Abstract
During fatiguing piano tasks, muscle fatigue develops differently between expert pianists. Differences in neuromotor strategies employed could explain a slower rate of fatigue development. The objective was to compare muscle activation and kinematic variabilities between ShortDuration (i.e., pianists with less endurance) and LongDuration groups. Results from 49 pianists showed that EMG activation variability of most shoulder and upper limbs muscles was greater for the ShortDuration group with time during two piano fatiguing tasks, namely Digital and Chord tasks. Segment acceleration variability, assessed using inertial measurement units, was also greater with time for the ShortDuration group at the right arm during the Digital task, and at the thorax and head during the Chord task. Finally, thorax lateroflexion variability increased with time for the LongDuration group (but not the ShortDuration group) during the Digital task. During the Chord task, wrist flexion variability was higher for the LongDuration group compared to the ShortDuration group. These results showed a direct effect of time on the pianists’ acceleration variability and EMG activation variability. In contrast, a protective effect of fatigue development could be attributed to kinematic variability. Results also suggest a higher risk of injury among pianists in the ShortDuration group.
Similar content being viewed by others
Introduction
Muscle fatigue caused by long hours of practice is critical in developing playing-related muscular disorders among pianists1. Muscle fatigue manifests in the upper trapezius during fast and slow music scores self-selected by pianists2, and in wrist and finger extensor muscles during repetitive piano tasks3,4. Expert pianists, with comparable years of experience, level of practice per day, maximum grip strength, laterality, age, and sex, do not show the same rate of fatigue development3. The pianists classified by Goubault et al.3 as the ShortDuration group showed a shorter time-to-task termination during repetitive piano tasks and a quicker myoeletric manifestation of fatigue compared to other pianists categorized as the LongDuration group. Previous studies showed that endurance, defined as the time until exhaustion, could be affected by different neuromotor strategies. Specifically, van Dieën et al.5 also discriminated two groups based on their endurance time during isometric contractions of the erector spinae, and showed a significant higher EMG activation variability in the endurance group. Oomen et al.6 also suggested that a lower motor variability could favor early state of fatigue and less endurance during a repetitive lifting task, and could increase the risk of development for muscular disorders. Consequently, comparing the neuromotor strategies employed by pianists of different endurance characteristics might help identify the factors slowing down the rate of fatigue development.
Laboratory studies have suggested that a greater muscle activation variability was associated with a slower rate of fatigue development7. Using high-density electromyography (EMG), Farina et al.7 showed that individuals with more heterogeneous muscle activity over time sustained static contraction longer than individuals with more uniform muscle activity distribution. Also, muscle fatigue causes adaptation; the biceps brachii, and brachialis EMG variability decreased during isokinetic, repetitive, concentric/eccentric elbow flexion8. Local fatigue can lead to an increased force variability in neighbor segments during isometric tasks, including maximal voluntary contraction and accurate force production before and after a 1-min maximal voluntary contraction of the index finger9. Also, the index finger showed a significant increase in force variability during accurate force production. Moreover, the variance of individual finger force increased for the four fingers involved in the tasks, while the total force variance showed only a modest change. This finding suggests an adaptive increase in force variability by non-fatigued segments as a strategy to slow down the rate of fatigue development, affecting accuracy during a multi-element performance. Since force fluctuations are greater as more motor units are recruited10,11, an association can be expected between EMG activity and its variability. This association was partially supported by Bosch et al.12, where the wrist extensor showed more cycle-to-cycle variability in EMG amplitude at an increased working pace. As muscle activations become more variable with fatigue, a direct impact on kinematics could be observed.
From an ergonomic viewpoint, motor variability may benefit the physiological and medical outcome of physical work as an intrinsic source of exposure variation13, allowing tissues to transiently recover from preceding exposures14,15. Although occupational studies suggest larger kinematic variability among experienced workers16, fatigue and recovery variability could be dependent on the measurement context17,18. Indeed, McDonald et al.17 showed that during simulated, repetitive, task-specific work, performance could be maintained throughout the post-fatigue work cycles via kinematic and muscular adaptations, such as reduced glenohumeral flexion and scapular rotation. Qin et al.18 observed increased kinematic variability adaptation over time in the shoulder joint to reduce the biomechanical loading on the fatigued shoulder region during a simulated, repetitive light assembly working task. On the other hand, a decreased kinematic variability over time in the more distal part of the upper limb was also observed, possibly to compensate for the increased variability caused by the shoulder joint while still maintaining task requirements18. These studies agree with the theory that stabilization and adaptation of voluntary movement against perturbations, like fatigue, relies on task-dependant synergies of degrees of freedom and a flexible binding19. This allows a stable linking of combinations of degrees of freedom that are essential to achieve the task, but releasing from tight control combinations of degrees of freedom that lead to the same task performance20,21. Therefore, kinematic adaptations observed to enhance endurance can happen in different body segments depending on the task performed. Kinematic variability strategies may differ between expert pianists and could explain the observed differences in time-to-task termination3.
This study aimed to understand the neuromotor strategies that slow down the development of fatigue in piano performance. To this end, we compared muscle activation and kinematic variabilities of expert pianists of different endurance times by assessing the effect of group (ShortDuration vs. LongDuration) and time (task Initiation vs. task Termination/Midpoint task) on the EMG and kinematic variability. We hypothesized that the LongDuration group would show greater EMG and kinematic variability than the ShortDuration group. We also hypothesized that EMG and kinematic variability would decrease with time.
Methods
The methods described below were part of a larger study where initial results were previously published in Goubault et al.3. Thus, the information in this section matches the methods section of that study.
Participants
Fourty-nine expert pianists were recruited. All participants were enrolled in undergraduate or graduate studies in piano performance or had at least a university (or equivalent) degree. Participants had to be free of any playing-related muscular disorders during the year preceding the experimentation. After receiving instructions on the protocol, participants read and signed written informed consent. The protocol was approved by the Université de Montréal’s Ethics Committee (CPER-18-086-D), and the research was performed in accordance with the Declaration of Helsinki. As described in our previous study, participants were categorized into two groups, ShortDuration and LongDuration, based on their duration on a Digital (repeating a right-hand 16-tone sequence) and a Chord (repeating a right-hand chord sequence) task3, as described in full details hereafter. Briefly, a k-means clustering and a silhouette validity index were used to identify the optimal number of sub-groups, i.e., two, based on the participants’ time-to-task termination, which varied across participants in the tasks described hereafter. Characteristics of participants are shown in Table 1.
Instrumentation
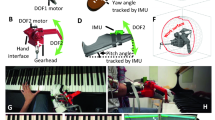
As in Goubault et al.3, participants were equipped with 49 monopolar EMG electrodes of 1.5 mm diameter (TMSi, EJ Oldenzaal, The Netherlands) separated by 2 cm and positioned on the right wrist flexor and extensor muscles according to a 7 × 7 array (Fig. 1). Before electrode placement, forearm skin was scrubbed with 70% isopropyl alcohol pads. A conductive gel was used to improve skin-electrode conductivity. Electrodes were attached to the skin with circular double-sided tape, and a medical elastic net bandage was used to hold the electrodes in place and minimize cable movements. Additionally, participants were equipped with two monopolar electrodes separated by 2 cm and positioned on the right biceps brachii, triceps brachii, anterior and lateral deltoids, and superior trapezius. Electrode locations were determined according to the Surface ElectroMyoGraphy for the Non-Invasive Assessment of Muscles project (SENIAM) recommendations22. Hair was removed with a razor when necessary, and skin was cleaned with alcohol swabs. The electrode positioning procedure was performed by a trained physiotherapist to ensure the reliability and physiological meaning of the data. It was then validated by a series of 10 submaximal voluntary contractions during which EMG signals were visually inspected in real-time on a display monitor. Briefly, the production of a burst of activation during a submaximal contraction was checked to ensure proper positioning of the electrodes with an appropriate signal-to-noise ratio. EMG signals were acquired using a 72-channel Refa amplifier (TMSi, EJ Oldenzaal, The Netherlands) at a sampling rate of 2048 Hz.
(A) close-up of the electrodes positioned on the forearm before medical elastic net bandage positioning, and (B) schematic view of the 7 × 7 array of electrodes positioned on the forearm and the underlying muscles, by Goubault et al.3, CC BY.
Participants were also equipped with 17 inertial measurement units (IMUs) (XSENS, Enschede, Netherlands) positioned on feet, shanks, thighs, pelvis, trunk, head, shoulders, arms, forearms, and hands, as recommended by the manufacturer. The IMUs positioning procedure was performed by the same researcher to ensure inter-participant reliability. IMU signals were acquired at a sampling rate of 60 Hz.
The participants played a grand piano (Disklavier DC7X Enspire Pro, Yamaha Canada Music Ltd, Toronto, Canada). A digital sound level meter (407,730 SLM, Extech Instruments, Nashua, USA) was placed at a 1.5 m distance from the back-left side of the piano bench to continuously monitor the sound intensity level.
Experimental procedures
The experimental procedure was identical to Goubault et al.3. The music scores were sent to the participants at least five days before the experiment. Participants were asked to practice the two excerpts to play accurately and comfortably during the experiment. Before collecting data, participants’ maximal handgrip force was measured using a handgrip dynamometer (Takei Scientific Instruments, Tokyo, Japan). Participants performed two 5-s maximal voluntary contractions with the right hand, interspersed with a 1-min rest period. Verbal encouragement was provided during contractions. Participants then performed a 5-min warm-up session on the piano, followed by a sound check to familiarize themselves with the required sound intensity level. Participants were then asked to perform the Digital and Chord tasks described hereafter without interruption. The order of the two piano tasks was randomly assigned, and a 15-min rest period was provided to participants in-between tasks. Both tasks were performed fast and loud to increase muscle activation levels23,24,25 and accelerate the development of muscle fatigue3. To this end, the tempo was established using a metronome connected to an earphone placed in the participants’ left ear. A screen was positioned in front of the piano for the pianist to adjust the playing volume when necessary. The Digital task corresponded to a 16-tone right-hand digital sequence on the first two measures of Exercise no 7 from C.L. Hanon’s ‘The Virtuoso Pianist’ (Appendix-A1), spanning a major sixth interval in the middle register of the keyboard. Every note was a sixteenth (very short in duration) with no rests between each note. Participants were asked to play this task loudly (minimum threshold = 78 dB) and fast (quarter note = 112 beats per minute [BPM]) while maintaining an accurate sound intensity. The Chord task corresponded to a right-hand chord sequence based on bar 119 of Franz Liszt’s Ballade no 2 in B minor S.171 (Appendix-A1). The chord sequence was composed of three notes played five times: once in the middle register (a staccato [detached] eight note) and twice (fast repetitions) in each of the middle-high and high registers of the keyboard. The rhythm of the two chords for each fast repetition was different, with the first chord as a sixteenth note and the second chord as a staccato eight note. There was the equivalent of an eighth note rest between all the registers which, given the performance tempo, was a very small period. After the high-register chords, the rest period was slightly longer (one quarter rest and one eight rest) before the pianist had to return to the middle register for the subsequent repetition. Participants were asked to play this task loudly (minimum threshold = 84 dB [middle register] and 94 dB [high register]) and fast (quarter note = 120 BPM) while maintaining dynamic and musical intention. The rate of perceived exertion was monitored every 30 s while participants were playing via the CR-10 Borg scale26. Participants were stopped if they reached two consecutive scores of 7 or more at the CR-10 Borg scale or after 12 min of continuous playing27. Two videos extract of both Digital and Chord tasks are available in supplemental files.
Data processing
Electromyography analysis
Monopolar EMG signals were converted into bipolar EMG signals by subtracting the two closest monopolar signals along the columns. EMG data were then filtered using a 10–400 Hz band-pass filter and a 60–120–180 ± 1 Hz stop-band filter to remove 60 Hz electrical contamination and its harmonics3. All filters were second-order zero-lag Butterworth filters. Data were then zero-aligned by subtracting the mean value. Muscle activation level was calculated using a 9 Hz low-pass filtering of the full-wave rectified EMG signals, normalized to the average of the 10 maximum amplitude values recorded during each task. Cycles, referring to each repetition of Digital and Chord excerpts, were time-normalized to handle possible variations between cycles before intra-participant variability calculation.
Kinematics analysis
Free-of-gravity segment accelerations (x, y, z) of the pelvis, thorax, neck, right scapula, shoulder, elbow, and wrist were filtered using a second-order zero-lag Butterworth band-pass filter with 0.5–15 Hz cut-off frequencies. The magnitude of segment acceleration was then calculated, and cycles were time-normalized.
Joint angles were directly extracted from XSENS MVN software (XSENS, Enschede, Netherlands), except for the pelvis and thorax, since only specific spine segments (i.e., L5, L3, T12, T8) are estimated with XSENS MVN software using interpolation of the pelvis, thorax, and head data. Therefore, pelvis and thorax angles were reconstructed using quaternion data and ‘yxz’ Euler angle sequence. Joints of interest were located at the pelvis (3 degrees-of-freedom [DoFs] in tilt-elevation-rotation), thorax (3 DoFs in flexion-lateroflexion-rotation), neck (3 DoFs in flexion-lateroflexion-rotation), and right scapula (3 DoFs in tilt-elevation-rotation), shoulder (3 DoFs in flexion-abduction-rotation), elbow (2 DoFs in flexion-pronation), and wrist (2 DoFs in flexion-abduction). For each DoF, the angle cycles were time-normalized.
Measure of variability
For the ShortDuration group, the interquartile range (IQR) was calculated for each muscle activation, segment acceleration, and joint angle at each frame for the first and last 15 cycles (task Initiation and Termination, respectively), as illustrated on Fig. 2. For the LongDuration group, the interquartile range (IQR) was calculated for each muscle activation, segment acceleration, and joint angle at each frame for the first (task Initiation) and mid (task Midpoint) 15 cycles, i.e., corresponding approximately to the task Termination time of the ShortDuration group. For each participant, the median IQR at Initiation and Term./Mid. were used as the intra-participant variability measure.
Statistical analysis
A two-way ANOVA on Group (ShortDuration vs. LongDuration) × Time (Initiation vs. Term./Mid.) with repeated measures on the Time was performed on the variability of EMG activation level, segment accelerations, and joint angles. T-test analysis was performed post-hoc as necessary. Data processing and statistical analyses were performed with Matlab R2019a (The MathWorks Inc., Natick, MA, USA).
Results
Three and two out of fourty-nine participants were removed from analysis for the Digital and Chord tasks, respectively, because of missing data. In addition, for angle analysis of the Digital task, five participants were removed because of important drift observed in angle data (i.e., more than 5°).
Muscle activation variability
Digital task. There was a significant Group-Time interaction for the EMG activation variability for 30 out of 42 forearm bipolar signals (Fig. 3). Paired t-test analysis showed that the EMG activation variability increased with Time for the ShortDuration group (p < 0.01), while it did not change for the LongDuration group (p > 0.05). The EMG activation variability significantly increased with Time for 6 out of 42 forearm bipolar signals. It was significantly higher for the ShortDuration than the LongDuration group for 5 out of 42 forearm bipolar signals.
(A) colormap representation of ANOVA results’ p-values for Group-Time interaction (left), main effects of Group (middle) and Time (right) for the Digital task. Columns of electrodes are labeled c1 to c7 as illustrated in Fig. 1. Notes. Non-significant values, i.e., p-values above or equal to 0.05 are represented in white. (B) Median value of activation levels’ IQR at Initiation (left) and Term./Mid., (right) for the ShortDuration (upper panel) and LongDuration (inferior panel) groups. Notes. *indicates significant Group-Time interaction; O indicates significant main effect of Group; + indicates significant main effect of Time; BI Biceps brachii; TR Triceps brachii; AD Anterior deltoid; LD Lateral deltoid; ST Superior trapezius.
In addition, there was a significant Group-Time interaction for the EMG activation variability of the biceps, anterior and lateral deltoids. Paired t-test analysis showed that the EMG activation variability increased with Time for the ShortDuration group (p < 0.001), while it did not change for anterior and lateral deltoids (p > 0.05) and slightly increased for the biceps (p = 0.01) for the LongDuration group. Moreover, the EMG activation variability of the triceps and superior trapezius significantly increased with Time and was significantly higher for the ShortDuration than the LongDuration group. Statistical results are shown in supplementary table S1 (Appendix-A2).
Chord task. There was a significant Group-Time interaction for the EMG activation variability for 12 out of 42 forearm bipolar signals (Fig. 4). Paired t-test analysis showed that the EMG activation variability did not change (p > 0.05) and increased with Time (p < 0.05) for 8 and 4 bipolar signals, respectively, for the ShortDuration group. EMG activation variability decreased (p < 0.05) and was stable with Time (p > 0.05) for 7 and 5 bipolar signals, respectively, for the LongDuration group. The EMG activation variability significantly decreased with Time for 9 out of 42 forearm bipolar signals. It was significantly higher for the ShortDuration than the LongDuration group for 2 out of 42 forearm bipolar signals. In addition, the EMG activation variability significantly decreased with Time for the biceps. Statistical results are shown in supplementary table S2 (Appendix-A2).
(A) colormap representation of ANOVA results’ p-values for Group-Time interaction (left), main effects of Group (middle) and Time (right) for the Digital task. Columns of electrodes are labeled c1 to c7 as illustrated in Fig. 1. Notes. Non-significant values, i.e., p-values above or equal to 0.05 are represented in white. (B) Median value of activation levels’ IQR at Initiation (left) and Term./Mid., (right) for the ShortDuration (upper panel) and LongDuration (inferior panel) groups. Notes. *indicates significant Group-Time interaction; O indicates significant main effect of Group; + indicates significant main effect of Time; BI Biceps brachii; TR Triceps brachii; AD Anterior deltoid; LD Lateral deltoid; ST Superior trapezius.
Segment acceleration variability
Digital task There was a significant Group-Time interaction on the variability of the right arm acceleration. Paired t-test analysis showed that the variability of the right arm acceleration increased significantly with Time for both ShortDuration and LongDuration groups (p < 0.001 and p = 0.008, respectively), with a greater increase for the ShortDuration group. Moreover, there was a significant increasing acceleration variability of the pelvis, thorax, head, shoulder, forearm, and hand with Time (Fig. 5). Statistical results are shown in supplementary table S3 (Appendix-A2).
IQR of acceleration magnitude at Initiation and Term./Mid. For the Digital task. The white dots represent the mean, the first whisker interval represents the 95% confidence interval of the mean, and the second whisker interval represents the standard deviation of the mean. Notes. *indicates significant Group-Time interaction; + indicates significant main effect of Time; no main effect of Group was observed.
Chord task There was a significant Group-Time interaction on the variability of the thorax and head accelerations. Paired t-test analysis showed that the acceleration variability of the thorax and head increased significantly with Time for the ShortDuration group (p = 0.02 and p = 0.04, respectively), while it did not change for the LongDuration group (p = 0.41 and p = 0.15, respectively). Moreover, there was a significant decreasing acceleration variability of the right forearm and hand with Time (Fig. 6). Statistical results are shown in supplementary table S4 (Appendix-A2).
IQR of acceleration magnitude at Initiation and Term./Mid. For the Chord task. The white dots represent the mean, the first whisker interval represents the 95% confidence interval of the mean, and the second whisker interval represents the standard deviation of the mean. Notes. * indicates significant Group-Time interaction; + indicates significant main effect of Time; no main effect of Group was observed.
Joint angle variability
Digital task There was a significant Group-Time interaction on the variability of the thorax lateroflexion. Paired t-test analysis showed that the variability of the thorax lateroflexion increased significantly with Time for the LongDuration group (p < 0.004), while it did not change for the ShortDuration group (p = 0.81). Moreover, there was a significant effect of Time on the angle variability of the pelvis (tilt, elevation, and rotation), thorax (flexion and rotation), neck (lateroflexion), scapula (elevation and rotation), shoulder (flexion and abduction), and elbow (flexion and pronation). For all these DoFs, the angle variability increased with Time (Fig. 7). Statistical results are shown in supplementary table S5 (Appendix-A2).
IQR of angle segment at Initiation and Term./Mid. for the Digital task. The white dots represent the mean, the first whisker interval represents the 95% confidence interval of the mean, and the second whisker interval represents the standard deviation of the mean. Notes. *indicates significant Group-Time interaction; + indicates significant main effect of Time; no main effect of Group was observed.
Chord task There was a significant effect of Group on the angle variability for wrist flexion, with a higher angle variability for the LongDuration group compared to the ShortDuration group. Additionally, there was a significant effect of Time on the angle variability of the thorax flexion, and neck lateroflexion; angle variability increased with Time (Fig. 8). Statistical results are shown in supplementary table S6 (Appendix-A2).
IQR of angle segment at Initiation and Term./Mid. for the Chord task. The white dots represent the mean, the first wisker interval represents the 95% confidence interval of the mean, and the second wisker interval represents the standard deviation of the mean. Notes. + indicates significant main effect of Time; o indicates significant main effect of Group; no Group-Time interaction was observed.
Discussion
The objective of the present study was to understand the neuromotor strategies that slow down the development of fatigue in piano performance. Results showed that EMG activation variability increased more for the ShortDuration group with Time during both Digital and Chord tasks, while it did not change, or decreased for the LongDuration group. Additionally, EMG activation variability of the biceps, anterior and lateral deltoids increased more for the ShortDuration group as compared to the LongDuration group during the Digital task. In terms of kinematics, segment acceleration variability increased more with fatigue for the ShortDuration group at distal segments (i.e., right arm) during the Digital task, and at proximal segments (i.e., thorax and head) during the Chord task. Moreover, proximal joint angle (i.e., thorax lateroflexion) variability increased with time for the LongDuration group during the Digital task, while it did not change for the ShortDuration group. During the Chord task, distal joint angle (i.e., wrist flexion) variability was higher for the LongDuration group compared to the ShortDuration group.
Variability and time-to-task termination
Results of the present study highlight a higher EMG activation variability with time for the ShortDuration group (i.e., the fatigued group), which partially contradicts previous studies suggesting either a protective effect of EMG variability against fatigue development7, or a decreasing EMG variability with fatigue8. However, in tasks that require a high level of accuracy, such as piano performance, motor variability impacting performance variables is undesirable. This could explain why the non-fatigued pianists of the LongDuration group maintained a low-level of EMG activation variability, whereas the fatigued pianists of the ShortDuration group had an increasing EMG activation variability, possibly to impede further fatigue development5,7. The increased EMG activation variability was accompanied by a greater increase of the right arm acceleration variability during the Digital task, and a greater increase of the thorax and head acceleration variability during the Chord task for the ShortDuration group, as compared to the LongDuration group. This possibly explains the decreased control of key-attack velocity due to fatigue for the ShortDuration group in Goubault et al.3. Interestingly, Madeleine et al.16 showed that experienced butchers had a larger variability than novices for several kinematic variables, but a reduced EMG variability compared to novices in simulated cutting tasks. This suggests that higher EMG variability among novices could be seen as a lack of adaptation to the work task, as seen among computer workers28. In the present study, the two groups had similar levels of piano experience and practice, and only differed in the time to exhaustion and myoelectric manifestation of fatigue3. Nonetheless, pianists from the LongDuration group adopted specific motor programmes, with an increased thorax lateroflexion and wrist flexion variability during the Digital and Chord task, respectively, that could have reduced the level of activity of some of the upper-limb muscles, and could possibly explain the greater time-to-task termination. An increased thorax angle variability was previously shown in repetitive fatiguing tasks27,29,30 suggesting a possible adaptation strategy at proximal joints used to compensate for upper-limb fatigue or fatigue-related discomfort during repetitive dynamic tasks. As pianists’ movements are individualized31, trunk movement could be a compensatory mechanism that potentially reduces the risk of injury. Nevertheless, these results suggest that pianists of the ShortDuration group could be at higher risk of injury than pianists of the LongDuration group, since they presented similar increasing EMG activation variability and decreasing joint angle variability as in Madeleine et al.16. However, future studies are needed to verify this hypothesis.
Variability and fatigue
Results of previous studies suggest a task dependency in the evolution of EMG activation variability with fatigue. Indeed, an increasing EMG activation variability has previously been observed in arm muscles (i.e., upper trapezius, anterior deltoid, and biceps) during a repetitive fatiguing task32,33. In contrast, a decreasing EMG activation variability was observed with fatigue in biceps brachii and brachialis during isokinetic, concentric/eccentric elbow flexion8. In the present study, the time affected both ShortDuration and LongDuration groups during the Digital and Chord tasks. The EMG activation variability increased with time for some wrist and finger flexor and extensor muscles and for the triceps and superior trapezius during the Digital tasks. During the Chord task, a decreasing EMG activation variability was observed with time for some wrist and finger flexor and extensor muscles as well as for the biceps. The notion of task dependency is reinforced with kinematics adaptations, with an increasing acceleration variability with time for most segments during the Digital task, and a decreasing acceleration variability with time for the right forearm and hand during the Chord task. It is also reportable that acceleration variability of the thorax, for instance, was qualitatively four times higher during the Chord task compared to the Digital task, suggesting that pianists used more their trunk motion during the Chord task to produce keystrokes. To produce keystrokes, expert pianists may use multi-joint upper-limb movements34,35. These multi-joint movements can also involve trunk motion, which can contribute to creating hand and finger keystroke velocities36, to facilitating and initiating upper-limb movements37 as well as to increasing upper-limb joint angle variability38. The same observation can be made for other segments, i.e., acceleration variability was higher at the upper-limb(s) and the head during the Chord task compared to the Digital task. The different movement characteristics of both tasks3 could explain the differences observed in the evolution of segment acceleration variability. The Chord task (1) requires movements of larger amplitude than the Digital task, and (2) is composed of sections formed by different fast rhythms with a short rest between each section (demanding therefore the production of higher bursts of keystroke force in short time periods). Therefore, during the Chord task, fatigue could be at the origin of the decreasing acceleration variability of distal segments because of less movement amplitude or a negative impact on the musical intention with the development of fatigue. On the contrary, the Digital task (1) requires movements of less amplitude with notes very closed to each other on the keyboard, and (2) is only composed of continuous fast (sixteenth) notes without rest between each note. This means that for keeping a high level of performance (i.e., no error and the right sound intensity and timing between notes as assessed in our previous study3), the room for maneuver of pianists in terms of acceleration variability is very low. Therefore, an increase of acceleration variability of the segments could be interpreted as a partial “loss of control” due to fatigue in that particular case, one that probably had a direct incidence on the variance of key velocity observed in our previous study3. Overall, even if the acceleration variability evolved in either direction with fatigue during both Digital and Chord tasks, it may be interpreted as a consequence of fatigue development instead of a strategy to impede further fatigue progress.
Music and variability
High inter-trial repeatability of EMG measurements was observed in string players (violinists) with less than 10% deviation of the mean muscular activities from their averages over the repetitions39,40,41. Another study showed low variability in maximum mouthpiece forces and kinematics among trumpet players42. The highly repeatable muscle activation and kinematic patterns in musicians, reflected in the present study by marginal variability of expert pianists muscle activation and movement patterns, could be explained by the years of practice required to become an expert40,43. Ericsson et al.44 estimated that expert musicians spent over 10,000 h of musical practice by the age of 21, leading to reorganization in certain sensory and motor systems and their interface45. The present study highlights that expert pianists can reproduce muscle activity and movement patterns and partially use variability with the development of fatigue when performing repetitive fatiguing Digital and Chord tasks.
Limitations
Some limitations have been taken into consideration when interpreting the results. First, the maximal voluntary contraction was not performed for all muscles. The EMG normalization was therefore determined using the average of the 10 maximum amplitude values recorded during each task. However, this should have a limited impact on the intra-participant EMG activation variability results as each bipolar signal is compared to itself within task Initiation and Term./Mid. Then, angular joints measured using IMUs should be interpreted with caution since fusion algorithms used to estimate sensor orientations can be subject to error accumulation over time caused by environmental magnetic perturbations and drift46,47,48. Nonetheless, drift should have a limited impact on the interpretation of the results since joint angle variability was calculated over 15 cycles, representing approximately 30-s of data recording.
Conclusion
Overall, this study highlights a direct effect of time on the EMG activation and segment acceleration variability of pianists, whereas a higher joint angle variability was observed in pianists having greater time-to-task termination. This suggests a direct effect of fatigue on EMG activation and segment acceleration variability, while a protective effect of fatigue development could be attributed to joint angle variability. Also, expert pianists with lower time-to-task termination during standard repeated Digital and Chord tasks exhibited an increased EMG activation variability, a greater increased acceleration variability and a lower wrist and trunk angle variability with time as opposed to expert pianists having higher time-to-task termination. This highlights different neuromotor strategies between pianists of different endurance characteristics, that could have an impact on the prevalence of injury. Future research should verify this hypothesis by assessing the EMG activation and kinematic variability effects from a training intervention aimed at increasing pianists’ endurance.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
Abbreviations
- EMG:
-
Electromyography
- IMU:
-
Inertial measurement units
- IQR:
-
Inter-quartile range
References
Ling, C.-Y., Loo, F.-C. & Hamedon, T. R. Playing-related musculoskeletal disorders among classical piano students at tertiary institutions in Malaysia: Proportion and associated risk factors. Med. Probl. Perform. Artists 33, 82–89 (2018).
Baeyens, J.-P. et al. Effects of rehearsal time and repertoire speed on upper trapezius activity in conservatory piano students. Med. Probl. Perform. Artists 37, 1–12 (2022).
Goubault, E. et al. Exhausting repetitive piano tasks lead to local forearm manifestation of muscle fatigue and negatively affect musical parameters. Sci. Rep. 11, 1–14 (2021).
McCarthy, K. C. Detection of Forearm Muscle Fatigue During Piano Playing Using Surface Electromyography (sEMG) Analysis (Carleton University, 2016).
van Dieën, J. H., Oude Vrielink, H. H. E. & Toussaint, H. M. An investigation into the relevance of the pattern of temporal activation with respect to erector spinae muscle endurance. Eur. J. Appl. Physiol. 66, 70–75 (1993).
Oomen, N. M. C. W., Graham, R. B. & Fischer, S. L. Exploring the relationship between kinematic variability and fatigue development during repetitive lifting. Appl. Ergon. 107, 103922 (2023).
Farina, D., Leclerc, F., Arendt-Nielsen, L., Buttelli, O. & Madeleine, P. The change in spatial distribution of upper trapezius muscle activity is correlated to contraction duration. J. Electromyogr. Kinesiol. 18, 16–25 (2008).
Bailey, C. A., Yoon, S. & Côté, J. N. Relative variability in muscle activation amplitude, muscle oxygenation, and muscle thickness: Changes with dynamic low-load elbow flexion fatigue and relationships in young and older females. J. Electromyogr. Kinesiol. 59, 102553 (2021).
Singh, T., Skm, V., Zatsiorsky, V. M. & Latash, M. L. Fatigue and motor redundancy: Adaptive increase in finger force variance in multi-finger tasks. J. Neurophysiol. 103, 2990–3000 (2010).
Moritz, C. T., Barry, B. K., Pascoe, M. A. & Enoka, R. M. Discharge rate variability influences the variation in force fluctuations across the working range of a hand muscle. J. Neurophysiol. 93, 2449–2459 (2005).
Taylor, A. M., Christou, E. A. & Enoka, R. M. Multiple features of motor-unit activity influence force fluctuations during isometric contractions. J. Neurophysiol. 90, 1350–1361 (2003).
Bosch, T., Mathiassen, S. E., Visser, B., de Looze, M. P. & van Dieën, J. H. The effect of work pace on workload, motor variability and fatigue during simulated light assembly work. Ergonomics 54, 154–168 (2011).
Mathiassen, S. E. Diversity and variation in biomechanical exposure: What is it, and why would we like to know?. Appl. Ergon. 37, 419–427 (2006).
Bongers, P. M., Kremer, A. M. & ter Laak, J. Are psychosocial factors, risk factors for symptoms and signs of the shoulder, elbow, or hand/wrist?: A review of the epidemiological literature. Am. J. Ind. Med. 41, 315–342 (2002).
Järvi, M. & Uusitalo, T. Job rotation in nursing: A study of job rotation among nursing personnel from the literature and via a questionnaire. J. Nurs. Manag. 12, 337–347 (2004).
Madeleine, P., Voigt, M. & Mathiassen, S. E. The size of cycle-to-cycle variability in biomechanical exposure among butchers performing a standardised cutting task. Ergonomics 51, 1078–1095 (2008).
McDonald, A. C., Tse, C. T. F. & Keir, P. J. Adaptations to isolated shoulder fatigue during simulated repetitive work. Part II: Recovery. J. Electromyogr. Kinesiol. 29, 42–49 (2016).
Qin, J., Lin, J.-H., Faber, G. S., Buchholz, B. & Xu, X. Upper extremity kinematic and kinetic adaptations during a fatiguing repetitive task. J. Electromyogr. Kinesiol. 24, 404–411 (2014).
Latash, M. L., Scholz, J. P. & Schöner, G. Toward a new theory of motor synergies. Mot. Control 11, 276–308 (2007).
Mattos, D. J. S., Latash, M. L., Park, E., Kuhl, J. & Scholz, J. P. Unpredictable elbow joint perturbation during reaching results in multijoint motor equivalence. J. Neurophysiol. 106, 1424–1436 (2011).
Busquets, A., Marina, M., Davids, K. & Angulo-Barroso, R. Differing roles of functional movement variability as experience increases in gymnastics. J. Sports Sci. Med. 15, 268–276 (2016).
Hermens, H. J., Freriks, B., Disselhorst-Klug, C. & Rau, G. Development of recommendations for SEMG sensors and sensor placement procedures. J. Electromyogr. Kinesiol. 10, 361–374 (2000).
Baeyens, J.-P. et al. Effects of rehearsal time and repertoire speed on extensor carpi radialis emg in conservatory piano students. Med. Probl. Perform. Artists 35, 81–88 (2020).
Chong, H. J., Kim, S. J. & Yoo, G. E. Differential effects of type of keyboard playing task and tempo on surface EMG amplitudes of forearm muscles. Front. Psychol. 6, 1–7 (2015).
Furuya, S., Aoki, T., Nakahara, H. & Kinoshita, H. Individual differences in the biomechanical effect of loudness and tempo on upper-limb movements during repetitive piano keystrokes. Hum. Mov. Sci. 31, 26–39 (2012).
Borg, G. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 14, 377–381 (1982).
Yang, C. et al. Changes in movement variability and task performance during a fatiguing repetitive pointing task. J. Biomech. 76, 212–219 (2018).
Delisle, A., Larivière, C., Plamondon, A. & Imbeau, D. Comparison of three computer office workstations offering forearm support: Impact on upper limb posture and muscle activation. Ergonomics 49, 139–160 (2006).
Bouffard, J., Yang, C., Begon, M. & Côté, J. Sex differences in kinematic adaptations to muscle fatigue induced by repetitive upper limb movements. Biol Sex Differ 9, 17 (2018).
Cowley, J. C. & Gates, D. H. Proximal and distal muscle fatigue differentially affect movement coordination. PLoS ONE 12, e0172835 (2017).
Turner, C., Visentin, P., Oye, D., Rathwell, S. & Shan, G. Pursuing artful movement science in music performance: Single subject motor analysis with two elite pianists. Percept. Mot. Skills 128, 1252–1274 (2021).
Samani, A., Srinivasan, D., Mathiassen, S. E. & Madeleine, P. Variability in spatio-temporal pattern of trapezius activity and coordination of hand-arm muscles during a sustained repetitive dynamic task. Exp. Brain Res. 235, 389–400 (2017).
Srinivasan, D., Sinden, K. E., Mathiassen, S. E. & Côté, J. N. Gender differences in fatigability and muscle activity responses to a short-cycle repetitive task. Eur. J. Appl. Physiol. 116, 2357–2365 (2016).
Furuya, S., Goda, T., Katayose, H., Miwa, H. & Nagata, N. Distinct inter-joint coordination during fast alternate keystrokes in pianists with superior skill. Front. Hum. Neurosci. 5, 1–13 (2011).
Furuya, S. & Kinoshita, H. Organization of the upper limb movement for piano key-depression differs between expert pianists and novice players. Exp Brain Res 185, 581–593 (2008).
Verdugo, F., Pelletier, J., Michaud, B., Traube, C. & Begon, M. Effects of trunk motion, touch, and articulation on upper-limb velocities and on joint contribution to endpoint velocities during the production of loud piano tones. Front. Psychol. 11, 1–22 (2020).
Verdugo, F., Begon, M., Gibet, S. & Wanderley, M. M. Proximal-to-distal sequences of attack and release movements of expert pianists during pressed-staccato keystrokes. J. Mot. Behav. 54, 316–326 (2022).
Turner, C., Goubault, E., Maso, F. D., Begon, M. & Verdugo, F. The influence of proximal motor strategies on pianists’ upper-limb movement variability. Hum. Mov. Sci. 90, 103110 (2023).
Duprey, S., Michaud, B. & Begon, M. Muscular activity variations of the right bowing arm of the violin player. Comput. Methods Biomech. Biomed .Eng. 20, 71–72 (2017).
Fjellman-Wiklund, A., Grip, H., Karlsson, J. S. & Sundelin, G. EMG trapezius muscle activity pattern in string players: Part I—is there variability in the playing technique?. Int. J. Ind. Ergon. 33, 347–356 (2004).
Ziane, C., Michaud, B., Begon, M. & Dal Maso, F. How do violinists adapt to dynamic assistive support? A study focusing on kinematics, muscle activity, and musical performance. Hum. Factors https://doi.org/10.1177/00187208211033450 (2021).
Devroop, K. & Chesky, K. Comparison of biomechanical forces generated during trumpet performance in contrasting settings. Med. Probl. Perform. Artists 17, 149–154 (2002).
Shumway-Cook, A. & Woollacott, M. H. Motor Control: Theory and Practical Applications (Lippincott Williams & Wilkins, 2001).
Ericsson, K. A., Krampe, R. T. & Tesch-Römer, C. The role of deliberate practice in the acquisition of expert performance. Psychol. Rev. 100, 363–406 (1993).
Pascual-Leone, A. The brain that plays music and is changed by it. Ann. N. Y. Acad. Sci. 930, 315–329 (2001).
Lebel, K., Boissy, P., Hamel, M. & Duval, C. Inertial measures of motion for clinical biomechanics: Comparative assessment of accuracy under controlled conditions—Changes in accuracy over time. PLoS ONE 10, e0118361 (2015).
Robert-Lachaine, X., Mecheri, H., Larue, C. & Plamondon, A. Validation of inertial measurement units with an optoelectronic system for whole-body motion analysis. Med. Biol. Eng. Comput. 55, 609–619 (2017).
Zhao, J. A review of wearable IMU (inertial-measurement-unit)-based pose estimation and drift reduction technologies. J. Phys. Conf. Ser. https://doi.org/10.1088/1742-6596/1087/4/042003 (2018).
Acknowledgements
We would like to thank the volunteers for their participation in the study, Justine Pelletier for her work on the design of the study and her participation in data collection, Valentin Degrave for his participation in data collection, the Piano department of the University of Montreal for its support in this study, the University of Montreal piano technician Marc St-Pierre for adjusting the piano before and during the experiment. This study was supported by Programme Audace du Fonds de Recherche du Québec (2018-AUDC-263189) and the Canada Foundation for Innovation (CFI).
Author information
Authors and Affiliations
Contributions
E.G. analyzed data and drafted the manuscript. C.T. and R.M. contributed to critical revision of the manuscript. M.B. and F.D.M., the lead scientists, helped in all facets of the project. F.V. defined the experimental set-up, performed the data collection and contributed to critical revision of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Goubault, E., Turner, C., Mailly, R. et al. Neuromotor variability partially explains different endurance capacities of expert pianists. Sci Rep 13, 15163 (2023). https://doi.org/10.1038/s41598-023-42408-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-42408-3
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.