Abstract
Oxygenated hemoglobin (OxyHem) in arterial blood may reflect disease severity in patients with systemic sclerosis (SSc). The aim of this study was to analyze the predictive value of OxyHem in SSc patients screened for pulmonary hypertension (PH). OxyHem (g/dl) was measured by multiplying the concentration of hemoglobin with fractional oxygen saturation in arterialized capillary blood. Prognostic power was compared with known prognostic parameters in SSc using uni- and multivariable analysis. A total of 280 SSc patients were screened, 267 were included in the analysis. No signs of pulmonary vascular disease were found in 126 patients, while 141 patients presented with mean pulmonary arterial pressure ≥ 21 mmHg. Interstitial lung disease (ILD) was identified in 70 patients. Low OxyHem ≤ 12.5 g/dl at baseline was significantly associated with worse survival (P = 0.046). In the multivariable analysis presence of ILD, age ≥ 60 years and diffusion capacity for carbon monoxide (DLCO) ≤ 65% were negatively associated with survival. The combination of low DLCO and low OxyHem at baseline could predict PH at baseline (sensitivity 76.1%). This study detected for the first time OxyHem ≤ 12.5 g/dl as a prognostic predictor in SSc patients. Further studies are needed to confirm these results.
Similar content being viewed by others
Introduction
Systemic sclerosis (SSc) is a rare, autoimmune disease characterized by diffuse fibrosis, inflammation and vasculopathy and remains one of the most complex and lethal diseases among connective tissue disorders1. The severity of organ involvement predicts the outcome in SSc patients2,3.
Pulmonary complications, mainly pulmonary arterial hypertension (PAH) and interstitial lung disease (ILD), remain the leading cause of mortality among SSc patients1. Consequently, screening and early diagnosis of both the disease and organ involvement are crucial for prognosis.
The establishment of risk stratification methods is essential to improve the survival through rapid medical and therapeutic interventions. The majority of available scoring models are yet to be validated1. Previous studies have already reported the prognostic meaning of impaired oxygen saturation (SaO2) during exercise for survival and disease progression among patients with SSc-ILD4,5. SaO2 may be influenced by both cardiac and pulmonary comorbidities. Furthermore, anemia has been shown to be a prognostic predictor of survival among patients with SSc6. A combination of these parameters might therefore be beneficial for risk stratification and prognosis.
Trudzinski et al. showed that apart from white blood cell count, oxygenated hemoglobin (OxyHem), the product of hemoglobin concentration and fractional oxygen saturation (SaO2) in arterial capillary blood gas analysis, is a suitable and easy to estimate novel biomarker for the prognosis of patients with chronic obstructive pulmonary disease (COPD)7. OxyHem measured in patients with stable COPD, but also during acute COPD exacerbations was independently associated with higher mortality among those patients8. OxyHem in arterial blood may also be a novel biomarker for the prognosis in patients with SSc.
Hence, the aim of this study was to analyze the prognostic value of OxyHem in SSc patients referred for screening for pulmonary hypertension (PH). Furthermore, the study investigated the association of OxyHem with the development of pulmonary vascular disease (PVD) among these patients.
Results
Baseline clinical and demographic characteristics
A total of 280 patients with SSc were screened for pulmonary hypertension. From this cohort, 13 patients met exclusion criteria, leading to a final study group of 267 patients (Fig. 1, Table 1). The mean age of the patients was 59.8 ± 13.4 years, 82% were female, 73.8% had limited cutaneous SSc (lcSSc) and 26.2% diffuse cutaneous SSc (dcSSc). The mean duration of SSc at baseline was 9.0 ± 9.3 years. Of these, 70 had significant ILD (26.2%) and 141 (52.8%) showed a mean pulmonary arterial pressure (mPAP) ≥ 21 mmHg (Fig. 1) while 126 had no signs of PH (41.6%). Of the 141 patients with PH 19 presented with PH due to left heart disease (Fig. 1). Patients with PAH received targeted PAH medication according to the contemporary guidelines9. Out of 267 patients, 46.4% were in World Health Organization functional class (WHO-FC) II, 30.3% in WHO-FC III and 2.6% in WHO-FC IV. The mean 6-min walking distance (6MWD) was 427.7 ± 102.5 m. Comorbidities were identified in 166 patients: 101 (37.8%) suffered from arterial hypertension, 58 (21.7%) had coronary heart disease, one (0.4%) had reported gastric antral vascular ectasia, three (1%) had lymphoma in the past history and 41 (15.4%) were obese, having a body mass index (BMI) > 30 kg/m2.
Clinical parameters characterizing patients with low OxyHem
The median OxyHem in this cohort was 12.89 g/dl (interquartile range: 151.8). Receiver operating characteristic (ROC) analyses for OxyHem were in line with the cutoff from the literature7 of 12.5 g/dl (ROC area under the curve P = 0.004). Overall, 94 SSc patients had an OxyHem ≤ 12.5 g/dl and 173 SSc patients OxyHem values above 12.5 g/dl (Table 2). Compared to patients with OxyHem > 12.5 g/dl, patients with low OxyHem were mainly female (n = 87, 92.6% vs. n = 132, 76.3%, P = 0.001), had mostly lcSSc (n = 75, 79.8% vs. n = 122, 70.5%, P = 0.011), a more impaired iron metabolism in terms of lower ferritin levels (75.3 ± 83.6 vs. 126.7 ± 150.6 µg/l, P = 0.004) in normal range, mean corpuscular volume of erythrocytes (MCV) (88.1 ± 8.7 vs. 90.3 ± 5.2 fl, P = 0.027) and mean corpuscular hemoglobin in erythrocytes (29.1 ± 3.0 vs. 30.1 ± 2.4 pg, P = 0.006). Moreover, they were clinically more limited in terms of WHO-FC (P = 0.001), shorter 6MWD (408.6 ± 101.3 vs. 437.8 ± 102.0 m, P = 0.036) and greater hemodynamic impairment with a higher pulmonary vascular resistance (PVR) (3.2 ± 2.6 vs. 2.4 ± 1.9 Wood Units (WU), P = 0.018) and mPAP (25.6 ± 11.7 vs. 21.6 ± 9.3 mmHg, P = 0.005) (Table 2). There was no significant difference in N-terminal pro brain natriuretic peptide (NTproBNP) between the two groups. Out of 94 patients with OxyHem ≤ 12.5 g/dl, 29 suffered from ILD (23.7%). In 59 patients with low OxyHem, mPAP was ≥ 21 mmHg (Fig. 1). Concomitant ILD was diagnosed in 70 patients, out of which 50 showed an mPAP ≥ 21 mmHg. Elevation of pulmonary arterial wedge pressure (PAWP) > 15 mmHg was detected in 22 patients, 19 showed an mPAP ≥ 21 mmHg (Fig. 1). Six patients had PH due to both left heart and lung disease. Pulmonary function test (PFT) parameters were significantly more impaired among patients with lower OxyHem including maximal vital capacity (VCmax), forced expiratory volume in one second (FEV1) and total lung capacity (TLC). In the laboratory analysis C-reactive protein (CRP) was higher in the low OxyHem group (P = 0.020) and these patients had significantly lower hemoglobin (Hb) (P < 0.0001). There was no difference in renal function between the two groups (Table 2).
Survival in SSc patients
Within the observation period of 3.2 ± 2.6 (median 2.7; interquartile range 3.5) years 47 patients died (17.6%): 22 due to PH (46.8%), 4 due to PH and/or ILD (8.5%), 3 due to ILD (6.4%), 7 due to cancer (14.9%), one due to pulmonary sepsis (2.1%) and 3 due to left heart disease (6.4%). Two patients underwent lung transplantation due to PH and ILD. In seven cases the cause of death remained unknown (14.9%). The mean estimated overall survival was 7.7 ± 0.3 standard error of the mean years (95% confidence interval 7.1 to 8.2) from baseline. Survival was similar in male and female patients (P = 0.624) and between patients with dcSSc and lcSSc (P = 0.694).
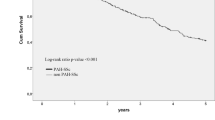
In the univariable Kaplan–Meier analysis, OxyHem ≤ 12.5 g/dl (P = 0.046), age at baseline of ≥ 60 years (P = 0.002), the presence of ILD (P < 0.0001), high PVR ≥ 2 WU (P = 0.002) and low DLCO ≤ 65% predicted (P < 0.001) were significantly associated with worse survival (Table 3). White blood cell (WBC) count ≥ 10,000/ml was analyzed together with OxyHem in the initially described COPD cohort7. It was also a significant prognostic parameter for survival in our cohort (P = 0.02).
Patients with low OxyHem ≤ 12.5 g/dl showed significantly worse survival than patients with OxyHem > 12.5 g/dl with 1-, 3-, 5-year survival of 98.7%, 82.9% and 71.2% versus 99.3%, 95.7% and 80.3%, respectively (Kaplan–Meier P = 0.046, Fig. 2). In the multivariable Cox-regression analysis, low DLCO, the presence of ILD and older age were identified as independent prognostic predictors for survival (combined P < 0.0001, Fig. 3).
OxyHem in pulmonary vascular disease
At baseline, patients with PH (mPAP ≥ 21 mmHg) had the highest rate of low OxyHem (41.8%), compared to patients with normal hemodynamics (27.8% low OxyHem; chi-square P = 0.016). Low OxyHem at follow-up was though not associated with hemodynamic status (P = 0.562) (Table 4). PH at baseline could be predicted with 41.8% sensitivity and 72.7% specificity by low OxyHem. When combining low DLCO and low OxyHem, prediction of PH at baseline had a sensitivity of 76.1% and a specificity of 50.6%. In comparison, step 1 of the DETECT algorithm10 reached 85.3% sensitivity and 37.0% specificity. In step 2 of the DETECT algorithm10, 79.4% sensitivity and 73.6% specificity could be reached for PH at baseline.
When using the three independent risk factors DLCO ≤ 65% predicted, age ≥ 60 years and presence of ILD, occurrence of PH with mPAP ≥ 21 mmHg during follow-up could be predicted with a sensitivity of 91.1% and a specificity of 34.6% when at least one of the three risk factors was met. The combination of DLCO ≤ 65% predicted and OxyHem ≤ 12.5 g/dl had a sensitivity of 58.7% and a specificity of 72.9% to predict PH during follow-up when at least one of the two risk factors was met (prediction of 44 out of 57; true negative 35 out of 66).
Discussion
This study detected for the first time that SSc patients with an OxyHem level ≤ 12.5 g/dl have a significantly worse survival than patients with an OxyHem level > 12.5 g/dl (P = 0.046). ILD, low DLCO, older age were robust prognostic factors associated with survival in the multivariable analysis. Furthermore, low OxyHem in combination with low DLCO at baseline were predictors of PH at baseline and during follow-up (sensitivity 76.1% and 58.7%, specificity of 50.6% and 72.9%, respectively). To our knowledge, this is the first study indicating the impact of OxyHem as predictor of survival among SSc patients.
OxyHem and SSc prognosis
Lower OxyHem was identified in patients with a shorter survival, highlighting the impact of oxygenation and the significance of oxygen transport among patients suffering from extended fibrosis and vasculopathy, that typically characterize SSc11. Patients with low OxyHem revealed more often clinical characteristics with worse survival in previously described risk stratification models, such as greater WHO-FC12, elevated CRP1, presence of ILD1, lower VCmax and DLCO13 as well as lower Hb and shorter 6MWD12,13. Patients with lcSSc and female patients had significantly more often a low OxyHem level in our cohort. As these patients would not be suspected to be at risk for an impaired outcome, this underlines the importance of evaluating patients also at low risk1,12,13,14,15,16.
SSc and oxygenation
The importance of oxygen saturation after exercise as a predictor of mortality among SSc-ILD patients was previously evaluated5. Exercise induced desaturation was also shown to be predictor of mortality among patients at risk for PH in the PHAROS registry, though SaO2 at rest did not differ between the groups17 and was found to be associated with 3-year-survival in the DIBOSA study18. Wu et al. reported that desaturation < 94% in 6MWD combined with the presence of arthritis was associated with disease progression in SSc-ILD4. In contrast, for the calculation of OxyHem, resting oxygen saturation can be used, which makes the parameter easier to determine and less exhausting for the patient compared to assessment of oxygen saturation during exertion.
PVD
In our cohort, OxyHem was associated with pulmonary vasculopathy only at baseline but not at follow-up. This might be caused by initiation of PAH targeted therapy after diagnosis. Hoeper et al. reported that oxygenation was not a prognostic marker for survival among prevalent patients with idiopathic PAH under therapy, which supports our results19. Recently, Valentin et al. (2021) showed that oxygen desaturation (SaO2) ≥ 3% at the time of the first follow-up after one year in combination with low DLCO was significantly associated with worse prognosis of PAH20, matching our results linking low OxyHem and low DLCO with early PVD. In this cohort, male sex did not correlate with survival consistent with results from the literature6,21,22.
Strengths and limitations of the study
This study provides important insights into prognostic predictors and stratification models in SSc patients screened for PH. The screening for PH was performed in an expert center including a thorough diagnostic workup, leading to a well characterized study cohort. OxyHem offers a parameter, which is pathophysiologically important in different organ involvements in SSc such as ILD, PH and cardiac diseases. While it was a significant predictor in the univariable analysis it was outperformed in the multivariable model. This could be due to the limited sample size or the strong effect of ILD on prognosis. Since we only included extensive ILD in our assessment, this could have also biased the results. One of the main limitations of this study is its retrospective character leading to a smaller data set due to missing parameters. Early prognostic markers need to be investigated in future, prospective studies. We also had no control cohort of healthy individuals or other well characterized patients to compare our results to. However, the prognostic value of OxyHem has already been described in COPD patients7. Furthermore, other organ manifestations of SSc, such as gastrointestinal manifestations and renal crisis, could not be considered due to missing data. Although the cohort size of our study is large for such a rare disease, sample size of subgroups was too small to differentiate the impact of OxyHem on survival separately in ILD and PH. Another limitation is that in our cohort blood gas analysis was performed with arterialized capillary blood from the earlobe, which might not be included in standard screening procedures by rheumatologists. However, this method has already shown good correlation with arterial puncture23. The meaning of OxyHem during the course of the disease including follow-up data should be addressed in future studies. Finally, our cohort included only SSc patients referred for screening for PH, which could have led to biased data.
Conclusions
The results of this study revealed that OxyHem may be a new biomarker which could be used for risk stratification in SSc and identification of patients with early PVD and PH, respectively. OxyHem is easy to calculate in daily routine and values ≤ 12.5 g/dl were significantly associated with a worse survival. Moreover, the combination of DLCO ≤ 65% predicted and low OxyHem could predict the development of early PVD in this cohort. Further prospective studies are needed to confirm the results.
Methods
Study population
SSc patients with dcSSc or lcSSc 24 referred to our center for PH at the Thoraxklinik Heidelberg gGmbH at Heidelberg University Hospital, Germany for screening from their rheumatologists between 2008 and 2020 were retrospectively analyzed in this single center, cross-sectional cohort study. All patients fulfilled the SSc classification criteria of the American College of Rheumatology/European League against Rheumatism25 and were followed yearly according to the current recommendations9. Individuals were excluded, if they were underaged, had rheumatic diseases other than SSc, had missing data for the calculation of OxyHem or were unable to provide informed consent. Data of part of this cohort has already been analyzed and published before26. The ethics committee of the Medical Faculty of Heidelberg University (internal number S-126/2021) had no objection against the conduct of this study. All patients provided written informed consent for the analysis of the data. The study complied with the Declaration of Helsinki in its current version.
Study design
Data from routinely performed examinations were extracted from the patient's files at the first presentation in the PH center. Patients were referred from their rheumatologists for screening for PH due to unexplained breathlessness and/or suspicion for PH. A detailed clinical work-up was performed in all patients. The routinely performed screening included medical history, recording of comorbidities such as arterial hypertension, coronary heart disease or obesity (BMI > 30 kg/m2), physical examination, electrocardiogram, PFT with DLCO, blood gas analysis obtained from arterialized capillary blood from earlobe, echocardiography at rest and during exercise, high resolution computed tomography scan of the lungs (HRCT) to investigate the presence of any pulmonary manifestations or diseases, WHO-FC, 6MWD test under standardized conditions27, laboratory examinations including blood count tests, NTproBNP, CRP, analysis for iron deficiency, renal function including glomerular filtration rate, and right heart catheterization at rest and/or during exercise.
The presence of cardiac disease, such as coronary heart disease, and pulmonary manifestations, such as PH or ILD, was documented and, in case of pathology, furtherly examined. ILD was only considered in case of extensive ILD defined as the presence of significant interstitial fibrosis in the HRCT of the lungs (> 20% of the parenchyma) or restrictive pattern in PFT (VCmax < 70% predicted with a normal ratio of FEV1 to VC) in case of missing quantification of interstitial fibrosis in the HRCT28. Manifest PAH was diagnosed according to valid guidelines at the time of the study (mPAP ≥ 25 mmHg, PAWP ≤ 15 mmHg and PVR > 3 WU) measured by RHC9. Signs of early PVD were defined as mPAP ≥ 21 mmHg. PH due to left heart disease was defined by an mPAP ≥ 21 mmHg with a PAWP > 15 mmHg29. PH due to lung disease was based on lung function parameters and in case of ILD HRCT imaging30.
Statistical methods
Statistical analyses were performed by a medical statistician (NB). Data are presented as mean ± standard deviation (SD) with 95% confidence interval of the mean. Frequency data are presented as n and %. The concentration of OxyHem, (g/dl) was calculated at baseline by the multiplication of Hb concentration in blood with the SaO2 measured during blood gas analysis as described before7.
Survival analysis was performed using the Kaplan-Meier and Cox-regression analysis. The first screening visit date was set as baseline for the survival analysis. Death was defined as death due to any cause. Thresholds for dichotomization of parameters were either obtained by the literature of known prognostic predictors or by ROC in case of unknown threshold or in need of threshold adjustment. Clinical parameters were compared with the two-sided student’s t-test for independent samples for patients above versus below the OxyHem threshold. Qualitative data was analyzed using the chi-square test, respectively. This study is of exploratory nature. P-values < 0.05 were considered as statistically significant and were not adapted for multiple testing.
Variables were evaluated by univariable analysis to identify their impact on survival. Parameters were then analyzed with multivariable Cox-regression analysis. OxyHem was compared to known prognostic factors in SSc, which were identified by literature search. These included sex, type of SSc, age ≥ 60 years at baseline31, presence of ILD1,15, PVR ≥ 2 WU26, DLCO ≤ 65% predicted13. In addition, WBC count was included as it showed significant predictive power aside from OxyHem in the OxyHem discovery cohort7. For the multivariable Cox-regression all parameters listed above were considered.
Survival analysis was performed in subsets of patients with signs of cardiovascular disease with either increased PVR ≥ 2 WU, or with mPAP ≥ 25 mmHg. All analyses have been performed using IBM SPSS version 27 (SPSS Statistics V27, IBM Corporation, Somers, New York).
Ethics approval and consent to participate
All data were pseudonymized, and the study was approved by the ethics committee of the medical faculty of Heidelberg University Hospital (internal number S-126/2021). The study complies with the Declaration of Helsinki in its current version. The patients participating in this study provided written informed consent.
Data availability
The datasets are available upon reasonable request to the corresponding author.
Abbreviations
- 6MWD:
-
6-Minute walking distance
- BMI:
-
Body mass index
- CRP:
-
C-reactive protein
- dcSSc:
-
Diffuse cutaneous systemic sclerosis
- DLCO:
-
Diffusion capacity for carbon monoxide
- FEV1 :
-
Forced expiratory volume in one second
- fl:
-
Femtoliters
- GFR:
-
Glomerular filtration rate
- Hb:
-
Hemoglobin
- HRCT:
-
High resolution computed tomography
- ILD:
-
Interstitial lung disease
- lcSSc:
-
Limited cutaneous systemic sclerosis
- MCV:
-
Mean corpuscular volume of erythrocytes
- mPAP:
-
Mean pulmonary arterial pressure
- NTproBNP:
-
N-terminal pro brain natriuretic peptide
- OxyHem:
-
Oxygenated hemoglobin
- PAH:
-
Pulmonary arterial hypertension
- PAWP:
-
Pulmonary arterial wedge pressure
- PFT:
-
Pulmonary function test
- PH:
-
Pulmonary hypertension
- PVD:
-
Pulmonary vascular disease
- PVR:
-
Pulmonary vascular resistance
- RHC:
-
Right heart catheterization
- ROC:
-
Receiver operating characteristic
- SaO2 :
-
Arterial oxygen saturation
- SSc:
-
Systemic sclerosis
- SD:
-
Standard deviation
- TLC:
-
Total lung capacity
- VCmax:
-
Maximal vital capacity
- VC:
-
Vital capacity
- WHO-FC:
-
World Health Organization functional class
References
Elhai, M. et al. Mapping and predicting mortality from systemic sclerosis. Ann. Rheum Dis. 76, 1897–1905. https://doi.org/10.1136/annrheumdis-2017-211448 (2017).
Elhai, M., Meune, C., Avouac, J., Kahan, A. & Allanore, Y. Trends in mortality in patients with systemic sclerosis over 40 years: A systematic review and meta-analysis of cohort studies. Rheumatology (Oxford) 51, 1017–1026. https://doi.org/10.1093/rheumatology/ker269 (2012).
Toledano, E. et al. A meta-analysis of mortality in rheumatic diseases. Reumatol. Clin. 8, 334–341. https://doi.org/10.1016/j.reuma.2012.05.006 (2012).
Wu, W. et al. Prediction of progression of interstitial lung disease in patients with systemic sclerosis: The SPAR model. Ann. Rheum Dis. 77, 1326–1332. https://doi.org/10.1136/annrheumdis-2018-213201 (2018).
Swigris, J. J. et al. Exercise peripheral oxygen saturation (SpO2) accurately reflects arterial oxygen saturation (SaO2) and predicts mortality in systemic sclerosis. Thorax 64, 626–630. https://doi.org/10.1136/thx.2008.111393 (2009).
Ruiter, G. et al. Iron deficiency in systemic sclerosis patients with and without pulmonary hypertension. Rheumatology 53, 285–292. https://doi.org/10.1093/rheumatology/ket331 (2014).
Trudzinski, F. C. et al. Associations of oxygenated hemoglobin with disease burden and prognosis in stable COPD: Results from COSYCONET. Sci. Rep. 10, 10544. https://doi.org/10.1038/s41598-020-67197-x (2020).
Hinke, C. F. et al. Prognostic value of oxygenated hemoglobin assessed during acute exacerbations of chronic pulmonary disease. Respiration 100, 387–394. https://doi.org/10.1159/000513440 (2021).
Galiè, N. et al. 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur. Heart J. 37, 67–119. https://doi.org/10.1093/eurheartj/ehv317 (2016).
Coghlan, J. G. et al. Evidence-based detection of pulmonary arterial hypertension in systemic sclerosis: The DETECT study. Ann. Rheum Dis. 73, 1340–1349. https://doi.org/10.1136/annrheumdis-2013-203301 (2014).
van Hal, T. W., van Bon, L. & Radstake, T. R. A system out of breath: How hypoxia possibly contributes to the pathogenesis of systemic sclerosis. Int J Rheumatol 2011, 824972. https://doi.org/10.1155/2011/824972 (2011).
Pokeerbux, M. R. et al. Survival and prognosis factors in systemic sclerosis: Data of a French multicenter cohort, systematic review, and meta-analysis of the literature. Arthritis Res. Ther. 21, 86. https://doi.org/10.1186/s13075-019-1867-1 (2019).
Nihtyanova, S. I. et al. Prediction of pulmonary complications and long-term survival in systemic sclerosis. Arthritis Rheumatol. 66, 1625–1635. https://doi.org/10.1002/art.38390 (2014).
Fransen, J. et al. Clinical prediction of 5-year survival in systemic sclerosis: Validation of a simple prognostic model in EUSTAR centres. Ann. Rheum Dis. 70, 1788–1792. https://doi.org/10.1136/ard.2010.144360 (2011).
Panopoulos, S. et al. Predictors of morbidity and mortality in early systemic sclerosis: Long-term follow-up data from a single-centre inception cohort. Autoimmun. Rev. 17, 816–820. https://doi.org/10.1016/j.autrev.2018.02.008 (2018).
Valentini, G. et al. The European Scleroderma Trials and Research group (EUSTAR) task force for the development of revised activity criteria for systemic sclerosis: Derivation and validation of a preliminarily revised EUSTAR activity index. Ann. Rheum Dis. 76, 270–276. https://doi.org/10.1136/annrheumdis-2016-209768 (2017).
Hsu, V. M. et al. Risk factors for mortality and cardiopulmonary hospitalization in systemic sclerosis patients at risk for pulmonary hypertension, in the PHAROS registry. J. Rheumatol. 46, 176–183. https://doi.org/10.3899/jrheum.180018 (2019).
Gadre, A. et al. Six-minute walk test as a predictor of diagnosis, disease severity, and clinical outcomes in Scleroderma-associated pulmonary hypertension: The DIBOSA study. Lung 195, 529–536. https://doi.org/10.1007/s00408-017-0034-1 (2017).
Hoeper, M. M., Pletz, M. W., Golpon, H. & Welte, T. Prognostic value of blood gas analyses in patients with idiopathic pulmonary arterial hypertension. Eur. Respir. J. 29, 944–950. https://doi.org/10.1183/09031936.00134506 (2007).
Valentin, S. et al. Outcomes of patients with decreased arterial oxyhaemoglobin saturation on pulmonary arterial hypertension drugs. Eur. Respir. J. https://doi.org/10.1183/13993003.04066-2020 (2021).
Al-Dhaher, F. F., Pope, J. E. & Ouimet, J. M. Determinants of morbidity and mortality of systemic sclerosis in Canada. Semin. Arthritis Rheum 39, 269–277. https://doi.org/10.1016/j.semarthrit.2008.06.002 (2010).
Volkmann, E. R. et al. Short-term progression of interstitial lung disease in systemic sclerosis predicts long-term survival in two independent clinical trial cohorts. Ann. Rheum Dis. 78, 122–130. https://doi.org/10.1136/annrheumdis-2018-213708 (2019).
Langlands, J. H. & Wallace, W. F. Small blood-samples from ear-lobe puncture: A substitute for arterial puncture. Lancet 2, 315–317. https://doi.org/10.1016/s0140-6736(65)90286-2 (1965).
LeRoy, E. C. & Medsger, T. A. Jr. Criteria for the classification of early systemic sclerosis. J. Rheumatol. 28, 1573–1576 (2001).
van den Hoogen, F. et al. 2013 classification criteria for systemic sclerosis: An American College of Rheumatology/European League against Rheumatism collaborative initiative. Arthritis Rheum 65, 2737–2747. https://doi.org/10.1002/art.38098 (2013).
Xanthouli, P. et al. Haemodynamic phenotypes and survival in patients with systemic sclerosis: The impact of the new definition of pulmonary arterial hypertension. Ann. Rheum Dis. 79, 370–378. https://doi.org/10.1136/annrheumdis-2019-216476 (2020).
Guyatt, G. H. et al. Effect of encouragement on walking test performance. Thorax 39, 818–822 (1984).
Goh, N. S. et al. Interstitial lung disease in systemic sclerosis: A simple staging system. Am. J. Respir. Crit. Care Med. 177, 1248–1254. https://doi.org/10.1164/rccm.200706-877OC (2008).
Humbert, M. et al. ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur. Heart J. https://doi.org/10.1093/eurheartj/ehac237 (2022).
Nathan, S. D. et al. Pulmonary hypertension in chronic lung disease and hypoxia. Eur. Respir. J. https://doi.org/10.1183/13993003.01914-2018 (2018).
Chung, L. et al. Survival and predictors of mortality in systemic sclerosis-associated pulmonary arterial hypertension: Outcomes from the pulmonary hypertension assessment and recognition of outcomes in scleroderma registry. Arthritis Care Res. (Hoboken) 66, 489–495. https://doi.org/10.1002/acr.22121 (2014).
Acknowledgements
This work is part of the doctoral thesis of OG. For the publication fee we acknowledge financial support by Deutsche Forschungsgemeinschaft within the funding programme "Open Access Publikationskosten" as well as by Heidelberg University. We thank the patients who participated in this study.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Designing of the study: E.G., P.X., F.C.T., N.Be., C.A.E. Acquisition of data: P.X., O.G., N.M., E.G., N.Bl., H.M.L., B.E., S.H., A.M.M., C.N. Analysis and interpretation of data: N.Be., E.G., P.X., O.G., A.M.M., N.Bl., H.M.L., C.A.E. Drafting of manuscript: P.X., N.Be., O.G., E.G., C.A.E. Revision of manuscript, substantial intellectual contribution to the work and final approval: all authors.
Corresponding author
Ethics declarations
Competing interests
PX has received personal fees from MSD and OMT, outside the submitted work. OG, NBe, FT and NM have nothing to declare. CAE received speaking fees from MSD, outside the submitted work. BE received travel fees, consulting fees, speaking fees, and/or honoraria from Actelion, MSD, Bayer and OMT (less than $10,000 each). SH has received personal fees from Bayer, MSD, Actelion and GSK, outside the submitted work. AMM has received personal fees from Bayer, MSD and Boehringer-Ingelheim outside the submitted work. CN has received consulting fees, speaking fees, and/or honoraria from Actelion, MSD, Boehringer, Novartis, Bayer, and AstraZeneca (less than $10,000 each). NBl has received consulting fees, speaker fees, and/or honoraria from AbbVie, BMS, Pfizer, Roche, Novartis, UCB, Janssen-Cilag, Novartis, SOBI, Actelion outside the submitted work. HML has received consulting fees, speaking fees, and/or honoraria from AbbVie, BMS, Pfizer, Cellgene, Medac, GSK, Roche, Chugai, Novartis, UCB, Janssen-Cilag, AstraZeneca, and Lilly (less than $10,000 each) and research support from AbbVie, MSD, BMS, Cellgene, Medac, GSK, Roche, Chugai, Novartis, UCB, Janssen-Cilag, AstraZeneca, Lilly, Baxter, SOBI, Biogen, Actelion, Bayer Vital, Shire, Octapharm, Sanofi, Hexal, Mundipharm, and Thermo Fisher. EG has received grants and personal fees from Actelion, Bayer AG, and MSD; grants from GSK, Novartis, and United Therapeutics; and personal fees from SCOPE, OrPha Swiss GmbH, and Zurich Heart House (less than $10,000 each).
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xanthouli, P., Gordjani, O., Benjamin, N. et al. Oxygenated hemoglobin as prognostic marker among patients with systemic sclerosis screened for pulmonary hypertension. Sci Rep 13, 1839 (2023). https://doi.org/10.1038/s41598-023-28608-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-28608-x
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.