Abstract
Emerging evidence suggest migraine is a response to cerebral energy deficiency or oxidative stress in the brain. Beta-hydroxybutyrate (BHB) is likely able to circumvent some of the meta-bolic abnormalities reported in migraine. Exogenous BHB was given to test this assumption and, in this post-hoc analysis, multiple metabolic biomarkers were identified to predict clinical improvements. A randomized clinical trial, involving 41 patients with episodic migraine. Each treatment period was 12 weeks long, followed by eight weeks of washout phase / second run-in phase before entering the corresponding second treatment period. The primary endpoint was the number of migraine days in the last 4 weeks of treatment adjusted for baseline. BHB re-sponders were identified (those with at least a 3-day reduction in migraine days over placebo) and its predictors were evaluated using Akaike’s Information Criterion (AIC) stepwise boot-strapped analysis and logistic regression. Responder analysis showed that metabolic markers could identify a “metabolic migraine” subgroup, which responded to BHB with a 5.7 migraine days reduction compared to the placebo. This analysis provides further support for a “metabolic migraine” subtype. Additionally, these analyses identified low-cost and easily accessible biomarkers that could guide recruitment in future research on this subgroup of patients.
This study is part of the trial registration: ClinicalTrials.gov: NCT03132233, registered on 27.04.2017, https://clinicaltrials.gov/ct2/show/NCT03132233
Similar content being viewed by others
Introduction
Migraine is a common, complex and debilitating neurological disorder1, but its primary pathogenic mechanisms are not yet completely understood. Despite having been referred to as a “hypoglycemic headache” in 1935 already2, the focus of clinical and basic research in the last decades was primarily on (neuro-) vasculature, cerebral excitability, and neurotransmission. In recent years, metabolism and mitochondrial (dys-)function have regained interest. Various lines of evidence suggest migraine is—at least partially—a metabolic as much as a neurological disease, in which the migraine attack is a warning signal to increased oxidative stress and / or (cerebral) hypometabolism3.
Magnetic resonance spectroscopy (MRS) studies in migraineurs consistently show decreased ATP levels or hypometabolism4,5,6,7,8,9,10,11,12,13,14,15. Mitochondrial function and oxidative stress markers have also been shown to be abnormal in higher-frequency migraine16. Additional support comes from early studies demonstrating metabolic changes induced by fasting, glucose or insulin administration, which were shown to be able to even trigger migraine attacks in susceptible patients16,17,18,19,20,21,22.
Several nutraceuticals23, such as riboflavin at high dose (200–400 mg/day)30,31,32,33,34,35,36; coenzyme Q10 (400 mg capsules or 300 mg liquid suspension)24,25,26,27,28,29, magnesium37 and alpha-lipoic acid (600 mg)38,39,40 have shown to prevent migraine attacks also suggesting a link between migraine and metabolism/or mitochondrial functioning.
Oxidative stress seems to be the common denominator of most migraine triggers41,42 and apart from clearly “metabolic” triggers (such as physical exercise, fasting, and stress), many of the seemingly unrelated triggers (like ovarian hormone changes, alcohol, weather changes, intense light, and strong odors) can negatively impact mitochondrial metabolism and/or oxidative stress (see reviews3,41). Mechanistically, nitrosative, oxidative, and electrophilic stress can activate transient receptor potential channels, expressed in meningeal nociceptive nerve terminals43,44, thereby providing a mechanism by which known migraine trigger factors which increase oxidative stress could lead to migraine pain.
Metabolic approaches to migraine prevention, such as a ketogenic diet (KD), which to some extent mimics the state of fasting, have been shown to be migraine protective44,45,46,47,48,49. The KD was developed over 100 years ago, after the observation that prolonged fasting has antiepileptic properties50. Like fasting, it promotes the hepatic production of ketone bodies (KBs). Recently the KD has received renewed interest, due to the observation that KBs could be beneficial for a variety of other neurological disorders as51,52,53 all brain cells have the capacity to use KBs as respiratory substrates54.
Out of the three physiologically relevant KBs β-hydroxybutyrate (BHB) constitutes up to 70%55 and acts also as a signaling molecule56. Consequently, it has the potential to positively influence other pathways commonly believed to be part of migraine pathophysiology57.
In complex and heterogenous diseases such as migraine, a therapy that can simultaneously target multiple possible pathogenic pathways seems advantageous and elevated KB levels have been shown to be well tolerated for extended periods of time, even up to several years47,58,59,60,61,62,63,64,65,66,67,68,69,70. However, a very strict KD, may be difficult to adhere to longer-term.
Our research group wanted to examine whether exogenously raised KBs would also be able to attenuate migraine frequency, if ingested daily, and carried out the first RCT exploring the effect of BHB as a prophylactic agent in episodic migraine patients71 where a non-statistically significant reduction of 1.9 migraine days over placebo was documented, however, some patients clearly reduced more days than other. In line with the already outline evidence supporting the existence of a “metabolic migraine subgroup”, we aimed to evaluate if metabolic health markers could identify patients responded to BHB supplementation.
Materials and methods
Trial design
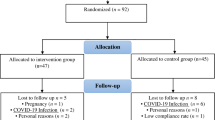
The trial conducted was a double-blind, randomized, placebo-controlled trial with a crossover design with 41 migraine patients meeting the ICHD-3 (International Classification of Headache Disorders version 3 Beta) Classification criteria73. The trial was registered at ClinicalTrials.gov (NCT03132233), approved by the local ethics committee Swissethics (EKNZ 2015-304) and the National Swiss Drug Agency (2016DR2109). The detailed methods can be found in the published study protocol72. In brief, the trial consisted of a four-week run-in period followed by randomization. Then a first treatment period of 12 weeks, followed by a washout period of 4 weeks. Afterward, a second run-in phase of 4 weeks and finished with the second treatment period of 12 weeks.
Study medication
The investigational medicinal product (IMP) used in this clinical trial was 9 gr of D-BHB (from 18 gr racemic BHB) in powdered calcium (Ca2 +)–magnesium (Mg2 +)–salt form (Ca–Mg–BHB) divided into three servings per day. The mineral load determined the maximal IMP dose. The placebo group received sachets containing Mannitol.
Clinical measures
At pre- and post- intervention visits the following assessments were additionally conducted: Migraine Disability Test (MIDAS)74, Headache Impact Test, version 6 (HIT-6)75 and blood draw for biomarker and safety analysis (albumin, Calcium, cortisol, alanine aminotransferase, pancreatic-amylase, alkaline phosphatase, aspartate aminotransferase, beta-hydroxybutyrate, bilirubin, creatine kinase, Chloride, Cholesterol, Cholesterol Quotient, Cortisol basal, high sensitivity-C reactive protein, globulin, fasting glucose, gamma-glutamyl transferase, glomerular filtration rate, uric acid, HbA1c, High density lipoprotein, urea, Potassium, creatinine, lactate plasma, lactate dehydrogenase, low-density lipoprotein (calculated with Friedewald’s equation), Magnesium, Sodium, Phosphate, total protein, triglycerides, leukocyte count, erythrocytes, hemoglobin, hematocrit, mean corpuscular volume, platelets, T3, T4, Insulin and thyroid stimulating hormone (TSH). All blood samples were taken after an overnight fast between 8 and 11 am and all markers were considered in the responder analysis. Further details on data collection are provided in the published study protocol72.
Statistical analysis
Data wrangling and statistical analyses for this purpose were performed using R version 4.0.3. and the packages: tidyverse, readxl, performance, tableone, gtools, MASS, bootStepAIC, lmtest, rpart and car. When relevant, data and all linear model residuals were tested for normality using stats::shapiro.test. A Friedman Test was used to analyze pharmacokinetics data and differences between responders and non-responders were analyzed using Mann–Whitney and Kruskal–Wallis’s rank sum tests. Baseline vs follow-up metabolite changes were analyzed using Wilcoxon tests.
To identify factors associated with positively responding to KBs supplementation, we evaluated the relevance of different combinations of independent predictors according with the explanatory capacity of each model Akaike’s Information Criterion (AIC), the consistency of their coefficient signs, and the consistency of their statistical relevance. This procedure was performed via a bootstrap AIC consistency diagnosis in which 100 independent samples drawn at random from the dataset using bootStepAIC::boot.stepAIC. To avoid collinearity, we analyzed potential models by grouping blood markers according with their corresponding physiological system (thyroid markers, liver function markers, blood cells markers, etc.) to identify the best predictor from each system and test its predictive contribution to different potential models. For evaluating the all-around performance of the combined models for predicting BHB response, we used performance::compare_performance which allows for simultaneously comparing AIC, Bayes Information Criterion (BIC), Root mean squared error (RMSE), and Tjur’s R2. Linear assumptions of the models we corroborated using performance::check_model.
Finally, we used supervised machine-learning regression trees to identify potentially useful cutoffs for relevant predictors using rpart::rpart.
Local ethical approval and consent to participate
All participants provided informed consent to participate in the trial. Ethical approval was granted by Swissethics, EKNZ PB 2016-00497. Also, all methods were performed in accordance with the relevant guidelines and regulations.
Results
Study population
A total of 9 out of 32 patients (28.13%) met our conservative criteria for BHB treatment response, ranging from 3- to 12-day reduction in migraine days from baseline compared to placebo. Treatment responders had an average of 5.78 (SD = 2.63) less migraine days compared to placebo.
To evaluate is pharmacokinetic differences were likely responsible for the differences in therapeutic success, a Friedman test was conducted between responders and non-responders for glucose and BHB (Fig. 1).
Table 1 describes the distribution and demographic and metabolic differences between responders and not responders at baseline.
After 3 months of BHB supplementation many of these markers changed into the direction of the non-responder levels. TSH dropped by 15%, triglycerides by 12%, fasting glucose by 7%, hs-CRP by 53% and endogenous BHB levels increased by 56% in the responder group. In addition, fasting insulin dropped by 11% and cortisol levels by 18%. Furthermore, ALT increased by 29%, phosphate by 12%, LDH by 7% and magnesium by 5% (see Fig. 2). Only the change in CRP was statistically significant (p = 0.002) after using Wilcoxon’s hypothesis testing.
After comparing multiple logistic regression models with 100 bootstrapped samples from all available measurements, we concluded that, because of their cost, availability and Beta coefficient consistency, C-reactive protein, Phosphorus and HbA1c are the most useful predictors of BHB supplementation responsiveness. All were statistically significant below the 0.05 threshold in > 95% of the bootstrap simulations and the sign of their estimate was consistent 100% of the times. Only variables with a 100% coefficient sign consistency were selected for the candidate models and, to avoid collinearity and physiological “redundance”, only one predictor per system was used. Table 2 summarizes the all-around performance of the candidate models according with their AIC, BIC, RMSE and R2.
The model including Pi + HS − CRP + FT3 + ALT accounts for 26% of the variance using only 4 predictors. Adding 2 more predictors; HbA1c and LDH, improved the explained variance and accounts for almost half of the variance explained by the model using all metabolites. After identifying potentially useful markers, we used a regression tree to then identify their potentially useful cut-off points.
As suggested by Fig. 3, using these cut-off points, the least and apparently most useful markers for identifying whether a person belongs to the responder group were inorganic phosphorous, hs-CRP (a marker for inflammation) and Hba1C (long-term blood sugar). This model’s linear assumptions were corroborated (Supplemental Information).
Discussion
Classifying sub-groups of migraineurs based on objective biomarkers is essential for improving clinical study designs, developing novel treatments and ultimately, improving clinical care. Hereby, we documented metabolic markers in BHB supplementation responders differed from those who did not respond. To our knowledge, this is the first work proposing blood biomarkers for predicting treatment response to migraine prophylaxis and could help pave the way not only for testing future anti-inflammatory/metabolic interventions, but also for reassessing already existing solutions76.
However, there were several limitations to this analysis. At trial onset, only the racemic BHB was available, which has ½ of the potency of the bioidentical D- BHB77. In addition, the dose of D-BHB (9 g per day) was low compared to the 185 g of KBs produced by the liver during fasting78. The upper limit was determined by the mineral’s upper daily intake requirements that the BHB was bound to. Due to these two factors, the potency of the current formulation was so low that nutritional ketosis (> 0.5 mmol/l BHB) was never reached79. Furthermore, mannitol was used as a placebo, but it may be not a be an ideal placebo because it shares one migraine relevant mechanism as it increases brain tissue oxygenation80,81. Not surprisingly, we identified 4 responding to mannitol.
Despite the trial’s limitations, we could find potentially useful and easily available predictors (and their potentially useful cut-offs) for identifying whether a person belongs to the responder group or not based on the independent markers of inorganic phosphorous, HS-CRP (a marker for inflammation) and Hba1C (long-term blood sugar) and this result is highly significant—despite our small sample size but should be prospectively validated in future studies.
In contrast, several markers of metabolism and inflammation were worse in the BHB responder group while no pharmacokinetic differences were found. Additionally, most of these markers improved or started looking more like the non-responder group after the intervention. Most notably hs-CRP (inflammation) more than halved and this change was significant, despite the small sample size (n = 9), thus, it is likely the rest of the paired analyses were underpowered. Together, these findings suggest that responders were responding because of their baseline pathophysiological differences and not because they were exposed to higher doses of BHB.
While the connection to energy metabolism with glucose, insulin, KBs, HbA1c and triglycerides seems evident, the other markers also have a strong connection to metabolism. Hs-CRP has been found to be a marker of impaired energy metabolism, in addition to a marker of acute systemic inflammation82, the thyroid is well known for its signalling role in energy homeostasis and energy metabolism83, and ALT has been shown to play a role in metabolic disease84. The exact mechanisms by which KB improve or prevent migraine can be multiple and additive ranging from restoring energy utilisation to ameliorating inflammation85.
It is necessary to mention we are not the first ones to find an association between treatment response to a metabolic migraine therapeutic and a biomarker. Over a decade ago the therapeutic response to high-dose riboflavin was shown to be associated with specific mitochondrial DNA (mtDNA) haplogroups (non- H mitochondrial DNA haplotypes)36, which is also indicative of a “metabolic migraine subgroup”. However, mtDNA haplogroups are, however, not as easily identified as the common three laboratory markers that we propose. These biomarkers could be used to guide the inclusion criteria of future clinical trials and aid the selection of “metabolic migraineurs” for future trials.
We should highlight that the utility of these individual predictors is context dependant and we provided data for cost–benefit assessments. For example, as shown in Table 2, adding FT3 and ALT to Pi + HS − CRP + HbA1c doubles the R2 but would likely complicate recruitment as there are not many point-of-care options for measuring them. Importantly, the identified cut-offs in this work need to be validated prospectively but, the fact that they are already close to those currently used in clinical practice suggest.
Finally, it is possible that these potentially relevant metabolic predictors are not migraine specific and could be useful for studies on other illnesses for which ketosis has been hypothesized to be beneficial such as diabetes, heart failure and epilepsy.
Conclusion
This study provides further support for a distinct “inflammatory/ metabolic migraine” subgroup with unique metabolic and inflammatory signatures. Three easy to measure blood markers (hs-CRP, HbA1c and phosphorus) could assist personalized metabolic migraine treatments and prophylactic interventions.
Data availability
The datasets generated and/or analysed during the current study are not publicly available due to parts of the data set still being analysed at present but are available from the corresponding author on reasonable request.
Abbreviations
- AE:
-
Adverse event
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- AIC:
-
Akaike’s information criterion
- BIC:
-
Bayes information criterion
- RMSE:
-
Root mean square error
- ATP:
-
Adenosine triphosphate
- BHB:
-
Beta-hydroxybutyrate
- BMI:
-
Body mass index
- Ca:
-
Calcium
- CI:
-
Confidence interval
- HS-CRP:
-
High-sensitivity C-reactive protein
- CTU:
-
Clinical trial unit
- HbA1c:
-
Glycosylated hemoglobin
- GMP:
-
Good manufacturing practice
- ICHD-3:
-
International classification of headache disorders version 3
- IMP:
-
Investigational medicinal product
- ITT:
-
Intention to treat
- HDL:
-
High density lipoprotein
- HIT:
-
Headache impact test
- IQR:
-
Interquartile range
- KB:
-
Ketone bodies
- KD:
-
Ketogenic diet
- LDH:
-
Lactate dehydrogenase
- LDL:
-
Low density lipoprotein
- MIDAS:
-
Migraine disability assessment
- Mg:
-
Magnesium
- 31P-MRS:
-
Phosphorus‐31 magnetic resonance spectroscopy
- Na:
-
Sodium
- NSAIDs:
-
Non-steroidal anti-inflammatory drugs
- Pi:
-
Phosphorous
- PP:
-
Per protocol
- RCT:
-
Randomized clinical trial
- ROS:
-
Reactive oxygen species
- TSH:
-
Thyroid stimulating hormone
- USB:
-
University hospital basel
- VAS:
-
Visual analog scale
References
Stovner, L. J., Hoff, J. M., Svalheim, S. & Gilhus, N. E. Neurological disorders in the Global Burden of Disease 2010 study. Acta Neurol. Scand. 129, 1–6 (2014).
Gray, P. A. & Burtness, H. I. Hypoglycemic headache*. Endocrinology 19, 549–560 (1935).
Gross, E. C., Lisicki, M., Fischer, D., Sándor, P. S. & Schoenen, J. The metabolic face of migraine—From pathophysiology to treatment. Nat. Rev. Neurol. 15, 627–643 (2019).
Barbiroli, B. et al. Abnormal brain and muscle energy metabolism shown by 31P magnetic resonance spectroscopy in patients affected by migraine with aura. Neurology 42, 1209–1214 (1992).
Kim, J. H. et al. Interictal metabolic changes in episodic migraine: A voxel-based FDG-PET study. Cephalalgia 30, 53–61 (2010).
Lodi, R. et al. Deficit of brain and skeletal muscle bioenergetics and low brain magnesium in juvenile migraine: An in vivo31p magnetic resonance spectroscopy interictal study. Pediatr. Res. 42, 866–871 (1997).
Lodi, R. et al. Deficient energy metabolism is associated with low free magnesium in the brains of patients with migraine and cluster headache. Brain Res. Bull. 54, 437–441 (2001).
Montagna, P. et al. 31P-magnetic resonance spectroscopy in migraine without aura. Neurology 44, 666–669 (1994).
Reyngoudt, H., Achten, E. & Paemeleire, K. Magnetic resonance spectroscopy in migraine: What have we learned so far?. Cephalalgia 32, 845–859 (2012).
Schulz, U. G. et al. Association between cortical metabolite levels and clinical manifestations of migrainous aura: An MR-spectroscopy study. Brain 130, 3102–3110 (2007).
Welch, K. M., Levine, S. R., D’Andrea, G., Schultz, L. R. & Helpern, J. A. Preliminary observations on brain energy metabolism in migraine studied by in vivo phosphorus 31 NMR spectroscopy. Neurology 39, 538–541 (1989).
Lodi, R. et al. Quantitative analysis of skeletal muscle bioenergetics and proton efflux in migraine and cluster headache. J. Neurol. Sci. 146, 73–80 (1997).
Barbiroli, B. et al. Complicated migraine studied by phosphorus magnetic resonance spectroscopy. Cephalalgia 10, 263–272 (1990).
Reyngoudt, H., Paemeleire, K., Descamps, B., De Deene, Y. & Achten, E. 31P-MRS demonstrates a reduction in high-energy phosphates in the occipital lobe of migraine without aura patients. Cephalalgia 31, 1243–1253 (2011).
Gross, E. C. et al. Mitochondrial function and oxidative stress markers in higher-frequency episodic migraine. Sci. Rep. 11, 4543 (2021).
Hockaday, J. M., Williamson, D. H. & Whitty, C. W. M. Blood-glucose levels and fatty-acid metabolism in migraine related to fasting. Lancet 297, 1153–1156 (1971).
Shaw, S. W., Johnson, R. H. & Keogh, H. J. Metabolic changes during glucose tolerance tests in migraine attacks. J. Neurol. Sci. 33, 51–59 (1977).
Dexter, J. D., Roberts, J. & Byer, J. A. The five hour glucose tolerance test and effect of low sucrose diet in migraine. Headache 18, 91–94 (1978).
Wang, X. et al. Are glucose and insulin metabolism and diabetes associated with migraine? A community-based, case-control study. J. Oral Facial Pain Headache 31, 240–250 (2017).
Rainero, I. et al. Insulin sensitivity is impaired in patients with migraine. Cephalalgia 25, 593–597 (2005).
Fava, A. et al. Chronic migraine in women is associated with insulin resistance: A cross-sectional study. Eur. J. Neurol. 21, 267–272 (2014).
Cavestro, C. et al. Insulin metabolism is altered in migraineurs: A new pathogenic mechanism for migraine?. Headache 47, 1436–1442 (2007).
Shaik, M. M. & Gan, S. H. Vitamin supplementation as possible prophylactic treatment against migraine with aura and menstrual migraine. Biomed. Res. Int. 2015, 469529 (2015).
Dahri, M., Hashemilar, M., Asghari-Jafarabadi, M. & Tarighat-Esfanjani, A. Efficacy of coenzyme Q10 for the prevention of migraine in women: A randomized, double-blind, placebo-controlled study. Eur. J. Integr. Med. 16, 8–14 (2017).
Dahri, M., Tarighat-Esfanjani, A., Asghari-Jafarabadi, M. & Hashemilar, M. Oral coenzyme Q10 supplementation in patients with migraine: Effects on clinical features and inflammatory markers. Nutr. Neurosci. 0, 1–9 (2018).
Sándor, P. S. et al. Efficacy of coenzyme Q10 in migraine prophylaxis: A randomized controlled trial. Neurology 64, 713–715 (2005).
Hajihashemi, P., Askari, G., Khorvash, F., Reza Maracy, M. & Nourian, M. The effects of concurrent Coenzyme Q10, L-carnitine supplementation in migraine prophylaxis: A randomized, placebo-controlled, double-blind trial. Cephalalgia https://doi.org/10.1177/0333102418821661 (2019).
Shoeibi, A. et al. Effectiveness of coenzyme Q10 in prophylactic treatment of migraine headache: An open-label, add-on, controlled trial. Acta Neurol. Belg. 117, 103–109 (2017).
Rozen, T. et al. Open label trial of coenzyme Q10 as a migraine preventive. Cephalalgia 22, 137–141 (2002).
Boehnke, C. et al. High-dose riboflavin treatment is efficacious in migraine prophylaxis: An open study in a tertiary care centre. Eur. J. Neurol. 11, 475–477 (2004).
Condò, M., Posar, A., Arbizzani, A. & Parmeggiani, A. Riboflavin prophylaxis in pediatric and adolescent migraine. J Headache Pain 10, 361–365 (2009).
Gaul, C., Diener, H.-C., Danesch, U., Migravent® Study Group, on behalf of the M. S. Improvement of migraine symptoms with a proprietary supplement containing riboflavin, magnesium and Q10: A randomized, placebo-controlled, double-blind, multicenter trial. J. Headache Pain 16, 516 (2015).
Schoenen, J., Jacquy, J. & Lenaerts, M. Effectiveness of high-dose riboflavin in migraine prophylaxis. A randomized controlled trial. Neurology 50, 466–470 (1998).
Rahimdel, A., Mellat, A., Zeinali, A., Jafari, E. & Ayatollahi, P. Comparison between intravenous sodium valproate and subcutaneous sumatriptan for treatment of acute migraine attacks; double-blind randomized clinical trial. Iran J. Med. Sci. 39, 171–177 (2014).
Thompson, D. F. & Saluja, H. S. Prophylaxis of migraine headaches with riboflavin: A systematic review. J. Clin. Pharm. Ther. 42, 394–403 (2017).
Di Lorenzo, C. et al. Mitochondrial DNA haplogroups influence the therapeutic response to riboflavin in migraineurs. Neurology 72, 1588–1594 (2009).
Chiu, H.-Y., Yeh, T.-H., Huang, Y.-C. & Chen, P.-Y. Effects of intravenous and oral magnesium on reducing migraine: A meta-analysis of randomized controlled trials. Pain Physician 19, E97-112 (2016).
Magis, D. et al. A randomized double-blind placebo-controlled trial of thioctic acid in migraine prophylaxis. Headache 47, 52–57 (2007).
Cavestro, C. et al. Alpha-lipoic acid shows promise to improve migraine in patients with insulin resistance: A 6-month exploratory study. J. Med. Food 21, 269–273 (2018).
Ali, A. M., Awad, T. G. & Al-Adl, N. M. Efficacy of combined topiramate/thioctic acid therapy in migraine prophylaxis. Saudi Pharm. J. 18, 239–243 (2010).
Borkum, J. M. Migraine triggers and oxidative stress: A narrative review and synthesis. Headache https://doi.org/10.1111/head.12725 (2015).
Benemei, S., Fusi, C., Trevisan, G. & Geppetti, P. The TRPA1 channel in migraine mechanism and treatment. Br. J. Pharmacol. 171, 2552–2567 (2014).
Kozai, D., Ogawa, N. & Mori, Y. Redox regulation of transient receptor potential channels. Antioxid. Redox Signal. 21, 971–986 (2014).
Strahlman, R. S. Can ketosis help migraine sufferers? A case report. Headache 46, 182 (2006).
Di Lorenzo, C. et al. Diet transiently improves migraine in two twin sisters: Possible role of ketogenesis?. Funct. Neurol. 28, 305–308 (2013).
Maggioni, F., Margoni, M. & Zanchin, G. Ketogenic diet in migraine treatment: A brief but ancient history. Cephalalgia 31, 1150–1151 (2011).
Schnabel, T. G. An experience with a ketogenic dietary in migraine. Ann. Intern. Med. 2, 341 (1928).
Di Lorenzo, C. et al. Migraine improvement during short lasting ketogenesis: A proof-of-concept study. Eur. J. Neurol. https://doi.org/10.1111/ene.12550 (2014).
Di Lorenzo, C. et al. Cortical functional correlates of responsiveness to short-lasting preventive intervention with ketogenic diet in migraine: A multimodal evoked potentials study. J. Headache Pain 17, 58 (2016).
Bailey, E. E., Pfeifer, H. H. & Thiele, E. A. The use of diet in the treatment of epilepsy. Epilepsy Behav. 6, 4–8 (2005).
Danial, N. N., Hartman, A. L., Stafstrom, C. E. & Thio, L. L. How does the ketogenic diet work? Four potential mechanisms. J. Child Neurol. 28, 1027–1033 (2013).
Stafstrom, C. E. & Rho, J. M. The ketogenic diet as a treatment paradigm for diverse neurological disorders. Front. Pharmacol. 3, 59 (2012).
Barañano, K. W. & Hartman, A. L. The ketogenic diet: Uses in epilepsy and other neurologic illnesses. Curr. Treat. Options. Neurol. 10, 410–419 (2008).
Edmond, J., Robbins, R. A., Bergstrom, J. D., Cole, R. A. & de Vellis, J. Capacity for substrate utilization in oxidative metabolism by neurons, astrocytes, and oligodendrocytes from developing brain in primary culture. J. Neurosci. Res. 18, 551–561 (1987).
Dedkova, E. N. & Blatter, L. A. Role of β-hydroxybutyrate, its polymer poly-β-hydroxybutyrate and inorganic polyphosphate in mammalian health and disease. Front. Physiol. 5, 260 (2014).
Puchalska, P. & Crawford, P. A. Multi-dimensional roles of ketone bodies in fuel metabolism, signaling, and therapeutics. Cell Metab. 25, 262–284 (2017).
Gross, E. C., Klement, R. J., Schoenen, J., D’Agostino, D. P. & Fischer, D. Potential protective mechanisms of ketone bodies in migraine prevention. Nutrients 11, 811 (2019).
Nei, M., Ngo, L., Sirven, J. I. & Sperling, M. R. Ketogenic diet in adolescents and adults with epilepsy. Seizure 23, 439–442 (2014).
Reid, C. A., Mullen, S., Kim, T. H. & Petrou, S. Epilepsy, energy deficiency and new therapeutic approaches including diet. Pharmacol. Ther. https://doi.org/10.1016/j.pharmthera.2014.06.001 (2014).
de Almeida Rabello Oliveira, M. et al. Effects of short-term and long-term treatment with medium- and long-chain triglycerides ketogenic diet on cortical spreading depression in young rats. Neurosci. Lett. 434, 66–70 (2008).
Henderson, S. T. et al. Study of the ketogenic agent AC-1202 in mild to moderate Alzheimer’s disease: A randomized, double-blind, placebo-controlled, multicenter trial. Nutr. Metab. 6, 31 (2009).
Klepper, J., Leiendecker, B., Riemann, E. & Baumeister, F. A. The ketogenic diet in German-speaking countries: Update. Klin. Padiatr. 216, 277–285 (2003).
Paoli, A., Bianco, A., Damiani, E. & Bosco, G. Ketogenic diet in neuromuscular and neurodegenerative diseases. Biomed. Res. Int. 2014, 474296 (2014).
Freeman, J. M. & Kossoff, E. H. Ketosis and the ketogenic diet, 2010: Advances in treating epilepsy and other disorders. Adv. Pediatr. 57, 315–329 (2010).
Liu, Y. C. & Wang, H.-S. Medium-chain triglyceride ketogenic diet, an effective treatment for drug-resistant epilepsy and a comparison with other ketogenic diets. Biomed. J. 36, 9–15 (2013).
Valayannopoulos, V. et al. Successful treatment of severe cardiomyopathy in glycogen storage disease type III with D, L-3-hydroxybutyrate, ketogenic and high-protein diet. Pediatr. Res. 70, 638–641 (2011).
Clarke, K. et al. Kinetics, safety and tolerability of (R)-3-hydroxybutyl (R)-3-hydroxybutyrate in healthy adult subjects. Regul. Toxicol. Pharmacol. 63, 401–408 (2012).
Kossoff, E. H., Cervenka, M. C., Henry, B. J., Haney, C. A. & Turner, Z. A decade of the modified Atkins diet (2003–2013): Results, insights, and future directions. Epilepsy Behav. 29, 437–442 (2013).
Newport, M. T., VanItallie, T. B., Kashiwaya, Y., King, M. T. & Veech, R. L. A new way to produce hyperketonemia: Use of ketone ester in a case of Alzheimer’s disease. Alzheimer’s Dement. 11, 99–103 (2015).
Douris, N. et al. Adaptive changes in amino acid metabolism permit normal longevity in mice consuming a low-carbohydrate ketogenic diet. Biochem. Biophys. Acta 1852, 2056–2065 (2015).
Putananickal, N. et al. Efficacy and safety of exogenous beta-hydroxybutyrate for preventive treatment in episodic migraine: A single-centred, randomised, placebo-controlled, double-blind crossover trial. Cephalalgia https://doi.org/10.1177/03331024211043792 (2021).
Gross, E. et al. Efficacy and safety of exogenous ketone bodies for preventive treatment of migraine: A study protocol for a single-centred, randomised, placebo-controlled, double-blind crossover trial. Trials 20, 61 (2019).
The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia : an international journal of headache 33, 629–808 (2013).
Benz, T. et al. Translation, cross-cultural adaptation and reliability of the German version of the migraine disability assessment (MIDAS) questionnaire. Health Qual. Life Outcomes 16, 42 (2018).
Martin, M., Blaisdell, B., Kwong, J. W. & Bjorner, J. B. The short-form headache impact test (HIT-6) was psychometrically equivalent in nine languages. J. Clin. Epidemiol. 57, 1271–1278 (2004).
Zhu, Y. et al. The efficacy and safety of calcitonin gene-related peptide monoclonal antibody for episodic migraine: A meta-analysis. Neurol. Sci. 39, 2097–2106 (2018).
Stubbs, B. J. et al. On the metabolism of exogenous ketones in humans. Front. Physiol. 8, 848 (2017).
Laffel, L. Ketone bodies: A review of physiology, pathophysiology and application of monitoring to diabetes. Diabetes Metab. Res. Rev. 15, 412–426 (1999).
Zhang, Y. et al. Ketosis proportionately spares glucose utilization in brain. J. Cereb. Blood Flow Metabol. 33, 1307–1311 (2013).
Wakai, A., Roberts, I. G., Schierhout, G. & Wakai, A. Mannitol for acute traumatic brain injury. Cochrane Database Syst. Rev. https://doi.org/10.1002/14651858.CD001049.pub2 (2005).
Schilte, C. et al. Mannitol improves brain tissue oxygenation in a model of diffuse traumatic brain injury*. Crit. Care Med. 43, 2212–2218 (2015).
Broekhuizen, R., Wouters, E. F. M., Creutzberg, E. C. & Schols, A. M. W. J. Raised CRP levels mark metabolic and functional impairment in advanced COPD. Thorax 61, 17–22 (2006).
McAninch, E. A. & Bianco, A. C. Thyroid hormone signaling in energy homeostasis and energy metabolism. Ann. N. Y. Acad. Sci. 1311, 77–87 (2014).
Hanley, A. J. G. et al. Liver markers and development of the metabolic syndrome: The insulin resistance atherosclerosis study. Diabetes 54, 3140–3147 (2005).
Norwitz, N. G., Jaramillo, J. G., Clarke, K. & Soto, A. Ketotherapeutics for neurodegenerative diseases. Int. Rev. Neurobiol. 155, 141–168. https://doi.org/10.1016/bs.irn.2020.02.003 (2020).
Acknowledgements
Many thanks to Ambros Gleissner for the data cleaning and merging.
Funding
This research was funded by the Swiss National Science Foundation (SNF), grant number 32003B_173193/1”.
Author information
Authors and Affiliations
Contributions
E.C.G. participated in the design of the study and its or organization, conduct and data acquisition, and was responsible for the main composition of the manuscript. N.P. and A.-L.O. participated in the conduct of the study. A.S.-M. covered all statistical aspects of the study and edited the manuscript. D.F. participated in the study design, its organization and edited the manuscript. J.S. provided additional text and citations and in-depth editing of the manuscript. All authors proofread the final manuscript prior to submission.
Corresponding author
Ethics declarations
Competing interests
ECG and DF are the inventors of the patent WO 2018/115158 Al held by the UKBB and University of Basel on the use of beta-hydroxybutyrate in migraine prevention. ECG is the founder of KetoSwiss AG. Other authors declare no conflicts of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gross, E.C., Putananickal, N., Orsini, AL. et al. Defining metabolic migraine with a distinct subgroup of patients with suboptimal inflammatory and metabolic markers. Sci Rep 13, 3787 (2023). https://doi.org/10.1038/s41598-023-28499-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-28499-y
This article is cited by
-
Komplementäre und ergänzende Verfahren in der Kopfschmerztherapie
Der Schmerz (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.