Abstract
Takayasu arteritis (TA) is a systemic disease affecting women of reproductive age. Similarly to other systemic autoimmune diseases, pregnancies in patients suffering from TA are at high risk for adverse outcomes; however, the precise incidence of adverse events has not been assessed in a systematic approach. The aim of this study was to evaluate the prevalence of adverse pregnancy outcomes in TA. Searches were conducted on PubMed, Cochrane Library, Scopus and Cinahl databases from inception to 25 May 2022. Three independent investigators extracted data and assessed the risk of bias using ROBINS-1 tool. We used a random effects model to calculate the prevalence of the adverse pregnancy outcomes in TA, namely miscarriage, hypertension and pre-eclampsia. We calculated the prevalence of the adverse outcomes in pregnancy for TA. We included 27 studies, with 825 pregnancies. The occurrence of miscarriage, hypertension and pre-eclampsia in patients with TA was 16% (CI 12–21%, p < 0.01), 37% (CI 30–45%, p < 0.01) and 14% (CI 8–23%, p < 0.01), respectively. The results of our meta-analysis indicate that pregnancies in patients with TA are at increased risk for adverse pregnancy outcomes compared to the general population, suggesting that pregnant women with TA should be closely monitored.
Trial registration: There was no registration for this systematic review.
Similar content being viewed by others
Introduction
Systemic vasculitides are rare autoimmune disorders characterized by necrotizing inflammatory infiltration of blood vessels and multisystem involvement. Vasculitides are classified into large, medium and small vessels diseases based on the size of the involved arteries1. Takayasu arteritis (TA) is a large vessels vasculitis, predominantly affecting the main branches of the aorta. It is more common in women of childbearing age, approximately 20–30 years old2,3.
It is widely recommended that women suffering from autoimmune systemic diseases need to plan their pregnancy during periods of remission or low disease activity in order to avoid unfavorable outcomes for both mother and fetus, as well as to prevent a severe relapse of the disease itself during this period4. During previous decades, young women with TA were advised to avoid pregnancy, due to elevated risk of adverse events. Better understanding of the pathophysiologic mechanisms and the expansion of therapeutic choices have resulted in efficient control of the disease, including lower rates of irreversible tissue damage, better quality of life and increased life expectancy5,6. Despite such advances, pregnancy in such patients is still considered as high risk and requires special management and follow up7.
Recent evidence support that pregnant women with TA are at increased risk of hypertension, miscarriage and intrauterine growth restriction (IUGR) neonates with this excess risk to be associated with high disease activity during conception8. On the other hand, observations suggest that TA tends to remain stable or improve during pregnancy, once conception was achieved during period of low disease activity9. However, the precise effect of TA on pregnancy outcomes compared to the general population has not been assessed in a systematic, holistic approach.
The aim of this study is to synthesize the data that refer to pregnancy in women with TA, in order to provide a comprehensive meta-analysis of the impact of the disease on the pregnancy course and outcomes in this population.
Materials and methods
This study is a systematic review and meta-analysis of proportions. The results are reported according to PRISMA guidelines10. We conducted a meta-analysis of proportions, as most of the studies included are observational-retrospective or prospective.
Search strategy and inclusion criteria
A search of the open access electronic databases PubMed, Scopus, Cochrane Library, and Cinahl until 25 May 2022 was conducted. The research algorithm included the terms “Takayasu arteritis”, “systemic vasculitis” and “pregnancy outcome”, both in all fields and MeSH (medical subject headings) terms. Precisely, the search strategy was the following: ((Takayasu arteritis) OR ((systemic vasculitis[MeSH Terms]) OR (systemic vasculitis))) AND ((pregnancy outcome) OR (pregnancy outcome[MeSH Terms])).
For this systematic review and meta-analysis, we included pregnant adult women (age > 18 years old) with TA, based on either ACR or Ishikawa classification criteria11,12. Other group of systemic vasculitides or secondary (infective, drug-induced or malignancy-induced) were excluded. We also, excluded studies that were not published in English, case reports and papers that did not provide arithmetic data to proceed in calculations.
We included observational-prospective or retrospective and case control studies. In some, there was comparison of pregnancy outcomes before and after vasculitis diagnosis. In these cases, we used only the data after the onset of the disease, as it was considered that before disease onset, the negative results of the vasculitis would, probably, be absent.
Data extraction
The data from the final studies were exported from two independent investigators (SP, DD) and included: first author, publication year, country, study type (prospective, retrospective, case control), number of patients and pregnancies, diagnostic criteria, mean age, mean duration of disease, medication, miscarriage (%), cesarian section (%), pre-term delivery (%), hypertension (%), pre-eclampsia (%) and IUGR neonates (%).
Study outcomes
The outcomes studied were miscarriage, defined as loss of pregnancy before 20th week of gestation (terminations of pregnancy either for social or medical reasons, e.g., patient on therapy with possible teratogenic agents, like cyclophosphamide, were not included in this category), hypertension (> 140/90 mmHg) and pre-eclampsia13,14,15.
Statistical analysis
A meta-analysis of proportions was conducted for every outcome, to calculate the total prevalence16. This meta-analysis type is used for statistical synthesis of studies that do not include control group. Although some of the studies included are case control, they were very few, so we proceeded in meta-analysis of proportions.
All calculations were conducted with RStudio (version 2022.02.3). The data were not normally distributed, so we made a logarithmic transformation. For heterogeneity evaluation we used the Ι2 index. When the value of this index is above 50%, it is considered that there is important heterogeneity between studies. Furthermore, for our calculations, we preferred the random effects model, which is more suitable for medical research. In cases that high heterogeneity was noticed, we found the outliers studies, according to the z-value. The limit of two was considered significant so, every study with z-value above that, was considered as an outlier that contributed much to the high heterogeneity. Studies that did not report results for a specific outcome were excluded from the synthesis16,17.
Results
Study search results
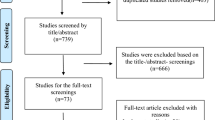
We conducted a thorough investigation of the literature and found 399 studies. After removal of the duplicates, 292 remained. Two independent investigators (SP, DD) assessed the rest at the level of title and abstract. Consensus was reached with a third investigator (AM), if any disagreement occurred, and 264 articles were excluded. The rest 28 were evaluated as a whole text. One paper was excluded, because there was no consensus on the numbers in text and in tables. We totally included 27 papers referring to TA8,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43. A flow diagram according to PRISMA guidelines follows (Fig. 1).
Study characteristics
Next, we present the basic characteristics of the studies included in the systematic review and meta-analysis (Table 1).
Risk of bias assessment
The included articles were assessed by two independent investigators (SP, AM) for risk of bias with the “ROBINS-1” tool, for non-randomized trials. This tool evaluates every study in seven domains, as presented in the figure below (Fig. 2). There was no article of low quality44.
Adverse pregnancy outcomes in Takayasu arteritis
Miscarriage was calculated in 16% (CI 12–21%, p < 0.01), as presented below (Fig. 3A). The two studies with missing values for this outcome (Matsumura and David) were excluded automatically23,36.
The prevalence of hypertension was assessed 37% (CI 30–45%, p < 0.01). Again, studies with missing data were excluded (Fig. 3B). According to z-values, we found that studies of Hidaka and Mandal contributed much to the heterogeneity19,26. We re-evaluated the I2 index after removing first Hidaka, and then Mandal study and the values were 69% and 67%, respectively. The prevalence of hypertension in TA without these two studies remained 37% (CI 30–44%, p < 0.01).
The prevalence of pre-eclampsia in TA pregnant women was calculated in 14% (CI 8–23%, p < 0.01), with relatively high heterogeneity (Fig. 3C). According to z-value, the study that diverged from the others was Mandal et al.26. The prevalence of pre-eclampsia after excluding this one was 12% and the I2 was 77.58%. Next, we present a figure with the aforementioned results.
Discussion
The findings of the current systematic review and meta-analysis indicate a significantly higher rate of maternal complications, namely hypertension, miscarriage and pre-eclampsia occurring in 37%, 16% and 14% of pregnancies in TA women, compared to the general population. Respectively, large-scale reviews report that in community, miscarriage presents in 15%, hypertension in 5–10% and pre-eclampsia in 3%, far less than our findings45,46,47. Such observations highlight the complexity of pregnancy in TA and underscore the multidisciplinary approach in these patients.
The anchor risk factor for unfavorable pregnancy outcomes according to Shiping He et al. seems to be high disease activity during pregnancy, followed by renal artery involvement and history of hypertension8. On the other hand, advanced maternal age or prolonged disease duration did not predispose to obstetric complications8. Gönenli et al., also reported that maternal age did not oppose as risk factor for poor pregnancy outcomes, in contrast to vascular damage, and recent diagnosis, commonly associated with uncontrolled inflammation and high disease activity40.
New onset and/or aggravation of hypertension appear to be the main maternal complications in our analysis, which concurs with previous studies8. This finding was not surprising, as in the majority of patients in the studies included, hypertension was the main symptom of the disease24,33,36. It is worth noting that high blood pressure during pregnancy presented even in women under anti-hypertensive medication, suggesting the necessity of close monitoring despite anti-hypertensive treatment21,24. Of note, the incidence of pre-eclampsia is lower compared to hypertension, potentially reflecting the optimal management of pregnant women with TA. Indeed, Abisror et al., demonstrated that the administration of low dose glucocorticoids, namely 5 mg of prednisolone, resulted in lower rates of pre-eclampsia, probably via reducing the inflammatory burden38. In any case, the acknowledgment of the risk coupled with the efficient control of blood pressure before and during pregnancy is of utmost importance for the achievement of favorable outcomes.
With regards to low incidence of miscarriages and abortions demonstrated in our analysis, some, but not all studies have reported similar results8,33. Apparently, such discrepancy is related with the heterogeneity of the studies especially the disease activity status during the conception and pregnancy period.
In view of treatment, the most common prescribed regimens were corticosteroids and/or anti-hypertensive agents. Furthermore, prophylactic prescription of low dose aspirin has been decribed, although Abisror et al. did not find any beneficial effect on fetomaternal outcomes18,27,38 Less frequently, low molecular weight heparin was prescribed8,20,30. Immunosuppressive drugs, compatible with pregnancy, mainly azathioprine, have also been administrateed29. Finally, vascular surgery repair of affected arteries was part of the therapeutic intervention and pregnancy planning in some cases before conception36.
To the best of our knowledge, this is the first meta-analysis to combine data from all published studies in large databases. Given that pregnancy in TA remains a challenge for rheumatologists and obstetricians, the current findings provide in a systematic approach the most frequent complications during pregnancy in TA women. It is vital for patients to conceive during remission, or at least low disease activity, in order to decrease the risk of adverse outcomes48. As pregnant women with TA are predisposed to pregnancy complications, diligent surveillance especially for blood pressure, and kidney function are necessary for optimal outcome49.
Still, our findings should be interpreted in the context of several limitations relevant to the substantial heterogeneity among the included publications. Indeed, studies varied significantly in terms of population characteristics-especially medication schemes and disease activity, sample size and study design. Although heterogeneity was high according to the I2 index, we should underline that in meta-analysis of proportions, high values are expected, due to differences in place and time of study conduction. Therefore, high values of I2 do not, necessarily indicate inconsistent studies16.
Prospective case–control studies, even with small numbers, are currently lacking from recent bibliography. That prevents us from performing a classic meta-analysis that would enable us to assess in larger samples the greater risk for adverse pregnancy outcomes that these patients present, in comparison with the general population. To date, most published studies are only retrospective or small observational ones.
Our study does not only report on the incidence of pregnancy complications but, it also showcases the need for further research. Conducting high-quality observational studies, as well as exploring the impact of pregnancy in less common systemic inflammatory diseases such as vasculitides, systemic sclerosis and inflammatory myopathies is deemed mandatory to provide a realistic guidance for clinicians. It is worth noting that, in contrast to other conditions, guidelines for the management of family planning, assisted reproduction and pregnancy in TA, are currently lacking, and therefore, observational large studies focusing on potential management strategies, could provide a guidance for clinical practice50.
Conclusion
Our study estimated high rates of hypertension, miscarriage, and pre-eclampsia in TA pregnant women. These findings indicate that women with TA are at higher risk of adverse pregnancy outcomes than in general population. Thus, more intense follow-up along with multidisciplinary approach is needed for these patients to reach an optimal delivery.
Data availability
The data supporting the conclusions of this article are in this article.
Change history
09 February 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41598-023-29324-2
Abbreviations
- IUGR:
-
Intrauterine growth restriction
- TA:
-
Takayasu arteritis
References
Jennette, J. C. et al. 2012 Revised International Chapel Hill consensus conference nomenclature of vasculitides. Arthritis Rheum. 65, 1–11 (2013).
Onen, F. & Akkoc, N. Epidemiology of Takayasu arteritis. Press. Med. 46, e197–e203 (2017).
Seyahi, E. Takayasu arteritis: An update. Curr. Opin. Rheumatol. 29, 51–56 (2017).
Merz, W. M., Fischer-Betz, R., Hellwig, K., Lamprecht, G. & Gembruch, U. Pregnancy and autoimmune disease. Dtsch. Arztebl. Int. 119, 145–156 (2022).
Torp, C. K. et al. Vasculitis therapy refines vasculitis mechanistic classification. Autoimmun. Rev. 20, (2021).
Westman, K., Flossmann, O. & Gregorini, G. The long-term outcomes of systemic vasculitis. Nephrol. Dial. Transplant 30(Suppl 1), i60–i66 (2015).
Ross, C., D’Souza, R. & Pagnoux, C. Pregnancy Outcomes in Systemic Vasculitides. vol. 22 (2020).
He, S. et al. Pregnancy outcomes in Takayasu arteritis patients. Semin. Arthritis Rheum. 55, 152016 (2022).
Padiyar, S., Manikuppam, P., Kabeerdoss, J., Rathore, S. & Danda, D. Update on pregnancy in Takayasu arteritis—A narrative review. Int. J. Rheum. Dis. 24, 758–765 (2021).
Page, M. J. et al. PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. BMJ 372, (2021).
Arend, W. P. et al. The American College of Rheumatology 1990 criteria for the classification of Takayasu arteritis. Arthritis Rheum. 33, 1129–1134 (1990).
Ishikawa, K. Diagnostic approach and proposed criteria for the clinical diagnosis of Takayasu’s arteriopathy. J. Am. Coll. Cardiol. 12, 964–972 (1988).
Dugas, C. & Slane, V. H. Miscarriage. StatPearls (2022).
Brouwers, S., Sudano, I., Kokubo, Y. & Sulaica, E. M. Arterial hypertension. Lancet (London, England) 398, 249–261 (2021).
Steegers, E. A. P., Von Dadelszen, P., Duvekot, J. J. & Pijnenborg, R. Pre-eclampsia. Lancet (London, England) 376, 631–644 (2010).
Barker, T. H. et al. Conducting proportional meta-analysis in different types of systematic reviews: A guide for synthesisers of evidence. BMC Med. Res. Methodol. 21, 1–9 (2021).
Wang N. How to Conduct a Meta-Analysis of Proportions in R: A Comprehensive Tutorial. John Jay Coll. Crim. Justice, 1–63. https://doi.org/10.13140/RG.2.2.27199.00161 (2016).
Tanacan, A., Unal, C., Yucesoy, H. M. H. M. M., Duru, S. A. S. A. & Beksac, M. S. M. S. S. Management and evaluation of pregnant women with Takayasu arteritis. Arch. Gynecol. Obstet. 299, 79–88 (2019).
Hidaka, N., Yamanaka, Y., Fujita, Y., Fukushima, K. & Wake, N. Clinical manifestations of pregnancy in patients with Takayasu arteritis: Experience from a single tertiary center. Arch. Gynecol. Obstet. 285, 377–385 (2012).
Ishikawa, K. & Matsuura, S. Occlusive thromboaortopathy (Takayasu’s disease) and pregnancy. Clinical course and management of 33 pregnancies and deliveries. Am. J. Cardiol. 50, 1293–1300 (1982).
Wong, V. C. W., Wang, R. Y. C. & Tse, T. F. Pregnancy and Takayasu’s arteritis. Am. J. Med. 75, 597–601 (1983).
Aso, T., Abe, S. & Yaguchi, T. Clinical gynecologic features of pregnancy in Takayasu arteritis. Heart Vessels 7, 125–132 (1992).
Matsumura, A., Moriwaki, R. & Numano, F. Pregnancy in Takayasu arteritis from the view of internal medicine. Heart Vessels 7, 120–124 (1992).
Sharma, B. K., Jain, S. & Vasishta, K. Outcome of pregnancy in Takayasu arteritis. Int. J. Cardiol. 75, S159–S162 (2000).
Suri, V. et al. Pregnancy and Takayasu arteritis: A single centre experience from North India. J. Obstet. Gynaecol. Res. 36, 519–524 (2010).
Mandal, D. et al. Takayasu arteritis in pregnancy: An analysis from eastern India. Arch. Gynecol. Obstet. 285, 567–571 (2012).
de Jesús, G. R. R. G. R. et al. Pregnancy may aggravate arterial hypertension in women with takayasu arteritis. Isr. Med. Assoc. J. 14, 724–728 (2012).
Tanaka, H., Tanaka, K., Kamiya, C., Iwanaga, N. & Yoshimatsu, J. Analysis of pregnancies in women with Takayasu arteritis: Complication of Takayasu arteritis involving obstetric or cardiovascular events. J. Obstet. Gynaecol. Res. 40, 2031–2036 (2014).
Alpay-Kanitez, N. et al. Favourable pregnancy outcome in Takayasu arteritis: A single-centre experience. Clin. Exp. Rheumatol. 33, S-7–10 (2015).
Singh, N., Tyagi, S., Tripathi, R. & Mala, Y. M. M. Maternal and fetal outcomes in pregnant women with Takayasu aortoarteritis: Does optimally timed intervention in women with renal artery involvement improve pregnancy outcome?. Taiwan. J. Obstet. Gynecol. 54, 597–602 (2015).
Mohammad, A. J. & Mandl, T. Takayasu arteritis in Southern Sweden. J. Rheumatol. 42, 853–858 (2015).
Assad, A. P. L., Da Silva, T. F., Bonfa, E. & Pereira, R. M. R. Maternal and neonatal outcomes in 89 patients with Takayasu Arteritis (TA): Comparison before and after the TA diagnosis. J. Rheumatol. 42, 1861–1864 (2015).
Comarmond, C. et al. Takayasu arteritis and pregnancy. Arthritis Rheumatol. 67, 3262–3269 (2015).
Zhang, Y., Li, Y. & Zhang, J. Clinical analysis: 13 cases of pregnancy complicated with Takayasu arteritis. Ginekol. Pol. 88, 654–661 (2017).
Gudbrandsson, B. et al. Takayasu arteritis and pregnancy: A population-based study on outcomes and mother/child-related concerns. Arthritis Care Res. 69, 1384–1390 (2017).
David, L. S. S. et al. Obstetric and perinatal outcomes in pregnant women with Takayasu’s arteritis: Single centre experience over five years. J. Turkish Ger. Gynecol. Assoc. 21, 15–23 (2020).
Pyo, J. Y. Y., Song, J. J. J., Park, Y.-B. & Lee, S.-W. Pregnancy morbidities in korean patients with takayasu arteritis: A monocentric pilot study. Yonsei Med. J. 61, 970–975 (2020).
Abisror, N. et al. Analysis of risk factors for complications and adverse obstetrical outcomes in women with Takayasu arteritis: A French retrospective study and literature review. Clin. Rheumatol. 39, 2707–2713 (2020).
Gupta, L. et al. Poor obstetric outcomes in Indian women with Takayasu arteritis. Adv. Rheumatol. 60, (2020).
Gönenli, M. G. et al. Pregnancy in Takayasu’s arteritis has a high risk of hypertension-related fetomaternal complications: A retrospective study of a Turkish cohort. Int. J. Rheum. Dis. 25, 140–146 (2022).
Velásquez Giraldo, I. et al. Pregnancy outcomes in patients with Takayasu’s arteritis: Case series. Rev. Colomb. Reumatol. (English Ed. 28, 213–217 (2021).
Nguyen, V., Wuebbolt, D., Pagnoux, C. & D’Souza, R. Pregnancy outcomes in women with primary systemic vasculitis: A retrospective study. J. Matern. Neonatal Med. 32, 1–7 (2019).
Fredi, M. et al. Systemic vasculitis and pregnancy: A multicenter study on maternal and neonatal outcome of 65 prospectively followed pregnancies. Autoimmun. Rev. 14, 686–691 (2015).
Sterne, J. A. et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 355, 1 (2016).
Quenby, S. et al. Miscarriage matters: the epidemiological, physical, psychological, and economic costs of early pregnancy loss. Lancet (London, England) 397, 1658–1667 (2021).
Shen, M. et al. Comparison of risk factors and outcomes of gestational hypertension and pre-eclampsia. PLoS One 12, 1 (2017).
Hutcheon, J. A., Lisonkova, S. & Joseph, K. S. Epidemiology of pre-eclampsia and the other hypertensive disorders of pregnancy. Best Pract. Res. Clin. Obstet. Gynaecol. 25, 391–403 (2011).
Borchers, A. T., Naguwa, S. M., Keen, C. L. & Gershwin, M. E. The implications of autoimmunity and pregnancy. J. Autoimmun. 34, J287–J299 (2010).
Burton, G. J., Redman, C. W., Roberts, J. M. & Moffett, A. Pre-eclampsia: Pathophysiology and clinical implications. BMJ 366, 1–15 (2019).
Andreoli, L. et al. EULAR recommendations for women’s health and the management of family planning, assisted reproduction, pregnancy and menopause in patients with systemic lupus erythematosus and/or antiphospholipid syndrome. Ann. Rheum. 76, 476–485 (2017).
Author information
Authors and Affiliations
Contributions
S.P. and T.D. conceived the idea and title of the manuscript. D.D., A.M. and S.P. analyzed the data, including study selection, data extraction and assessment of bias. S.P. made the statistical analysis. All authors contributed to writing this document. T.D. supervised the task from initiation. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: The original version of this Article contained errors in the Abstract. Full information regarding the corrections made can be found in the correction for this Article.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Partalidou, S., Mamopoulos, A., Dimopoulou, D. et al. Pregnancy outcomes in Takayasu arteritis patients: a systematic review and meta-analysis. Sci Rep 13, 546 (2023). https://doi.org/10.1038/s41598-023-27379-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-27379-9
This article is cited by
-
A Cohort Study of Pregnancy and Fetal Complications Among Patients with Rheumatologic Disorders
The Journal of Obstetrics and Gynecology of India (2024)
-
Successful pregnancies in a patient with Takayasu arteritis and antiphospholipid syndrome, maintained on infliximab corticosteroid-free regimen: case-based review
Rheumatology International (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.