Abstract
Human cooperation (paying a cost to benefit others) is puzzling from a Darwinian perspective, particularly in groups with strangers who cannot repay nor are family members. The beneficial effects of cooperation typically increase nonlinearly with the number of cooperators, e.g., increasing returns when cooperation is low and diminishing returns when cooperation is high. Such nonlinearity can allow cooperation between strangers to persist evolutionarily if a large enough proportion of the population are already cooperators. However, if a lone cooperator faces a conflict between the group’s and its own interests (a social dilemma), that raises the question of how cooperation arose in the first place. We use a mathematically tractable evolutionary model to formalise a chronological narrative that has previously only been investigated verbally: given that ancient humans interacted mostly with family members (genetic homophily), cooperation evolved first by kin selection, and then persisted in situations with nonlinear benefits as homophily declined or even if interactions with strangers became the norm. The model also predicts the coexistence of cooperators and defectors observed in the human population (polymorphism), and may explain why cooperators in behavioural experiments prefer to condition their contribution on the contributions of others (conditional cooperation in public goods games).
Similar content being viewed by others
Introduction
How did humans evolve the ability to cooperate in groups with strangers? Social situations often involve a conflict between the group’s interests and an individual’s interests, i.e., they are social dilemmas1, and paying a cost to benefit others is puzzling from a ‘selfish’ Darwinian perspective. Nevertheless, humans not only regularly cooperate with strangers in daily life, they even cooperate in lab-based games that have been specifically designed to ensure the conflict2. In a lab-based linear public goods game (PGG), anonymous participants (i.e., mutual strangers) can contribute money to a common pool, which is then multiplied and split between them regardless of who contributed. The multiplier is chosen such that the group’s payoff is maximised when everyone contributes, but an individual’s payoff is maximised by not contributing; yet consistently, some participants will contribute3,4.
One possible resolution to this puzzle is that most real-life PGGs are in fact not linear5,6,7, and because cooperating with strangers in a nonlinear PGG can maximise individual payoff, individuals learn the social heuristic to cooperate and apply it to all seemingly similar PGGs8,9,10. The lab-based games above are ‘linear’ because there is a linear relationship between the level of contribution and the benefit from the public good (constant multiplier). In a linear PGG, when an individual switches from defect to cooperate, that individual’s payoff decreases by the same amount regardless of the number of cooperators in the game. Thus, the strength of the social dilemma is independent of cooperator frequency. However, if the benefit from the public good varies as a nonlinear function of the number of cooperators, then the switching gains may vary such that—at certain cooperator frequencies—the social dilemma is absent entirely. For example, in the threshold PGG (Fig. 1), a public good is provided when the number of cooperators in the group meets a minimum threshold5,11,12. Once the threshold is achieved, no defector has an incentive to cooperate, but nor will any cooperator switch to defect, because switching would cause the loss of the public good and thus decrease the former cooperator’s own payoff (\(Z < W\) in Fig. 1)13. This game-theoretic analysis of the switching gains translates mathematically to the evolutionary dynamics14: in an infinitely large, well-mixed population, if the population already has a sufficiently large proportion of cooperators, then cooperation may persist. Thus, such nonlinear PGGs may be both a source of the social heuristics that misfire in lab-based linear PGGs15 and also explain how cooperation persists evolutionarily.
A hypothetical threshold public goods game involving a prehistoric hunt (for modern examples, see21,22). A minimum number of hunters (\(\tau = 5\)) must cooperate to successfully surround and kill an animal or their efforts are wasted. A cooperator’s payoff is W if the threshold is met and X if it is not (blue line). All members share the meat (\(n=8\)); therefore, the highest payoff goes to defectors (red line) regardless of whether the hunt is successful (payoff Y) or not (payoff Z). However, if an individual is likely to be the pivotal hunter, i.e., the hunter that brings the group above the threshold for a successful hunt, then it is incentivised to cooperate because the payoff to a defector when the threshold is not met is less than the payoff to a cooperator when the threshold is met (\(Z < W\)).
However, while nonlinearity may explain the evolutionary persistence of cooperation, cooperating is only adaptive if a large enough proportion of the population are already cooperators, which raises the question of how cooperation arose in the first place6,16,17. Rare cooperative ‘mutants’ can only invade a population of defectors if the payoff to a lone cooperator is higher than the payoff it would have received if it had defected instead14. In other words, cooperation can never evolve in a nonlinear PGG that presents a social dilemma to a lone cooperator (in contrast to, e.g., a by-product mutualism18).
One escape is if groups in the past tended to form between family members, i.e., genetic homophily, allowing cooperators to first invade by kin selection. Once cooperators built up sufficient numbers in the population, grouping with family was no longer necessary; cooperation can persist in certain nonlinear games due to their dynamical properties discussed above, even as the social environment shifts from interacting with kin to nonkin. The verbal argument that the invasion and persistence of cooperation in one-shot games can arise via a combination of kin selection and nonlinear benefits has been made before16, and some simulation results also support this intuition6,17, but a mathematically tractable model is needed to provide a rigorous basis for this narrative. One challenge is to account for genetic association between group members beyond pairs of individuals (Hamilton’s r), i.e., triplets, quadruplets, and so on (reviewed in19).
Here, we model how genetic homophily affects the evolutionary invasion and persistence of cooperation in such a nonlinear PGG. We treat the level of homophily as an extrinsic factor that reflects the prevailing social environment at a given point in time. To analyse the model, we use the higher-order genetic association approach of Ohtsuki20, which provides a systematic mathematical framework to account for genetic association between more than two group members. Our main technical innovation is to detail three different homophilic group-formation models, which allows us to obtain analytical results for the evolutionary dynamics. This facilitates easy identification of bifurcation points and regions of parameter space in which cooperation and defection are both evolutionarily stable, with the current state depending on the history of the system. We show that homophily can indeed facilitate the invasion of cooperation, and moreover, that cooperation can persist if homophily subsequently declines, in some cases even if homophily disappears altogether. Thus, we provide a rigorous explanation for how cooperation arose and persisted as humans expanded their social networks from kin to nonkin and strangers.
Methods
We focused on the threshold PGG (e.g., Fig. 1), which is the nonlinear PGG that provides the clearest example of the evolutionary properties of interest (i.e., cooperators can persist but cannot invade). Previous authors show that the dynamical qualities of the threshold game are preserved in more general sigmoid games14,16,17,23,24, and we also obtain qualitatively similar results under homophily (see Supplementary).
In the threshold PGG, the public good is produced when the number of Cooperators in a group of size n meets or exceeds a threshold \(\tau\), where \(1< \tau < n\)5,11,12. We defined the payoff structure with four parameters: W and Y are the payoffs to cooperators and defectors, respectively, if the threshold is met; and X and Z are the corresponding payoffs if the threshold is not met (Table 1). We imposed three constraints: (1) \(\text {min}(W, X, Y, Z) = X\), which represents the risk of cooperation; (2) \(Z<W\), which combined with the first constraint (in particular, \(X < Z\)) ensures that just meeting the threshold is Pareto optimal; and (3) \(Z < Y\), which means Defectors benefit from the public good without contributing (free-riding). The constraints can be summarised as \(X<Z, Z<W, Z<Y\). These constraints offered more flexibility than the one-parameter model common in the theoretical literature (e.g., \(W = 1-c\), \(X=-c\), \(Y=1\), \(Z=0\))25,26, reflecting the greater range explored in the experimental literature27,28,29.
To characterise the evolutionary dynamics, we used the higher-order genetic association approach of Ohtsuki20, which quantifies the change in the proportion of Cooperators in the population over time, \(\Delta p\) (Supplementary S1). The model assumes an infinitely large population of haploid individuals, and we refer to subsets of individuals that are identical by descent as “families”. Each individual has a genetically determined pure Cooperate or Defect strategy, and its payoff from the threshold game determines the number of clonal offspring it contributes to the next generation. The model assumes that all offspring join a global pool, so competition in this model is global and there is no local competition30. Our approach is equivalent to the replicator-dynamics approach under random group formation, but it can also account for the genetic association between group members under homophilic group formation. Homophily is imposed as an extrinsic factor in our model that increases the probability that kin group together, and thus indirectly increases the probabilities that Cooperators group together and Defectors group together. This modifies the probability distribution of outcomes, and thus the expected payoff given a particular strategy, and therefore influences \(\Delta p\).
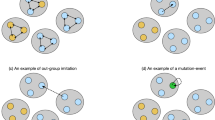
Defining a homophilic group-formation process allows us to derive the probability distribution of different group compositions—i.e., how the group is divided into families that are identical by descent, and thus share the same strategy—and therefore we can obtain analytic expressions for \(\Delta p\). We explored three different homophilic group-formation models (details in Supplementary S2) where groups are formed sequentially and current members tend to recruit or attract kin (Fig. 2). These models were chosen primarily as plausible simplifications of reality, but also selected because they are different enough to potentially have different qualitative effects on the model (Supplementary S8). We defined the homophily level \(h = [0,1]\) as the expected proportion of new recruits that are kin of an existing member. We are interested in the effect of a changing tendency to group with kin over time, with this tendency being imposed extrinsically and attributable to a changing social organisation that we do not explicitly model. Therefore, we varied the homophily level by treating the parameters describing kin recruitment as control parameters. The three group-formation models are as follows:
-
1.
Leader driven: The leader is chosen at random from the population. The leader recruits/attracts nonkin with probability \(q \in [0,1]\) and kin with probability \(1-q\), and therefore \(h \equiv 1-q\). This model was inspired by foraging-band formation among Nambikwara people (Brazil), where the group leader provides the focal point around which the group forms31.
-
2.
Members attract: The initial member is chosen at random. Current group members have equal weighting 1 of attracting a new member who is kin, but nonkin members are also attracted to the group itself with collective weighting \(\alpha \in [0, \infty )\), and therefore \(h \equiv \left( \sum _{i=1}^{n-1} i/(\alpha +i) \right) /\left( n-1 \right)\) represents the expected proportion of kin recruited during group formation. This model was chosen because it describes a plausible mechanism while also providing a relatively simple description of the family partition probabilities, i.e., Ewens’ sampling formula32.
-
3.
Members recruit: The initial member is chosen at random. Current group members have an equal chance to recruit the next member. Members recruit nonkin with probability \(q \in [0,1]\) and kin with probability \(1-q\), and therefore \(h \equiv 1-q\). This model was created as an alternative to the members-attract model and represents an opposite extreme of which member drives recruitment to the leader-driven model.
Examples of a group of five individuals forming according to the rules of the three homophilic group-formation models: (a) leader driven, (b) members attract, and (c) members recruit. Current members sequentially recruit or attract new members (lines) who tend to be kin (same colour). The grey group in (b) represents attraction of nonkin to the group itself.
Results
If groups are formed randomly, Cooperators can never invade a population of Defectors because the payoff to a lone Cooperator in a group of Defectors is lower than the payoff to Defectors (Supplementary S4; e.g., Fig. S1). However, if group formation is sufficiently homophilic, Cooperators can invade.
Homophilic group-formation promotes cooperation by promoting both the invasion and persistence of Cooperators (Fig. 3 shows results for the members-recruit group-formation model; qualitatively similar results are obtained for other models, Supplementary S5). Mathematically, as homophily is increased from zero, three successive and biologically important bifurcations occur. First, when homophily is increased above some threshold \({\hat{h}}\) (Supplementary S6), the dynamics can transition from zero to two interior equilibria: an unstable polymorphism \(p_u^*\) and a stable polymorphism \(p_s^*\) (e.g., Fig. 3a). Therefore, Cooperators can coexist with Defectors under homophily where it was not possible under random group formation. Further increases in homophily move the unstable polymorphism towards the all-defect equilibrium (\(p_u^* \downarrow\)) and the stable polymorphism towards the all-cooperate equilibrium (\(p_s^* \uparrow\)). This both increases the proportion of Cooperators in the stable polymorphic population and lowers the initial proportion of Cooperators required to invade and persist. The second and third bifurcations occur when each of two interior equilibria collide with the corresponding trivial equilibria (\(p_s^* \rightarrow p_1^*=1\) at \(h=h_1\) and \(p_u^* \rightarrow p_0^*=0\) at \(h=h_0\), Supplementary S6), which causes the two trivial equilibria to flip between stable and unstable, which flips the invasibility of each strategy. For \(h > h_1\), Defectors can no longer invade a population of Cooperators (\(p_1^*\) stable), and for \(h > h_0\), Cooperators can invade a population of Defectors (\(p_0^*\) unstable).
Two examples of how genetic homophily affects the evolutionary dynamics in our model, showing possible trajectories of cooperation as human homophily (tendency to interact with genetically related others) decreased over time due to changing social environments (blue lines). The evolutionary dynamics separates into qualitatively different regimes depending on the homophily level: \(h < {\hat{h}}\), Cooperators cannot persist (dark shading); \(h < h_1\), Defectors can both invade and persist (red shading); \(h > h_0\), Cooperators can invade (blue shading). In the human ancestral past (e.g., \(>1.6\) Ma33), homophily was high (point A), which under the model would have allowed cooperation to invade (B). As interactions with nonkin increased (h decreased), Cooperation persisted even into the region where it could not invade (C). Depending on the parameter values, Cooperation can either persist even if homophily disappears entirely (i.e., all interactions become nonkin; D) or be lost below a certain level of homophily (E). The example illustrated uses the members-recruit group-formation model (\(h \equiv 1-q\)) with parameter values \(n=8\), \(W=2\), \(Y=3\), \(Z=0\); (a) \(\tau =5\), \(X=-1\); (b) \(\tau =4\), \(X=-0.5\). See Supplementary S5 for qualitatively similar results from other group-formation models and for sigmoid PGGs.
Under random group formation, the stable coexistence of Cooperators and Defectors can be lost if the group size increases26 or the threshold \(\tau\) is changed to an intermediate value25. Introducing homophilic group formation can reverse these effects and cause the stable polymorphism to reappear (Supplementary S7) in the same way as illustrated in Fig. 3a. The leader-driven group-formation model is the most conducive to cooperation, particularly at moderate group size (e.g., Supplementary Fig. S6): Defectors need lower homophily to invade and persist (\(h_1\) is lower), Cooperators can invade at lower homophily (\(h_0\) is lower), and Cooperators can persist at lower homophily (\({\hat{h}}\) is lower). For the leader-driven and members-attract models, as the group size increases, the number of individuals in the group that are members of the largest family in the group asymptotes to a fixed proportion. In contrast, for the members-recruit model, the proportion asymptotes to 0, and therefore the members-recruit model is the least conducive to cooperation (Supplementary S8).
Discussion
Over the course of human evolution, social interactions have expanded from kin to nonkin and strangers34,35,36,37,38, and that general trend continues today. For example, hominin material transport distances increased from \(\sim\)13 km before 1.6 Ma (comparable to chimpanzee home-range sizes) to hundreds of kilometres after 130 ka, indicating exchange networks33 and relationships beyond kin39. Today, hunter-gatherers maintain social networks of hundreds of unrelated individuals40,41 who congregate seasonally for communal hunting and social activities22,42, and in industralised societies, falling kin-network density is motivating new bases for social identity43.
When we imposed this pattern of declining genetic homophily on the results from our model, several chronological narratives emerged (blue arrows, Fig. 3). When homophily was high in the ancestral past, selection favoured cooperation, allowing the faculties that coordinate collective action to evolve (A \(\rightarrow\) B, Fig. 3). The tendency to associate with kin declined over time, but cooperation was not necessarily lost. Cooperation persisted if a sufficiently high degree of homophily was retained (B \(\rightarrow\) C), or if the game payoffs allowed cooperation to persist in the absence of homophily (C \(\rightarrow\) D). In some cases in the model, the loss of homophily led to the loss of cooperation in that game (C \(\rightarrow\) E), which cannot be recovered by simply reversing that incremental loss of homophily. Either homophily must be greatly increased to allow cooperation to reinvade (E \(\rightarrow\) A \(\rightarrow\) B), or a large proportion of Cooperators must invade the population together (above the separatrix, dashed line, Fig. 3).
Three main pieces of evidence suggest that our narrative is plausible. First, real-world benefits from hunting typically have sigmoid-like relationship with the number of hunters17, which qualitatively matches the shape of our model’s payoff functions. Collaborative foraging in general has been linked to the early stages of cooperation’s evolution44, including confrontational scavenging45,46, which also likely has a nonlinear benefit function47. Hunter-gatherers today often share meat among members of hunting groups formed on the basis of cultural rather than genetic kinship21,42,48, reflecting the later stages of our narrative. Second, if we are correct that cooperative social heuristics are derived from such real-life nonlinear PGGs, then participants in lab-based linear PGGs should behave as though they are playing nonlinear PGGs—and indeed they do. Cooperative participants prefer to condition their contributions on the contribution level of others4,49,50, which is only rational in nonlinear games. Conditional cooperation is also correlated with the belief that the payoff-maximising strategy depends on the contribution of others51, and chat logs from computer-networked games reveal a common misperception of linear PGGs as some type of coordination problem52. Third, the polymorphic coexistence of cooperators and defectors predicted by both our threshold game and qualitatively similar sigmoid games14,16,17,23 is consistent with the observation of polymorphism in human cooperative types17. Nonlinear games with a continuous strategy can also possess an interior equilibrium, i.e., a mixed strategy53, which is consistent with experimental participants’ tendency to avoid the extreme strategies of 100% defection or cooperation15,54.
Our model’s key contribution is to demonstrate the continuity of the evolutionary persistence of cooperation as interactions shifted from kin to strangers. The idea that cooperation first arose through kin selection is not new16,55,56,57, and some authors have emphasised that nonlinearity can permit cooperation to persist in one-shot PGGs with strangers without enforcement nor assortment23, but the connections between the two are typically explained verbally. A complete explanation for cooperation in social dilemmas must address both invasion and persistence of cooperative types6, and both past homophily and nonlinearity in our model are necessary: a linear game with past homophily may allow invasion but not persistence; and a nonlinear game without homophily may allow persistence but not invasion. We were able to obtain the necessary analytic expressions for the evolutionary dynamics (that could then be solved numerically) because we used the higher-order genetic association approach20. One barrier until now has been calculating family partition probabilities (\(F_{n \rightarrow {\textbf{n}}}\)), and our group-formation models may be of use to future workers (code and tutorials in Supplementary S9), including those who wish to build more realism into the models (e.g., Nambikwara leaders are not chosen at random but by reputation31).
We modelled homophilic group formation through direct recognition of family members, which in early hominins likely operated through co-residence duration58 and physical phenotype matching59,60, and later also through (culturally defined) kinship frameworks61; however, limited dispersal can also lead to homophilic group formation without kin recognition62. For example, Lehmann et al.63 explored the invasion and persistence of punishment using Wright’s infinite islands approach64, where the population is split into discrete patches with occassional dispersal of individuals between them. Although limited dispersal promotes homophily, it also promotes competition between kin, which can cancel out homophily’s positive effects on cooperation65. Therefore, the evolution of cooperation by limited dispersal typically requires some additional mechanism to export competition (reviewed in66), whereas offspring in our model always compete globally with offspring from other groups30. Several authors have shown that spatial clustering allows cooperation to invade threshold PGGs specifically6,17, and such spatially explicit lattice models can also reveal interesting, complex behaviour67; however, an analytical treatment of lattice models is difficult.
We wish to highlight some previous theoretical works with similar themes to our own. First, two conceptually similar papers modelled repeated interactions (in contrast to our one-shot game) with contingent strategies: threshold-triggered defection57 and also punishment56. They too found that high relatedness can facilitate the invasion of cooperative strategies that could otherwise only persist but not invade. In contrast to our model, they used the infinite-islands approach discussed above, and the cancellation effect of kin competition was averted by the additional assumption that population size on each ‘island’ was elastic (budding was also discussed). Thus, genetic assortment is present in both their and our approaches, and both export competition, but the scenario considered and assumptions needed differ. Second, Peña et al.68 studied the convergence stability of mixed strategies of discrete actions in nonlinear games. They assumed weak selection and that mutations caused only a small deviation in strategy, which allowed them to omit higher-order genetic associations (beyond dyadic relatedness). They found that a PGG with relatedness was equivalent to a game between randomly drawn players with rescaled payoffs (their Eq. 15). Consequently, the effects of relatedness in their model differ from those in our model, where our mutant invader plays a new discrete strategy (e.g., a Cooperator invading a population of Defectors) and the higher-order genetic associations must be accounted for. Finally, for a study of relatedness in nonlinear PGGs with continuous levels of investment (in contrast to our discrete strategies), we refer readers to Coder Gylling and Brännström69.
The two mechanisms that can promote cooperation between nonkin that have received the most attention in the literature—repeated interactions and reputation effects—involve violations of typical lab-based PGG assumptions (i.e., one-shot, anonymous interactions), and our model suggests the assumption of linearity also deserves scrutiny. Repeated interactions have a strong effect on cooperative behaviour in group games70, and may partly explain conditional cooperation in a 2-player games71,72; however, tit-for-tat like cooperative strategies can only persist in many-player linear PGGs under restrictive conditions73 (but see74). There is good evidence that reputational mechanisms influence cooperation within groups70,75,76,77,78; however, for reputation to serve as a complete explanation for cooperation in anonymous lab-based games, one must assume that the cue responses governing cooperation cannot easily distinguish between anonymous and reputation-relevant interactions, which is controversial15,40,79. In contrast, it seems plausible that humans have not evolved proximate mechanisms that can properly interpret payoff functions. Even in the conceptually simple threshold game, the calculation required to determine whether a cooperative symmetric Nash equilibrium exists is difficult26,80 (Supplementary S4.2.4), and experimental participants instead use a process of trial-and-error to converge on the equilibrum12,28,81,82,83. If most real-life PGGs are nonlinear, then relying on social heuristics is likely the only practical option available. Therefore, we agree with Raihani and Bshary15 that real-life nonlinear PGGs may be a significant source of social heuristics, and with Archetti et al. 7 that nonlinearity may be an important part of the explanation for cooperation between strangers.
Social heuristics provide a proximate explanation, and evolution an ultimate explanation, for why humans cooperate, and our model closes this gap for social dilemmas analogous to nonlinear PGGs. The gap remains open for other mechanisms of cooperation persistence; for example, Orma people have relatively high PGG contributions3 likely because their real-life public-goods practice includes punishment84, but how punishment first arose remains an open question63,85,86,87. In cultures with real-life monetary nonlinear PGGs, we may expect experimental participants to start with a modest expectation that cooperating will be payoff maximising and learn not to cooperate instead88,89,90. Combining social heuristics with an evolutionary approach may provide additional insights. For example, the positive assortment of cooperative Hadza campmates91 is probably due to social-learning dynamics rather than assortative preferences92,93,94. If each dry-season camp is roughly analogous to a single group-game in our model, then some years the camp will successfully converge on cooperation and some years it will not, which determines the ‘mood’ of camp members when they participate in the experiment (cf.,95), but cooperation persists in the population because it is not selected against in the long term.
Our model abstracts and simplifies the narrative, and we wish to highlight some key points where greater realism might be added in the future. Cooperative activities like hunting likely involved nontrivial cognitive faculties (e.g., joint intentionality44, communication abilities33,96, and understanding roles and expectations97) that provided a substrate for cooperation with nonkin—our model ignores cognitive evolution, but including it could enhance the persistence of cooperation. We also treated homophily level as an extrinsic parameter; however, it would be interesting to study a model where homophily is an intrinsic trait98. We speculate that the opportunity to cooperate with nonkin may have entered a positive feedback with declining homophily, increasing the number of potential cooperative partners and therefore the number of games played and benefits produced. We can contrast this with the evolutionary maladaption hypothesis99,100,101, where ancestral cooperation was indiscriminate but—due to limited dispersal—typically directed at kin, and was only extended to nonkin later and maladaptively due to e.g., rapidly increasing population size. In our model, individuals who cooperate with nonkin are not necessarily maladapted; therefore, their behaviour was not selected against. The key challenge would have been to overcome kin bias, potentially by developing new cues for cooperation (e.g., sharing a common fate102,103). The exact mechanisms (including cultural104) are outside the scope of our model, but we note that contemporary humans often cooperate with nonkin using cooptations of kin psychology105,106,107. For example, socially constructed notions of kinship61,108 coopt both the kinship framework that coordinates cooperation109,110 and the kin-psychological impulse to do so111. Interestingly, homophily in the broader sense of generalised attraction to similar others112,113 likely also has roots in kin psychology105,114, and the human tendency to empathise115 and cooperate with similar others even based on meaningless traits116,117 may have provided a starting point for nongenetic bases of group identity118, which functions by triggering the expectation that others will coordinate their cooperation with one’s own (cf.,21,75).
In conclusion, we propose that cooperation in human societies first arose due to kin selection, but as humans expanded their interactions to nonkin, it persisted due to the dynamical properties of certain nonlinear PGGs. Cooperative one-shot encounters with strangers occur frequently in modern society. We hope that as our understanding of what promotes or impedes such cooperation grows, we will become better equipped to tackle modern social and environmental dilemmas119.
Data Availability
Functions and scripts used to generate the results, along with some tutorials, are archived in the Github repository: https://github.com/nadiahpk/homophilic-threshold-PGG, DOI: https://doi.org/10.5281/zenodo.7344398.
References
Van Lange, P. A., Joireman, J., Parks, C. D. & Van Dijk, E. The psychology of social dilemmas: A review. Organ. Behav. Hum. Decis. Process. 120, 125–141 (2013).
Ledyard, J. O. Public goods: A survey of experimental research. In Handbook of Experimental Economics (eds Kagel, J. & Roth, A.) 111–193 (Princeton University Press, Princeton, 1995).
Henrich, J. et al. “Economic man’’ in cross-cultural perspective: Behavioral experiments in 15 small-scale societies. Behav. Brain Sci. 28, 795–815 (2005).
Thöni, C. & Volk, S. Conditional cooperation: Review and refinement. Econ. Lett. 171, 37–40 (2018).
Archetti, M. & Scheuring, I. Game theory of public goods in one-shot social dilemmas without assortment. J. Theor. Biol. 299, 9–20 (2012).
Vásárhelyi, Z. & Scheuring, I. Invasion of cooperators in lattice populations: Linear and non-linear public good games. Biosystems 113, 81–90 (2013).
Archetti, M., Scheuring, I. & Yu, D. The non-tragedy of the non-linear commons. arXiv:2020.04022 (2020).
Rand, D. G. et al. Social heuristics shape intuitive cooperation. Nat. Commun. 5, 1–12 (2014).
Rand, D. G. Cooperation, fast and slow: Meta-analytic evidence for a theory of social heuristics and self-interested deliberation. Psychol. Sci. 27, 1192–1206 (2016).
Capraro, V. The dual-process approach to human sociality: A review. Available at SSRN 3409146 (2019).
Palfrey, T. R. & Rosenthal, H. Participation and the provision of discrete public goods: A strategic analysis. J. Public Econ. 24, 171–193 (1984).
Cadsby, C. B. & Maynes, E. Voluntary provision of threshold public goods with continuous contributions: Experimental evidence. J. Public Econ. 71, 53–73 (1999).
Gradstein, M. & Nitzan, S. Binary participation and incremental provision of public goods. Soc. Choice Welf. 7, 171–192 (1990).
Peña, J., Lehmann, L. & Nöldeke, G. Gains from switching and evolutionary stability in multi-player matrix games. J. Theor. Biol. 346, 23–33 (2014).
Raihani, N. J. & Bshary, R. Why humans might help strangers. Front. Behav. Neurosci. 9, 39. https://doi.org/10.3389/fnbeh.2015.00039 (2015).
Bach, L. A., Helvik, T. & Christiansen, F. B. The evolution of n-player cooperation-threshold games and ESS bifurcations. J. Theor. Biol. 238, 426–434 (2006).
Boza, G. & Számadó, S. Beneficial laggards: Multilevel selection, cooperative polymorphism and division of labour in threshold public good games. BMC Evol. Biol. 10, 1–12 (2010).
Clutton-Brock, T. Cooperation between non-kin in animal societies. Nature 462, 51–57 (2009).
Ohtsuki, H. Evolutionary games in Wright’s island model: Kin selection meets evolutionary game theory. Evol. Int. J. Organ. Evol. 64, 3344–3353 (2010).
Ohtsuki, H. Evolutionary dynamics of n-player games played by relatives. Philos. Trans. R. Soc. B: Biol. Sci. 369, 20130359 (2014).
Alvard, M. S. & Nolin, D. A. Rousseau’s whale hunt? Coordination among big-game hunters. Curr. Anthropol. 43, 533–559 (2002).
Balme, J. Communal hunting by Aboriginal Australians: Archaeological and ethnographic evidence. In Manipulating Prey: Development of Large-Scale Kill Events Around the Globe (eds Carlson, K. & Bemet, L.) 42–62 (University of Colorado Press, Boulder, Colorado, 2018).
Archetti, M. & Scheuring, I. Coexistence of cooperation and defection in public goods games. Evol. Int. J. Organ. Evol. 65, 1140–1148 (2011).
Archetti, M. How to analyze models of nonlinear public goods. Games 9, 17 (2018).
De Jaegher, K. High thresholds encouraging the evolution of cooperation in threshold public-good games. Sci. Rep. 10, 1–10 (2020).
Nöldeke, G. & Peña, J. Group size and collective action in a binary contribution game. J. Math. Econ. 88, 42–51 (2020).
Dawes, R. M., Orbell, J. M., Simmons, R. T. & Van De Kragt, A. J. Organizing groups for collective action. Am. Polit. Sci. Rev. 80, 1171–1185 (1986).
Rapoport, A. & Eshed-Levy, D. Provision of step-level public goods: Effects of greed and fear of being gypped. Organ. Behav. Hum. Decis. Process. 44, 325–344 (1989).
Gross, J. & De Dreu, C. K. Individual solutions to shared problems create a modern tragedy of the commons. Sci. Adv. 5, eaau7296 (2019).
West, S. A. et al. Cooperation and the scale of competition in humans. Curr. Biol. 16, 1103–1106 (2006).
Lévi-Strauss, C. The social and psychological aspects of chieftainship in a primitive society: The Nambikwara. Trans. N. Y. Acad. Sci. 2 (1944).
Ewens, W. J. The sampling theory of selectively neutral alleles. Theor. Popul. Biol. 3, 87–112 (1972).
Marwick, B. Pleistocene exchange networks as evidence for the evolution of language. Camb. Archaeol. J. 13, 67–81 (2003).
Kuhn, S. L., Stiner, M. C., Reese, D. S. & Güleç, E. Ornaments of the earliest Upper Paleolithic: New insights from the Levant. Proc. Natl. Acad. Sci. 98, 7641–7646 (2001).
Gamble, C., Gowlett, J. & Dunbar, R. The social brain and the shape of the Palaeolithic. Camb. Archaeol. J. 21, 115–136 (2011).
Brooks, A. S. et al. Long-distance stone transport and pigment use in the earliest Middle Stone Age. Science 360, 90–94 (2018).
Sehasseh, E. M. et al. Early middle stone age personal ornaments from Bizmoune Cave, Essaouira, Morocco. Sci. Adv. 7, eabi8620 (2021).
Ringbauer, H., Novembre, J. & Steinrücken, M. Parental relatedness through time revealed by runs of homozygosity in ancient DNA. Nat. Commun. 12, 1–11 (2021).
Moutsiou, T. Changing scales of obsidian movement and socialnetworking. In Unravelling the Palaeolithic: Ten years of research at the Centre for the Archaeology of Human Origins (CAHO, University of Southampton), 85–95 (British Archaeological Reports, 2012).
Hill, K. R. et al. Co-residence patterns in hunter-gatherer societies show unique human social structure. Science 331, 1286–1289 (2011).
Bird, D. W., Bird, R. B., Codding, B. F. & Zeanah, D. W. Variability in the organization and size of hunter-gatherer groups: Foragers do not live in small-scale societies. J. Hum. Evol. 131, 96–108 (2019).
Kelly, R. L. The Lifeways of Hunter-Gatherers: The Foraging Spectrum (Cambridge University Press, Cambridge, 2013).
David-Barrett, T. Herding friends in similarity-based architecture of social networks. Sci. Rep. 10, 1–6 (2020).
Tomasello, M., Melis, A. P., Tennie, C., Wyman, E. & Herrmann, E. Two key steps in the evolution of human cooperation: The interdependence hypothesis. Curr. Anthropol. 53, 673–692 (2012).
Thompson, J. C., Carvalho, S., Marean, C. W. & Alemseged, Z. Origins of the human predatory pattern: The transition to large-animal exploitation by early hominins. Curr. Anthropol. 60, 1–23 (2019).
Pobiner, B. L. The zooarchaeology and paleoecology of early hominin scavenging. Evol. Anthropol. Issues News Rev. 29, 68–82 (2020).
Bickerton, D. & Szathmáry, E. Confrontational scavenging as a possible source for language and cooperation. BMC Evol. Biol. 11, 1–7 (2011).
Alvard, M. Genetic and cultural kinship among the Lamaleran whale hunters. Hum. Nat. 22, 89–107 (2011).
Fischbacher, U., Gächter, S. & Fehr, E. Are people conditionally cooperative? Evidence from a public goods experiment. Econ. Lett. 71, 397–404 (2001).
Chaudhuri, A. Sustaining cooperation in laboratory public goods experiments: A selective survey of the literature. Exp. Econ. 14, 47–83 (2011).
Burton-Chellew, M. N., El Mouden, C. & West, S. A. Conditional cooperation and confusion in public-goods experiments. Proc. Natl. Acad. Sci. 113, 1291–1296 (2016).
Cox, C. A. & Stoddard, B. Strategic thinking in public goods games with teams. J. Public Econ. 161, 31–43 (2018).
Motro, U. Co-operation and defection: Playing the field and the ESS. J. Theor. Biol. 151, 145–154 (1991).
Kümmerli, R., Burton-Chellew, M. N., Ross-Gillespie, A. & West, S. A. Resistance to extreme strategies, rather than prosocial preferences, can explain human cooperation in public goods games. Proc. Natl. Acad. Sci. 107, 10125–10130 (2010).
Axelrod, R. & Hamilton, W. D. The evolution of cooperation. Science 211, 1390–1396 (1981).
Boyd, R., Schonmann, R. H. & Vicente, R. Hunter-gatherer population structure and the evolution of contingent cooperation. Evol. Hum. Behav. 35, 219–227 (2014).
Schonmann, R. H. & Boyd, R. A simple rule for the evolution of contingent cooperation in large groups. Philos. Trans. R. Soc. B: Biol. Sci. 371, 20150099 (2016).
Lieberman, D., Tooby, J. & Cosmides, L. Does morality have a biological basis? An empirical test of the factors governing moral sentiments relating to incest. Proc. R. Soc. Lond. B 270, 819–826 (2003).
Weisfeld, G. E., Czilli, T., Phillips, K. A., Gall, J. A. & Lichtman, C. M. Possible olfaction-based mechanisms in human kin recognition and inbreeding avoidance. J. Exp. Child Psychol. 85, 279–295 (2003).
DeBruine, L. M., Jones, B. C., Little, A. C. & Perrett, D. I. Social perception of facial resemblance in humans. Arch. Sex. Behav. 37, 64–77 (2008).
Voorhees, B., Read, D. & Gabora, L. Identity, kinship, and the evolution of cooperation. Curr. Anthropol. 61, 194–218 (2020).
Kay, T., Keller, L. & Lehmann, L. The evolution of altruism and the serial rediscovery of the role of relatedness. Proc. Natl. Acad. Sci. 117, 28894–28898 (2020).
Lehmann, L., Rousset, F., Roze, D. & Keller, L. Strong reciprocity or strong ferocity? A population genetic view of the evolution of altruistic punishment. Am. Nat. 170, 21–36 (2007).
Rousset, F. Genetic Structure and Selection in Subdivided Populations Vol. 40 (Princeton University Press, Princeton, 2013).
Taylor, P. D. Altruism in viscous populations—an inclusive fitness model. Evol. Ecol. 6, 352–356 (1992).
West, S. A., Pen, I. & Griffin, A. S. Cooperation and competition between relatives. Science 296, 72–75 (2002).
Gokhale, C. S. & Park, H. J. Eco-evolutionary spatial dynamics of nonlinear social dilemmas. In Advances in Dynamic Games (eds Ramsey, D. & Renault, J.) 187–200 (Springer International Publishing, Birkhäuser, Cham, 2020).
Peña, J., Nöldeke, G. & Lehmann, L. Evolutionary dynamics of collective action in spatially structured populations. J. Theor. Biol. 382, 122–136 (2015).
Coder Gylling, K. & Brännström, Å. Effects of relatedness on the evolution of cooperation in nonlinear public goods games. Games 9, 87 (2018).
Yamakawa, T., Okano, Y. & Saijo, T. Detecting motives for cooperation in public goods experiments. Exp. Econ. 19, 500–512 (2016).
Kiyonari, T., Tanida, S. & Yamagishi, T. Social exchange and reciprocity: Confusion or a heuristic?. Evol. Hum. Behav. 21, 411–427 (2000).
Skyrms, B. The stag hunt. Proc. Address. Am. Philos. Assoc. 75, 31–41 (2001).
Boyd, R. & Richerson, P. J. The evolution of reciprocity in sizable groups. J. Theor. Biol. 132, 337–356 (1988).
Takezawa, M. & Price, M. E. Revisiting, “The evolution of reciprocity in sizable groups’’ continuous reciprocity in the repeated n-person prisoner’s dilemma. J. Theor. Biol. 264, 188–196 (2010).
Yamagishi, T. & Mifune, N. Does shared group membership promote altruism? Fear, greed, and reputation. Ration. Soc. 20, 5–30 (2008).
Mifune, N., Hashimoto, H. & Yamagishi, T. Altruism toward in-group members as a reputation mechanism. Evol. Hum. Behav. 31, 109–117 (2010).
De Dreu, C. K., Balliet, D. & Halevy, N. Parochial cooperation in humans: Forms and functions of self-sacrifice in intergroup conflict. Adv. Motiv. Sci. 1, 1–47 (2014).
Balliet, D., Wu, J. & De Dreu, C. K. Ingroup favoritism in cooperation: A meta-analysis. Psychol. Bull. 140, 1556 (2014).
Fehr, E. & Henrich, J. Is strong reciprocity a maladaptation? In Genetic and Cultural Evolution of Cooperation (ed. Hammerstein, P.) 55–82 (MIT Press, Cambridge, MA, 2003).
Rapoport, A. Provision of public goods and the MCS experimental paradigm. Am. Polit. Sci. Rev. 79, 148–155 (1985).
Offerman, T., Sonnemans, J. & Schram, A. Value orientations, expectations and voluntary contributions in public goods. Econ. J. 106, 817–845 (1996).
Hsu, L.-C. Effects of framing, group size, and the contribution mechanism on cooperation in threshold public goods and common resources experiments. Acad. Econ. Pap. 31, 1–31 (2003).
Cartwright, E., Stepanova, A. & Xue, L. Impulse balance and framing effects in threshold public good games. J. Public Econ. Theory 21, 903–922 (2019).
Binmore, K. Economic man-or straw man?. Behav. Brain Sci. 28, 817–818 (2005).
Boyd, R. & Mathew, S. A narrow road to cooperation. Science 316, 1858–1859 (2007).
Mathew, S. & Boyd, R. When does optional participation allow the evolution of cooperation?. Proc. R. Soc. B: Biol. Sci. 276, 1167–1174 (2009).
Roberts, G. When punishment pays. PLoS ONE 8, e57378 (2013).
McAuliffe, W. H., Burton-Chellew, M. N. & McCullough, M. E. Cooperation and learning in unfamiliar situations. Curr. Dir. Psychol. Sci. 28, 436–440 (2019).
Andreozzi, L., Ploner, M. & Saral, A. S. The stability of conditional cooperation: Beliefs alone cannot explain the decline of cooperation in social dilemmas. Sci. Rep. 10, 1–10 (2020).
Burton-Chellew, M. N. & West, S. A. Payoff-based learning best explains the rate of decline in cooperation across 237 public-goods games. Nat. Hum. Behav. 5, 1330–1338 (2021).
Apicella, C. L., Marlowe, F. W., Fowler, J. H. & Christakis, N. A. Social networks and cooperation in hunter-gatherers. Nature 481, 497–501 (2012).
Henrich, J. Human cooperation: The hunter-gatherer puzzle. Curr. Biol. 28, R1143–R1145 (2018).
Smith, K. M., Larroucau, T., Mabulla, I. A. & Apicella, C. L. Hunter-gatherers maintain assortativity in cooperation despite high levels of residential change and mixing. Curr. Biol. 28, 3152–3157 (2018).
Smith, K. M. & Apicella, C. L. Partner choice in human evolution: The role of cooperation, foraging ability, and culture in Hadza campmate preferences. Evol. Hum. Behav. 41, 354–366 (2020).
Peysakhovich, A. & Rand, D. G. Habits of virtue: Creating norms of cooperation and defection in the laboratory. Manage. Sci. 62, 631–647 (2016).
Van de Kragt, A. J., Orbell, J. M. & Dawes, R. M. The minimal contributing set as a solution to public goods problems. Am. Polit. Sci. Rev. 77, 112–122 (1983).
Sedikides, C. & Skowronski, J. J. On the evolutionary functions of the symbolic self: The emergence of self-evaluation motives. In Psychological Perspectives on Self and Identity (eds Tesser, A. et al.) 91–117 (American Psychological Association, Washington DC, 2000). https://doi.org/10.1037/10357-004.
Newton, J. The preferences of Homo Moralis are unstable under evolving assortativity. Int. J. Game Theory 46, 583–589 (2017).
Burnham, T. C. & Johnson, D. D. The biological and evolutionary logic of human cooperation. Anal. Kritik 27, 113–135 (2005).
Hagen, E. H. & Hammerstein, P. Game theory and human evolution: A critique of some recent interpretations of experimental games. Theor. Popul. Biol. 69, 339–348 (2006).
El-Mouden, C., Burton-Chellew, M., Gardner, A. & West, S. A. What do humans maximize? In Evolution and Rationality: Decisions, Cooperation and Strategic Behaviour (eds Okashi, S. & Binmore, K.) 23–49 (Cambridge University Press, Cambridge, 2012). https://doi.org/10.1017/CBO9780511792601.003.
Kramer, R. M. & Brewer, M. B. Effects of group identity on resource use in a simulated commons dilemma. J. Pers. Soc. Psychol. 46, 1044 (1984).
Brewer, M. B. & Kramer, R. M. Choice behavior in social dilemmas: Effects of social identity, group size, and decision framing. J. Pers. Soc. Psychol. 50, 543 (1986).
Henrich, J. & Muthukrishna, M. The origins and psychology of human cooperation. Annu. Rev. Psychol. 72, 207–240 (2021).
Dunbar, R. I. The anatomy of friendship. Trends Cogn. Sci. 22, 32–51 (2018).
Richerson, P. J. & Boyd, R. Complex societies. Hum. Nat. 10, 253–289 (1999).
Chang, C.-C., Kristensen, N. P., Le Nghiem, T. P., Tan, C. L. & Carrasco, L. R. Having a stake in the future and perceived population density influence intergenerational cooperation. R. Soc. Open Sci. 8, 210206 (2021).
Wiessner, P. Indoctrinability and the evolution of socially defined kinship. In Indoctrinability, Ideology and Warfare: Evolutionary Perspectives (eds Eibl-Eibesfeldt, I. & Salter, F.) 133–150 (Berghahn Books, Oxford, 1998).
Read, D. From experiential-based to relational-based forms of social organization: A major transition in the evolution of Homo sapiens. Proc. Br. Acad. 158, 199–229 (2010).
Read, D. Cultural kinship as a computational system: From bottom-up to top-down forms of social organization. Comput. Math. Organ. Theory 18, 232–253 (2012).
Korchmaros, J. D. & Kenny, D. A. Emotional closeness as a mediator of the effect of genetic relatedness on altruism. Psychol. Sci. 12, 262–265 (2001).
McPherson, M., Smith-Lovin, L. & Cook, J. M. Birds of a feather: Homophily in social networks. Ann. Rev. Sociol. 27, 415–444 (2001).
Ebenau, A., von Borell, C., Penke, L., Ostner, J. & Schülke, O. Personality homophily affects male social bonding in wild Assamese macaques, Macaca assamensis. Anim. Behav. 155, 21–35 (2019).
Curry, O. & Dunbar, R. I. Do birds of a feather flock together?. Hum. Nat. 24, 336–347 (2013).
Krebs, D. Empathy and altruism. J. Pers. Soc. Psychol. 32, 1134–1146 (1975).
Billig, M. & Tajfel, H. Social categorization and similarity in intergroup behaviour. Eur. J. Soc. Psychol. 3, 27–52 (1973).
Burger, J. M., Messian, N., Patel, S., Del Prado, A. & Anderson, C. What a coincidence! The effects of incidental similarity on compliance. Pers. Soc. Psychol. Bull. 30, 35–43 (2004).
Moffett, M. W. Human identity and the evolution of societies. Hum. Nat. 24, 219–267 (2013).
Van Lange, P. A. M. & Rand, D. G. Human cooperation and the crises of climate change, COVID-19, and misinformation. Annu. Rev. Psychol. 73, 379–402. https://doi.org/10.1146/annurev-psych-020821-110044 (2022).
Acknowledgements
We thank Jorge Peña and Marco Archetti for interesting discussions, and thank Lynette Loke and Fanhua Kong for comments on early drafts. NPK and RA were funded by the Ministry of Education, Singapore, under a Tier 1 Grant (WBS A-0004766-00-00). HO acknowledges the support by JSPS KAKENHI Grant Numbers JP19H04431 and JP20K06812.
Author information
Authors and Affiliations
Contributions
All authors contributed to conceptualisation, mathematical analysis, review and editing. N.P.K. conceived the central concept, performed most of the mathematical analysis, coding, and literature review, and wrote the original draft. H.O. primarily oversaw the methodology and mathematical analysis, and R.A.C. oversaw writing, contributed code and analysis, and acquired funding.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kristensen, N.P., Ohtsuki, H. & Chisholm, R.A. Ancestral social environments plus nonlinear benefits can explain cooperation in human societies. Sci Rep 12, 20252 (2022). https://doi.org/10.1038/s41598-022-24590-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-24590-y
This article is cited by
-
Assortment by Group Founders Always Promotes the Evolution of Cooperation Under Global Selection But Can Oppose it Under Local Selection
Dynamic Games and Applications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.