same high percent identity coverage, indicating that the rbcLa gene is highly conserved amongst Prunus species. Prunus species identified from BLAST present in the study area include Prunus virginiana (“bitterberry” or “chokecherry”) and Prunus padus (“bird cherry”). Trifolium repens was identified frequently throughout the season using both the rbcLa and trnH barcodes and was targeted almost exclusively by female Ae. albopictus mosquitoes.
Discussion and perspectives
The first part of this work revealed that several ornamental plants (8 out of the 11 tested here) can serve as potential sources of carbohydrates to two of the most invasive mosquito species, Ae. aegypti and Ae. albopictus. Because of the selected plants' short, seasonal blooming period, we could not go beyond testing three individuals of each plant species for each of the mosquito species. While this limits our ability to conclude with confidence about mosquitoes’ preferences between these plant species, it is striking that mosquitoes were attracted to the majority of them. Indeed, the uncertainty around whether the selected individual plants represent the larger population of their species means that, by chance, we could have sampled individuals that were less attractive to mosquitoes. This was not the case, suggesting that the range of plant species that can serve as mosquito hosts is likely larger than previously thought.
The association between the chemical composition of the representative plants we tested and their ability to elicit mosquito attraction and feeding further suggests that mosquitoes can exploit many plants to sustain their metabolism. In addition, this corroborates previous findings [e.g.,5,26,27], indicating that combinations of chemicals at specific ratios are more important in explaining mosquito attraction than the simple presence or absence of a given individual chemical. For example, high numbers of landings and feedings on goldenrod were observed for both mosquito species. Occurrences of mosquitoes visiting goldenrod have been recorded as early as 190728, and multiple genera have been observed visiting this plant in the field (personal observations). But the most abundant compound in goldenrod’s scent, ɑ-pinene, has been tested against Ae. aegypti and Ae. albopictus for larvicidal or mosquitocidal effects29,30, while its role in mediating attraction or repellency in mosquitoes remains unclear. Similarly, Germacrene D, which has been shown to repel Ae. albopictus31, was present in high abundance in goldenrod’s scent (15.1%). We observed a similar trend with Celosia, marigold, and wave petunia, on which mosquitoes landed despite the presence of β-bisabolene in the scent of Celosia, which is present in essential oils that repel Ae. aegypti32. Caryophyllene was the most abundant compound of marigold’s scent and is a major constituent in an essential oil repellent to Ae. aegypti but has not been tested with Ae. albopictus, potentially inducing higher visitation activity from this species33. The scent of yarrow was dominated by cis-Verbenol and ß-phellandrene. When mixed with (-)-limonene, ß-phellandrene was found to be repellent against Ae. albopictus mosquitoes31. Cis-verbenol is also present in essential oils that exhibit larvicidal activity against Ae. albopictus and Ae. aegypti34. However, the capacity of these compounds to attract or repel Ae. aegypti and Ae. albopictus has yet to be tested.
We did not observe feeding activity on wave petunia, yet individuals of both species tested positive for nectar consumption and exhibited the highest carbohydrate concentrations in this study. It is, however, possible that mosquitoes fed on stems/leaves containing sugars either outside of the camera’s frame, or outside of the camera’s recording time at night when petunias emit a stronger scent1,35,36. In the headspace of Lantana, we identified β-ocimene as the most abundant volatile, which has been shown to elicit dose-dependent repellent responses in multiple aedine species, including Ae. aegypti37,38. Yet, we observed multiple landings from both species. This could be attributed to the ratio of different volatiles in Lantana’s scent profile; benzaldehyde was also a major constituent in Lantana scent and has been hypothesized to attract mosquitoes when present in volatile blends39. In the scent profile of butterfly bush, ɑ-farnesene was a prominent volatile; its enantiomer β-farnesene has demonstrated a low degree of repellence in Ae. aegypti mosquitoes40. However, because mosquitoes have enantioselectivity for odor compounds, β-farnesene may not be representative of a response to α-farnesene41, as implied by the high number of attempted feedings observed from both mosquito species. Vice versa, plants such as Guara released high amounts of nonanal, a VOC known to play a role in attracting Ae. aegypti to orchids7, while seeing the lowest visitation activity from mosquitoes. Finally, β-ocimene is a compound with a high degree of ubiquity in plant scents and was the most abundant compound in the scents of Mexican heather (79.24%) and Scaevola (56.93%)42. Some Ae. albopictus were observed feeding on Scaevola and tested positive for sugar consumption, but no mosquito was seen feeding on nor tested positive for crop nectar with Mexican heather.
Mosquito attraction to the plants did not necessarily imply successful sugar feeding. Almost 20% of Ae. albopictus in plant visitation assays with Celosia tested positive for fructose consumption but contained a low concentration of total carbohydrates. Likewise, we observed high landing and feeding activity from Ae. albopictus on marigold, but no mosquitoes tested positive for fructose consumption. We also observed high visitation activity on yarrow, which is generally described as mosquito-repellent43, but low nectar consumption rates were found in the total carbohydrate assays. This could be attributed to the morphology of Celosia (tall, feathery inflorescence) and marigold (wide, bowl-like) flowers not being adapted for a mosquito’s proboscis to probe and feed on. Though yarrow seems to provide a low nutritional reward, it is clear that this flower is not aversive to mosquitoes. Conversely, we observed a low number of feedings from both mosquito species on Guara, yet several mosquitoes tested positive for sugar feeding.
Altogether, our plant visitation assays and subsequent chemical analysis suggest that given the broad range of chemical associations present in plant scents, more plants than previously suspected could serve as hosts. Since the chemical analysis supports the idea that mosquito attraction is likely mediated by combinations of chemicals rather than their simple presence/absence, more work is required to fully comprehend the chemical basis of mosquito-plant interactions. In addition, this prompted us to employ a reversed approach and, rather than test each individual plant for mosquito attraction, identify the plants fed upon by local, field-caught mosquitoes.
Using plant DNA barcoding, we identified 26 unique plant species that various local mosquito species, as well as Ae. albopictus, feed on in an urban environment and noticed variations depending on the time of year. Overall, the range of plants we identified was broad and encompassed 18 plant families. While most plant species were identified in 1–2 samples, two genera occurred at higher frequencies (in > 12 samples): Prunus and Trifolium. Prunus represents a group of trees and shrubs that are cultivated for their fruits and decorative qualities and Trifolium are extremely common in lawns and along roadsides in the United States and produces a substantial amount of nectar in florets that are assumedly shallow enough for a mosquito’s proboscis to reach44,45. In a study by Kicel et al.46, 1-octen-3-ol was determined as a major constituent in the scent profile of Trifolium repens’ stems and leaves. This compound also emanates from human skin and has been reported as a kairomone (i.e., attractant) for Aedes mosquito species (Ae. albopictus included), especially when acting synergistically with CO247,48. Due to the prevalence of Trifolium repens in urban locations containing an abundance of hosts, Aedes mosquitoes are likely responding to both 1-octen-3-ol and CO2 when host-seeking in these areas49. In addition, occurrences of Trifolium repens have been documented in every continent, and it is able to grow in a range of climates from arctic to tropical, potentially contributing to Ae. albopictus’ wide establishment as an invasive disease vector50,51.
Another plant host that appeared more than once in our results using both barcodes was the maple tree (Acer spp.). Maple trees are some of the most frequently planted ornamental trees in North America52. These trees produce flowers and generally bloom from March to June. Interestingly, mosquitoes containing maple tree DNA were captured in July and August. Therefore, it is possible that the captured Ae. albopictus mosquitoes obtained carbohydrates by feeding on extra floral nectaries or from the leaves of the maple1,36. Alternatively, mosquitoes have been observed to feed on phloem sap, and may be doing so in this case due to the maple tree’s high sap production53,54. Due to its commonality in urban settings, maple trees may be contributing to this mosquito’s success in establishing itself as an invasive species.
Mosquitoes have been reported feeding on plant fruits in as early as 175855. Observations of mosquitoes feeding on pears and watermelons have been made in the 1900s, and the data presented here supports this with observed amplifications of Pyrus communis (pear) and Citrullus mucosospermus (watermelon) DNA56. We also identified Solanum lycopersicum (tomato), Cucumis sativus (cucumber), and Cucurbita pepo (pumpkin) as mosquito plant hosts. To our knowledge, this is the first published evidence that Ae. albopictus, or any mosquito species may feed on these plants. Furthermore, these plant hosts were identified only from mosquitoes captured in August and September, highlighting the importance of phenological studies for establishing mosquito sugar feeding activity in the wild. Previous studies have shown An. arabiensis larvae will feed on the pollen of maize plants in sub-Saharan Africa; gravid female An. arabiensis will lay their eggs near it so as to provide food for their offspring57. In the present study, Cx. pipiens used Zea mays (maize) as a plant host, indicating that the attractive blend could potentially be used as a multi-species attractant.
Success rates in species discrimination have been shown to vary between the rbcLa and trnH barcodes58,59, and here we show variations in the plant families they can identify. When comparing the ability of rbcLa, trnH, and matK to accurately identify legume (Fabaceae) species, trnH ranked the highest and was only slightly more accurate than rbcLa60. In the present study, trnH identified a higher amount of legume species, but had a similar percent identity to legume DNA sequences amplified by rbcLa, supporting the conclusion from Sanchez et al.60. A similar study was done previously to compare barcodes in discriminating Cucurbitaceae species, and trnH was once again found to exhibit the highest accuracy61. The trnH barcode identified multiple Cucurbitaceae species in the present study, whereas rbcLa identified none. The rbcLa barcode identified 18 different DNA sequences belonging to the Rosaceae family and Prunus genus but could not discriminate these sequences at the species level.
Overall, this study highlights the importance of expanding our understanding of mosquito phytophagy, particularly for invasive species. We show that Ae. aegypti and Ae. albopictus can leverage ornamental plants for sugar-feeding and identified a broader than suspected range of plants fed upon by mosquitoes in the wild. We found that mosquitoes can find sugar resources throughout the season which indicate the necessity to further explore their sugar feeding habits in other regions of the US and beyond to better control mosquito populations. Ornamental plants therefore likely help establish mosquitoes’ presence in urban, heavily populated areas. The data presented in this work sets the stage for future systematic surveys of mosquitoes’ host plant selection and could inform the public of plants that may or may not be attracting mosquitoes to the areas they are being planted. In areas with a higher risk of mosquito-borne disease transmission, the experiments described here can be used to enact field removal of plants and to develop baits / repellents to mitigate the spread of disease. Elucidating the mosquito-plant relationship is pivotal for maintaining the development of effective and environmentally friendly disease vector control techniques.
Materials and methods
Insects
The Rockefeller Ae. aegypti strain (MRA-734, MR4, AATCC®, Manassas, VA, USA) and the ATM95 Ae. albopictus strain (ATM-NJ95, AATCC®, Keyport, NJ, USA) were used for this study. These strains are maintained in our laboratory following previously published methods7. Larvae were reared in 26 × 35 × 4 cm covered trays that were filled with deionized water. The trays were kept in a climatic chamber at 26° ± 0.5 °C and 60 ± 10% humidity under light:dark cycles of 12 h:12 h. The diet of the larvae consisted of Hikari Tropic First Bites (Petco, San Diego, CA, USA). Prior to starting the plant visitation experiment, around 100 pupae were placed into mosquito breeding containers (BioQuip, Rancho Dominguez, CA, USA—1425, 1425DG) on the day of pupation. Upon emergence, female and male mosquitoes were starved for 1–2 days before being individually selected with forceps. To do so, the containers were placed in a cool environment to immobilize mosquitoes and isolate them for the following plant visitation assays.
Plant visitation assays
Plant visitation protocol
Two BugDorm-1 insect cages (BugDorm, DP1000) were placed on top of a 26 × 35 × 4 cm tray containing DI water to help minimize risks of desiccation. These cages were then placed in a secondary larger acrylic cage (17 × 22 × 32 in) which further provided a warm and humid environment for the mosquitoes. Each assay was conducted with both mosquito species separately to allow for better comparisons between the two species (e.g., survival, feeding). Within each cage, a GoPro camera (Hero5 Black), a water-containing cup covered by a wet paper towel for humidity, and the plant of interest were placed (Fig. 1A). Eleven ornamental plants purchased from local nurseries were tested: wave petunia (Petunia petunia x atkinsiana), red impatiens (Impatiens walleriana), marigold (Tagetes spp.), Celosia (spp.), butterfly bush (Buddleja spp.), Guara (spp.), Lantana (spp.), Mexican heather (Cuphea hyssopifolia), Scaevola (spp.), goldenrod (Solidago spp.), and yarrow (Achillea millefolium). All methods including plant collection were performed in accordance with the relevant guidelines and regulations. In addition, an iButton (Maxim, DS1923) was programmed and added to the cage to record humidity and temperature throughout the assays. Ten females and ten males of either Ae. aegypti or Ae. albopictus (3 replicates of 20 mosquitoes per mosquito species using, each time a different individual plant from the same species) were released into different cages. Assays were conducted at the end of the day between 4:30 and 5:30 pm, which has been previously reported as a peak sugar-feeding activity time for Ae. aegypti and the beginning of the sugar-feeding rhythm for Ae. albopictus62,63. In the following morning, between 8:30 and 9:30 am (total: 16 h), alive mosquitoes were collected using a Bug Vacuum (Redeo, XCQ-B), sorted by sex, and stored for subsequent sugar analysis at – 70 ºC. Dead mosquitoes were tallied and removed before beginning another assay to assess survival. Three replicates, totaling thirty mosquitoes per sex per mosquito species, were conducted for each plant species (N = 1320 mosquitoes).
Video analysis
GoPro cameras were used to record the mosquito behavior for a duration of two hours. Due to the consistent timing of initializing the assays and beginning the recording, we ensured that the videos captured the peak of sugar-feeding activity in these day-active mosquitoes (between 4:30 p.m. and 7:30 p.m.). Each video was analyzed by counting the total number of mosquito landings and feedings on the flower of interest. Landings were defined as a mosquito flying to the flower and idling on it for any amount of time. A single feeding was defined as the vertical movement of a mosquito’s head in a flower (i.e., probing), which is a characteristic motion of nectar-feeding. The sex of each mosquito landing or feeding was also noted for each landing and feeding event.
Data analysis
The effect of the plant species, mosquito species and sex on the number of observed landings and feedings was analyzed by means of Generalized Linear Mixed Models assuming a Poisson error distribution for the number of landings and a quasibinomial error distribution for the proportion of feedings per landing, with a logit link. The replicate number was used as a random effect in the model, and non-significant effects were conserved. The analysis was performed in R (version 4.1.1) using the package lme4 (version 1.1.27.1) and pairwise comparisons were performed using the package emmeans (version 1.7.1.1)64,65,66,67.
Scent collection and GC–MS analyses
Scent collection
To collect the headspace of each plant species, the inflorescence of the plant was enclosed in a nylon oven bag (Reynolds Kitchens, USA) that was taped tight around the stem. Two tygon tubes (Cole-Parmer, USA) were connected on one side to a diaphragm air pump (Gast, Benton Harbor, MI, USA), while the other side was inserted at the small opening of the bag. Air flow was then initiated by connecting the pump to a 6 V battery (Power-Sonic Batteries, USA). One tube pumped ingoing air into the bag through a charcoal filter cartridge (1 L/min), which served to remove any contaminants from the pump or the surrounding environment. The other tube pulled air out of the bag (1 L/min) through a headspace trap composed of a borosilicate Pasteur pipette (VWR, Radnor, PA, USA) containing 100 mg of Porapak powder Q 80–100 mesh (Waters Corporation, Milford, MA, USA). After 24 h of headspace collection, traps were eluted with 600 μL of 99% purity hexane (Sigma Aldrich, 227064-2L). The samples were sealed in 2 mL amber borosilicate vials (VWR, Radnor, PA) with Teflon-lined caps (VWR, Radnor, PA) and were subsequently stored at − 70 °C to remain stable until analysis by GC–MS. For controls, samples were taken concurrently from empty oven bags as in Lahondère et al.7. For each plant species, between 7 and 12 collection replicates were performed using several individuals from the same species.
GC–MS analysis
Twenty microliters from each plant scent sample were pipetted from the preceding method and placed into a vial with an insert (VWR, Radnor, PA) to be analyzed with a GC–MS (Thermo Fisher Scientific, Trace 1310) equipped with a 30 m column (Thermo Fisher Scientific, I.D. 0.25 mm, #36096-1420). Helium was used as the carrier gas at a constant flow of 1 cc/min. After each sample was prepared for the species of interest, they were loaded into the machine using an autosampler (TriPlus RSH, Thermo Fisher Scientific). The oven temperature was set at 45 °C, held for 4 min, followed by a heating gradient ramping to 230 °C, held for 6 min (total run: 28.5 min).
Chromatogram peaks were integrated using the Chromeleon software MS quantitative processing method (Thermo Fisher Scientific) and tentatively identified using the online NIST library. Major peaks found with consistently high abundances across multiple samples for each ornamental were then recorded for comparison across ornamental species. An internal standard, made of 100 ng/µL of heptyl acetate (Sigma-Aldrich, CAS #112-06-1), was added to each sample to calculate the concentrations of each compound based on a calibration curve. External synthetic standards, if available commercially, were used to confirm the chemical identity as in Lahondère et al.7.
Carbohydrate content assays
Nectar detection in the mosquito crop
Carbohydrate contents were measured by sequentially using the cold and warm anthrone methods described by van Handel67. First, mosquitoes were crushed with a glass rod in culture glass tubes (Sigma-Aldrich, C1048-72EA) containing 300 μL of cold anthrone reagent to detect for fructose consumption, as fructose is a monosaccharide that would only be present in the mosquito if it fed on a plant. The anthrone reagent was prepared by combining 150 mL water in a 1 L Erlenmeyer flask on ice with 380 mL sulfuric acid (Fisher, CAS #7664-93-9), in which 750 mg of anthrone (Sigma-Aldrich, CAS #90-44-8) was then dissolved. The samples were kept idle at room temperature (25 °C) for 30 min, after which their color was compared against a negative control (yellow) and positive control (dark green) containing a mosquito which was fed with a 20% fructose solution (Fig. 1D).
Quantitative carbohydrate assays
Following this, glass tubes containing the cold anthrone samples were filled with anthrone reagent to a 5 mL mark, heated for 17 min at 92 °C in a dry bath, and then cooled before being vortexed for 15–20 s. A sample without a mosquito was prepared additionally as a control and blank for the spectrophotometer (Perkin Elmer Lambda 20 UV/Visible Spectrophotometer). The optical density (OD) of each sample was then determined at 625 nm. For samples with an OD625 above one, 200 μL of sample was diluted with 800 μL anthrone reagent that had been heated as above, giving a dilution factor of 5. The carbohydrate content was quantified using the OD values and a calibration line that had been created by performing the above procedure with samples containing 25, 50, 100, 150, and 200 μg of glucose solution.
Data analysis
The effect of the plant species, mosquito species and sex on the number of measured amount of carbohydrates and proportion of positive-testing mosquitoes was analyzed by means of Generalized Linear Models assuming a Gaussian error distribution for the amount of carbohydrates and a binomial error distribution for the proportion of positive-testing mosquitoes per assay. Similarly, the effect of the plant species, mosquito species and sex on the proportion of surviving mosquitoes was analyzed by means of Generalized Linear Models assuming a binomial error distribution for the proportion of survivors per assay. The analysis was performed in R (version 4.1.1) using the package lme4 (version 1.1.27.1) and pairwise comparisons were performed using the package emmeans (version 1.7.1.1)64,65,66.
Plant DNA barcoding assay
Mosquito trapping
Throughout the field season of 2021 (Late May–End of October), mosquitoes were collected weekly from ten residential yards in Blacksburg, VA (USA) containing a high diversity of ornamental and wild plant species. Mosquitoes were captured once a week using BG-2 Sentinel Traps (BioQuip) baited with an attractive lure and carbon dioxide released by dry ice placed in an adjacent cooler (BioQuip). The traps were deployed late afternoon and retrieved mid-morning (total: 16–17 h), which prevented mosquitoes from drying out or digesting crop sugars, which usually occurs within 24 h after ingestion25. To further limit sugar digestion and degradation, mosquitoes were transported from the field to the lab on ice and stored at – 70 ºC.
Mosquito sample processing and DNA extraction
Mosquitoes collected from the field were identified by morphological traits under a microscope68, and washed in phosphate-buffered saline (Sigma-Aldrich, #806552-1L) to remove plant material contaminants, after which they were crushed in a microcentrifuge tube containing 100 μL of 0.3 M sodium acetate (Thermo Scientific, AM9740) and 200 μL of absolute molecular grade ethanol (Sigma-Aldrich, E7023-500 mL) as in Wanjiku et al.69. After incubation for 30 min at − 20 °C, homogenates were centrifuged at 4 °C for 10 min at 12,000 g. Following centrifugation, 200 µL of the sample supernatant was tested for the presence of fructose using the cold anthrone method described above. After a 24-h drying period, pellets were extracted using the RED Extract-N-AMP plant DNA extraction kit (Sigma-Aldrich, XNAP-1KT), according to the manufacturer’s instructions. Leaves from Trifolium repens collected in the field had their DNA extracted in a similar fashion and were used as a positive control.
PCR and Sanger sequencing
DNA was extracted from samples that gave a positive result for the anthrone test and genes of interest were amplified using PCR. Each sample was composed of 200 ng of DNA, PCR grade water (Fisher, AM9935), 10 μL of MyTaqHSmix (Bioline BIO-25045), 0.4 μM of forward and reverse primers targeting the chloroplast ribulose-1,5 biphosphate carboxylase/oxygenase large chain gene (rbcLa) (Primer R: GCTTCGGCACAAAAKARGAARCGGTCTC; Primer F: TATGTAGCTTAYCCMTTAGACCTTTTTGAAGA) or trnh-psbA intergenic spacer region (trnH) (trnH: CGCGCATGGTGGATTCACAATCC; psbA: GTTATGCATGAACGTAATGCT) to reach a sample volume of 22 μL. PCR cycling conditions for the rbcLa barcode were conducted as in Wanjiku et al.69 with an initial denaturation of 95 °C for 1 min, 35 cycles of denaturation at 95 °C for 15 s, annealing at 50 °C for 40 s, and extension at 72 °C for 1 min followed by one cycle of final extension at 72 °C for 10 min. For the trnH barcode, cycling conditions followed Nyasembe et al.37 with a denaturation of 94 °C for 1 min, followed by 45 cycles of denaturation at 94 °C for 1 min, annealing at 55 °C for 40 s and 72 °C for 1 min, and a final extension at 72 °C for 10 min. 1% agarose gels and a 1 Kb DNA ladder (Genesee Scientific, 42-432) were used to visualize and confirm the size of the PCR products. Samples with clear, single bands of the correct size on the agarose gel were sent to Genewiz (South Plainfield, NJ, USA) for purification and Sanger sequencing. Sequences were then assembled using CLC main workbench (Qiagen) and compared to the GenBank database using the Basic Local Alignment Search Tool (BLAST) to search for candidate host plant species; only sequencing results with more than 85% homology were considered as potential candidates.
Data availability
Data are available here: https://github.com/mosquito-hub/mosquito_sugar_feeding.git.
References
Foster, W. A. Mosquito sugar feeding and reproductive energetics. Annu. Rev. Entomol. 40(1), 443–474 (1995).
Baker, H. & Baker, I. Amino-acids in nectar and their evolutionary significance. Nature 241, 543–545 (1973).
Nicolson, S. W. & Thornburg, R. W. Nectar chemistry. In Nectaries and Nectar 215–264 (Springer, 2007).
Rivera-Perez, C., Clifton, M. E. & Noriega, F. G. How micronutrients influence the physiology of mosquitoes. Curr. Opin. Insect Sci. 23, 112–117 (2017).
Nyasembe, V. O. & Torto, B. Volatile phytochemicals as mosquito semiochemicals. Phytochem. Lett. 8, 196–201 (2014).
Nyasembe, V. O., Teal, P. E., Mukabana, W. R., Tumlinson, J. H. & Torto, B. Behavioural response of the malaria vector Anopheles gambiae to host plant volatiles and synthetic blends. Parasit. Vect. 5, 234 (2012).
Lahondere, C. et al. The olfactory basis of orchid pollination by mosquitoes. Proc. Natl. Acad. Sci. 117(1), 708–716 (2020).
Jaleta, K. T., Hill, S. R., Birgersson, G., Tekie, H. & Ignell, R. Chicken volatiles repel host-seeking malaria mosquitoes. Malar. J. 15(1), 354 (2016).
Bokore, G. E. et al. Grass-like plants release general volatile cues attractive for gravid Anopheles gambiae sensu stricto mosquitoes. Parasit. Vect. 14(1), 1–15 (2021).
Meza, F. C. et al. Behavioural and electrophysiological responses of female Anopheles gambiae mosquitoes to volatiles from a mango bait. J. Chem. Ecol. 46(4), 1–10 (2020).
Liu, N. Insecticide resistance in mosquitoes: Impact, mechanisms, and research directions. Annu. Rev. Entomol. 60, 537–559 (2015).
Gu, Z. Y. et al. Efficacy of orally toxic sugar baits against contact-insecticide resistant Culex quinquefasciatus. Acta Trop. 202, 105256 (2020).
Fiorenzano, J. M., Koehler, P. G. & Xue, R. D. Attractive toxic sugar bait (ATSB) for control of mosquitoes and its impact on non-target organisms: A review. Int. J. Env. Res. Public Health 14(4), 398 (2017).
Khallaayoune, K. et al. Attractive toxic sugar baits: Control of mosquitoes with the low-risk active ingredient dinotefuran and potential impacts on nontarget organisms in Morrocco. Env. Entomol. 42, 1040–1045 (2013).
Revay, E. E. et al. Formulation of attractive toxic sugar bait (ATSB) with safe EPA-exempt substance significantly diminishes the Anopheles sergentii population in a desert oasis. Acta Trop. 150, 29–34 (2015).
Qualls, W. A. et al. Indoor use of attractive toxic sugar bait (ATSB) to effectively control malaria vectors in Mali, West Africa. Malar. J. 14(1), 1–8 (2015).
Fryzlewicz, L., Van-Winkle, A. & Lahondere, C. Development of an attractive toxic sugar bait for the control of Aedes j. japonicus (Diptera: Culicidae). J. Med. Entomol. 59(1), 308–313 (2022).
Müller, G. C., Junnila, A. & Schlein, Y. Effective control of adult Culex pipiens by spraying an attractive toxic sugar bait solution in the vegetation near larval habitats. J. Med. Entomol. 47(1), 63–66 (2010).
Qualls, W. A., Xue, R., Revay, E. E., Allan, S. A. & Müller, G. C. Implications for operational control of adult mosquito production in cisterns and wells in St. Augustine, FL using attractive sugar baits. Acta Trop. 124(2), 158–161 (2012).
Leta, S. et al. Global risk mapping for major diseases transmitted by Aedes aegypti and Aedes albopictus. Int. J. Infect. Dis. 67, 25–35 (2018).
Ryan, S. J., Carlson, C. J., Mordecai, E. A. & Johnson, L. R. Global expansion and redistribution of Aedes-borne virus transmission risk with climate change. PLoS Negl. Trop. Dis. 13(3), e0007213 (2019).
Tian, J. et al. Effect of common ornamental plants on the survivorship and fecundity of the Aedes albopictus (Diptera: Culicidae). Florida Entomol. 102(1), 36–42 (2019).
Martinez-Ibarra, J. A., Rodriguez, M. H., Arredondo-Jimenez, J. I. & Yuval, B. Influence of plant abundance on nectar feeding by Aedes aegypti (Diptera: Culicidae) in southern Mexico. J. Med. Entomol. 34(6), 589–593 (1997).
Li, Y. et al. Urbanization increases Aedes albopictus larval habitats and accelerates mosquito development and survivorship. PLoS Negl. Trop. Dis. 8(11), e3301 (2014).
Fikrig, K. et al. Sugar feeding patterns of New York Aedes albopictus mosquitoes are affected by saturation deficit, flowers, and host seeking. PLoS Negl. Trop. Dis. 14(10), e0008244 (2020).
Najar-Rodriguez, A. J., Galizia, C. G., Stierle, J. & Dorn, S. Behavioral and neurophysiological responses of an insect to changing ratios of constituents in host plant-derived volatile mixtures. J. Exp. Biol. 213(19), 3388–3397 (2010).
VanderGiessen, M., Tallon, A. K., Damico, B., Lahondère, C. & Vinauger, C. Soap application alters mosquito-host interactions. Iscience 26, 5 (2023).
Knab, F. Mosquitoes as flower visitors. J. N. Y. Entomol. Soc. 15(4), 215–219 (1907).
Sarma, R., Adhikari, K. & Khanikor, B. Evaluation of efficacy of pinene compounds as mosquitocidal agent against Aedes aegypti Linn (Diptera: culicidae). Int. J. Trop. Insect Sci. 42(3), 2567–2577 (2022).
Russo, E. B. & Marcu, J. Cannabis pharmacology: The usual suspects and a few promising leads. Adv. Pharmacol. 80, 67–134 (2017).
Liakakou, A. et al. Isolation of volatile compounds with repellent properties against Aedes albopictus (Diptera: Culicidae) using CPC technology. Molecules 26(11), 3072 (2021).
Boonyuan, W. et al. Excito-repellency of essential oils against an Aedes aegypti (L.) field population in Thailand. J. Vect. Ecol. 39(1), 112–122 (2014).
da Silva, R. C. S. et al. (E)-caryophyllene and α-humulene: Aedes aegypti oviposition deterrents elucidated by gas chromatography-electrophysiological assay of Commiphora leptophloeos leaf oil. PLoS One 10(12), e0144586 (2015).
Lee, D. C. & Ahn, Y. J. Laboratory and simulated field bioassays to evaluate larvicidal activity of Pinus densiflora hydrodistillate, its constituents and structurally related compounds against Aedes albopictus, Aedes aegypti and Culex pipiens pallens in relation to their inhibitory effects on acetylcholinesterase activity. Insects 4(2), 217–229 (2013).
Fenske, M. P. et al. Circadian clock gene LATE ELONGATED HYPOCOTYL directly regulates the timing of floral scent emission in Petunia. Proc. Natl. Acad. Sci. 112(31), 9775–9780 (2015).
Clements, A. N. The Biology of Mosquitoes. Volume 2: Sensory reception and behaviour (CABI publishing, 1999).
Nyasembe, V. O., Tchouassi, D. P., Pirk, C. W. W., Sole, C. L. & Torto, B. Host plant forensics and olfactory-based detection in Afro-tropical mosquito disease vectors. PLoS Negl. Trop. Dis. 12(2), e0006185 (2018).
Dube, F. F. et al. Fresh, dried or smoked? Repellent properties of volatiles emitted from ethnomedicinal plant leaves against malaria and yellow fever vectors in Ethiopia. Malar. J. 10(1), 375 (2011).
Tucker, K. R., Steele, C. H. & McDermott, E. G. Aedes aegypti (L.) and Anopheles stephensi Liston (Diptera: Culicidae) susceptibility and response to different experimental formulations of a sodium ascorbate toxic sugar Bait. J. Med. Entomol. 59(5), 1710–1720 (2022).
Cantrell, C. L., Ali, A. & Jones, A. M. P. Isolation and identification of mosquito biting deterrents from the North American mosquito repelling folk remedy plant, Matricaria discoidea DC. PloS One 13(10), 2065 (2018).
Wheelwright, M., Catherine, R. W. & Riabinina, O. Olfactory systems across mosquito species. Cell Tissue Res. 383(1), 75–90 (2021).
Farré-Armengol, G., Filella, I., Llusià, J. & Peñuelas, J. β-Ocimene, a key floral and foliar volatile involved in multiple interactions between plants and other organisms. Molecules 22(7), 1148 (2017).
Tunón, H., Thorsell, W. & Bohlin, L. Mosquito repelling activity of compounds occurring in Achillea millefolium L (asteraceae). Econ. Botany 48(2), 111–120 (1994).
Potter, D. A., Redmond, C. T., McNamara, T. D. & Munshaw, G. C. Dwarf white clover supports pollinators, augments nitrogen in clover-turfgrass lawns, and suppresses root-feeding grubs in monoculture but not in mixed swards. Sustainability 13(21), 11801 (2021).
Jakobsen, H. B. & Kritjánsson, K. Influence of temperature and floret age on nectar secretion in Trifolium repens L.. Ann. Botany 74(4), 327–334 (1994).
Kicel, A., Wolbis, M., Kalemba, D. & Wajs, A. Identification of volatile constituents in flowers and leaves of Trifolium repens L.. J. Essential Oil Res. 22(6), 624–627 (2010).
Xu, P., Zhu, F., Buss, G. K. & Leal, W. S. 1-Octen-3-ol-the attractant that repels. F1000Research 2015, 4 (2015).
Shone, S. M., Ferrao, P. N., Lesser, C. R., Glass, G. E. & Norris, D. E. Evaluation of carbon dioxide–and 1-Octen-3-Ol–baited centers for disease control Fay-Prince traps to collect Aedes Albopictus. J. Am. Mosquito Control Assoc. 19(4), 445 (2003).
Upchurch, R. P., Peterson, M. L. & Hagan, R. M. Effect of soil-moisture content on the rate of photosynthesis and respiration in Ladino Clover (Trifolium repens L.). Plant Physiol. 30(4), 297 (1955).
Gibson, P. B. & Cope, W. A. White clover. Clover Sci. Technol. 25, 471–490 (1985).
Seebens, H. et al. No saturation in the accumulation of alien species worldwide. Nat. Commun. 8(1), 1–9 (2017).
Korányi, D. & Markó, V. Host plant identity and condition shape phytophagous insect communities on urban maple (Acer spp.) trees. Arthropod-Plant Interact. 16(1), 129–143 (2022).
Stone, C.M. & Foster, W.A. Plant-sugar feeding and vectorial capacity. In Ecology of parasite-vector interactions 35–79 (Wageningen Academic Publishers, 2013).
Nasci, R. S. Toxorhynchites rutilus septentrionalis feeding on tree sap. J. Am. Mosquito Control Assoc. 2(4), 141 (1986).
Swammerdam, J. The book of nature, or, the history of insects: Reduced to distinct classes, confirmed by particular instances (1758).
Peach, D. A. & Gries, G. Mosquito phytophagy–sources exploited, ecological function, and evolutionary transition to haematophagy. Entomol. Exper. Appl. 168(2), 120–136 (2020).
Wondwosen, B. et al. Sweet attraction: Sugarcane pollen-associated volatiles attract gravid Anopheles arabiensis. Malar. J. 17(1), 1–9 (2018).
Cabelin, V. L. D. & Alejandro, G. J. D. Efficiency of matK, rbcL, trnH-psbA, and trnL-F (cpDNA) to molecularly authenticate Philippine ethnomedicinal Apocynaceae through DNA barcoding. Pharmacogn. Mag. 12, S384 (2016).
Liu, Z. et al. Applying DNA barcodes for identification of plant species in the family Araliaceae. Gene 499(1), 76–80 (2012).
Loera-Sánchez, M., Studer, B. & Kölliker, R. DNA barcode trnH-psbA is a promising candidate for efficient identification of forage legumes and grasses. BMC Res. Notes 13(1), 1–6 (2020).
Pang, X. et al. Utility of the trnH–psbA intergenic spacer region and its combinations as plant DNA barcodes: A meta-analysis. PloS One 7(11), e48833 (2012).
Gillett, J. D., Haddow, A. J. & Corbet, P. S. The sugar-feeding-cycle in a cage-population of mosquitoes. Ent. Exp. Appl. 5(3), 223–232 (1962).
Yee, W. L. & Foster, W. A. Diel sugar-feeding and host-seeking rhythms in mosquitoes (Diptera: Culicidae) under laboratory conditions. J. Med. Entomol. 29(5), 784–791 (1992).
Bates, D. et al. Package ‘lme4’. http://lme4.r-forge.r-project.org (2009).
Lenth, R. et al. Emmeans: Estimated marginal means, aka least-squares means (Version 1.3.4). Emmeans Estim. Marg. Means Aka Least‐Sq. Means. https://CRAN.R-project.org/package=emmeans (2019).
R Core Team. R Foundation for Statistical Computing. R: A Language and Environment for Statistical Computing. https://www.R-project.org/ (2017).
Van Handel, E. Rapid determination of glycogen and sugars in mosquitoes. J. Am. Mosquito Control 1(3), 299–301 (1985).
Kline, D. L. Traps and trapping techniques for adult mosquito control. J. Am. Mosquito Control Assoc. 22(3), 490–496 (2006).
Wanjiku, C. D., Tchouassi, P., Sole, C. L., Pirk, C. & Torto, B. Plant sugar feeding patterns of wild-caught Aedes aegypti from dengue endemic and non-endemic areas of Kenya. Med. Vet. Entomol. 35(3), 417–425 (2021).
Acknowledgements
We thank Darren Dougharty and Shajaesza Diggs for their support with mosquito colony care. We would also like to thank Danny Eanes for his technical support as well as the residents of Blacksburg who provided their backyards for mosquito collection. Special thanks to Barbara Glaser for her help with relaying the information about our research project. We are grateful to members of the Lahondère laboratory and Vinauger laboratory for their feedback on the present study. This research was funded by the Eppley Foundation for Research, the USDA NIFA (VA-160160 and 2020-68018-30674), the Department of Biochemistry, The Fralin Life Science Institute, the Global Change Center at Virginia Tech. The following reagent was obtained through BEI Resources, NIAID, NIH: Aedes aegypti, Strain ROCK, MRA-734, contributed by David W. Severson and Aedes albopictus, Strain ATM-NJ95, Eggs, NR-48979. Figure 1A and D were done using Biorender.com.
Author information
Authors and Affiliations
Contributions
F.U., M.F., V.P. and C.L. performed experiments. C.L., C.V. and F.U. wrote the manuscript and prepared the figures. C.V. conducted the statistical analyses for Part 1 of the study. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Upshur, I.F., Fehlman, M., Parikh, V. et al. Sugar feeding by invasive mosquito species on ornamental and wild plants. Sci Rep 13, 22121 (2023). https://doi.org/10.1038/s41598-023-48089-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-48089-2
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.