Abstract
In this study, we hypothesised that the actual development stage (i.e., current age of the ecosystem) is a determining factor for the magnitude of methane production and emissions in young, northern high-latitude peatlands. We demonstrate that the earliest development of peat soil imposes a sink-to-source shift in the greenhouse warming potential of emerging peatlands in response to climate change that holds feedback mechanisms of importance for short-term (< 100 years) climate warming.
Similar content being viewed by others
Introduction
The northern high latitudes represent a substantial atmospheric source of methane emissions and will be subject to temperature and precipitation increases with projected climate change1. Northern peatlands are a key contributor to this Arctic atmospheric methane source, and it is pivotal to understand the spatial and temporal factors controlling the observed spatial variability in their emissions. Microclimate and plant species composition are well-known drivers that influence the scale of net emissions2. The storage of soil organic carbon (SOC) in cold, waterlogged peatlands is especially facilitated by low microbial decomposition rates in relation to the input of plant organic matter3. The response of the microbial metabolism to increasing temperature is highly variable and difficult to monitor.
We investigated multiple datasets from a peatland located in the Zackenberg valley, NE Greenland4, which has been subject to a large number of previous methane flux studies4 and a wide range of related experiments and process-based modelling efforts5,6,7. We focus on the particular hypothesis that the peat age and peatland development stage strongly influence the spatial variability in methane fluxes, and we use radiocarbon dating of the basal peat age in distributed locations of the fen to study this. If the hypothesis is true, this will in turn add additional explanatory power to ecosystem models representing the spatial distribution of methane emissions.
Results and discussion
Despite different focusses in previous methane flux studies from Zackenberg, they all include comparable control measurements in undisturbed plots4. When comparing these studies, the fluxes vary by a factor of more than five over relatively short distances (less than 100 m). Although part of this variability can be explained by interannual fluctuations, a substantial variation in base fluxes remains when the temporal aspect is excluded. Base flux is here defined as the flux measured in undisturbed (by experimental treatments, i.e., control plots) locations of our studied fen peatland on comparable dates (July and August only) and times of day and obtained using similar flux chamber methodologies. An overview of these different methodologies is provided in Scheller et al.4.
In addition to direct physical drivers such as temperature and soil moisture, vascular plant species composition and density are known to act as drivers/controls of base fluxes2. Detailed information from multispectral analyses of the vascular plant cover (and possibly species-level recognition) may be used for extrapolation purposes that relate to the part of the methane flux variability associated with this factor (Fig. 1). However, the substantial variability observed from multiple flux studies summarized in Fig. 1 and Table 1 comes from periods with similar microclimatic conditions and locations sharing a homogeneous vegetation cover (dominated by Dupontia psilosantha and Eriophorum scheuchzeri wet tundra peatland). This suggests that other factors, such as the soil/peat development stage, may also contribute to explaining the documented flux variability.
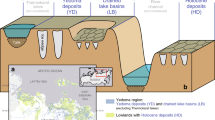
Angled view of the DEM-based orthophoto illustrating the Zackenberg fen site across a 400 m × 450 m area. The map includes the locations of all cores (Table 1a), CH4 flux study sites (Table 1b) and EC tower (Table S2) together with an indication of the main surface water flow direction. Additionally, an NDVI map (August 2020) characterising the local plant greenness overlays the map. The areas surrounding the main flow are found to hold the older, deeper peat deposits and hence higher methane emissions. In support of this, the older peat age determinations at C3, C6, C7, and C8 are all located centrally to the hydrological flow lines and coincide with the vicinity of high mcrA gene counts (for C3) and fluxes measured. F3 and F6 are both located approximately 300 m from the edge of the map. Note that the surface topography, infrastructure, and subsurface processes are not to scale. Image source: Greenland Ecosystem Monitoring, 2020.
The spatial distribution (Fig. 1) and magnitude variability (Table 1b) of 12 published studies focusing on various aspects of methane fluxes in the Zackenberg fen peatland area over the time period 1997–2021 was previously investigated4. The base fluxes reported by all these studies characterising the fen peatland area span a range from < 1 to > 8 mg CH4 m−2 h−1. Such an extensive range suggests that multiple processes are involved, and there is a continued need to better understand the underlying mechanisms controlling this peatland spatial variability.
To examine the flux variability in more detail, we collected soil cores randomly from the fen peatland area and analysed them for 14C age and methanogenic activity (Table 1a and Table S1). Additional earlier published 14C age data from the same fen area were also included. This particular fen peatland reveals recent ecosystem development dating back approximately one thousand years and with large areas only emerging as peatland ecosystems (vegetated wet tundra as opposed to barren or floodplain) since the Little Ice Age (fourteenth to eighteenth century). As a result, this cold period has left a very short time for actual peat development. Most northern peatlands date back much further in time (averaging 4–5 k years8) and include deeper peat deposits. However, current growing season methane emissions in several in parts of the Zackenberg fen are already comparable in strength to the emissions from deeper peatlands in southern parts of the Arctic9.
This recent development of the studied tundra landscape may resemble the general future for the entire Arctic domain as a consequence of predicted climate warming16. These consequences also include further permafrost thawing and subsequent changes in productivity and peat formation16.
The fen peatland in Zackenberg has been reported as a CO2 sink in all years except one (2018) associated with a substantial late snowmelt period (due to an anomalously snow-rich winter) as measured over more than 10 years using the eddy covariance (EC) technique7 (Fig. 1 and Table S2). It is further expected that the CO2 sink strength of this fen system will continue to increase, with an additional 10 g CO2 m−2 year−1 by the end of 210017. The central part of the fen (Location 74.48°N, −20.56°E) is now also a certified Class 2 station in the Integrated Carbon Observing System (ICOS), and 2021 included the first-ever complete winter season (including the November–April period during which Zackenberg Research Station is unattended). The annual C uptake is higher than that in another more southern tundra fen site in SE Greenland due to its documented nutrient-richer conditions7. The annual CO2 uptake varies between −90 g CO2 m−2 year−1 and + 21 g CO2 m−2 year−1, with an average of −50 g CO2 m−2 year−1 7 over a decadal scale of measurements (Table S2). Therefore, the consideration of methane as a key component of the greenhouse gas (GHG) balance of this fen peatland ecosystem needs to be aligned with the significant variability in the CO2 sink functioning.
We hypothesised that the early stages of organic soil and peat formation and development could initiate a rapid increase in methane emissions relative to a barren landscape. The spatial difference among sites related to basal peat age could then also be a factor determining the observed local-scale differences in base fluxes.
Microbial methane cycling includes methane-producing, methanogenic, methane-releasing, and methanotrophic processes, which have been studied in peatlands before18. While methane oxidation can offset peatland methane emissions3, we investigated whether the abundance of methyl-coenzyme M reductase (mcrA), a key methanogenesis metabolism gene, as a proxy for the abundance of methanogenic activity19, increases with the onset of early organic layer/peat development. The highest mcrA gene counts were found to coincide with the oldest BP age and highest methane emissions, although no statistical correlation could be found.
Relatively older samples are situated close to the main hydrological flow lines crossing the fen (Fig. 1) and are associated with the presence of methane released from vascular plants but also the presence of peat-forming mosses in the ground layer that in turn stimulate the initiation of peat development. All these factors combined may then lead to a spatial pattern associated with higher methanogenesis gene counts in the older parts of the peat (Table 1a). This may potentially further contribute to the spatial pattern of genes that suggest/align with a spatial pattern of overall methane fluxes (Table 1b). The above-described independent lines of evidence led us to assume that these flow lines were the likely initiation centres for the onset of fen peatland development, which would fit with early and more recent studies on early formation patterns20,21.
The combined in situ observations (Table 1) provide the possibility of evaluating the total GHG balance associated with the early development stages over a few hundred years of an emerging the peatland ecosystem in the high Arctic. This process also suggests the potential development towards conditions already prevailing at lower latitudes. The observed and predicted trends towards warmer and wetter conditions in the Arctic will lead to more widespread wet tundra peatland development associated with the “greening” of the tundra22. The development of the GHG balance of these changing landscapes will, therefore, also hold important implications for further climate development and hence warrant inclusion in Earth system models23.
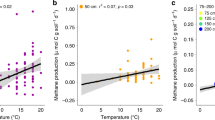
The results presented in Table 1 and Fig. 2 show the relationship among vegetation cover (as proxy from plant greenness), basal peat age and base flux of methane. Although the connection between vegetation composition and cover has been well documented before15, the combination with very young peat age data is new. Our data suggest that the oldest parts of the fen peatland complex are not only closer to the flow lines but also associated with higher fluxes despite similar vegetation cover. Similarly, the results indicate the highest mcrA gene abundance in the two oldest, deepest and lowest SOC basal peat samples (Table S1). We interpret this variability in methanogenic abundance as follows: under more anaerobic conditions, methanogenesis dominates despite the low energy yield for the microorganisms, while more energy-efficient anaerobic processes dominate in shallow, carbon-rich samples, lowering the methanogen abundance24.
Annual methane emissions plotted against basal peat ages from closest vicinity locations (red dots, left axis). Furthermore, the GHG balance was calculated using a standard global warming potential (GWP) calculation (100 year GWP CH4: 28) for methane in CO2 equivalents combined with the annual fen CO2 flux range from the 11-year eddy covariance dataset (upper + 23 g CO2-C m−2 year−1, lower – 90 g CO2-C m−2 year−1, respectively; Table S2) (light and dark green dots, right axis). Note the age range (0–100 years) depending on CO2 sink strength where the fen ecosystem shifts from being a sink to a source of warming-inducing gases.
An analysis of the flux emission magnitude as a function of the age of peatland development shows a rapid increase in methane emissions towards a base level obtained after 100 years (Fig. 2). By further (1) combining our data with the range of contemporary observed CO2 balance from the same ecosystem7 and (2) applying a standard global-warming potential (GWP) conversion of CH4 emissions to CO2-equivalents25, our analysis reveals that the GHG balance likely shifts from an initial sink to a source of C as the methane emissions strengthen after less than 100 yrs. The reverse shift (from source to sink) is well documented for subarctic fen peatland ecosystems over timescales from 1000 to 9000 years e.g. 26. Here, we find an initial sink-to-source shift that may take place at a much faster pace and within a timeframe covered by present-day climate projections from, e.g., the state-of-the-art CMIP6 Earth System Models27,28 (regardless of the SSP emission scenario) at a 100–300-year timescale. Consequently, the positive feedback mechanism shown here at the centennial scale (which is shaped by the combined climate and peatland development) will most likely be replaced over a the millennial timescale by a gradual cooling effect as the peatlands develop and accumulate greater amounts of peat26,29. On the shorter centennial time scale, it should also be kept in mind that from a climate feedback perspective, the natural ecosystem emission dynamics need to be considered in light of the current and future impacts of anthropogenic emissions30.
This study has clear limitations in the number of soil samples obtained versus available flux data locations. However, assuming the documented process will be applicable to widespread greening and wetting of natural high Arctic tundra areas, this rapid conversion over just 100 years points to a highly dynamic shift in greenhouse forcing from natural ecosystems with possible implications for climate projections in the 100–300-year timeframe. Further detailed studies from Zackenberg and other similar sites with early peat development will be necessary.
Methods
Spatial mapping
An unmanned aerial vehicle (UAV) recorded data on 3 August 2020 at a spatial resolution of 5 × 5 cm. In addition, the normalised difference vegetation index (NDVI) map based on multispectral data indicates variability in plant photosynthetic activity, while the natural colour orthophoto serves as an overview of the study area. Maps of the stream order were drawn using ESRI ArcMap 10.8.
Fen core sampling & physical soil analyses
In 2020, five fen cores were sampled, of which the deepest 2 cm were separated and stored at 4 °C until later laboratory analyses. The weight-based relative soil organic matter and water content were determined by loss on ignition at the Department of Ecoscience, Risø, Aarhus University, and radiocarbon dating was performed at the Department of Geology, Lund University, as described previously22,31.
mcrA quantitative gene abundance
Genomic DNA from the methanogen Methanosarcina barkeri DSM 8687 (Leibniz Institute DSMZ—German Collection of Microorganisms and Cell Cultures GmbH, Germany) in the five fen cores was extracted according to the manufacturer’s instructions in triplicate with a DNeasy® Powerlyzer® Microbial Kit (Qiagen, Hilden, Germany) and quantified with a Qubit® 2.0 Fluorometer (Thermo Fisher Scientific, Life Technologies, Roskilde, Denmark). Simultaneously, the mcrA (methyl-coenzyme M reductase) gene copy numbers were determined by counting cells (100 × 1.30 plan-neofluar Zeiss oil immersion) in an anaerobic Methanosarcina barkeri culture after cells of known optical density were fixated in 4% formaldehyde solution and stained with 1:10 diluted Acridine Orange. With the M. barkeri DNA extracts as standards, the samples’ mcrA gene abundance was measured with qPCR utilising 10 µM primers mlas (5’-GGTGGTGTMGGDTTCACMCARTA-3’) and mcrA-rev (5’-CGTTCATBGCGTAGTTVGGRTAGT-3’)16, 5 × HOT FIREPol® EvaGreen® qPCR Supermix Polymerase (Solis Biodyne, TAG Copenhagen A/S, Copenhagen, Denmark) at 95 °C for 12 min., followed by 35 cycles of 15 s at 95 °C, 30 s at 55 °C and 30 s at 72 °C.
Methane flux studies
The data presented in the top two rows (F1 and F2) of Table 1b report mean values measured from automatic chambers between July and August in each year during the 2006–2019 period. F1 represents the inner 6 chambers, and F2 represents the outer 4 (Mastepanov et al., in prep.). The remaining studies (F3-6) used unmanipulated chamber measurements during June–August in different years. All studies used the closed-chamber technique, although with different setups (i.e., gas analysis, chamber design, sample time, plot number, and measurement frequency; see Table 2 in Scheller et al.4).
Net ecosystem exchange (NEE) of CO2
The available CO2 data from the Zackenberg fen consist of high temporal resolution measurements between 2008 and 2018 using the eddy covariance (EC) technique7. The EC system was equipped with a closed-path infrared gas analyser LI-6262 and 3-D sonic anemometer Gill R2 until August 2012, when it was upgraded to an enclosed-path LI-7200 and Gill HS. The sonic anemometer in Zackenberg was installed at a height of 3 m (the air intake was attached at the same level). High-frequency (10 Hz) CO2 concentration and wind component data were processed according to FLUXNET/ICOS community technique standards7. The aggregated 30 min NEE fluxes were further quality-checked and postprocessed using a gap-filling technique based on marginal distribution sampling from the ReddyProc R tool32. Detailed information on the EC system setup, flux preprocessing, quality checks of the systems, and data postprocessing (NEE gap-filling) can be found in reference 7. The CO2 fluxes reported in Table S2 follow the standard micrometeorological sign of convention, which is CO2 uptake and release as negative and positive fluxes, respectively.
Data availability
The long-term flux datasets analysed and used during the current study are available in the open-source repository Greenland Ecosystem Monitoring Database https://data.g-e-m.dk. The radiocarbon and molecular datasets are available from the corresponding author upon reasonable request.
References
AMAP. Arctic Climate Change Update 2021: Key Trends and Impacts. Summary for Policy-Makers. Vol. 16 (Tromsø, 2021).
Ström, L., Mastepanov, M. & Christensen, T. R. Species-specific effects of vascular plants on carbon turnover and methane emissions from wetlands. Biogeochemistry 75, 65–82. https://doi.org/10.1007/s10533-004-6124-1 (2005).
Tveit, A., Schwacke, R., Svenning, M. M. & Urich, T. Organic carbon transformations in high-Arctic peat soils: Key functions and microorganisms. ISME J. 7, 299–311. https://doi.org/10.1038/ismej.2012.99 (2013).
Scheller, J. H., Mastepanov, M., Christiansen, H. H. & Christensen, T. R. Methane in Zackenberg Valley, NE Greenland: Multidecadal growing season fluxes of a high-Arctic tundra. Biogeosciences 18, 6093–6114. https://doi.org/10.5194/bg-18-6093-2021 (2021).
Falk, J. M., Schmidt, N. M., Christensen, T. R. & Strom, L. (2015) Large herbivore grazing affects the vegetation structure and greenhouse gas balance in a high arctic mire. Environ Res Lett. doi: https://doi.org/10.1088/1748-9326/10/4/045001.
Strom, L. et al. Controls of spatial and temporal variability in CH4 flux in a high arctic fen over three years. Biogeochemistry 125, 21–35. https://doi.org/10.1007/s10533-015-0109-0 (2015).
López-Blanco, E. et al. Multi-year data-model evaluation reveals the importance of nutrient availability over climate in arctic ecosystem C dynamics. Environ Res Lett 15, 094007. https://doi.org/10.1088/1748-9326/ab865b (2020).
Gorham, E. Northern peatlands—Role in the carbon-cycle and probable responses to climatic warming. Ecol. Appl. 1, 182–195 (1991).
Mastepanov, M. et al. Revisiting factors controlling methane emissions from high-Arctic tundra. Biogeosciences 10, 5139–5158. https://doi.org/10.5194/bg-10-5139-2013 (2013).
Palmtag, J. et al. Storage, landscape distribution, and burial history of soil organic matter in contrasting areas of continuous permafrost. Arct. Antarct. Alp. Res. 47, 71–88. https://doi.org/10.1657/Aaar0014-027 (2015).
Strom, L., Tagesson, T., Mastepanov, M. & Christensen, T. R. Presence of Eriophorum scheuchzeri enhances substrate availability and methane emission in an Arctic wetland. Soil. Biol. Biochem. 45, 61–70. https://doi.org/10.1016/j.soilbio.2011.09.005 (2012).
Falk, J. M., Schmidt, N. M. & Ström, L. Effects of simulated increased grazing on carbon allocation patterns in a high arctic mire. Biogeochemistry 119, 229–244. https://doi.org/10.1007/s10533-014-9962-5 (2014).
Tagesson, T. et al. Land-atmosphere exchange of methane from soil thawing to soil freezing in a high-Arctic wet tundra ecosystem. Global Change Biol. 18, 1928–1940. https://doi.org/10.1111/j.1365-2486.2012.02647.x (2012).
Christensen, T. R. et al. Trace gas exchange in a high-arctic valley 1. Variations in CO2 and CH4 flux between tundra vegetation types. Global Biogeochem. Cy. 14, 701–713 (2000).
Joabsson, A. & Christensen, T. R. Methane emissions from wetlands and their relationship with vascular plants: An Arctic example. Global Change Biol. 7, 919–932 (2001).
AMAP. AMAP Arctic Climate Change Update 2021: Key Trends and Impacts. viii+148 (AMAP, 2021).
López-Blanco, E. et al. The future of tundra carbon storage in Greenland—Sensitivity to climate and plant trait changes. Sci. Total Environ. 846, 157385. https://doi.org/10.1016/j.scitotenv.2022.157385 (2022).
Høj, L., Olsen, R. A. & Torsvik, V. L. Effects of temperature on the diversity and community structure of known methanogenic groups and other archaea in high Arctic peat. ISME J. 2, 37–48. https://doi.org/10.1038/ismej.2007.84 (2008).
Freitag, T. E. & Prosser, J. I. Correlation of methane production and functional gene transcriptional activity in a peat soil. Appl. Environ. Microb. 75, 6679–6687. https://doi.org/10.1128/Aem.01021-09 (2009).
Clymo, R. S. The limits to peat bog growth. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 303, 605–654 (1984).
Yu, Z., Loisel, J., Brosseau, D. P., Beilman, D. W. & Hunt, S. J. Global peatland dynamics since the Last Glacial Maximum. Geophys. Res. Lett. . https://doi.org/10.1029/2010GL043584 (2010).
Box, J. E. et al. Key indicators of Arctic climate change: 1971–2017. Environ Res Lett 14, 045010. https://doi.org/10.1088/1748-9326/aafc1b (2019).
Müller, J. & Joos, F. Committed and projected future changes in global peatlands—Continued transient model simulations since the Last Glacial Maximum. Biogeosciences 18, 3657–3687. https://doi.org/10.5194/bg-18-3657-2021 (2021).
Ernakovich, J. G., Lynch, L. M., Brewer, P. E., Calderon, F. J. & Wallenstein, M. D. Redox and temperature-sensitive changes in microbial communities and soil chemistry dictate greenhouse gas loss from thawed permafrost. Biogeochemistry 134, 183–200. https://doi.org/10.1007/s10533-017-0354-5 (2017).
Masson-Delmotte, V. P., Zhai, A., Pirani, S.L., Connors, C., Péan, S., Berger, N., Caud, Y., Chen, L., Goldfarb, M.I., Gomis, M., Huang, K., Leitzell, E., Lonnoy, J.B.R., Matthews, T.K., Maycock, T., Waterfield, O., Yelekçi, R. Y., & Zhou, B. (eds). Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. (Cambridge University Press, 2021).
Piilo, S. R. et al. Spatially varying peatland initiation, Holocene development, carbon accumulation patterns and radiative forcing within a subarctic fen. Quatern. Sci. Rev.. https://doi.org/10.1016/j.quascirev.2020.106596 (2020)
O’Neill, B. C. et al. The scenario model intercomparison project (ScenarioMIP) for CMIP6. Geosci. Model Dev. 9, 3461–3482. https://doi.org/10.5194/gmd-9-3461-2016 (2016).
Arndal, M. F. et al. Seasonal variation in gross ecosystem production, plant biomass, and carbon and nitrogen pools in five high arctic vegetation types. Arct. Antarct. Alp. Res. 41, 164–173. https://doi.org/10.1657/1938-4246-41.2.164 (2009).
Frolking, S., Roulet, N. T. & Fuglestvedt, J. How northern peatlands influence the Earth’s radiative budget: Sustained methane emission versus sustained carbon sequestration. J. Geophys. Res. Biogeosci. 111, G01008. https://doi.org/10.1029/2005JG000091 (2006).
Christensen, T. R., Arora, V. K., Gauss, M., Höglund-Isaksson, L. & Parmentier, F.-J.W. Tracing the climate signal: Mitigation of anthropogenic methane emissions can outweigh a large Arctic natural emission increase. Sci. Rep.-UK 9, 1146. https://doi.org/10.1038/s41598-018-37719-9 (2019).
Christensen, T. R. et al. Multiple ecosystem effects of extreme weather events in the Arctic. Ecosystems 24, 122–136. https://doi.org/10.1007/s10021-020-00507-6 (2021).
Reichstein, M., Moffat, A. M., Wutzler, T., & Sickel, K. REddyProc: Data Processing and Plotting Utilities of (Half-)Hourly Eddy-Covariance Measurements. R Package Version 0.8-2/r14 (Version R Package Version 0.8-2/r14). (2016).
Funding
This study was funded by the Danish Energy Agency and the Danish Environmental Protection Agency through the Greenland Ecosystem Monitoring Program. The study was also supported by the Greenland Research Council.
Author information
Authors and Affiliations
Contributions
T.R.C. wrote the draft manuscript. M.S. produced the molecular and peat age data. J.S. and D.A.R. provided the spatial and hydrological mapping. M.M., M.J.K. and E.L.B. supplied the flux data. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Christensen, T.R., Scheller, J., Scheel, M. et al. Rapid shift in greenhouse forcing of emerging arctic peatlands. Sci Rep 13, 2828 (2023). https://doi.org/10.1038/s41598-023-29859-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-29859-4
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.