Abstract
The aim of this study was to evaluate the foraminal enlargement and its influence on microcrack formation and apical transportation in root canals with apical curvature. Eighteen maxillary lateral incisors with apical curvature were selected by using micro-CT images. Root canals were randomly divided in two groups (n = 9) according to root canal preparation using two working lengths: 1 mm short of the apical foramen (control group) and 1 mm beyond the apical foramen (foraminal enlargement). For both groups Reciproc Blue R40 was used for root canal instrumentation. Specimens were scanned by nano-CT (UniTOM HR) before and after root canal preparation. Percentage, length, and width of microcracks, and apical transportation were assessed. Kappa, chi‐square and McNemar tests were used for qualitative analyses while paired and unpaired t-test were used for quantitative analyses (α = 0.05). For both groups, rather similar and low percentages of microcracks were observed before root canal preparation (P > 0.05). The foraminal enlargement promoted new microcracks, not observed in the control group. An increase in microcrack length was observed when the foraminal enlargement was performed (P < 0.05). Higher apical transportation was observed when foraminal enlargement was performed (P < 0.05). Foraminal enlargement using a heat-treated reciprocating file size 40 promoted microcracks and higher apical transportation than root canal preparation up to 1 mm short of apical foramen.
Similar content being viewed by others
Introduction
Root canal preparation directly influences long-term endodontic prognosis1. However, the apical limit of the root canal instrumentation is still a controversial topic in endodontics1,2. Although the most accepted working length is 1 mm short of the apical foramen1, the foraminal enlargement technique has also been proposed by some authors2,3,4. This procedure aims to clean the apical foramen4 in order to improve the root canal disinfection2,4. However, it is still not well defined if foraminal enlargement can promote microcracks and increase transportation of the apical foramen5,6,7,8.
The presence of dentinal microcracks may influence long-term survival of the tooth9, since microorganisms may proliferate in crack lines, leading to the establishment of biofilm on the root surface10. In addition, microcracks could potentially promote a vertical root fracture11, especially in roots with reduced dentin thickness12. Apical transportation may also negatively affect cleaning and filling of the apical root portion, resulting in failure to control infection and to promote root canal sealing13.
Micro-computed tomography (micro-CT) has been used to evaluate dentin microcracks, since it is a high precision tool that enables the location of such defects14. However, this method has limitations especially for visualizing small microcracks, with lower accuracy than operating microscope with transillumination15. Accordingly, to detect small structures or to perform high accuracy measurements, a higher precision tool is required14. Nano-computed tomography (nano-CT) devices have small focal spot and increased signal-to-noise ratio, making possible to achieve a maximum spatial resolution in the submicrometer range16.
Therefore, the aim of the present study was to evaluate the influence of foraminal enlargement on microcrack formation and apical transportation in maxillary lateral incisors with apical curvature by using nano-CT. The null hypothesis was that the enlargement of apical foramen would not cause microcracks and/or apical transportation.
Materials and methods
Ethical implications
The following methods were carried out in accordance with the Declaration of Helsinki and this study was approved by the Research Ethics Committee of the School of Dentistry of Araraquara/UNESP (protocol number: 29320820.8.0000.5416). All teeth used in this study were obtained from the Human Teeth Bank of the School of Dentistry of Araraquara/UNESP.
Specimen selection
Human maxillary lateral incisors, extracted for reasons not related to this study, were collected. with apical curvature (25°–35°)17 previously stored in 0.1% thymol solution were used. The inclusion criteria comprised teeth with complete apical formation, absence of root fractures, calcifications or internal resorptions. Roots were inspected under a stereomicroscope at × 12 magnification to exclude those with any external dentinal defect and roots with immature apices. A digital radiographic system (RVG 6100; Kodak Dental Systems, NY) was used to select teeth according to inclusion criteria. Image J program (National Institutes of Health, Bethesda, MD, USA) was used to assess the degree of curvature in radiographic images. All selected teeth were scanned using a micro-CT device (SkyScan 1276; Bruker-micro-CT, Kontich, Belgium) at a low-resolution (35 µm voxel size). The three-dimensional images allowed a more accurate selection, and the inclusion criteria confirmation, as well as the selection of only teeth that did not present microcracks in the apical third. After exclusion of teeth that did not reach the inclusion criteria, 18 maxillary lateral incisors with apical curvature were selected. Specimens were stored individually in numbered Eppendorf tubes at 100% humidity throughout the execution of the research.
Root canal preparation
Conventional access cavities were performed, and root canals were explored using a size #10K-file (Dentsply Sirona, Ballaigues, Switzerland). When the tip of the instrument was visible through the main foramen, the working length (WL) was determined. In order to simulate the periodontal ligament, the specimens were assembled in an apparatus with acrylic resin and each tooth was embedded in condensation silicone (Oranwash, Zhermack SpA, Badia Polesine, Italy). The apparatus was immersed in distilled water during the operative steps to prevent dehydration of the specimens. The same dentist, specialist in endodontics, prepared all the teeth.
Lateral incisors were randomly divided into two experimental groups (n = 9), by using a stratified random sampling method, considering the preoperative volume of the root canals. For the control group, a conventional preparation was used, meaning that the WL was defined as 1 mm short of the apical foramen. For the foramen enlargement group, the WL was defined as 1 mm beyond the apical foramen. For both groups, Reciproc Blue R40 (VDW GmbH, Munich, Germany) files were operated by an electric motor VDW SILVER (VDW GmbH) set in the “RECIPROC ALL” function. Each instrument was gradually inserted into the canal using in-and-out movements, at the three levels (cervical, middle and apical) up to WL. Root canal irrigation was performed with 5 mL of 2.5% sodium hypochlorite (NaOCl), using a 30G side-vented needle (NaviTip, Ultradent Products, South Jordan, UT) adapted to a 5 mL syringe (Ultradent Products). As final irrigation, 2 mL of 17% EDTA followed by 5 mL of distilled water were used.
Nano-CT analysis
For nano-CT image acquisition, a high-resolution protocol (2 µm voxel size) was used to scan all teeth before and after instrumentation by using UniTOM HR device (Tescan, Brno, Czech Republic). A tungsten target was employed, and the voltage and current applied were 65 kV and 123 µA, respectively, with an exposure time of 310 ms, 0.5 mm Al filter and 360° rotation around the vertical axis. Images were reconstructed using Acquila reconstruction software (Acquila v.2900), and superimposed with geometric alignment by DataViewer software program (Data Viewer v.1.5.1, Bruker). Quantitative and qualitative analyzes were performed using a CTAn software program (CTAn v.1.14.4, Bruker). To create the three-dimensional images, the CTVox software (CTVox v.3.2; Bruker) was used.
Analyses were performed in the 2-mm apical region of the roots. For qualitative analyses of microcracks, cross-sectional images before and after instrumentation were simultaneously assessed. Then, these were compared by two blinded examiners at two different time points. Microcracks were defined as lines or defects extending from inside the root canal to the dentin or from the outer root surface into the dentin. The distribution of microcracks was expressed as a percentage of the total cross-sectional images18. The qualitative analyses of microcracks were based on their length and width. For length measurement, the extension of slices containing the microcrack was recorded, knowing that each slice represents exactly 0.002 mm, the number of slices was multiplicated for this value, and the result was considered as the microcrack length in mm. For width measurement, the extension of the microcracks was measured in each 10 cross-sectional slices, corresponding to 0.020 mm.
Measurement of the apical transportation were obtained before and after instrumentation on cross-sectional images of the roots. As proposed by Gambill et al.19, the following formula was used to calculate the root canal transportation: (X1–X2) − (Y1–Y2). X1 represented the shortest distance from the outside of the curved root to the periphery of the canal before instrumentation, Y1 represented the shortest distance from the inside of the curved root to the periphery of the canal before instrumentation, X2 represented the shortest distance from the outside of the curved root to the periphery of the instrumented canal, and Y2 represented the shortest distance from the inside of the curved root to the periphery of the instrumented canal. Ten cross-sectional images, determined by the arithmetical mean value, were measured in the last 2 mm of apex of each root.
Statistical analysis
For sample calculation, G* Power 3.1.7 for Windows program (Heinrich-Heine-Universität Dusseldorf, Dusseldorf, Germany) was used. Chi‐square and t test were used with an Alpha type error of 0.05 and Beta power of 0.95. Previous studies were used to determine the specific effect size for percentage of microcracks, 0.9320; and root canal transportation, 3.1121. A total of 8 specimens was indicated as being the ideal size required.
All outcome data were analyzed with the GraphPad Prism 7.00 statistical software package (GraphPad Software, La Jolla, CA, USA).
For qualitative analyses of microcracks, results were expressed as the percentage for each group. Kappa statistics was used for intra- and inter-examiner agreement. McNemar test was used to determine significant differences before and after instrumentation and chi‐square test was used for comparations between groups. For the qualitative analysis of microcracks and transportation, the normality of the data was tested using the Shapiro–Wilk test. The paired t-test was used for comparisons between before and after instrumentation. For comparisons between the groups non-paired t test was used. The level of significance was 5% for all the analysis.
Results
High intra and inter-examiner agreement was found with Kappa values higher than 0.91 for all evaluations.
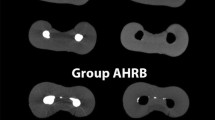
Table 1 shows the mean of slices presenting microcracks, taking into account all evaluated samples. These results indicate a low percentage of microcracks before preparation, being similar between groups (P > 0.05). The enlargement of apical foramina caused new microcracks with a higher percentage when compared to the preparation 1 mm short of apical foramen (P < 0.05). New microcracks were not found in the control group (P > 0.05). Figure 1 shows the appearance of new microcracks only after foraminal enlargement, as this finding was not detected in the control group.
Table 2 indicates the mean values and standard deviations of the length and width of microcracks observed before and after instrumentation. An increase in the microcracks length could be observed after foraminal enlargement (P < 0.05), while in the control group the length values were similar before and after instrumentation (P > 0.05). The width of cracks was similar between groups and before and after instrumentation for both groups (P > 0.05).
Figure 2 indicates significant higher apical transportation after foramen enlargement in comparison with the control group (P < 0.05). Figure 3 shows the presence of microcracks and apical deformations in 3D reconstructed images of nano-CT.
Discussion
The results of this ex-vivo study suggest that the enlargement of apical foramen using a heat-treated reciprocating file size #40 promoted the formation of microcracks and apical transportation. Therefore, the null hypothesis was rejected.
The current literature is still controversial regarding whether the limit of root canal preparation affect treatment prognosis1,2. Moreover, a lack of consistency is observed in relation to the association between the working length and the presence of microcracks5,6,7,8. These conflicting results may be attributed to the use of different methodological designs, teeth morphology and analytical methods15. Therefore, this is the first study to assess the influence of working length on microcrack formation and on root foramen transportation by using nano-CT system.
A recent study did not find any correlation between the working length and the formation of new dentinal microcracks after preparation, however with a different methodological approach, especially regarding the size of apical preparation8. In this previous study, a lower apical preparation was performed until the size 0.25 mm by using Reciproc Blue R25. Nevertheless, the use of larger apical preparation has been recommended to improve the effect of irrigating solution22, and to enhance root canal disinfection23. In addition, the current state-of-art of the heat-treated NiTi instruments, transversal sections and tapers results in safer root canal preparation of curved root canals24. The aforementioned statements justify the use of a larger apical preparation with the reciprocating NiTi file Reciproc Blue R40 in the present study. On the other hand, the higher size of preparation may have influenced microcrack formation after foraminal enlargement.
During sample selection, micro-CT images were used to select only those teeth that did not have microcracks. However, the high-resolution nano-CT images allowed accurate location of the microcracks before and after root canal preparation. In both groups, the microcracks presented small size, achieving an average length of 0.22 mm and an average width of 0.27 mm. When the foraminal enlargement was performed, microcracks were longer after preparation, while in the control group, preoperative microcracks remained unchanged. These microcracks can be a potential site for bacterial infection6, significantly decreasing the long-term prognosis of the teeth.
In both groups, a small percentage of microcracks were observed before root canal instrumentation, as also observed in previous studies25,26,27,28. These microcracks can be attributed to the forces during extraction and excessive occlusal functional loads or even tooth age29. Dentin dehydration during experimental steps also have been referred as a risk factor for microcracks appearance30. However, to prevent the dentin dehydration the teeth were stored in 0.1% thymol and during nano-CT scans the specimens were covered with parafilm paper. Additionally, besides the facts that each specimen has acted as its own control, in the control group no new microcracks were found after instrumentation.
The foraminal enlargement caused more transportation in the root apex than the instrumentation performed 1 mm short of apical foramen. Reciproc Blue files are submitted to a heat treatment that improves their flexibility31, making them suitable for instrumentation of curved root canal32. However, previous studies have shown some transportation, even using instruments with heat treatment8,33. Some authors have also pointed out that this transportation can occur more frequently in preparations 1 mm beyond the foramen8,34. Root canal transportation may negatively affect the outcomes of endodontic treatments35, especially in the apical region that represents a critical zone to maintain infection of the root canal system36. In addition, as can been observed in Fig. 3, the transportation is still associated with deformations of the apical foramen8. In agreement, a previous study also observed higher apical foramen deformations after foraminal enlargement34. Moreover, it should be emphasized that such deformations may affect the sealing of the root canal filling.
Even with the inherent limitations related to an ex-vivo investigation, our results support the concept of restricting the root canal instrumentation procedures to the root canal limit, especially when larger files are used. Although some authors have recommended the enlargement of apical foramen to improve the apical cleaning37, this might increase microcrack formation and canal transportation. Further studies are necessary to verify the influence of foraminal enlargement on filling adaption as well on the root canal cleaning.
Conclusion
In this ex-vivo study, the evaluation of ultra-resolution images showed a higher number of microcracks and apical transportation with the foraminal enlargement technique using heat-treated reciprocating files when compared to conventional preparation until 1 mm short of apical foramen.
Data availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
References
Ricucci, D. Apical limit of root canal instrumentation and obturation, part 1. Literature review. Int. Endod. J. 31, 384–393. https://doi.org/10.1046/j.1365-2591.1998.00184.x (2021).
Yaylali, I. E., Teke, A. & Tunca, Y. M. The effect of foraminal enlargement of necrotic teeth with a continuous rotary system on postoperative pain: A randomized controlled trial. J. Endod. 43, 359–363. https://doi.org/10.1016/j.joen.2016.11.009 (2017).
Borlina, S. C. et al. Influence of apical foramen widening and sealer on the healing of chronic periapical lesions induced in dogs’ teeth. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. Endod. 109, 932–940. https://doi.org/10.1016/j.tripleo.2010.01.028 (2010).
de Souza Filho, F. J., Benatti, O. & de Almeida, O. P. Influence of the enlargement of the apical foramen in periapical repair of contaminated teeth of dog. Oral. Surg. Oral. Med. Oral. Pathol. 64, 480–484. https://doi.org/10.1016/0030-4220(87)90157-5 (1987).
Adorno, C. G., Yoshioka, T. & Suda, H. The effect of working length and root canal preparation technique on crack development in the apical root canal wall. Int. Endod. J. 43, 321–327. https://doi.org/10.1111/j.1365-2591.2010.01684.x (2010).
Adorno, C. G., Yoshioka, T. & Suda, H. Crack initiation on the apical root surface caused by three different nickel-titanium rotary files at different working lengths. J. Endod. 37, 522–525. https://doi.org/10.1016/j.joen.2010.12.002 (2011).
de Oliveira, B. P. et al. Micro-computed tomographic analysis of apical microcracks before and after root canal preparation by hand, rotary, and reciprocating instruments at different working lengths. J. Endod. 43, 1143–1147. https://doi.org/10.1016/j.joen.2017.01.017 (2017).
Vieira, M. et al. Morphologic changes of apical foramen and microcrack formation after foraminal enlargement: A scanning electron microscopic and micro-computed tomographic analysis. J. Endod. 46, 1726–1732. https://doi.org/10.1016/j.joen.2020.07.017 (2020).
Sim, I. G., Lim, T. S., Krishnaswamy, G. & Chen, N. N. Decision making for retention of endodontically treated posterior cracked teeth: A 5-year follow-up study. J. Endod. 42, 225–229. https://doi.org/10.1016/j.joen.2015.11.011 (2016).
Shen, Y., Stojicic, S. & Haapasalo, M. Antimicrobial efficacy of chlorhexidine against bacteria in biofilms at different stages of development. J. Endod. 37, 657–661. https://doi.org/10.1016/j.joen.2011.02.007 (2011).
Wilcox, L. R., Roskelley, C. & Sutton, T. The relationship of root canal enlargement to finger-spreader induced vertical root fracture. J. Endod. 23, 533–534. https://doi.org/10.1016/S0099-2399(97)80316-0 (1997).
Lertchirakarn, V., Palamara, J. E. & Messer, H. H. Patterns of vertical root fracture: factors affecting stress distribution in the root canal. J. Endod. 29, 523–528. https://doi.org/10.1097/00004770-200308000-00008 (2003).
Vaudt, J., Bitter, K., Neumann, K. & Kielbassa, A. M. Ex vivo study on root canal instrumentation of two rotary nickel-titanium systems in comparison to stainless steel hand instruments. Int. Endod. J. 42, 22–33. https://doi.org/10.1111/j.1365-2591.2008.01489.x (2009).
Aksoy, C. et al. Evaluation of XP-endo shaper, reciproc blue, and ProTaper universal NiTi systems on dentinal microcrack formation using micro-computed tomography. J. Endod. 45, 338–342. https://doi.org/10.1016/j.joen.2018.12.005 (2019).
Campello, A. F. et al. Accuracy of microcomputed tomography in detecting dentinal cracks: A correlative study with scanning electron and operative microscopy. Scanning. https://doi.org/10.1155/2021/5571123 (2021).
Kampschulte, M. et al. Nano-computed tomography: Technique and applications. Rofo 188, 146–154. https://doi.org/10.1055/s-0041-106541 (2016).
Schneider, S. W. A comparison of canal preparations in straight and curved root canals. Oral. Surg. Oral. Med. Oral. Pathol. 32, 271–275. https://doi.org/10.1016/0030-4220(71)90230-1 (1971).
Pinto, J. C. et al. Influence of voxel size on dentinal microcrack detection by micro-CT after root canal preparation. Braz. Oral. Res. 35, e074. https://doi.org/10.1590/1807-3107bor-2021.vol35.0074 (2021).
Gambill, J. M., Alder, M. & del Rio, C. E. Comparison of nickel-titanium and stainless steel hand-file instrumentation using computed tomography. J. Endod. 22, 369–375. https://doi.org/10.1016/S0099-2399(96)80221-4 (1996).
Rödig, T. et al. Incidence of microcracks after preparation of straight and curved root canals with three different NiTi instrumentation techniques assessed by micro-CT. Aust. Endod. J. 45, 394–399. https://doi.org/10.1111/aej.12339 (2019).
Pedullà, E. et al. Shaping ability of two nickel-titanium instruments activated by continuous rotation or adaptive motion: A micro-computed tomography study. Clin. Oral. Investig. 20, 2227–2233. https://doi.org/10.1007/s00784-016-1732-4 (2016).
Srikanth, P. et al. Minimal apical enlargement for penetration of irrigants to the apical third of root canal system: A scanning electron microscope study. J. Int. Oral Health. 7, 92–96 (2015).
Rodrigues, R. et al. Influence of the apical preparation size and the irrigant type on bacterial reduction in root canal-treated teeth with apical periodontitis. J. Endod. 43, 1058–1063. https://doi.org/10.1016/j.joen.2017.02.004 (2017).
Pivoto-João, M. M. B., Tanomaru-Filho, M., Pinto, J. C., Espir, C. G. & Guerreiro-Tanomaru, J. M. Root canal preparation and enlargement using thermally treated nickel-titanium rotary systems in curved canals. J. Endod. 46, 1758–1765. https://doi.org/10.1016/j.joen.2020.08.007 (2020).
Ceyhanlı, K. T., Erdilek, N., Tatar, I. & Çelik, D. Response to comments on to our published article entitled: “Comparison of ProTaper, RaCe and Safesider instruments in the induction of dentinal microcracks: a micro-CT study”. Int. Endod. J. 49, 212–215. https://doi.org/10.1111/iej.12497 (2016).
De-Deus, G. et al. Micro-computed tomographic assessment on the effect of ProTaper next and twisted file adaptive systems on dentinal cracks. J. Endod. 41, 1116–1119. https://doi.org/10.1016/j.joen.2015.02.012 (2015).
Bayram, H. M. et al. Micro-computed tomographic evaluation of dentinal microcrack formation after using new heat-treated nickel-titanium systems. J. Endod. 43, 1736–1739. https://doi.org/10.1016/j.joen.2017.05.02 (2017).
Pop, I. et al. Synchrotron light-based μCT to analyse the presence of dentinal microcracks post-rotary and reciprocating NiTi instrumentation. Clin. Oral Investig. 19, 11–16. https://doi.org/10.1007/s00784-014-1206-5 (2015).
PradeepKumar, A. R. et al. Preexisting dentinal microcracks in nonendodontically treated teeth: An ex vivo micro-computed tomographic analysis. J. Endod. 43, 896–900. https://doi.org/10.1016/j.joen.2017.01.026 (2017).
De-Deus, G. et al. Dentinal microcrack development after canal preparation: A longitudinal in situ micro-computed tomography study using a cadaver model. J. Endod. 43, 1553–1558. https://doi.org/10.1016/j.joen.2017.04.027 (2017).
De-Deus, G. et al. Blue thermomechanical treatment optimizes fatigue resistance and flexibility of the reciproc files. J. Endod. 43, 462–466. https://doi.org/10.1016/j.joen.2016.10.039 (2017).
Belladonna, F. G. et al. Micro-computed tomography shaping ability assessment of the new blue thermal treated reciproc instrument. J. Endod. 44, 1146–1150. https://doi.org/10.1016/j.joen.2018.03.008 (2018).
Hu, W., Whitten, B., Sedgley, C. & Svec, T. Effect of three NiTi files on transportation of the apical foramen. Int. Endod. J. 47, 1064–1071. https://doi.org/10.1111/iej.12249 (2014).
Silva Santos, A. M., Portela, F. M. S. F., Coelho, M. S., Fontana, C. E. & De Martin, A. S. Foraminal deformation after foraminal enlargement with rotary and reciprocating kinematics: A scanning electronic microscopy study. J. Endod. 44, 145–148. https://doi.org/10.1016/j.joen.2017.08.013 (2018).
Peters, O. A. et al. Determining cutting efficiency of nickel-titanium coronal flaring instruments used in lateral action. Int. Endod. J. 47, 505–513. https://doi.org/10.1111/iej.12177 (2014).
Siqueira, J. F. & Rôças, I. N. Clinical implications and microbiology of bacterial persistence after treatment procedures. J. Endod. 34, 1291–301.e3. https://doi.org/10.1016/j.joen.2008.07.028ui.iou (2008).
Brandão, P. M. et al. Correction to: Influence of foraminal enlargement on the healing of periapical lesions in rat molars. Clin. Oral. Investig. 23, 2001–2003. https://doi.org/10.1007/s00784-018-2628-2 (2019).
Acknowledgements
This study was entire supported by São Paulo Research Foundation—FAPESP (2019/22885-0).
Author information
Authors and Affiliations
Contributions
J.C.P., K.F.V., R.J., M.T.F. contributed to the study design, data collection, data analysis, and drafting of the manuscript. J.C.P., K.F.V., A.F.L., M.S.P., J.M.G.T., R.J., M.T.F., contributed to the study design, data analysis, and editing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pinto, J.C., de Faria-Vasconcelos, K., Leite, A.F. et al. Effect of foraminal enlargement on microcrack formation and apical transportation: a nano-CT assessment. Sci Rep 13, 4881 (2023). https://doi.org/10.1038/s41598-023-31595-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-31595-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.